Abstract
Background:
Breast cancer is the most common malignancy in women and the leading cause of cancer-related death. Although most early-stage patients are cured, 20% to 30% develop metastases, significantly reducing survival rates. Recent research highlights the role of iron in cancer progression, although its full impact on breast cancer metastasis is not yet fully understood.
Objectives:
The aim of this study is to investigate the association between plasma iron levels at diagnosis of early-stage breast cancer and the risk of developing metastatic disease.
Design:
Retrospective single-center study.
Methods:
Patients with stage I to III breast cancer, diagnosed between 2007 and 2017, and with serum iron, transferrin saturation, and ferritin values available within 1.5 months before or after diagnosis were included. Cox proportional hazard models were applied to determine the association between iron levels and risk of metastasis.
Results:
In total, 1113 patients were included, 10% of them developed distant metastasis over a median follow-up period of 7 years. In multivariable analysis adjusting for age, stage, and subtype, transferrin saturation and serum iron were significantly associated with an increased risk of breast cancer metastasis. For each 10% increment of transferrin saturation at baseline, there was a 19% increase in metastatic risk (hazard ratio [HR] = 1.19; 95% confidence interval [CI] = [1.02-1.38]). Similarly, a serum iron increment of 10 µg/dL led to a 6% increase in risk (HR = 1.06; 95% CI = [1.01-1.12]). Ferritin was found not to be associated with metastatic risk (HR = 0.99; 95% CI = [0.98, 1.01]). There was no significant association with metastatic site or breast cancer subtype when adjusting for age and stage.
Conclusion:
Elevated transferrin saturation and serum iron at early breast cancer diagnosis are associated with increased risk for metastatic disease but not with location of metastases or breast cancer subtype. Further research is needed to understand the underlying mechanisms and to explore the potential of iron-targeted therapies.
Introduction
Breast cancer is the most common type of malignancy diagnosed in women worldwide, accounting for approximately 25% of all female cancer cases. Moreover, it is the leading cause of mortality in female cancer.1,2 Most patients with early breast cancer are cured after local treatment and systemic therapy. However, about 20% to 30% develop secondary metastases, often many years after primary breast cancer treatment. Once breast cancer spreads to distant organs, the 5-year survival rate drops to approximately 25%. 2 Current treatment options for metastases, including chemotherapy, immunotherapy, and endocrine treatment, have limitations in both efficacy and toxicity. The process of metastasis is still not fully understood. Therefore, identifying factors that contribute to the development of distant metastases is crucial in improving clinical outcomes.
Recent research has highlighted an important role of iron in the metastatic process. Iron is a nutrient essential for critical cellular processes such as DNA synthesis, metabolic enzyme function, and response to increased oxidative stress. 3 However, iron excess leads to formation of oxygen radicals and has been linked to tissue damage and increased cancer risk. 4 Cancer cells display an increased reliance on iron uptake and metabolism compared with normal cells. 5 Moreover, iron seems to play an important role in all stages of cancer, including tumor induction, growth, and metastasis.6 -8
Despite the growing body of literature linking iron addiction and ferroptosis to breast cancer, the overall contribution of physiological iron availability to breast tumor formation and progression is not well understood. This retrospective study aims to investigate the potential association between elevated iron levels at diagnosis of early breast cancer and the subsequent risk of developing metastatic disease. Serum iron, ferritin, and transferrin saturation were examined to determine any potential differences in predictive value. In addition, our study explored whether the potential impact of iron content differs according to breast cancer subtype and/or location of metastases (lung, liver, bone, etc.).
Methods
Patient selection
A retrospective single-center study was conducted using patient data extracted from the large database of the Multidisciplinary Breast Center at the University Hospital in Leuven, Belgium. This database contains information on more than 15 000 breast cancer patients diagnosed since 2000. All patients with stage I to III invasive breast cancer, diagnosed between January 1, 2007, and December 31, 2017, were selected. The following inclusion criteria were applied: female sex, unilateral and unifocal breast cancer, and first therapy for breast cancer (surgery, chemotherapy, or hormonal therapy) administered at the University Hospital of Leuven. In addition, serum iron (µg/dL), ferritin (µg/L), and transferrin saturation (%) assessed by the “routine” clinical laboratory within 1.5 months before or after diagnosis have to be available. When multiple blood samples were available in the defined period, 1 sample was selected based on (1) sample preferentially taken before breast cancer diagnosis or treatment and (2) with the shortest time interval to the diagnosis. Exclusion criteria were missing information on tumor stage or receptor status. Staging was based on pathological staging for patients who received upfront surgery and on clinical staging for patients receiving neoadjuvant systemic treatment. This was integrated into a “combined staging” allowing to include staging in multivariable models for both settings. The Leuven Breast Cancer Database also contains detailed follow-up information for all patients, including date and localization of distant metastases, which will be used to determine the distant metastases rate in this study.
Breast cancer subtype definition
Gene expression data to assess genomic breast cancer subtype were not systematically available. A surrogate definition was used based on the St. Gallen recommendations of 2011 to classify breast cancer subtypes. 9 This classification is based on tumor grade and the presence or absence of the estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor 2 (HER2) receptors in the tumor cells, as assessed by immunochemistry. In line with our institutional guidelines, ER and PR negativity were defined as expression in less than 1% of tumor cells, and HER2 positivity was defined as immunohistochemistry (IHC) 2+ or 3+ with positive fluorescence in situ hybridisation (FISH) testing. The following 5 surrogate phenotypes are considered (Table A1, Appendix 1): “Luminal A-like” (ER and/or PR positive, HER2 negative and grade 1 or 2); “Luminal B-like” (ER and/or PR positive, HER2 negative and grade 3); “Luminal HER2-like” (ER and/or PR positive, HER2 positive); “HER2-like” (ER/PR negative, HER2 positive); and “Basal-like” (ER/PR/HER2 negative). This last subtype is also referred to triple-negative breast cancer (TNBC).
Statistical methodology
Descriptive statistics are provided by frequencies, using percentages for categorical variables and medians with interquartile ranges for continuous variables. Kaplan-Meier estimates were applied to evaluate metastatic rates. Cox proportional hazard models were used to investigate the impact of iron content on metastasis. Multivariable models were applied to correct for the confounding variables age, stage, and subtype. There was no adjustment for the use of (neo)adjuvant chemotherapy, as this largely depends on these confounders. Results are presented as hazard ratios (HRs) with 95% confidence intervals (CIs) for an increment of 10 units of iron content. To assess the possible association with site-specific metastasis, the following locations were investigated: lung, liver, bone, abdominal nonliver, lymph nodes, brain, and cutaneous and other locations. This analysis was only corrected for age and stage (as an ordinal variable) due to the low number of events per metastasis site. Finally, to analyze whether the influence of iron content on secondary metastases depends on breast cancer subtype, an interaction effect was modeled between iron content and subtype. All tests were 2-sided and evaluated at a 5% significance level. Analyses have been performed using SAS software (version 9.4 of the SAS System for Windows).
Results
Study population and clinicopathological characteristics
Figure 1 displays the selection strategy of the study cohort, resulting in a final data set of 1113 patients. The characteristics of the study population are presented in Table 1. The median age was 58 years. Most women were postmenopausal (63%) at breast cancer diagnosis. In total, 809 (73%) patients underwent upfront surgery, and 304 (27%) received neoadjuvant systemic therapy (chemotherapy and/or endocrine treatment). Among hormone receptor-positive patients, 93% received adjuvant endocrine therapy. Luminal A-like tumors were the largest group, representing 46% of all phenotypes. In our cohort, the subtype was unknown in 8 patients. For the majority (n = 830, 75%), iron values were available before start of any treatment, while in 283 (25%), they were only available after first treatment initiation.
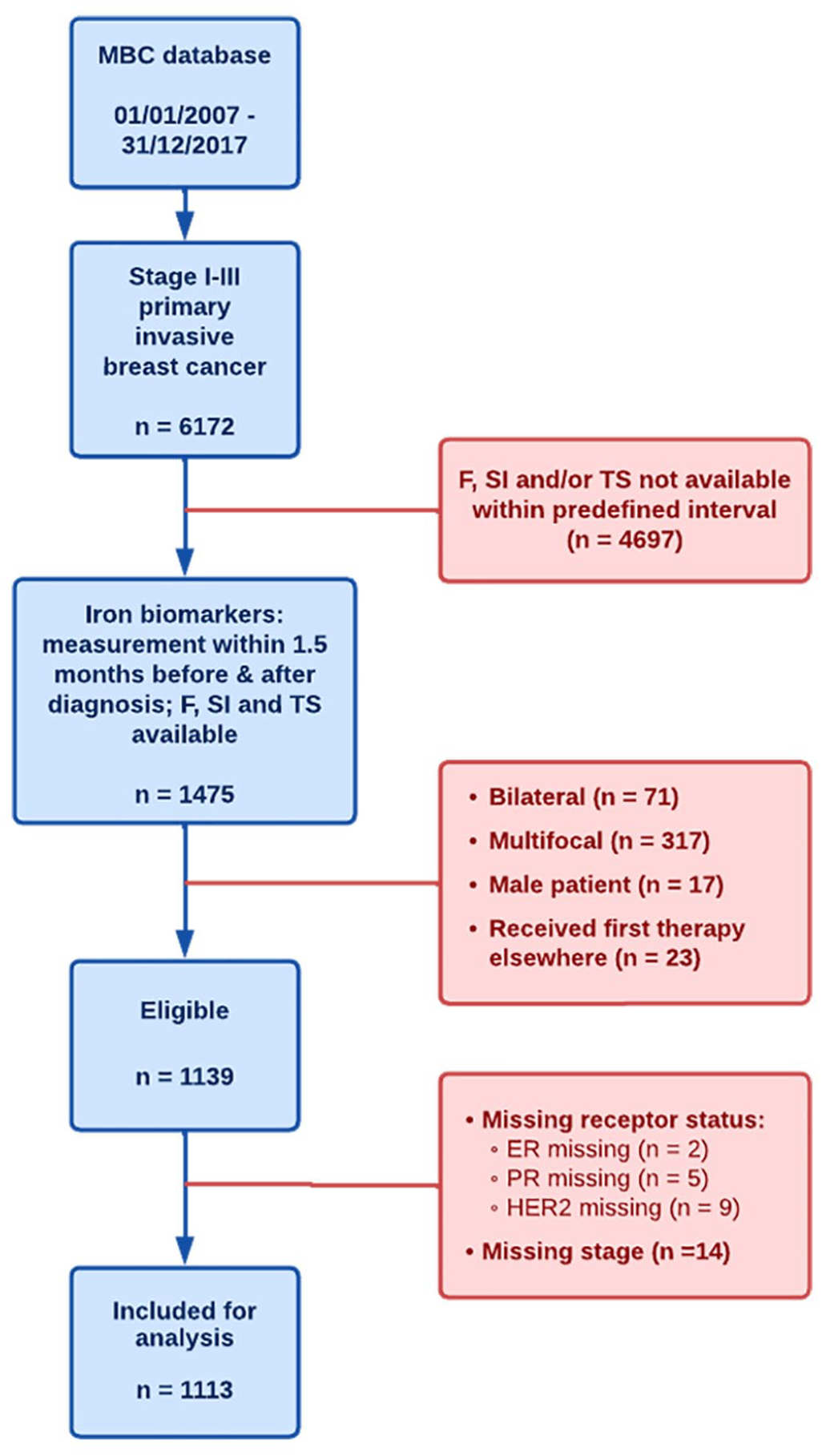
CONSORT diagram showing the patient selection process of the clinical study.
Descriptive statistics of the study population.

Note: The data are presented either as the number of occurrences (n) with the percentage of the total population in brackets (%) or as the median (M) with the interquartile range (IQR) in brackets, depending on the parameter.
With a median follow-up of 83 months, 111 patients (10%) developed distant metastases. Metastatic rate was calculated as the time between diagnosis and detection of metastases, with cases without metastases being censored at their last follow-up. The estimated 5-year and 10-year metastasis-free survival rates were 91% (95% CI = [89.5, 92.9]) and 88% (95% CI = [85.4, 90.1]), respectively. The most common sites of distant metastases were bone (63 patients, ie, 56.8% of all patients developing metastases), followed by liver (37 patients, 33.3%), lymph nodes (33 patients, 29.7%), lung (27 patients, 24.3%), and brain (19 patients, 17.1%).
Risk of secondary metastases by plasma iron content at breast cancer diagnosis
Multivariate Cox proportional HRs for iron biomarkers and metastases are shown in Table 2. Transferrin saturation and, to a lesser extent, serum iron at diagnosis were significantly associated with an increased risk of breast cancer metastasis. Specifically, for each 10% increment of transferrin saturation, there was a 19% increase in metastatic risk (HR = 1.19; 95% CI = [1.02-1.38]). Similarly, a serum iron increment of 10 µg/dL led to a 6% increase in risk (HR = 1.06; 95% CI = [1.01-1.12]). In contrast, ferritin was found not to be associated with metastatic risk (HR = 0.99; 95% CI = [0.98, 1.01]). The results of this analysis were adjusted for potential confounding factors (age, stage, and subtype). A sensitivity analysis was also conducted, which excluded patients whose iron biomarkers were assessed after the onset of primary treatment (given the potential influence of treatment/surgery on iron content). In the subgroup of patients with iron measurements before any treatment (n = 830), there were 84 distant metastatic events. Similar HRs for transferrin saturation (HR = 1.15; 95% CI = [0.97-1.37]), serum iron (HR = 1.04; 95% CI = [0.98,1.10]), and ferritin (HR = 0.99; 95% CI = [0.97, 1.01]) were observed as compared with the entire cohort, but none of these associations remained significant.
Risk of metastasis by iron content.

Abbreviations: CI, confidence interval; HR > (<)1: increased (decreased) risk with increasing predictor level; HR estimated for an increase with 10 units of the iron content; Analyses corrected for age and stage (categorical), and subtype; P < .05 considered statistically significant.
The impact of serum iron at breast cancer diagnosis on the location of secondary metastases was also assessed. Table 3 shows that the effect of the iron biomarkers on the risk of secondary metastases does not significantly differ across the specific metastasis locations (when corrected for tumor stage and patient age).
Cox proportional hazard models for iron biomarkers and metastases in specific locations.

Abbreviations: HR, hazard ratio; CI, confidence interval; HR > (<)1: increased (decreased) risk with increasing predictor level; HR estimated for an increase with 10 units of the iron content; analyses corrected for age and stage (ordinal); P < .05 considered statistically significant.
To analyze whether the influence of iron content on secondary metastases depends on breast cancer subtype, an interaction effect was modeled between iron content and subtype. Table 4 shows that the association of the 3 different iron content parameters with risk of metastasis does not depend on the subtype (adjusted for stage and age).
Cox proportional hazard models for iron biomarkers and metastasis in different tumor subtypes.

Abbreviations: HR, hazard ratio; CI, confidence interval; HR > (<)1: increased (decreased) risk with increasing predictor level; HR estimated for an increment of 10 units of iron content; analyses corrected for age and stage (categorical); P < .05 considered statistically significant.
Discussion
This retrospective, single-center study aimed to investigate the role of body iron content in the development of metastases in breast cancer. Our results suggest that transferrin saturation and, to a lesser extent, serum iron concentrations measured at diagnosis were significantly associated with an increased risk of breast cancer metastasis. Specifically, for each 10% increment of baseline transferrin saturation, there was a 19% increase in metastatic risk within the follow-up period of 7 years. A baseline serum iron increment of 10 µg/dL led to a 6% increase in risk. In contrast, ferritin was not associated with the risk of metastasis. This may possibly be related to ferritin being an acute phase protein, which increases with infection or inflammation, and is not necessarily associated with body iron content.
Our study is the first to demonstrate an association between iron biomarkers at diagnosis and the risk of developing distant metastases in breast cancer patients. Therefore, external validation of our findings is necessary. Previous studies have mainly focused on the impact of iron biomarkers on the risk of developing a primary breast cancer, with varying results. A meta-analysis by Chang et al 10 reported that high serum iron is associated with an increased risk of developing breast cancer, whereas no significant associations were observed with transferrin saturation nor with ferritin. Results of a study conducted by Chua et al 11 showed a significant association for both serum iron and transferrin saturation with the risk of breast cancer development.
Our study also explored whether the observed link between increased iron levels and enhanced metastasis formation was confined to particular metastasis locations, but no site-specific associations could be demonstrated.
Next, our results suggest that the influence of iron content on metastasis does not depend on breast cancer subtype. Only 1 clinical study investigating a potential interaction of phenotype was previously published. Graff et al 12 reported no significant difference in the association between ferritin level and ER/PR-positive or ER/PR negative breast cancer risk. Conversely, there is an in vitro study indicating a more important effect of iron in ER-positive breast cancer phenotypes. 13
There are several possible ways in which iron can play a role in metastasis. It has been widely documented that iron could play a role in forming a favorable micro-environment for the malignant cells.8,14,15 Excess iron can lead to the formation of oxygen radicals. This can give rise to DNA damage and (additional) mutations, triggering tumor heterogeneity. 16 Such heterogeneity allows cancer cells to survive in diverse environments and become resistant to systemic therapy.6,17 Furthermore, these oxygen radicals can promote angiogenesis, which is crucial for growth and spread of cancer cells. 18
During metastasis formation, cancer cells need to overcome oxidative stress. 19 Interestingly, it was recently shown that overexpression of Mitoferrin 1 (SLC25A37) or Mitoferrin 2 (SLC25A28), the 2 major mitochondrial iron importers, stabilized the protein expression of a mitochondrial glutathione transporter SLC25A39. 20 Thus, the correlation of transferrin saturation and circulating iron levels with metastasis that we observed in this study may be explained by an increase in the antioxidants capacity of cancer cells through mitochondrial glutathione import. Further functional studies may provide insights into this possibility.
Miller et al 21 revealed that about 30 different genes involved in iron metabolism were significantly associated with breast cancer prognosis. The results of our study may corroborate the potential use of transferrin saturation as a prognostic indicator for patients with early breast cancer. Moreover, our findings are consistent with the study by Chua et al, 11 showing a higher risk of cancer-related death in women with increased transferrin saturation levels.
Our results align with a potential role of iron in the development of metastatic disease in breast cancer. An older randomized study 22 suggested that reducing iron through phlebotomy may be associated with lower cancer risk and improved survival rates. Conversely, Edgren et al 23 found no significant association between blood donation frequency and cancer risk. Hemochromatosis has also been linked with increased incidence of breast cancer occurrence. 24 Currently, several iron-related therapies are being investigated. Iron chelators aim to remove iron molecules from the body through binding, resulting in depletion of iron.5,6,15 However, they may have side effects such as gastrointestinal symptoms or fatigue and were shown to be more effective in hematologic malignancies. 7 Alternatively, ferroptosis inducers have the potential to inhibit tumor growth and metastasis.25,26 However, these inducers may also lead to DNA damage and cell death in healthy bone marrow cells. 27 Although these therapies are still under development, they hold promise and deserve further investigation as potential novel treatment strategies for breast cancer patients.
Our study has some limitations. First, the follow-up period of 7 years is relatively short. It is known that the development of metastases can occur up to 20 years after initial diagnosis of breast cancer. 28 Another limitation is the relatively low number of metastatic events, with only 111 patients out of 1113 developing secondary metastases. The evaluation of iron content in relation to metastases location and/or subtype may be hampered by insufficient number of events. In about 25% of patients, iron values were only available after starting first treatment (surgery in most cases). We performed a separate analysis excluding these patients. The results of this analysis were not statistically significant but the HRs were similar to the original findings. Moreover, breast surgery is generally considered as minor surgery with limited blood loss, and mainly, ferritine is known as an acute phase protein that can be influenced by surgery/inflammation. Ferritine was least associated without outcome in our data. Finally, serum iron content was only available for a limited percentage of the whole breast cancer population documented in the Leuven Breast Cancer Database. This is mainly due to the fact that there was no standard procedure to collect data on serum iron levels until the year 2016. The assessment of iron content may have been linked to the specialist who first saw the patient (eg, medical oncologist vs surgeon), but it is not possible to further dissect this potential selection bias.
Conclusions
In conclusion, our study shows for the first time that transferrin saturation and serum iron at early breast cancer diagnosis are positively associated with the risk of secondary metastatic disease. A relation with location of metastases or breast cancer subtype could not be demonstrated. Further research should investigate the underlying mechanisms and explore the potential of iron-targeted therapies in the clinic.
Footnotes
Appendix 1
Overview of classification into subtypes based on the St. Gallen recommendations.

| ER and/or PR | HER2 | Tumor grade | |
|---|---|---|---|
| Luminal A-like | + | − | 1 or 2 |
| Luminal B-like | + | − | 3 |
| Luminal HER2-like | + | + | / |
| HER2-like | − | + | / |
| Basal-like | − | − | / |
Acknowledgements
The authors would like to thank C. Remmerie for her contribution in collecting the data.
