Abstract
Background:
The management of early breast cancer patients with metastatic sentinel lymph nodes (SLNs) remains a critical decision point in balancing survival outcomes with surgical morbidities. This study addresses whether sentinel lymph node biopsy (SLNB) with or without regional node irradiation (RNI) can offer comparable survival outcomes to axillary lymph node dissection (ALND).
Objectives:
To evaluate overall survival (OS), disease-free survival (DFS), locoregional recurrence (LRR), and mortality outcomes of SLNB (with or without RNI) compared with ALND in patients with metastasis-positive SLNs.
Design:
We have performed an updated meta-analysis of randomized controlled trials (RCTs) comparing SLNB alone or with RNI vs. ALND for early breast cancer patients with metastatic SLNs.
Data Sources and Methods:
Eligibility criteria included RCTs comparing SLNB ± RNI vs ALND for metastasis-positive SLN. PubMed, EMBASE, the Cochrane library, and online registers were searched for articles comparing SLNB alone vs. ALND for metastasis-positive SLN. Articles were evaluated for risk of bias using Cochrane’s revised tool (RoB). The main summary measures using the random effects model were hazard ratio and risk ratio.
Results:
Seven RCTs were included in the meta-analysis. Overall survival and DFS were superior in the SLNB group compared to the ALND group. Locoregional recurrence and mortality were comparable between both the groups. After stratifying RCTs with regards to adjuvant RNI, these parameters were still comparable to ALND.
Conclusion:
This meta-analysis suggests that SLNB, with or without RNI, offers comparable OS and DFS to ALND for early breast cancer patients with metastasis-positive SLNs. However, the inclusion of studies that did not differentiate between micrometastases and macrometastases may introduce bias, particularly when assessing the impact of RNI. The role of RNI remains debated specially in those with macrometastases, and future research should focus on stratified analyses to clarify this. While SLNB represents a viable alternative to ALND, further trials are needed to define the optimal role in subgroups with high-risk tumor biology.
Keywords
Introduction
Traditional management for early breast cancer patients with a clinically and radiologically node-negative axilla is sentinel lymph node biopsy (SLNB), followed by axillary lymph node dissection (ALND) in metastatic SLN.1-4 Emerging evidence, including systematic reviews and meta-analyses, suggests that SLNB alone or with regional nodal irradiation (RNI) may suffice in carefully selected patients, sparing the potential complications of ALND (lymphedema, limited shoulder mobility, and neuropathic pain) without compromising oncological outcomes.5-11
Owing to this, international guidelines recommended against the completion ALND in patients with metastatic SLN.1,12,13 However, one of the reviews showed a statistically significant difference in locoregional recurrence (LRR) in favor of ALND. 14 Hence, despite these recommendations, debates persist regarding the optimal management approach for patients with metastatic SLNs, particularly concerning the role of RNI in enhancing regional control and potentially improving survival outcomes. The need for a clear understanding of whether SLNB alone, SLNB with RNI, or ALND is preferable remains critical. Moreover, there is a lack of consensus on the effects of RNI in preventing recurrence when ALND is omitted, as well as its impact on long-term survival.
This study aims to address these gaps by conducting an updated meta-analysis of randomized controlled trials (RCTs) comparing SLNB with or without RNI against ALND in early-stage breast cancer patients with metastatic SLNs. By synthesizing the latest evidence, this analysis seeks to provide a comprehensive assessment of overall survival (OS), disease-free survival (DFS), LRR, and mortality outcomes, contributing valuable insights into the effectiveness and safety of SLNB as a potential alternative to ALND in specific patient populations.
Methods
Search strategy
The authors designed a priori search protocol (Supplementary document 1) to identify evidence relevant to this meta-analysis per Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines for updated systematic reviews 15 (Supplementary file 2). MedLine (via PubMed), EMBASE, Cochrane library, online clinical trial registers, and websites (Clinicaltrials.gov, Ctri.nic.in) were reviewed from its inception till 30th April 2024. Reviewers (MMI, SA, and SKY) conducted the comprehensive electronic search (Appendix 1). Titles and abstracts were screened for eligibility by both reviewers. Full texts were retrieved after excluding irrelevant studies, duplicate studies, review articles, case reports, editorials, and letters to the editor/correspondences. After reading the full text, each article was coded as eligible, ineligible, or doubtful. All disagreements were resolved via discussion among all reviewers through online consultations.
Inclusion criteria
Study designs: RCTs
Participants: All patients of early breast cancer whose SLNB was positive for metastasis
Intervention: SLNB alone or with RNI
Comparator: Patients undergoing ALND after SLNB was positive for metastasis
Outcome: Primary outcome measures were OS and DFS. Secondary endpoints were LRR and death.
In case of multiple papers based on the same or overlapping data sets, then only the article with the most extended follow-up of patients was included
Exclusion criteria
Study designs: Prospective observation studies, case-control studies, review articles, case reports, editorials, and letters to the editor/correspondence
Participants: Studies not comparing SLNB/RNI vs ALND or studies that have used only one type and not compared the two groups
Outcome: Studies not providing required data of OS, DFS, LRR, and death
Quality assessment
The methodologic quality of included studies was assessed using Cochrane’s revised tool to assess the risk of bias in randomized trials (RoB 2 tool). 16 Two reviewers evaluated individual bias items by assigning the above “Risk of bias” criteria as either “low,” “high,” or “unclear” risk.
Data extraction, analysis, and synthesis
Data extraction was carried out by the first author (MMI) and independently adjudicated by one other author (SKY) using a pilot data extraction form developed to address the research question. Disagreements were resolved by discussion among the reviewers. The extracted data included sample size, pathological definition of SLN metastasis, LRR, OS, DFS, and treatment regimens.
Statistical methods
Hazard ratio (HR) was used as a summary statistic for outcomes (OS and DFS). We calculated the risk ratio (RR) and corresponding 95% confidence interval (95% CI) for dichotomous outcomes. A statistically significant overall effect was defined as P ⩽ .05 or a 95% CI that did not cross the line of no effect (ie, 1 for RR).
Visual inspection of the forest plots and I² statistic, which quantifies inconsistency impacting meta-analysis, were used to describe heterogeneity. An I² statistic of 75% or more indicates a high degree of heterogeneity, and in such a situation, we did not pool data for meta-analysis. 17
Data analysis was performed using the Cochrane Collaboration’s Review Manager Software (RevMan version 5.3). Publication bias was assessed by visual inspection of the funnel plot.
Level of evidence
The grading of recommendation, assessment, development, and evaluation (GRADE) instrument was used for recommendation strength in the included studies in the meta-analysis.18,19 The quality of the evidence was characterized as high, moderate, low, or very low. The GRADE was assessed using an online tool from the website http://gradepro.org. Two review authors (SA and SKY) independently rated the quality of each outcome.
Results
As per our pre-defined criteria, seven articles were eligible for inclusion (Figure 1). All included studies were RCTs utilizing a parallel group non-inferiority design.20-26 The AMAROS trial recently published a 10-year follow-up, which was included in this meta-analysis rather than this group’s previous publication. 21 The characteristics of the included studies are listed in Table 1. A total of 7030 patients were included in the studies, 3585 in the SLNB/RNI group, and 3445 in the ALND group. The median duration of follow-up was 78.8 months (interquartile range 69.6 months). All studies reported outcomes directly comparing SLNB alone or with RNI vs ALND. Patients received postoperative adjuvant systemic therapy according to guidelines used at each center.

PRISMA 2020 flow diagram for updated systematic reviews which included searches of databases, registers, and other sources.
Study characteristics.

Risk of bias assessment
Each risk of bias item for individual studies and across all studies are depicted in Figures 2 and 3. Three studies did not report methods for random sequence generation and were rated as high risk of bias,21-23 three had an unclear risk of bias,20,21,25 and one had a low risk of bias. 26 All studies had unclear risk of bias for allocation concealment 20-22,25,26 except IBCSG (no risk) and OTOASAR (high risk). All studies had a low risk of bias for blinding participants except SENOMAC and SINODAR (high risk). All studies had a low risk of bias for other parameters.

Risk of bias graph: each risk of bias item presented as percentages across all included studies.

Risk of bias summary: each risk of bias item for individual studies.
Meta-analysis of primary outcomes
Five trials provided data to calculate the HR. Overall HR for death was 0.84 (95% CI: 0.73-0.97, P = .01), suggesting OS was superior in the SLNB group (Figure 4A). The heterogeneity (I2) was 0%, indicating low heterogeneity. Sensitivity analysis was conducted to assess the effect of each RCT on the OS, and it showed that the results were the same, ie, OS was superior in the SLNB group (the highest P-value was .05 after removing OTOASAR RCT). We also analyzed HR for OS based on further axillary treatment in the SLNB arm with or without adjuvant RNI. The SENOMAC trial did not provide adjuvant RNI data; hence, it was excluded. On stratifying RCTs with SLNB alone (no RNI) vs ALND (ACOSOGZ0011 and IBCSG), OS was still superior in SLNB alone group but statistically non-significant (HR 0.85, 95% CI 0.68-1.06, P = .14) (Figure 4B). Adjuvant RNI to axilla was given in two studies (AMAROS and OTOASAR), and although non-significant, OS was superior in the SLNB + RNI group as compared with ALND (HR 0.76, 95% CI 0.57-1.07, P = .11) (Figure 4C).

Forest plot for overall survival (A) overall SLNB group vs ALND, (B) SLNB alone vs ALND, and (C) SLNB + RT vs ALND.
Five trials were included in DFS synthesis as they provided sufficient data to pool the results. Overall HR for death was 0.85 (95% CI: 0.74-0.97, P = .02, (I2) = 0%.), suggesting DFS was superior in the SLNB group (Figure 5A). The sensitivity analysis conducted to assess the effect of each trial showed that if AMAROS trial is excluded from the analysis, results are rendered nonsignificant (P = .10). Similarly, we analyzed HR for DFS based on whether adjuvant RNI was given after SLNB or not in the experimental arm. Again, the SENOMAC trial did not provide adjuvant RNI data; hence, it was excluded. On stratifying RCTs with SLNB alone (no RNI) vs ALND (ACOSOGZ0011 and IBCSG), DFS was still superior in SLNB alone group but statistically non-significant (HR 0.85, 95% CI 0.68-1.06, P = .17) (Figure 5B). Adjuvant RNI to axilla was given in two studies (AMAROS and OTOASAR), although non-significant, OS was superior in SLNB + RNI group as compared with ALND (HR 0.84, 95% CI 0.68-1.02, P = .08) (Figure 5C).
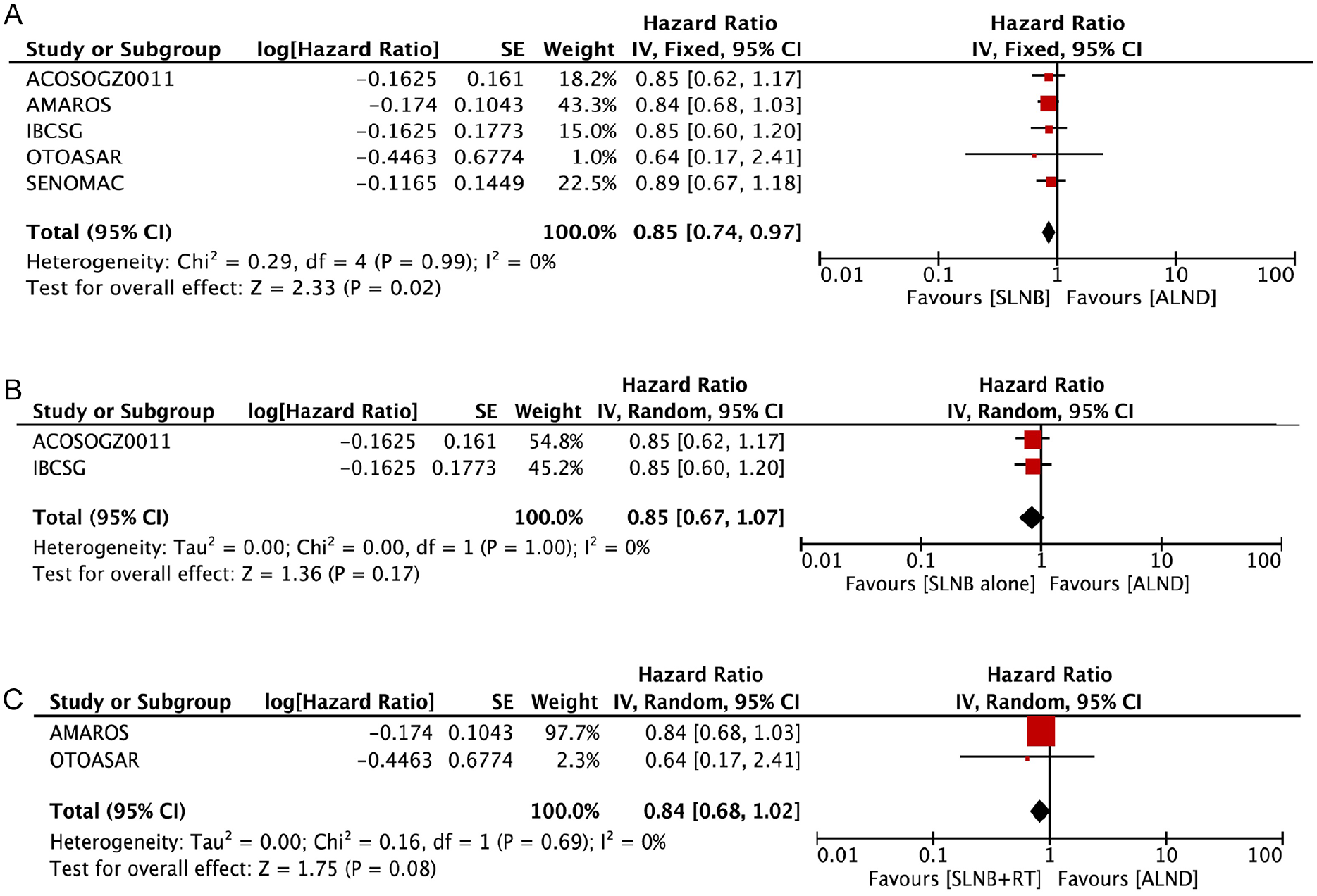
Forest plot for disease free survival (A) overall SLNB group vs ALND, (B) SLNB alone vs ALND, and (C) SLNB with RT vs ALND.
Meta-analysis of secondary outcomes
Seven studies were included in the meta-analysis of LRR, and it was slightly higher in the SLNB group. The overall RR was 1.18 (95% CI 0.87-1.60), but it was not significant (P = .29, (I2) = 0%.) (Figure 6A). In the sensitivity analysis, omission of any RCT did not render the results statistically significant. Similar to OS and DFS, we analyzed trials based on SLNB alone vs ALND (AATRM, ACOSOGZ0011, IBCSG, and SINODAR) and SLNB + RNI vs ALND (AMAROS and OTOASAR) (Figure 6B and C). The SENOMAC trial was excluded as it did not provide RNI data. Sentinel lymph node biopsy alone and SLNB + RT both had similar LRRs to those of ALND.

Forest plot for locoregional recurrence (A) overall SLNB group vs ALND, (B) SLNB alone vs ALND, and (C) SLNB with RT vs ALND.
Six studies were included in the meta-analysis of mortality (the total number of deceased patients per trial). The AATRM trial was excluded as it did not provide mortality data. Mortality was higher in the ALND group, but it was not significant statistically (RR 0.90, 95% CI 0.77-1.04, P = .15, (I2) = 0%.) (Figure 7A). The exclusion of the AMAROS trial renders the RR (0.80, 95% CI 0.67-0.97) statistically significant with a P-value equal to .02. We analyzed trials based on SLNB alone vs ALND (ACOSOGZ0011, IBCSG and SINODAR) and SLNB + RNI vs ALND (AMAROS and OTOASAR) (Figure 7B and C). The SENOMAC trial was excluded as it did not provide RNI data. Sentinel lymph node biopsy alone and SLNB + RNI both had similar mortality compared with ALND.

Forest plot for mortality (A) overall SLNB group vs ALND, (B) SLNB alone vs ALND, and (C) SLNB with RT vs ALND.
Visual inspection of the funnel plot did not reveal any publication bias (Figure 8). Overall, the quality of the evidence from the outcomes evaluated by the GRADE system was high for OS, DFS, LRR, and mortality, suggesting high confidence in the estimated effect from the assessed outcomes (Table 2).

Funnel plot.
GRADE assessment.
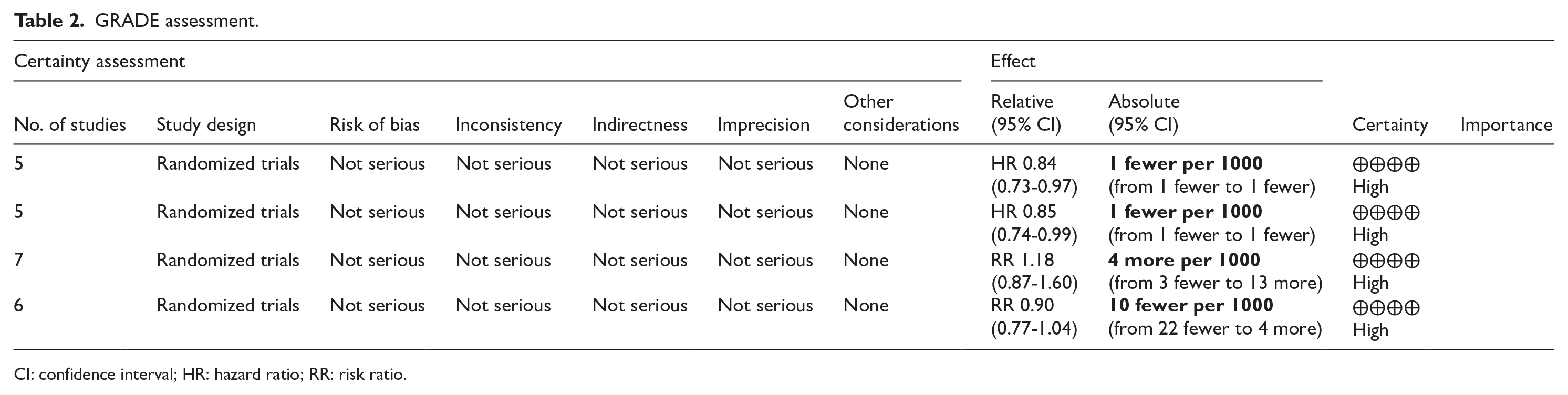
CI: confidence interval; HR: hazard ratio; RR: risk ratio.
Discussion
Our updated systematic review and meta-analysis confirms that SLNB is associated with superior OS and DFS compared to ALND, with no significant difference in LRR between the two groups. This updated analysis further reveals that OS, DFS, LRR, and mortality outcomes are comparable to ALND, even when adjuvant regional node irradiation (RNI) is not administered.
Systematic reviews and meta-analyses are essential tools an essential tool in evidence-based medicine as they identify, select, appraise, and synthesize evidence from relevant studies on a particular topic. By including all available evidence, it provides a more comprehensive understanding of the research landscape compared with individual studies. Meta-analysis, a component of systematic review, statistically combines data from multiple studies to provide a more precise estimate of the effect size which enhances the statistical power of the findings. In addition, systematic reviews reveal gaps in existing research thereby highlighting areas where further investigation is needed. This helps guide future research priorities and clinical practice.
In the last couple of decades, the standard axillary management of early breast cancer has evolved, and SLNB has replaced ALND for the treatment of clinically node-negative axilla. 27 The concept of further de-escalation of axillary surgery in metastatic SLN was first introduced widely by milestone RCT of the American College of Surgeons Oncology Group (ACOSOG) Z0011). 21 After the ACOSOGZ0011 trial, several other trials showed improved OS and DFS with SLNB ± RNI compared with ALND in early-stage breast cancer.20,22-26 As expected, locoregional standards for axillary management became the hottest research topic, and multiple meta-analyses were performed.8,11,28-33 Some of these could not provide conclusive recommendations as they were based on limited RCTs.2-4,28-32 Meta-analysis by Peristeri and Harissis 14 included five RCTs and provided high-strength evidence that SLNB was associated with better OS and DFS. However, they also reported an increased risk of LRR with SLNB; similar results were found by other systematic reviews and meta-analyses.8-10 However, these studies included prospective cohort and retrospective studies as well, thereby introducing the possibility of selection biases and heterogeneity, Our updated meta-analysis employed a more rigorous methodology (inclusion of only RCTs), comprehensive search strategy (including two latest RCTs and updated results of a previous trial), and assessment for the strength of the evidence (by GRADE assessment). We have also analyzed data for SLNB alone and SLNB + RNI vs. ALND, as sufficient number of trials were available. Our analysis suggests that ALND can be safely omitted in patients with limited SLN metastasis, defined variably across studies as either micrometastases or macrometastases up to 1 to 2 positive sentinel lymph nodes. Given these differences in inclusion criteria, further research is needed to refine patient selection criteria for axillary de-escalation.
The selection of patients for the omission of ALND is debatable. The high-risk groups like triple-negative, HER2 positives, SLN with extracapsular extension and cT2 breast cancer patients were underrepresented in all trials except SENOMAC. In addition, although all except the Z0011 trial have included mastectomy, the number of patients with mastectomy was significantly less compared with BCS. The radiotherapy nodal targets were not explicit in Z0011, and in SENOMAC, 89.9% had nodal RT in the SLNB group. The awaited result of POsitive Sentinel NOde: adjuvant therapy alone versus adjuvant therapy plus Clearance or axillary radiotherapy (POSNOC) RCT may help physicians in objectively selecting patients for axillary de-escalation (observation vs RT) in SLN-positive early breast cancer patients. 34
The major limitations of our meta-analysis were the limited number of RCTs, some of which have short follow-up periods as delayed recurrences are common in breast cancer. However, an updated follow-up of 10 years of AMAROS trial favors the SLNB group. 15 Another limitation is that these studies should have reported pathologists’ experience and detailed pathological evaluation techniques. Diagnosis of metastasis is increased with experienced pathologists, and hence, our research findings may not be applicable in settings where this is lacking. Another key limitation is the inclusion of studies that did not distinguish between micrometastases and macrometastases in positive sentinel lymph nodes. This is particularly relevant when assessing the impact of RNI, as its benefit remains unclear for patients with macrometastases. Finally, the underrepresentation of certain high-risk patient subgroups, such as those with HER2-positive or triple-negative breast cancer, extracapsular extension, or larger tumors, limits the generalizability of our findings.
While our study suggests that RNI may serve as an alternative to ALND in selected cases, our meta-analysis did not demonstrate a statistically significant survival advantage with RNI. Further research is needed to better define the role of RNI in patients with positive SLNs who forego ALND. Future research could explore whether certain subgroups benefit more from RNI than others. Additional long-term studies focusing on patient-reported outcomes, such as quality of life, functional outcomes, and lymphedema rates, are essential. Such research would provide a more comprehensive understanding of the real-world impact of these approaches and could further inform patient-centered care decisions. Further trials to address high-risk subgroups such as patients with triple-negative breast cancer, HER2-positive tumors, and those with larger tumors or extracapsular extension remain underrepresented in trials. Targeted trials are needed to evaluate the safety and efficacy of omitting ALND in these subpopulations.
Conclusion
This meta-analysis suggests that SLNB, with or without RNI, offers comparable OS and DFS to ALND for early breast cancer patients with metastasis-positive SLNs. However, the inclusion of studies that did not differentiate between micrometastases and macrometastases may introduce bias, particularly when assessing the impact of RNI. Role of RNI remains debated specially in those with macrometastases, and future research should focus on stratified analyses to clarify this. While SLNB represents a viable alternative to ALND, further trials are needed to define the optimal role in subgroups with high-risk tumor biology.
Supplemental Material
sj-docx-1-bcb-10.1177_11782234251335409 – Supplemental material for Sentinel Lymph Node Biopsy vs. Axillary Lymph Node Dissection for Early-Stage Breast Cancer and Sentinel Lymph Node Metastasis: An Updated Systematic Review and Meta-Analysis With Special Focus on Locoregional Recurrence and Regional Node Irradiation
Supplemental material, sj-docx-1-bcb-10.1177_11782234251335409 for Sentinel Lymph Node Biopsy vs. Axillary Lymph Node Dissection for Early-Stage Breast Cancer and Sentinel Lymph Node Metastasis: An Updated Systematic Review and Meta-Analysis With Special Focus on Locoregional Recurrence and Regional Node Irradiation by Sanjit Kumar Agrawal, M. Mohammed Imran, Shivank Sethi, Sanjay Kumar Yadav, Pawan Agarwal and Dhananjaya Sharma in Breast Cancer: Basic and Clinical Research
Supplemental Material
sj-docx-2-bcb-10.1177_11782234251335409 – Supplemental material for Sentinel Lymph Node Biopsy vs. Axillary Lymph Node Dissection for Early-Stage Breast Cancer and Sentinel Lymph Node Metastasis: An Updated Systematic Review and Meta-Analysis With Special Focus on Locoregional Recurrence and Regional Node Irradiation
Supplemental material, sj-docx-2-bcb-10.1177_11782234251335409 for Sentinel Lymph Node Biopsy vs. Axillary Lymph Node Dissection for Early-Stage Breast Cancer and Sentinel Lymph Node Metastasis: An Updated Systematic Review and Meta-Analysis With Special Focus on Locoregional Recurrence and Regional Node Irradiation by Sanjit Kumar Agrawal, M. Mohammed Imran, Shivank Sethi, Sanjay Kumar Yadav, Pawan Agarwal and Dhananjaya Sharma in Breast Cancer: Basic and Clinical Research
Footnotes
Appendix 1
Ethical Considerations
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Institutional ethical approval was not applicable.
Consent for Publication
Not applicable
Author Contributions
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data may be accessed upon reasonable request to the corresponding author.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
