Abstract
Background:
The application of sentinel lymph node biopsy (SLNB) has expanded from early breast cancer to locally advanced breast cancer with neoadjuvant chemotherapy (NAC). For patients with negative axillary lymph nodes, performing SLNB before or after NAC remains controversial.
Objectives:
To evaluate the diagnostic feasibility and reliability of SLNB after NAC in breast cancer patients with negative axillary nodes at initial diagnosis.
Design:
To calculate pooled identification rate (IR) and false negative rate (FNR) of SLNB after NAC on breast cancer patients with initially negative axillary nodes by enrolling relevant studies and perform subgroup analysis by the type of tracer and the number of biopsied sentinel lymph nodes in average.
Data sources and methods:
The PubMed, Embase, Cochrane, Web of Science, and Scopus databases from January 1, 2002, to March 1, 2022, were searched for studies. The QUADAS-2 tool and MINORS item were employed to evaluate the quality of the included studies. I2 and Q tests were used to evaluate the heterogeneity among the studies. Random-effects model and fixed-effects model were employed to calculate the pooled IR, FNR, and 95% confidence interval (CI). Publication bias was evaluated, and sensitivity analysis was performed. Subgroup analysis was performed according to the type of tracer (single/double) and the number of biopsied sentinel lymph nodes in average (⩽2/>2).
Results:
A total of 21 studies covering 1716 patients were enrolled in this study (IR = 93%, 95% CI = 90-96; FNR = 8%, 95% CI = 6-11).
Conclusion:
The SLNB after NAC can serve as a feasible and reliable approach in breast cancer patients with negative axillary lymph node. In our study, no significant impact of tracer was found on the IR and FNR of SLNB, and the number of biopsy nodes >2 leads to the decreased FNR of SLNB.
Keywords
Introduction
According to the latest cancer data published by the World Health Organization’s International Agency for Research on Cancer (IRAC) in 2020, breast cancer has been ranked as the highest cancer worldwide, surpassing the 2.2 million new lung cancer cases with 2.26 million new breast cancer cases. 1 Since the 1970s, NAC has been widely adopted to patients with operable breast cancer. More and more patients with locally advanced breast cancer are offered the opportunity of breast-conserving surgery as NAC is applied to shrink tumors and reduce clinical stage. 2 Information about the sensitivity to drugs can also be obtained during neoadjuvant chemotherapy (NAC), so as to guide subsequent adjuvant therapy and improve prognosis.
Axillary staging is a significant element for breast cancer treatment. In the NSABP-B-32 trial, covering 5611 early stage breast cancer patients with negative SLNs, no significant difference was found in disease-free survival (DFS) and overall survival (OS) between patients undergoing axillary lymph node dissection (ALND) or omitting ALND. 3 In 2005, the ASCO guidelines recommend SLNB to early stage breast cancer patients with clinical negative axillary nodes, 4 which have developed to the standard axillary surgery for early stage breast cancer patients with cN0. In Z0011 trial where patients with cN0 and 1-2 positive SLNs were randomly divided into the SLNB and ALND groups, no significant difference was revealed in regional recurrence, distant metastasis, and OS between these 2 groups, 5 so was in the AMAROS trial, suggesting that ALND can be omitted in cN0 patients with 1-2 positive SLNs. Meanwhile, the incidence of lymphedema in the SLNB group was significantly decreased compared with the ALND group (11% vs 23%). 6
Over the years, ALND has served as the standard axillary surgery for breast cancer patients after NAC. As for the present, SLNB after NAC has been discussed by a growing body of studies in patients with clinically negative axillary lymph nodes at initial diagnosis.7-9 The 2016 ASCO guidelines recommend SLNB for operable breast cancer patients with preoperative/neoadjuvant systemic therapy. 10 It has been proposed higher significance of SLNB in predicting local recurrence after NAC compared with before NAC, as well as more guiding significance in axillary treatment. 11 It is noticeable that SLNB after NAC allows for once operation, avoiding the physical injury and economic problems caused by 2-time operations. However, it has been suggested a lower IR and higher FNR of SLNB after NAC due to the possibility of lymphatic channel fibrosis that redirects the tracer flow away from the true SLNs, 12 which is the main reason for the unacceptably high FNR of SLNB after NAC in those cN1-cN2 stage patients. Therefore, some suggested higher accuracy and a better definition of clinical staging by performing SLNB before NAC.
At present, the IR and FNR of SLNB after NAC in breast cancer patients with clinically negative axillary lymph nodes have been assessed in several trials. Herein, we carried out a meta-analysis by collecting data from these trials to evaluate the diagnostic feasibility and safety of SLNB after NAC.
Materials and Methods
Literature search strategy
The systematic review was conducted according to the preferred reporting item guidelines for systematic review and meta-analysis (PRISMA). In this study, PubMed, Embase, Cochrane, Web of Science, and Scopus databases were searched from January 1, 2002, to March 1, 2022. The following free text terms and Medical Subject Headings (Mesh) terms were used: (“breast neoplasm” OR “breast cancer”) AND (“sentinel lymph node biopsy” OR “SLNB” OR “sentinel lymph node dissection”) AND (“neoadjuvant chemotherapy” OR “preoperative chemotherapy” OR “neoadjuvant treatment”).
Study inclusion criteria
The screening and exclusion of studies were conducted by 2 reviewers independently, with the inconsistent results determined by the third researcher. The inclusion criteria were as follows: (1) breast cancer patients with negative clinically axillary lymph nodes at initial diagnosis; (2) all patients received NAC; (3) all patients received SLNB after NAC, followed by routine ALND regardless of the results of SLNB; and (4) the clear IR and FNR of SLNB were provided. Inflammatory breast cancer, history of chemoradiotherapy, and prior axillary surgery were excluded.
Study quality assessment
Two reviewers independently evaluated the quality of included studies using Quality Assessment of Diagnostic Accuracy Studies 2 (QUADAS-2) tool. 13 The inconsistent results were determined by the third researcher. The tool covered 4 domains: patient selection, index test, reference standard, and flow and timing. The risk of bias will be assessed for all domains, and the applicability concerns for the first 3 domains. In each domain, the risk will be accessed as “low” if the answers to all questions are “yes”; “high” if at least 1 answer is “no”; and “unclear” if there is no sufficient evidence for at least 1 question. Meanwhile, the 12 items of the MINORS item were used for quality evaluation. Each item is 2 points, with a total score of 24 points, and studies with 15 points or more get enrolled in our research.
Data extraction and definitions
The following features of the enrolled studies were independently extracted by 2 reviewers: author, year, center, design, country, sample size, method of pre-NAC axilla assessment, tracer, average number of biopsied SLNs, and SLNB pathology method. Inconsistent results would be determined by the third researcher. The extracted outcome indexes referred to IR and FNR of SLNB. The IR was defined as the total number of patients who were successfully identified SLNs divided by sample size. Taking ALND as the gold standard, false negative means no breast cancer metastasis according to the pathology result of SLNB, while identified by the pathology result of ALND. The FNR was defined as the number of false negative patients divided by the sum of true positive patients (SLNB+ and ALD+) and false negative patients (SLNB− and ALD+).
Data analysis
The statistical analysis was performed in STATA 17.0 version. The heterogeneity among the enrolled studies was assessed by Q test and quantified by I 2 . 14 The I 2 ⩾ 50% indicated the heterogeneity among, and the sensitivity was analyzed using random-effect model. Otherwise, fixed-effects model would be employed. Forest plot was generated to show the effect sizes of each study and the pooled results (95% CI). Publication bias was estimated by funnel plot. An inverted symmetrical funnel shape indicated no publication bias of the included study, whereas the asymmetrical or incomplete funnel plot indicated publication bias. Egger test was also performed to evaluate the potential publication bias where P < .1 indicated statistically significant. Sensitivity analysis was performed to evaluate the effect of excluding any study on the overall effect. Subgroup analysis was carried out based on the tracer type (single/double) and the average number of biopsied sentinel nodes (⩽2/>2).
Results
Literature screening results
The depiction of literature screening process was detailed in Figure 1. A total of 2171 articles were obtained after the initial searching. The remaining 1044 articles were assessed after excluding 626 duplicate articles, 443 irrelevant articles, and 58 articles without full text. The reasons for exclusion mainly distributed in all patients were diagnosed as positive axillary lymph nodes; the number of patients with clinically negative axillary lymph nodes was not reported; some patients did not receive neoadjuvant therapy; some patients received SLNB before neoadjuvant therapy; some patients did not receive ALND; the IR and FNR of SLNB were not reported. Eventually, 21 articles covering 1716 patients were enrolled in the analysis.

Flow diagram of literature search.
Characteristics and quality assessment of included studies
The characteristics of the enrolled studies were depicted in Table 1. The 21 included articles were published from 2003 to 2021, respectively, from China (5), the USA (3), Japan (3), France (2), Brazil (2), Belgium (1), Korea (1), Italy (1), Spain (1), Czech (1), and Israel (1). Among them, 18 were single-center studies and 3 were multi-center studies; 14 were prospective and 7 were retrospective, with the sample sizes ranging from 29 to 172, 81.7 in average. Palpation was applied alone in 3 studies as a diagnostic method for axillary lymph node staging before neoadjuvant therapy, imaging diagnosis (with or without palpation) in 11 studies, and fine-needle biopsy (FNA) based on imaging in 4 studies. The methylene blue dye was used alone as SLNB tracer in 4 studies, radioisotope alone in 1, dual tracers in 7, radioisotope tracer and lymphatic imaging in 4, and dual tracers and lymphatic imaging in 5. Eleven studies reported the average number of SLNs biopsied, of which 6 had an average number of SLNs ⩽2 and >2 in 5. Hematoxylin-eosin (HE) staining alone was adopted in 9 studies and HE staining plus immunohistochemistry in 11.
Characteristics of the included studies.

Abbreviations: NAC, neoadjuvant chemotherapy; SLN, sentinel lymph node; SLNB, sentinel lymph node biopsy B, blue dye; R, radioisotope; L, lymphoscintigraphy; HE, hematoxylin–eosin; IHC, immunohistochemistry; FNA, fine-needle biopsy.
In 21 included studies, only Classe, Rebollo, Pecha, Yu, and Hunt gave clear definition of isolated tumor cells (ITCs), micrometastasis, and macrometastasis, whereas other 16 studies did not mention that. In these 5 studies, positive axillary lymph node was defined as metastatic deposits measured larger than 0.2 mm in size according to the AJCC staging manual.
The depiction of specific results of the quality assessment of the studies was detailed in Table 2, and the results of the quality assessment are summarized in Figure 2. There reported the coexistence of uncertainties and high risks in the patient selection part, and the quality was generally considerable in the other 3 parts of index test, reference standard, flow, and timing. Meanwhile, Table 3 describes the results of the quality assessment of included studies according to MINORS item.
Results of quality evaluation for the included studies according to the QADAS2 tool.


Summary of quality evaluation results.
MINORS quality evaluation for included studies.

Meta-analysis
The IR of included studies ranged from 80.6% to 97.6%. Considering the high heterogeneity (I 2 = 76.50%, P < .001), random-effects model was adopted for analysis (pooled IR = 93%, 95% CI = 90-96 [Figure 3]).

Forest plot of IR.
The FNR of included studies ranged from 0% to 17.65%. Considering the low heterogeneity (I 2 = 13.79%, P = .28), fixed-effect model was adopted for analysis (pooled FNR = 8%, 95% CI = 6-11 [Figure 4]).

Forest plot of FNR.
Publication bias
A significant publication bias was indicated by the funnel plot of IR. Egger test showed no publication bias in IR (P = .073; Figure 5).
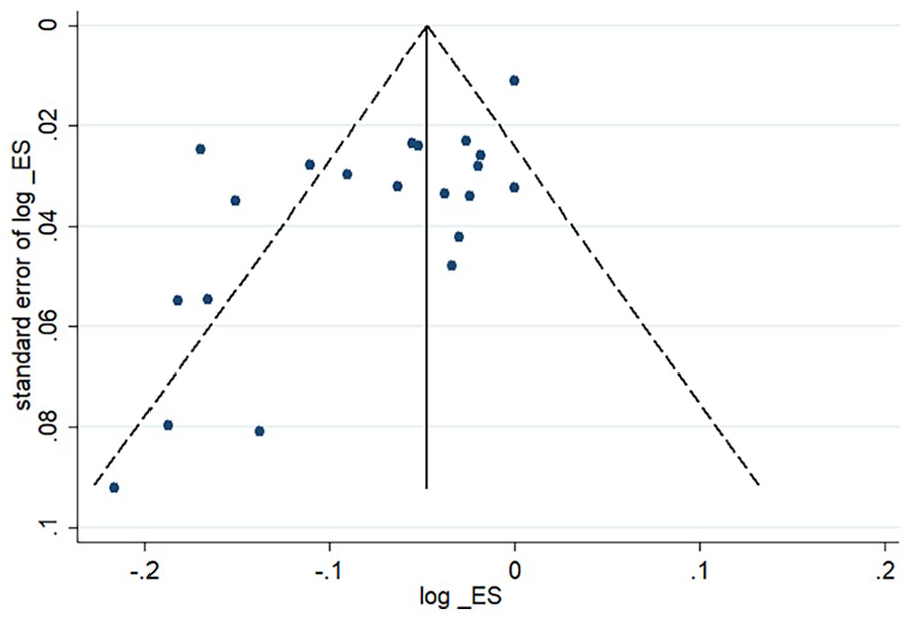
Funnel plot of IR.
No significant publication bias was observed in the funnel plot of FNR. Egger test showed no publication bias in FNR (P = .241; Figure 6).

Funnel plot of FNR.
Subgroup analysis
Subgroup analysis based on tracer revealed a pooled IR of 94% (95% CI = 92-96)9,15-18,33 and 93% (95% CI = 91-96)8,19-32 for single or dual tracers, and a pooled FNR of 7% (95% CI = 3-12) and 8% (95% CI = 5-11), respectively. Subgroup analysis based on the average number of SLNs biopsied showed a pooled IR of 95% (95% CI = 90-100)8,16,17,24-26 and 95% (95% CI = 92-97)9,15,19,20,29 for average SLNs ⩽2 or >2, and a pooled FNR of 10% (95% CI = 4-18) and 5% (95% CI = 2-9), respectively.
Sensitivity analysis
Sensitivity analysis of IR revealed that the effect size of outcome indicators did not significantly change after excluding any study, confirming the stability and credibility of the results of meta-analysis of IR.
Sensitivity analysis of FNR showed no significant change in the effect size of outcome indicators after excluding any study, indicating the stability and credibility of results of meta-analysis of FNR.
Discussion
This meta-analysis reviewed relevant studies in the last 2 decades to evaluate the diagnostic feasibility and reliability of SLNB after NAC in breast cancer patients with clinically negative axillary lymph nodes. The pooled IR was 93% (95% CI = 90-96) and the pooled FNR of 8% (95% CI = 6-11), meeting the acceptable criteria for SLNB in early breast cancer studies (IR > 90%, FNR < 10%), 34 indicating the feasibility and reliability of SLNB after NAC in patients with clinically negative axillary lymph nodes at initial diagnosis.
In Arm B of SENTINA trial, where patients received SLNB before and after NAC, the IR was only 60.8%, and the FNR was up to 51.6%. 7 Since the rather low accuracy of secondary SLNB, figuring out SLNB before or after NAC is important for axillary surgical treatment of breast cancer. A study on the timing of SLNB reported that there was no difference in IR of SLNB before or after NAC in breast cancer patients with cN0 (98% vs 95%; P = .032), whereas patients undergoing SLNB after NAC tended to exhibit negative SLNs (67% vs 54%; P = .001), followed by a correspondingly lower incidence of axillary surgery or radiation (33% vs 45%; P = .006). 35 In the MDACC retrospective study covering 3746 breast cancer patients with cN0, similar results of IR and FNR of SLNB before and after NAC are revealed (98.7% vs 97.4%, P = .017; 4.1% vs 5.9%, P = .39), without difference in regional recurrence, disease-free, or OS after clinical stage adjustment. 29 The evidence from randomized controlled trials comparing regional recurrence and long-term survival in patients with cN0 who underwent SLNB before or after NAC remains lacked, whereas non-randomized controlled trials and retrospective trials exhibited no difference in IR and FNR of SLNB before or after NAC. Obviously, SLNB after NAC can avoid physical injury and alleviate economic issues due to twice operations. So, the confirmation of feasibility and reliability of SLNB after NAC promotes it to serve as the ideal treatment for patients with cN0. This study confirms that it is reliable and feasible to perform SLNB after NAC in breast cancer patients with cN0 at initial diagnosis, which is of considerable significance for axillary treatment and tumor management of breast cancer.
There have published 7 meta-analyses about SLNB in breast cancer patients after NAC, with 4 focused on patients with positive axillary lymph nodes, 2 on patients with clinically negative axillary lymph nodes, and 1 on patients with positive or negative axillary lymph nodes. In the previous study, IR and FNR of SLNB after NAC in patients with clinically negative axillary lymph nodes were 94% (95% CI = 92-97) and 7% (95% CI = 2-12), 36 96% (95% CI = 95-97) and 6% (95% CI = 3-8), 37 and 94% (95% CI = 0.92-0.96) and 7% (95% CI = 0.05-0.09). 38 Our research basically reached the consistency with the foregoing researches, supporting the application of SLNB after NAC to guide axillary treatment in breast cancer patients with clinically negative axillary lymph nodes.
NASBP-B-27 trial revealed the IR of SLNB using methylene blue alone, radioisotope alone, or dual tracer of 78.1%, 88.9%, and 87.6%, respectively. 3 ACOSOG Z1071 trial indicated a significant decrease in FNR of SLNB using dual tracers compared with single tracer (10.8% vs 20.3%). 5 In this study, no significant difference was found between IR and FNR of SLNB with single/dual tracers, possibly due to the difference between axillary lymph nodes status of the included patients and aforementioned studies. Another similar meta-analysis based on patients with clinically negative axillary lymph nodes reported an IR of 97% and 91% with single/dual tracers, which revealed a broad consistency with our study. 38
In Arm C of SENTINA trial, factors such as the number of biopsied SLNs and tumor location posed a significant impact on the FNR. 7 The FNR was 17.7% in patients with 1 SLNs removed, 10% in those with 2 SLNs removed, and 6.9% in patients with 3 SLNs removed (P < .001). ACOSOG Z1071 trial also revealed the significantly reduced FNR when the number of biopsied SLNs was ⩾3. 39 Several other trials also recommended the excision of at least 2 SLNs.40,41 In the subgroup analysis of our study, the average number of biopsied SLNs in group 1 was 1.5 and the FNR of 10%, and the average number of biopsied SLNs in group 2 was 2.7 and the FNR of 5%. Accordingly, we suggest that the amount of SLNs to be biopsied should be at least 2 for the best accuracy of SLNB. In some cases, only one sentinel lymph node can be found due to the anatomy of patient. Some methods can be applied to biopsy more SLNs before removing them thus make sure the accuracy, such as applying dual tracers or prolonging the tracing time.
Conclusions
This meta-analysis evaluated the diagnostic feasibility and reliability of SLNB after NAC in breast cancer patients with negative axillary lymph nodes at initial diagnosis, with the pooled IR of 93% (95% CI = 90-96) and FNR of 8% (95% CI = 6-11). Correspondingly, it suggested that SLNB after NAC in patients with clinically negative axillary lymph nodes could be a feasible and accurate approach. Axillary lymph node dissection can be omitted in those with a negative SLNB result.
In this study, no significant influence of tracer on the IR and FNR of SLNB was found revealed, accompanied by the decreased FNR of SLNB when the number of biopsy nodes was >2. Therefore, the amount of SLNs biopsied is suggested to be at least 2 for the best accuracy of SLNB.
Limitations
In this study, to avoid sampling error derived from small sample size, only studies with sample size >20 were involved, which may affect the results. At the same time, only studies published in English were enrolled, which may lead to publication bias.
Supplemental Material
sj-docx-1-bcb-10.1177_11782234241255856 – Supplemental material for The Feasibility and Reliability of Sentinel Lymph Node Biopsy After Neoadjuvant Chemotherapy in Breast Cancer Patients With Negative Axillary Lymph Nodes—A Meta-analysis
Supplemental material, sj-docx-1-bcb-10.1177_11782234241255856 for The Feasibility and Reliability of Sentinel Lymph Node Biopsy After Neoadjuvant Chemotherapy in Breast Cancer Patients With Negative Axillary Lymph Nodes—A Meta-analysis by Mengjie Yu, Yu Liu, Zenan Huang, Qingqing Zhu and Yong Huang in Breast Cancer: Basic and Clinical Research
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
