Abstract
Background:
Oncotype-Dx (ODx) is a 21-gene assay used as a prognostic and predictive tool for hormone receptor (HR)-positive and human epidermal growth factor receptor 2 (HER2)-negative, node-negative, or 1 to 3 lymph node-positive early breast cancers (EBCs). The cost of the test, which is not available in low-middle income countries (LMICs), is not within the means of most individuals. The Ki-67 index is a marker of tumor proliferation that is cost-effective and easily performed and has been substituted in many cases to obtain prognostic information.
Objective:
We aimed to identify the correlation between the ODx recurrence score (RS) and the Ki-67 index in HR-positive EBCs and to determine whether Ki-67, like the ODx, can help facilitate clinical decision-making.
Design:
Systematic review correlating Ki-67 index and ODx in HR-positive and HER2-negative EBCs as per Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines.
Data sources and methods:
We searched different databases between January 2010 and May 2023 and included retrospective/prospective cohorts, clinical trials, case-control, and cross-sectional studies involving HR-positive and HER2-negative EBCs correlating the Ki-67 index and ODx RS categories.
Results:
Of the 18 studies included, 16 indicated a positive or weakly positive correlation between ODx and the Ki-67 index. The combined P value of the included studies is <0.05 (P = .000), which shows a statistical significance between the 2. Our review also discusses the potential of machine learning and artificial intelligence (AI) in Ki-67 assessment, offering a cost-effective and reproducible alternative.
Conclusion:
Even although there are limitations, studies indicate a favorable association between ODx and the Ki-67 index in specific situations. This implies that Ki-67 can offer important predictive details, especially regarding the likelihood of relapse in HR-positive EBC. This is particularly significant in LMICs where financial constraints often hinder the availability of costly diagnostic tests.
Plain Language Summary
The study explored the correlation between the expensive Oncotype-Dx (ODx) test and the more affordable Ki-67 index in predicting outcomes for certain breast cancers. Results from 16 out of 18 studies indicated a significant link between the 2 tests, suggesting Ki-67 could be a cost-effective alternative, especially in low- to middle-income countries.
Introduction
Invasive breast cancer (IBC) consists of a heterogeneous group of tumors with differences in prognosis and response to systemic therapy. This heterogeneity is demonstrated not only by the classification based on immunophenotyping but also by the intrinsic classification, which is well described. 1 Intrinsic phenotyping, based on genomic microarray of tumors, has established 5 categories of breast cancer (BC). These are luminal A, luminal B, human epidermal growth factor receptor 2 (HER2)-enriched, basal-like, and normal.2,3 Differentiation of luminal A from luminal B is important, as each subtype is unique in its prognosis and response to chemotherapy.
The luminal-A subtype is the most common subtype, comprising up to 75% of IBCs. It is characterized by its low-grade potential with an excellent response to hormonal treatment, 3 whereas the luminal-B subtype is aggressive with poor response to hormonal therapy and sensitivity to chemotherapy. The differentiation of these subtypes thus influences the management of these patients. Unfortunately, the lack of availability of intrinsic phenotyping and its application in mainstream practice is a limiting factor in decision-making regarding adjuvant systemic therapy.
According to the St Gallen experts’ consensus, luminal-A IBCs are estrogen receptor (ER)-positive with progesterone receptor (PR) expression of ⩾20%. They are HER2-negative and have a Ki-67 ⩽ 14% with a low RS based on the ODx genetic assay. In contrast, luminal-B IBC is ER-positive, HER2-negative, PR-negative, or <20%, with a Ki-67 ⩾ 20% and high RS based on multigenic assessment (whenever available). 4
There are 2 prospectively validated and commercially available genomic assays available for patients with HR-positive and HER2-negative EBCs, with the purpose to stratify the low-risk patient subgroup where chemotherapy can be omitted. These are the 21-gene Oncotype-Dx (ODx) and 70-gene MammaPrint assay. 5 The 21-gene ODx assay has been validated based on prospective trials in node-negative and in 1 to 3 (1-3) lymph node-positive, HR-positive and HER2-negative EBC. Oncotype-Dx test results when incorporated in a formula generate a RS (0-100). For postmenopausal women, those with RS ⩽ 26 are deemed low risk, whereas those with RS ⩾ 26 are high risk and candidates for endocrine treatment with additional chemotherapy. 5 While for premenopausal the cut off changes, a RS ⩽ 15 predicts a low risk of recurrence, therefore, only endocrine treatment is offered to this subgroup. Although a score between 16 and 25 predicts an intermediate-risk category, therefore, benefit of additional chemotherapy along with endocrine therapy is present. Those having RS ⩾ 26 are categorized as high risk for future recurrence and are candidates for both chemotherapy and endocrine therapy. 5 Besides the score, the age, and the premenopausal status play an important role in assigning treatment in patients with an intermediate score, which applies to women who are aged 50 years or younger. 5 Therefore, genomic assay assessment is crucial for deciding treatments among HR-positive and HER2-negative EBC patients. 5
Although ODx has proven to be an essential tool in decision-making regarding the benefits of chemotherapy, its application is limited in low-middle income countries (LMICs), as the cost is a major limiting factor (the test is outsourced to the United States and is paid for in USD). Due to a lack of ability to obtain this test, clinicians have resorted to reliance on traditional markers, like the Ki-67 Proliferative Index (PI) in addition to the other clinic-pathologic factors to distinguish between the low-risk and high-risk EBCs.6,7
Ki-67 is a proliferative gene that encodes a nuclear protein that facilitates cellular proliferation and growth during the M-phase of the cell cycle.8,9 Some studies have evaluated the correlation between the Ki-67 index and ODx RS for the use of Ki-67 as a readily available and inexpensive marker to predict the risk of recurrence instead of the very expensive ODx. 10 However, there are conflicting results regarding the use of Ki-67 as a predictor of the RS when compared to ODx among patients who fall in the intermediate-risk category for recurrence.
The heterogeneity of IBC and its variable response to chemotherapy in HR-positive EBCs highlights the need for molecular testing to ensure the judicious use of chemotherapy. The high costs and limited availability of tests like the ODx create a challenge for clinicians in LMICs. Therefore, the purpose of this systematic review is to determine whether a correlation exists between the Ki-67 index and ODx RS to establish the former’s utility as a surrogate marker for identifying high-risk EsBC permitting a cost-effective and readily available alternative to ODx.
Material and Methods
Literature review
This systematic review of original articles was performed in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. Two reviewers independently conducted the literature search across 4 databases: PubMed, Google Scholar, Embase, and Web of Science. Boolean operators were used alongside the following keywords to capture relevant articles: “HR-positive EBC”, OR “HR-positive and HER2-negative EBC”, ODx RS, and “Ki-67 score” OR “levels”. We also hand-searched (both forward and backward citation search) and retrieved any additional articles meeting our study question. All literature published in the English language between January 2010 and May 2023 was included in our review. In addition, the following inclusion criteria were applicable: (1) the study design included cohort studies, case-control, cross-sectional studies, and randomized controlled trials; (2) the studies that report the correlation, association, or comparison between ODx RS and Ki-67 levels in HR-positive, HER2-negative, lymph node-positive/negative EBCs; (3) no restriction on sample size, but the studies should report the sample size; (4) studies that use either old or new cutoff points for Ki-67 index and ODx RS; (5) studies that employ various statistical methods such as Spearman rank correlation, multivariable analysis, Fisher method, and chi-square distribution, to evaluate the correlation between Ki-67 and ODx RS; (6) studies that provide P values, confidence intervals, and other statistical measures related to the correlation between Ki-67 levels and ODx RS; and (7) studies that report on clinicopathologic factors, age, tumor size, tumor grade, ER/PR/HER2 status, and other relevant factors along with the correlation between Ki-67 and ODx RS. Unpublished or non-peer-reviewed studies, closed-access studies, and studies correlating ODx RS with biomarkers other than Ki-67 levels were excluded from our review.
In addressing and mitigating study limitations, our research team undertook a structured approach to ensure transparency, rigor, and comprehensiveness. Initially, the team engaged in a systematic discussion to evaluate the significance and potential impact of each identified limitation on the study’s overall findings. Prioritization of these limitations was conducted through a collaborative process, involving voting, iterative discussions, and consensus-building among team members. Recognizing the scarcity of data in the existing literature and the variability in reported outcomes, we opted to allow flexibility in the cut off values for both ODx and Ki-67 levels. This decision aimed to facilitate a more comprehensive exploration of potential correlations. In addition, to address our primary objective of establishing correlations, we made the deliberate choice to include studies that employed varied statistical methods in our review. This inclusive approach was designed to capture a broader range of evidence and perspectives on the correlation between ODx and Ki-67 levels. By integrating these steps into our methodology, we strived to proactively identify, assess, and address potential limitations, thereby reinforcing the validity, reliability, and generalizability of our study results.
Study selection and data extraction
Once duplicate articles and gray material eliminated manually, 2 reviewers independently conducted the screening to minimize the risk of bias. After title and abstract screening, a full-text review was performed to assess whether the articles met the inclusion criteria. Subsequently, 2 reviewers extracted the data from the included studies onto an Excel sheet with pre-defined columns. The flow of our extracted articles is displayed using the PRISMA flow diagram 2020 in Figure 1.

PRISMA flow diagram for studies correlating Oncotype-Dx Recurrence Score and Ki-67 Index.
Statistical analysis
The studies were evaluated for bias independently by 2 authors. Whether individual studies reported bias assessment was determined and has been specified in Tables 1 and 2. Because of the heterogeneity of study designs and the unavailability of a standardized tool for retrospective reviews, the reviewers identified study limitations through consensus. To identify the risk of bias in all included studies, Fisher method was used in which each P values obtained were combined manually after identifying the chi-square distribution for each study first and then a combined P value was obtained using calculator Stat Trek; http://courses.atlas.illinois.edu/spring2016/STAT/STAT200/pchisq.html and reconfirmed by using calculator https://stattrek.com/online-calculator/chi-square.11,12 The P values from 17 studies were used for statistical analysis. For two-tailed P values, they were converted to one-tailed P values first and then used for statistical analysis. One study did not report the P value; therefore, it was not included in the final analysis.
Study characteristics and comparative analysis of Ki-67 index, Oncotype-Dx recurrence score cutoffs, and prognostic scores of included studies.

Abbreviations: LVI, lymphovascular invasion; NR, not reported; PI, perineural invasion; PPH3, phosphohistone H3.
Studies with positive correlation between Oncotype-Dx recurrence score and Ki-67 index.
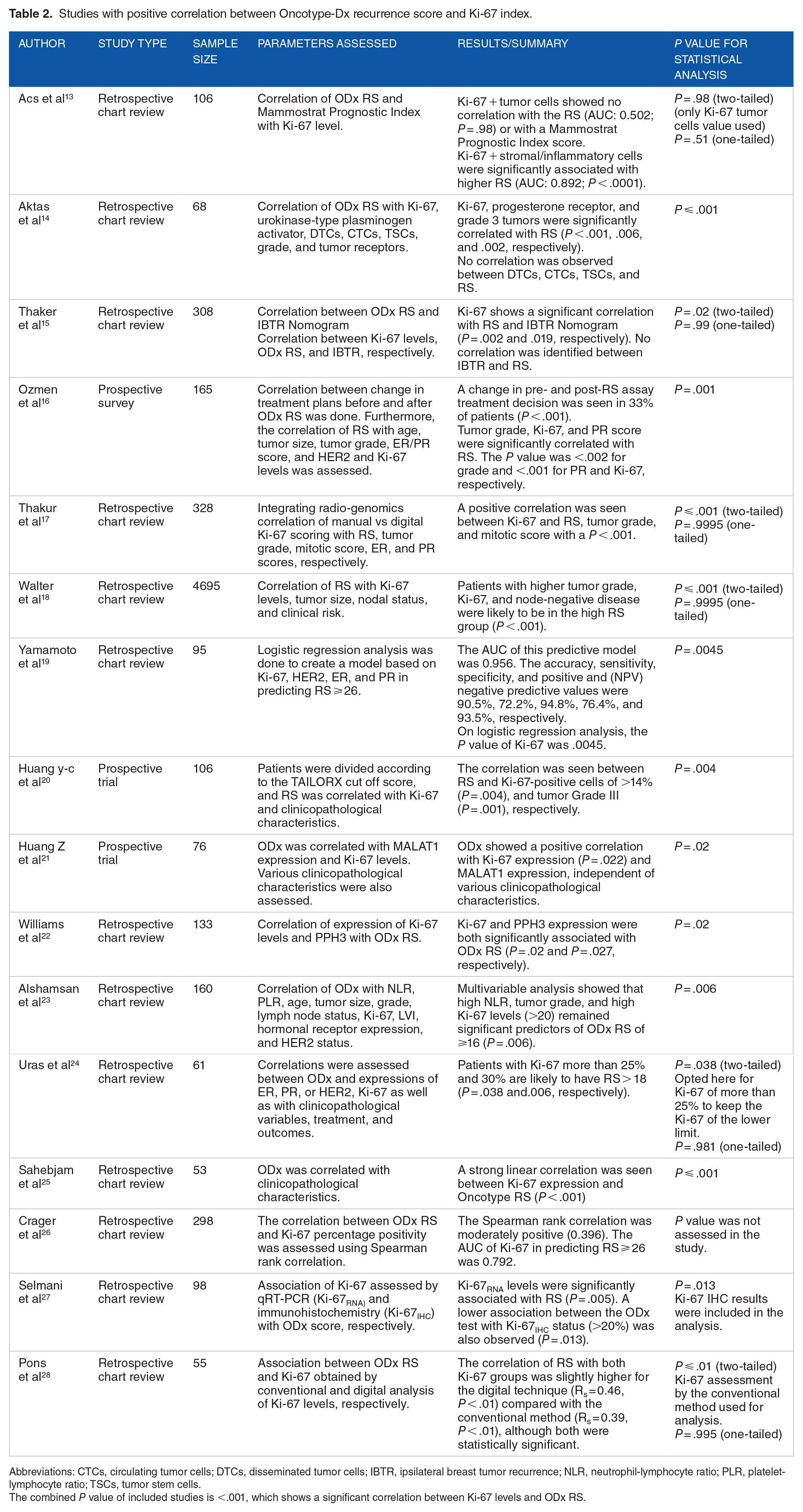
Abbreviations: CTCs, circulating tumor cells; DTCs, disseminated tumor cells; IBTR, ipsilateral breast tumor recurrence; NLR, neutrophil-lymphocyte ratio; PLR, platelet-lymphocyte ratio; TSCs, tumor stem cells.
The combined P value of included studies is <.001, which shows a significant correlation between Ki-67 levels and ODx RS.
Results
Eighteen studies, from 2010 through 2023, were included in this systematic review. Study characteristics and comparative analysis have been summarized in Table 1, whereas the correlations and results are summarized in Tables 2 and 3 below. The risk of bias among the studies is addressed using the QUADAS tool of risk assessment for systematic reviews, and further stratification of the parameters contributing to the risk is mentioned in Tables 1 and 2. 30 Among these, 15 were characterized as retrospective chart reviews primarily involving the use of patient tumor samples retrieved from storage. The remaining 3 included 2 prospective trials and 1 survey-based study evaluating the effect of ODx RS on treatment decisions. All these studies were selected because they evaluated the association between ODx RS and Ki-67 in some capacity.
Studies with negative correlation between Oncotype-Dx recurrence score and Ki-67 index.

The sample size ranged between 53 and 4695, with the median sample size being 106. Only 2 out of the 18 studies indicated no correlation between the ODx RS and Ki-67 index. The remaining observed varying degrees of correlation depending on the statistical methods used. The combined P value of included studies in our analysis was <.001, which shows that there is evidence of rejection of the null hypothesis in studies and there is a correlation between Ki-67 levels and ODx RS.
The ODx RS categories reported in the literature varied between different studies especially since the TAILORX trial in 2018 recommended new cut off points. As expected, 7 studies10,13,14,16,17,25 from 2011 through 2018 used the old cutoffs, that is, ODx score of ⩽18 for low risk, 18 to 30 for intermediate risk, and ⩾31 for high risk. Two studies from 2022,27,28 also used the older cutoffs, likely because their patient cohorts were taken from before 2018. Six18,19,23,26,29,31 studies used the new cutoffs which categorize <15 as low risk, 16 to 25 as intermediate risk, and ⩾26 as high risk, while 3 studies20,21,24 used both criteria for their analyses. One study treated the ODx RS as a continuous variable. 15
Similarly, for Ki-67 index score cutoffs were different across studies. Two studies relied on Ki-67 cutoffs of ⩽14% as low and >14% as high.20,21 Eight studies used the modified St Gallen criteria of <20% as low and ⩾20 as high.10,16,18,22-24,26,27 One study used 15 as the cut off albeit without any justification, 14 whereas another determined their own institutional cutoffs (⩽24 for low) based on their patient population. 28 Two studies also used <10 and >25 as cutoffs for low and high Ki-67, respectively.25,31 This heterogeneity in cutoffs indicates an absence of standardization.
In some cases, the presence of an association was tested between the 2 categorical variables. However, we also included studies that analyzed Ki-67 percentages as either a continuous or ordinal variable. These studies either used Spearman rank correlation or multivariable analysis to evaluate the presence of a correlation.
Discussion
The utility of ODx in HR-positive EBC is well established in the literature;5,32,33 however, the clinical applicability of Ki-67 instead of ODx for decision-making in this patient cohort regarding the benefit of adjuvant chemotherapy is still questionable. As a marker of mitotic activity, Ki-67 levels are associated with cellular proliferation and hence tumor aggressiveness. The prognostic significance of the Ki-67 score in HR-positive EBC has been emphasized in the literature with higher scores correlating with an increased risk of recurrence. 34
However, the complexity of tumor biology dictates that multiple genes are implicated in determining metastatic potential and hence subsequent risk of distant recurrence. In this regard, the ODx is superior since it evaluates the expression of multiple genes including those associated with metastasis such as matrix metalloproteinase-11 (MMP11) and Cathepsin L2 (CTSL2). 21 The ODx RS also incorporates Ki-67 and other proliferation-related genes into its calculation with the algorithm giving the greatest weightage to the proliferation group. 22 A study in our review indicates, however, that there is a possibility that stromal cell Ki-67 protein expression influences the calculation of ODx score since cDNA from the entire tissue sample is used. 13 This can serve as a potential source of discordance between Ki-67 index-based risk categorization and ODx since stromal cells are not counted by the pathologist during Ki-67 determination (Figure 2).

Genes whose mRNA expression profiles are assessed by the ODx assay. All these genes are overexpressed in BCs. However, overexpression of certain genes may also be expressed in stromal cells (fibroblasts, lymphocytes, macrophages) present within the tumor microenvironment. The ODx assay uses whole-tissue mRNA; hence, tumors with an inflammatory stroma may have higher expression profiles for certain genes, for example, Ki-67.
Co-expression of other markers such as MALAT1 and phosphohistone H3 was also assessed in 2 studies. These markers can help strengthen the prognostication when used in conjunction with the Ki-67 index and other clinicopathologic characteristics allowing clinicians to forgo ODx testing if a lower risk of recurrence is indicated. However, caution must be taken when relying on these results as they have not yet been validated using large-scale diagnostic studies.22,21
Various studies recommended the judicious use of ODx, by relying on a stepwise approach. Risk stratification using Ki-67 has been recommended initially, as low Ki-67 levels correlated well with a low ODx RS in HR-positive EBCs. However, this association tended to break down at higher Ki-67 levels. Therefore, clinicians may consider the use of ODx if the Ki-67 index and other markers indicate tumor aggressiveness, rather than using this score for every patient.19,20,26 Furthermore, validated equations have been developed to estimate the ODx RS. The Magee equations incorporate various clinicopathologic and immunohistochemical parameters, including Ki-67, to provide a score that has been shown to correlate with the ODx RS. The incorporation of these equations into the decision-making algorithm may be cost-effective for LMICs. 35
One study also indicated a discrepancy between the expression of Ki-67 RNA levels and the level of Ki-67 Immunohisto Chemistry (IHC) staining. 27 It identified low agreement between results using 2 different techniques of Ki-67 reporting. On univariate analysis, Ki-67 tumor RNA levels had a higher hazard ratio of event-free survival as compared to the Ki-67 assessment via the IHC method among their patients; also, the Ki-67 RNA was better correlated to ODx RS statistically, suggesting Ki-67 reporting via qRT-PCR is a better surrogate option to ODx RS when compared to Ki-67 reporting using conventional methods. In addition, this discordance may be a possible reason for the breakdown of the correlation between ODx and Ki-67, with high Ki-67 RNA levels being detected by the ODx RS, increasing the score.
Another study concluded that ODx testing could be considered an option for patients with high Ki-67 proliferation index or high-grade tumors because the score might classify them as low risk sparing these individuals’ chemotherapy in HR-positive EBC. 28 Rigorous risk stratification, therefore, is necessary to ensure that the benefits of adjuvant chemotherapy are not withheld, and additional toxicities are not incurred. A major discrepancy in the studies we reviewed was the variation in risk stratification cutoffs for the Ki-67 index.10,14,16,18,20,24,26-29,31 Some articles classified Ki-67 of less than 10 as low risk, whereas others used 20 or even 15 as a cutoff. Variations in these cutoffs may be due to changes in the standard criteria over time, for example, St Gallen updated their cutoff for Ki-67 from less than 14% to 20% to differentiate between the luminal-A and luminal-B subtypes based on survival outcomes. 4 The International Ki-67 in Breast Cancer Working Group (IKWG) has determined that the Ki-67 index be categorized as low at 5% or less and high for values 30% or more. These findings are based on studies indicating high interobserver variability in the 5 to 30 range. 36 Adopting a standardized cut off value determined using robust studies may increase the validity of Ki-67 as a prognostic test.
Harnessing machine learning and artificial intelligence for reliable Ki-67 index calculation
The use of machine learning or artificial intelligence (AI) may help eliminate interobserver variability as indicated by certain studies included in our review.17,28,37 Image analysis algorithms can provide an efficient and effective method to calculate the Ki-67 index and this, alongside other clinicopathologic information can subsequently be fed into a machine learning model to predict ODx RSs in EBCs. Although these procedures require access to software and technical expertise, effective use of machine learning can help overcome limitations associated with conventional methods, making them a cost-effective alternative to expensive gene-based assays. The robustness of the output from these algorithms is dependent on preanalytical factors such as the adequacy of tumor tissue in the section and completeness of patient data, however. It offers standardized data collection, accurate image analysis, predictive modeling, and quality assurance, simplifying processes and enhancing patient results when incorporated into clinical routines.38,39 Xie et al, 39 in a multicentric observational study involving 771 pairs of stained slides employed a scale-invariant feature transform (SIFT)-based AI system, achieving a remarkable 93% accuracy in identifying cancer tissues and 91.5% accuracy in calculating the Ki-67 index, showcasing the potential of AI to enhance accuracy and repeatability in Ki-67 assessment. In addition, studies by Li et al, 40 Fulawka et al, 41 Cai et al, 42 and Zehra et al 43 further highlight the consistent and reproducible results achieved through AI assistance in Ki-67 labeling, offering improved diagnostic accuracy and efficiency in BC evaluation.
LMIC implications
Despite some limitations, studies have proved a positive correlation between ODx and Ki-67 levels in certain contexts in early EBCs. This suggests that Ki-67 can provide valuable prognostic information along with clinicopathologic risk factors in HR-positive EBC. This is particularly relevant for LMICs where financial limitations often restrict access to expensive diagnostic tests. The process of acquiring tissue specimens transporting them to the United States and subsequently running the ODx assay is a costly undertaking in comparison with performing simple immunohistochemistry and subsequent cell counting (Figure 3).
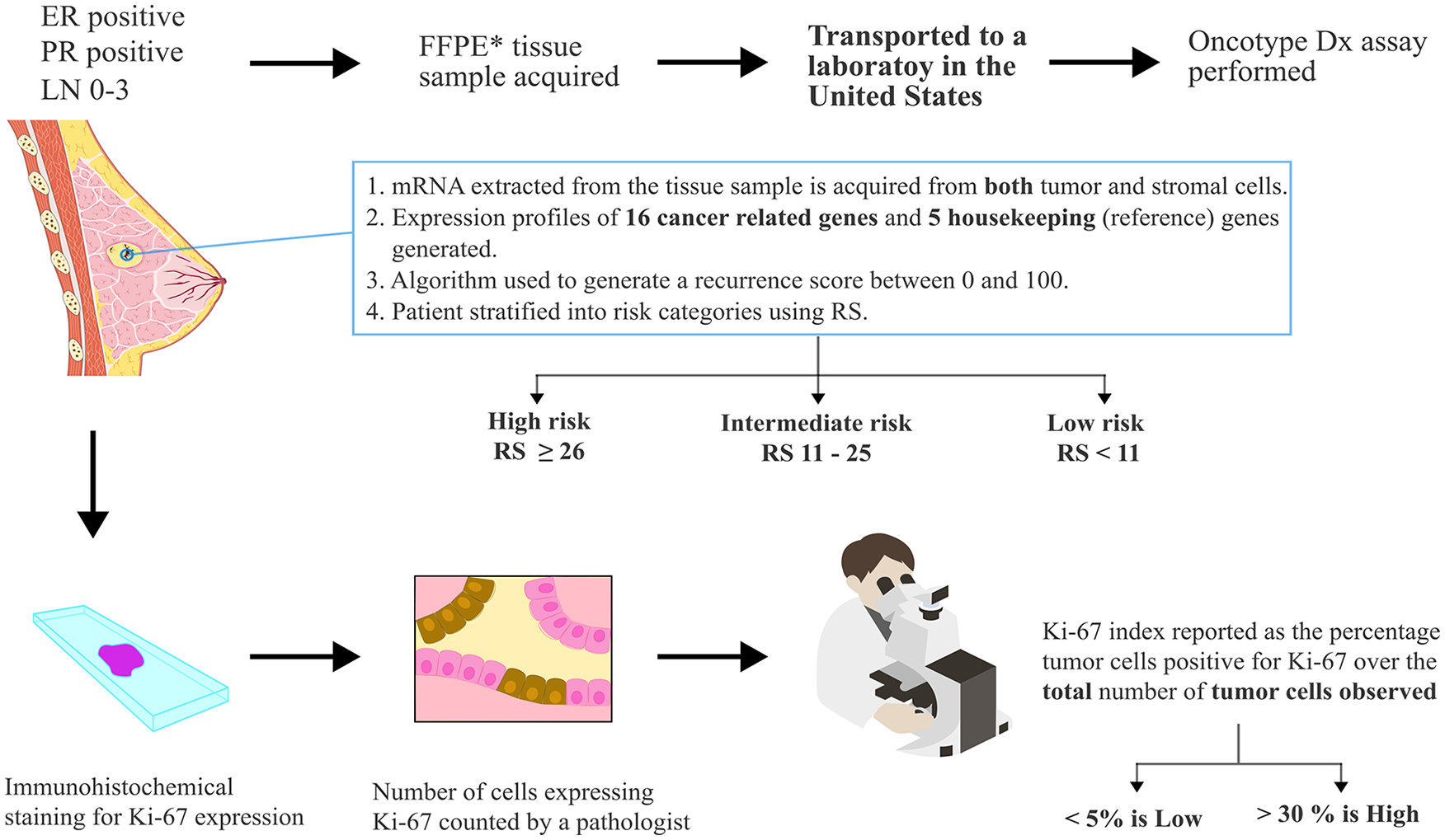
The procedure for obtaining a sample and subsequent testing is described for both the Oncotype-Dx and Ki-67 index. FFPE—formalin-fixed paraffin-embedded. The IKWG has determined that the Ki-67 index be categorized as low at 5% or less and high for values 30% or more.
Using Ki-67 as a surrogate marker could provide valuable prognostic information at a lower cost as an initial screening tool to identify patients who are more likely to benefit from genomic testing. Similarly, the correlation between Ki-67 levels and ODx RS opens the possibility of risk-adapted treatment strategies in LMICs, sparing low-risk patients from unnecessary interventions. Also, Ki-67 can be assessed using standardized laboratory techniques, making it more accessible and feasible in resource-limited settings compared with complex genomic tests like ODx. This allows for easier implementation and integration into existing health care systems.
Limitations
Most of the studies had small sample sizes and retrospective designs and different cut off values for both ODx and Ki-67 levels, which may limit the generalizability of their findings. Larger prospective studies with diverse populations could provide more robust evidence. Furthermore, a few studies reported interobserver variability and interlaboratory variation. This variability can impact the consistency and reliability of Ki-67 as a prognostic marker in place of ODx RS. Therefore, we do not propose that Ki-67 be used as a replacement for ODx. Instead, the clinician uses an algorithmic approach and determines the ideal circumstance to perform the ODx.
Some studies did not directly assess the correlation between ODx and Ki-67 levels. Instead, they examined the association of these markers with other variables or assessed their correlation indirectly. Direct comparisons between ODx and Ki-67 will provide more definitive information. Another limitation of the review is some studies reported positive correlations, whereas others found no or poor correlations. These discrepancies in the correlation between ODx and Ki-67 levels in studies indicate the complexity and heterogeneity of the relationship between these markers, highlighting the need for further research and the role of AI in future studies. Also, the influence of confounding variables, such as the presence of stromal cells, on the correlation between ODx and Ki-67 should be carefully considered and controlled for in future studies.
Another major issue found in retrospective studies is about factors that drove the clinician to order an ODx assay. If the decision was made based on prior knowledge of clinicopathologic parameters and ancillary biomarkers like Ki-67, a significant selection bias is introduced. Most studies did not comment on this particular factor.
Conclusions
This review highlights the complexities associated with using Ki-67 levels as a surrogate for ODx RS. Although some studies found a positive correlation between ODx and Ki-67 levels, others showed no or poor correlation. The decision to use ODx testing or Ki-67 levels as prognostic markers should be based on a comprehensive evaluation of multiple factors, including tumor characteristics, patient preferences, and available resources. Overall, the research on the correlation between ODx and Ki-67 levels offers promising implications for LMICs. It provides insights into the potential use of Ki-67 as a cost-effective and accessible marker in risk stratification and treatment decision-making, allowing for optimized resource allocation and personalized care in resource-limited settings. It is important to note that the field of oncology research is dynamic, and novel studies may provide further insights and updates on the relationship between ODx and Ki-67.
Footnotes
Acknowledgements
The authors would like to acknowledge Servier Medical Art for providing pictures (including an image of the breast and tumor cell icons). These figures were modified using text annotation and inclusion of additional details. Servier is licensed under a Creative Commons Attribution 3.0 Unparted License (https://creativecommons.org/licenses/by/3.0/). They would also like to acknowledge Database Center for Life Science (DBCLS) for providing an image of the light microscope which has been used in our figure. DBCLS (https://togotv.dbcls.jp/en/pics.html) is licensed under CC-BY 4.0 Unported ![]() .
.
Declarations
Authors note
The review was not registered in any database retro/prospectively. The review protocol was not made beforehand.
