Abstract
Background:
Contrast-enhanced mammography (CEM) is an emerging breast imaging modality. Clinical data is scarce.
Objectives:
To summarize clinical evidence on the use of iopromide in CEM for the detection or by systematically analyzing the available literature on efficacy and safety.
Design:
Systematic review and meta-analysis.
Data sources and methods:
Iopromide-specific publications reporting its use in CEM were identified by a systematic search within Bayer’s Product Literature Information (PLI) database and by levering a recent review publication. The literature search in PLI was performed up to January 2023. The confirmatory-supporting review publication was based on a MEDLINE/EMBASE + full text search for publications issued between September 2003 and January 2019. Relevant literature was selected based on pre-defined criteria by 2 reviewers. The comparison of CEM vs traditional mammography (XRM) was performed on published results of sensitivity and specificity. Differences in diagnostic parameters were assessed within a meta-analysis.
Results:
Literature search: A total of 31 studies were identified reporting data on 5194 patients. Thereof, 19 studies on efficacy and 3 studies on safety. Efficacy: in 11 studies comparing iopromide CEM vs XRM, sensitivity was up to 43% higher (range 1%-43%) for CEM. Differences in specificity were found to be in a range of −4% to 46% for CEM compared with XRM. The overall gain in sensitivity for CEM vs XRM was 7% (95% CI [4%, 11%]) with no statistically significant loss in specificity in any study assessed. In most studies, accuracy, positive predictive value, and negative predictive value were found to be in favor of CEM. In 2 studies comparing CEM with breast magnetic resonance imaging (bMRI), both imaging modalities performed either equally well or CEM tended to show better results with respect to sensitivity and specificity. Safety: eight cases of iopromide-related adverse drug reactions were reported in 1022 patients (0.8%).
Conclusions:
Pertinent literature provides evidence for clinical utility of iopromide in CEM for the detection or confirmation of breast cancer. The overall gain in sensitivity for iopromide CEM vs XRM was 7% with no statistically significant loss in specificity.
Introduction
X-ray mammography (XRM) today represents the primary diagnostic tool for breast cancer (BC) screening and for the evaluation of patients with suspected lesions in the breast. XRM visualizes breast pathologies by measuring attenuation differences between tumors and lesions and the physiologic fibro-glandular breast tissue. The sensitivity of XRM is limited particularly in women with dense breasts. 1 Contrast-enhanced breast magnetic resonance imaging (bMRI) features higher sensitivity than XRM.2 -4 Contrast-enhanced bMRI visualizes lesions by enhancing the tumor’s neo-angiogenesis and the leakage of contrast into the tumor tissue. 5 Like MRI, contrast-enhanced mammography (CEM) is able to assess both imaging principles by visualizing tumor morphology as well as its vascularization. 6 CEM is an advancement of digital XRM providing 2 images at 2 energy levels which are subsequently combined electronically to highlight the areas of enhancement. CEM requires intravenous administration of an iodine-based contrast medium prior to image acquisition.
According to the American College of Radiology (ACR) Appropriateness Criteria, CEM may be appropriate for supplemental BC screening of intermediate- and high-risk women with non-dense breasts and average- and intermediate-risk women with dense breasts. So far ACR does not recommend CEM for other clinical settings, eg, palpable breast masses, initial workup and surveillance for local recurrence and distant metastases in asymptomatic women. 7 However, in 2022, ACR acknowledged in a supplement to ACR BI-RADS® Mammography 2013 that CEM has been shown to be more sensitive than mammography or ultrasound for the detection of malignancy. 8
The European Society of Breast Imaging (EUSOBI) discussed 3 studies9 -11 and concluded that based on preliminary results, CEM can be considered as an alternative to bMRI in the case of contraindications to MRI in general or to gadolinium-based contrast injection. 5 Most recently, EUSOBI recommended supplemental screening in women with extremely dense breasts to be performed preferably with bMRI, because for the time being, level I evidence is available only for bMRI. 12 The recent EUSOBI recommendation, however, mentions that other imaging modalities, like CEM, may eventually offer practical advantages over bMRI. 12
The Royal College of Radiologists’ Guidance on screening and symptomatic breast imaging (fourth edition) stated that CEM may also be used in the symptomatic setting, where available. 13 Along that line, also the German Guideline Program in Oncology (GGPO) stated that CEM is an option if MRI is not feasible, especially for dense breasts. The guideline quoted a number of studies showing comparable diagnostic accuracy for CEM vs bMRI. 14
Jochelson et al 15 excellently summarized that CEM is an emerging breast imaging modality that helps improve diagnostic accuracy when routine breast imaging produces inconclusive findings and that may serve as an alternative to bMRI.
Iopromide (UltravistTM) is a low osmolar non-ionic X-ray contrast medium (LOCM) with iodine as active ingredient. 16 Initially approved in February 1985, iopromide features a clinical track record of more than 37 years. As of June 30, 2022, more than 324 million have been administered to patients in more than 118 countries. 17 Numerous studies have documented the overall safety profile18 -24 in a wide range of indications (including but not limited to computed tomography, angiography, urography, cholangiopancreatography, and arthrography).
On January 20, 2023, iopromide received the European Union approval for CEM. The approved indication is “for use in adult women in CEM to evaluate and detect known or suspected lesions of the breast, as an adjunct to XRM (with or without ultrasound) or as an alternative to MRI when MRI is contraindicated or unavailable.”
The objective of this article was to summarize clinical evidence on the use of iopromide in CEM for the detection or confirmation of BC by systematically analyzing the available literature on efficacy and safety.
Data Sources and Methods
Databases
As a first step, iopromide-specific publications were searched in Bayer’s Product Literature Information (PLI) database up to January 2023.
As a second step, additional iopromide-specific publications were retrieved from Zanardo et al, 25 who published a systematic review on CEM based on a MEDLINE/EMBASE + full-text search from September 2003 to January 2019 (Figure 1). Full texts with the search term CEM were evaluated by 2 reviewers.

Literature search strategy—identification of studies in databases and literature. CEM indicates contrast-enhanced mammography; CESM, contrast-enhanced spectral mammography; LOCM, low osmolar non-ionic X-ray contrast medium; PLI, product literature information; XRM, traditional mammography.
The PLI automatically (in rare cases also manually) imports data containing information from biomedical journals dealing with Bayer Pharma and Consumer Health (CH) products from 4 large, commercially and publicly available databases: MEDLINE and EMBASE (until today), Derwent Drug File and BIOSIS (until December 2019). In addition, search hits from Reactions Weekly (case reports) or Medmeme (conference abstracts) complemented PLI. The scientific areas of interest were as follows: clinical pharmacology, efficacy and pharmacokinetics, adverse effects in case reports, clinical adverse effects, pre-clinical toxicology, and pharmacokinetics. Duplicate records and reviews were deleted manually.
The search strategy is shown in Figure 1.
Statistics
Sensitivity and specificity of CEM and XRM and the respective numbers of patients with malignant/benign lesions (N) were extracted from 11 papers (Table 4). The number of patients correctly classified (m) were derived by multiplying N with the given sensitivity or specificity.
The risk difference per publication was computed using the N’s and m’s as basis for a generalized linear model with identity link and a binomial distribution. In case of sensitivities or specificities of 100%, a small value (.0001) was subtracted from m to make computations feasible. SAS Version 9.4 was used for this analysis step.
A meta-analysis based on the data for sensitivity and specificity was computed per method (CEM or XRM) and for the computed risk-differences using the R-package “meta.” 26
Results
Results of the literature search
Twenty-four publications reporting data specifically on iopromide were identified by the company’s PLI database (Figure 1). In addition, 7 papers were found in the full-text literature search by Zanardo et al. 25 In total, 31 studies were included (Figure 1).
Nineteen studies reported efficacy results for the detection or confirmation of BC (Tables 1 and 4), 3 studies reported safety results (Table 2), and 11 studies reported other findings, not relevant for efficacy or safety analysis (Table 3). 2 studies provided data for efficacy and safety analysis27,28 (Figure 1).
Efficacy studies on CEM using iopromide for the detection or confirmation of BC.
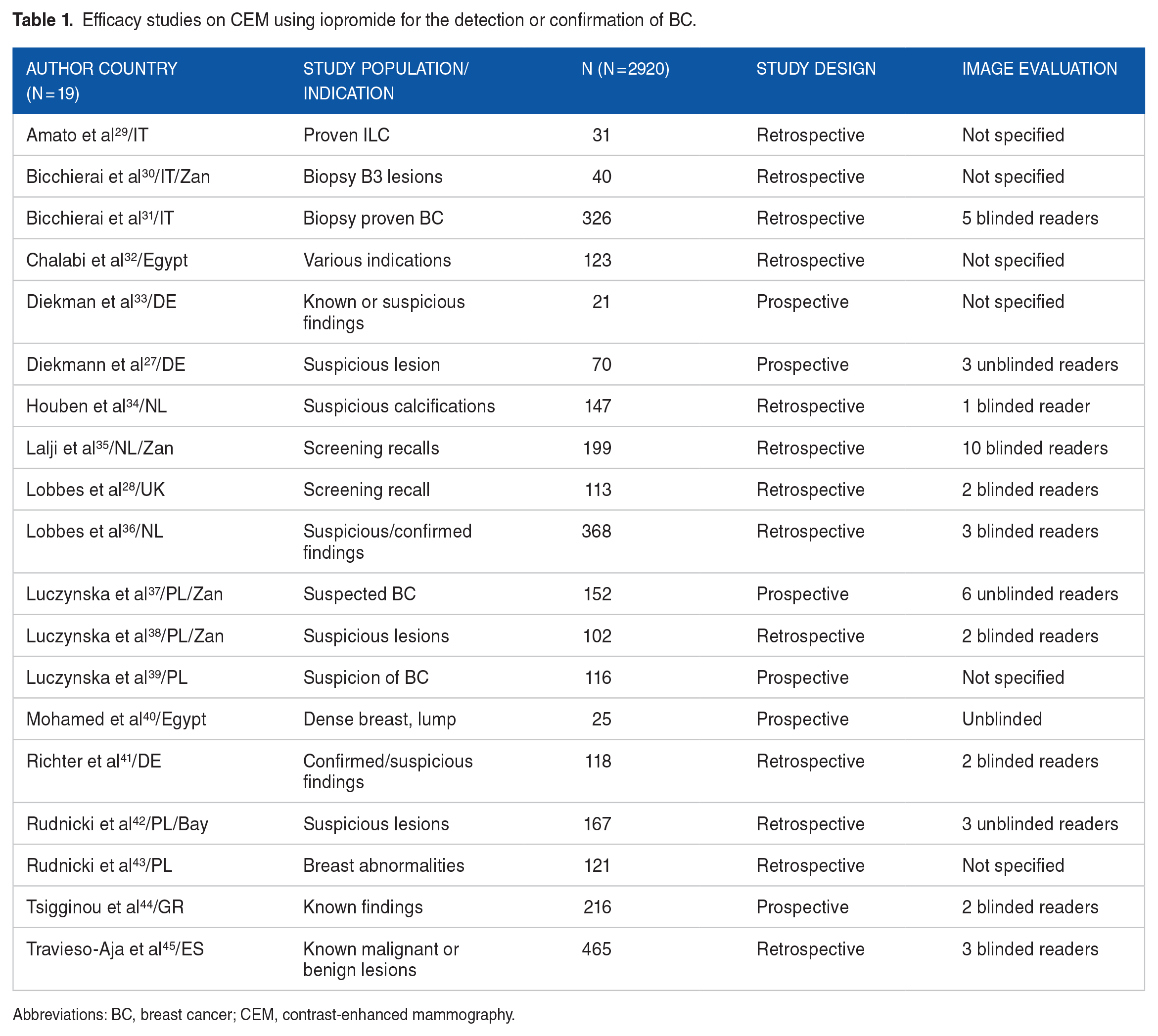
Abbreviations: BC, breast cancer; CEM, contrast-enhanced mammography.
Safety studies on CEM using iopromide.

Abbreviations: BC, breast cancer; CEM, contrast-enhanced mammography.
Studies on CEM using iopromide, not specifically reporting efficacy or safety (=reason for exclusion).

Abbreviations: BC, breast cancer; CEM, contrast-enhanced mammography.
Patient population
Mean age of patients was specified in 24 of the 31 studies, covering 4348 of 5194 patients (84%). Considering the respective patient counts for the 24 studies, the mean age of the included patients was 57.2 years (the average of the specified mean ages without correction for the patient numbers was 56.1 years). Patients were referred to imaging for screening purposes and for characterization of suspicious clinical findings.
Efficacy
A total of 19 studies reported efficacy of iopromide in contrast-enhanced mammography. Six had a prospective and 13 a retrospective design. Nine studies were read by blinded readers and 4 by unblinded readers. The other studies did not specify reading procedure (Table 1).
CEM vs XRM
Eleven studies reported efficacy results of iopromide CEM vs XRM. Sensitivity was always higher for CEM, ranging from 62% to 100%. The advantage ranged from 1% 44 up to 44% 40 (Table 4, Figure 2). Overall, CEM provided a gain in sensitivity of 7% (95% CI [4%, 11%]) in the meta-analysis. No hints for heterogeneity were found for the difference in sensitivities for the studies included (P = .06) (Figure 3).
Efficacy results—CEM vs XRM, bMRI and uncontrolled.

Abbreviations: BC, breast cancer; BIRADS, Breast Imaging Reporting & Data System; bMRI, breast magnetic resonance imaging; CEM, contrast-enhanced mammography; NPV, negative predictive value; PPV, positive predictive value; XRM, traditional mammography.
For Figure 3, we recalculated the number of true positives for sensitivity. This resulted in differences of 43% for sensitivity and −4% for specificity.
Values given for detection of index lesion, identification of adjunctive lesions and correct determination of extension for masses, non-mass enhancement and masses associated with non-mass enhancement.

Sensitivities of CEM vs XRM. CEM indicates contrast-enhanced mammography; XRM, traditional mammography.

Differences of sensitivities of CEM vs XRM. CEM indicates contrast-enhanced mammography; CI, confidence interval; XRM, traditional mammography.
A similar diagnostic advantage of CEM was seen for specificity in most studies. The differences were found to be in a range of −3% 40 to 46% 28 (Table 4, Figures 4 and 5). Overall, CEM was found to provide a gain in specificity of 16% (95% CI [6%, 26%]). However, a hint for heterogeneity was found for the difference in specificities (P < .01). Therefore, no clear pattern on the advantage was found. Nevertheless, at least no disadvantage of CEM vs XRM can be concluded based on the study-level differences (Figure 5).

Specificities of CEM vs XRM. CEM indicates contrast-enhanced mammography; XRM, traditional mammography.

Differences of specificities of CEM vs XRM. CEM indicates contrast-enhanced mammography; CI, confidence interval; XRM, traditional mammography.
Accuracy, and positive and negative predictive values (PPV, NPV) were not recorded by all authors. However, those who did record found more favorable results for CEM (Table 4). Three cases with intra-individual comparison of both modalities are shown on Figure 6.

Cases showing intra-individual comparison of iopromide CEM and XRM. CEM indicates contrast-enhanced mammography; DCIS, Ductal carcinoma in situ; XRM, traditional mammography.
Four case studies with intra-individual comparisons of XRM vs CEM images are shown on Figure 6.
CEM vs bMRI
2 studies reported efficacy results of iopromide CEM vs bMRI. Sensitivity was similar 43 or higher for CEM. 38 Specificity was higher 43 or not reported. 38 Accuracy, PPV, and NPV were in favor of CEM in both studies (Table 4).
CEM uncontrolled
Six efficacy studies presented uncontrolled findings. Sensitivity was in the range of 25% to 100%, 29 specificity from 61% 42 to 98% 30 (Table 4).
Safety
Adverse events (AEs)/adverse drug reactions (ADRs) related to iopromide administration were reported in 3 of the 31 studies (Table 2). In total, 8 adverse drug reactions were reported from 7 patients: 1 case of severe allergic reaction that required intensive care and led to discontinuation of the procedure, 27 1 moderate 46 and 5 mild cases of urticaria,28,46 and 1 moderate shortness of breath. 46 In relation to the total number of 1022 patients examined in these studies, this corresponds to 0.8% (Table 5).
Safety results of studies on contrast-enhanced mammography using iopromide.
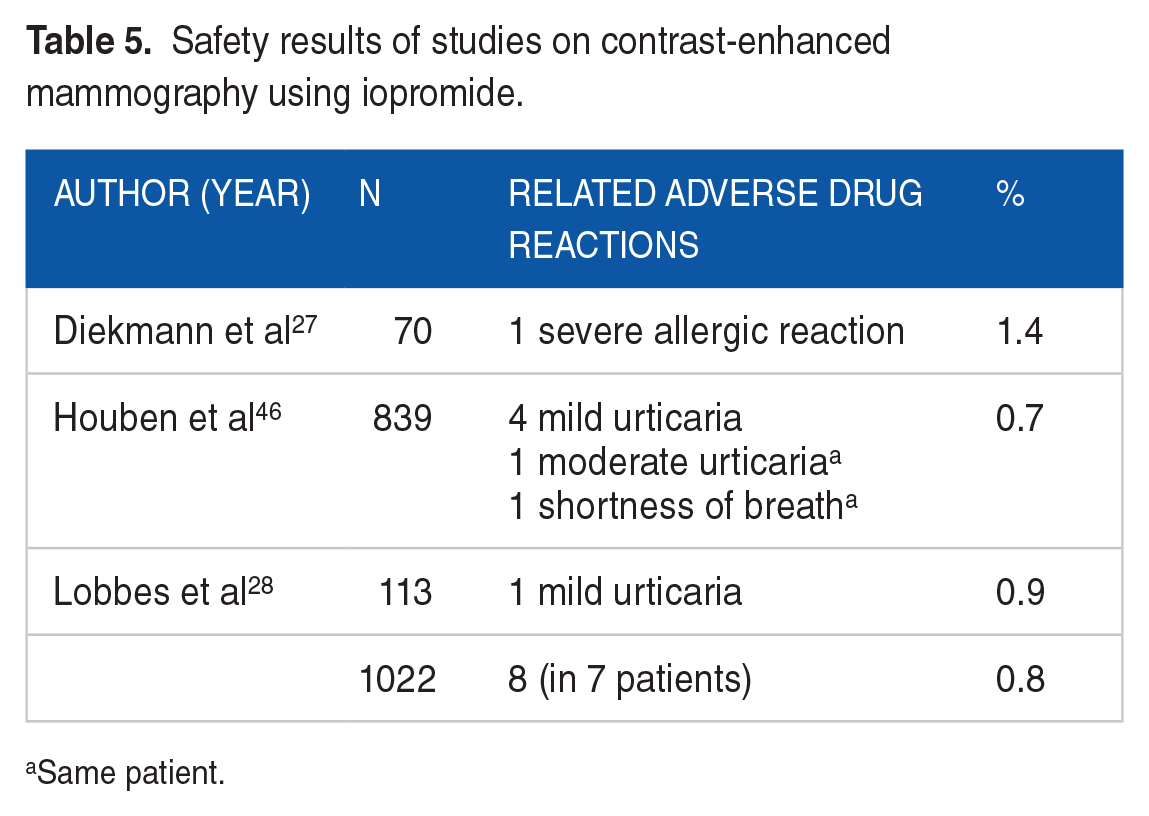
Same patient.
In the remaining 28 studies, no AEs or ADRs to iopromide were reported. However, in 11 studies, patients with known risk of allergic reactions or CIN were excluded upfront, in another 15 studies exclusion criteria to known risks of allergic reactions, CIN or renal insufficiency were not explicitly mentioned. 1 study excluded patients for other reasons than known risk of allergic reactions or CIN.
Most of the AEs were reported in context of hypersensitivity reactions and are in line with the well-established safety profile of iopromide. No new safety concerns were identified during this analysis. The benefit risk profile remains favorable and unchanged.
Discussion
We systematically analyzed the usage of iopromide for CEM for detection or confirmation of BC and found strong evidence for higher diagnostic performance compared with XRM and similar performance compared with bMRI.
The PLI database is a robust source as it aggregates data from 4 leading international literature databases. The PLI retrieved more publications than MEDLINE/EMBASE alone (applied by Zanardo et al 25 ) as this system is specifically designed for the company’s global pharmacovigilance department (PV). As PV is obliged to report any ADR all over the world, also country-specific journals are utilized, identifying publications in other languages apart from English. This ensures a high degree of completeness of the literature search (Figure 1).
All 31 studies on iopromide included at total of 5194 patients, 2920 and 1022 patients in the efficacy and safety analysis, respectively (2 studies reported efficacy and safety). In 11 studies (n = 1435 patients) neither efficacy nor safety was reported so they were excluded from the analysis. All patients were included either for screening or for characterization of suspicious findings.
Tagliafico et al 9 published a similar review and meta-analysis focusing on sensitivity and specificity on 8 studies including 920 patients presenting 994 lesions. Unfortunately, the type of LOCM was not specified. Zanardo et al 25 included 14 012 patients from 84 studies looking at a large range of LOCMs. However, Zanardo et al 25 investigated primarily technique, protocols, and adverse reactions, while this analysis put emphasis on efficacy and safety.
In 11 studies, sensitivity and specificity of CEM vs XRM were almost always in favor of CEM with an overall gain of 7% in sensitivity and no hints for a loss in specificity. This held also true for accuracy, PPV, and NPV (Table 4). These findings support results from other groups on other LOCMs.
Tennant et al 10 performed a review of 100 CEM examinations (LOCM not specified) vs low-energy images and reported CEM showing increased sensitivity (95% vs 84%, P < .025) and specificity (81% vs 63%, P < .025). They conclude that CEM provides improved efficacy in BC screening and staging as primary mammographic investigation.
Likewise, Mori et al 58 focused on 72 women with dense breasts who underwent CEM with iohexol and XRM. Sensitivity was 86.2% for CEM and 53.4% for XRM (<.001), and specificity 94.1% and 85.9% (<.016), respectively. They concluded that CEM offers superior clinical performance compared with XRM and may decrease false negatives especially for women with dense breasts.
Kim et al 59 compared both imaging modalities in BC screening on 64 cases. Median sensitivity and specificity improved with the addition of CEM (iohexol). Sensitivity: from 86% (XRM alone) to 100% (XRM + CEM), specificity, from 85% to 88%.
While other publications point in the same direction,11,60 Lorek et al 61 criticized that CEM overestimates the tumor size on average by 5 mm.
The 2 studies on efficacy of iopromide CEM vs bMRI showed similar 43 or higher sensitivity for CEM 38 while accuracy, PPV and NPV were in favor of CEM (Table 4).
Three intra-individual comparison studies of CEM with other LOCMs vs bMRI are of interest in this context.62 -64 1 retrospective study by Xing et al 64 on 235 patients administered iohexol and 2 prospective studies, 1 on 178 patients by Fallenberg (iobitridol) 62 and 1 on 84 patients by Kim (iohexol) 63 reported a similar trend: Almost identical sensitivities for both modalities and a slightly higher specificity for CEM. In contrast, Pötsch et al 65 found in a meta-analysis of 7 studies investigating 1137 lesions higher sensitivity for bMRI (97%) vs CEM (91%), however, at the cost of lower sensitivity, 69% vs 74%.
The 6 uncontrolled studies on iopromide showed a sensitivity of 25% to 100% 29 and a specificity of 61% 42 to 98% 47 (Table 4).
A recent systematic review and meta-analysis comprising a total of 945 patients of 8 studies using 5 different LOCMs by Suter et al 66 calculated a sensitivity of 85% and a specificity of 77%. Tagliafico et al 9 found a sensitivity of 98% and a sensitivity of 58% in a meta-analysis of 8 studies with 920 patients. Although the range is quite broad, our results seem to corroborate these findings.
A total of 8 AEs of iopromide-related AEs/ADRs were reported in 7 patients from three studies comprising 1022 patients. This corresponds to a reporting rate of 0.8%. Skin reactions like urticaria, erythema, rash, and pruritus are usually the most frequent events. The most severe AEs are hypersensitivity reactions. 67 The safety findings reported here in patients receiving iopromide-enhanced CEM are well in line with other studies focusing on AEs/ADRs after intravenous iopromide administration for other indications, ie, 1.5%, 18 0.7%, 19 and 2.8%. 20 These are also comparable with results of studies on a broad spectrum of different LOCMs, 0.37%, 22 0.34%, 68 and 0.97%. 69 The reported AEs/ADRs related to contrast media administration are expected to be independent of the imaged region of the body.
A frequently raised concern about CEM is the increased radiation dose compared with XRM. James et al 70 retrospectively compared the radiation dose of CEM using high- and low-energy projections with the radiation dose received during XRM in 173 patients with varying breast thickness and density. At a mean breast thickness of 63 mm, the average glandular dose was 3.0 and 2.1 mGy for CEM and XRM, respectively. They conclude that this increase of 0.9 mGy falls below the dose limit of 3 mGy set by Mammography Quality Standards Act regulations (MQSA). Similarly, Gennaro et al 71 reported that the dose for CEM is about 30% higher compared with standard XRM, an increase close to that of digital tomosynthesis, but it is the upper limit according to MQSA.
CEM is assumed to become an emerging new imaging modality providing higher diagnostic efficacy compared with XRM and similar efficacy compared with bMRI in BC screening and diagnostic work up. In addition, CEM also offers the opportunity for CEM-guided biopsy, a new technique that can be successfully used as an alternative to MRI-guided breast biopsy. 72 As CEM requires only limited technical upgrades of the broadly available XRM equipment and can be implemented easily into the current XRM workflow, it might be a cost-effective alternative to bMRI, in particular as there is some evidence that patients prefer CEM over bMRI.73,74 CEM might also be a good option when MRI technique is not available.
Limitations
Some limitations need to be addressed:
This review was not based on the patient individual data of the original studies but published results.
As for any literature review, the retrospective nature of the data and the variability of information collected and reported is a major limitation.
As this review focused on studies on iopromide, the full picture regarding use of LOCMs in CEM is not reflected.
Each study only provided 1 pair of sensitivity/specificity results, so that the meta-analyses for the diagnostic parameters might be biased due to, eg, reader experience and selection of readers. In addition, the data sources with regard to prospective and retrospective data and blinding of readers did vary across studies, which increases uncertainty and heterogeneity in the results (Table 1).
Except for 3 studies, all other were performed in Europe.
Studies included mostly women with a high-risk predisposition for BC. It is not clear if this might impact the rate of AEs related to iopromide administration.
No long-term safety data after repetitive administrations were reported.
Studies using iopromide for other imaging modalities (eg, breast CT) were not included.
No dose-finding studies were found.
Potentially varying diagnostic performances between different commercially available mammography systems were not assessed. An impact of the mammography system on diagnostic performance cannot be excluded.
Conclusions
Pertinent literature supports iopromide to be an effective and safe agent for CEM for the detection or confirmation of BC. The overall gain in sensitivity for iopromide CEM vs XRM was 7% without compromising specificity.
Footnotes
Acknowledgements
Not applicable.
