Abstract
Rationale and Objectives:
According to the Breast Imaging and Reporting Data System (BI-RADS), one of the main limitations of MRI is diagnosing the non-mass enhancement (NME). The NME lesion is challenging since it is unique to the MRI lexicon. This study aims to report our experience with NME lesions diagnosed by MRI referred for MRI-guided biopsies and discuss the management and follow-up of these lesions.
Materials and Methods:
We retrospectively evaluated all MRI-guide breast biopsies. We included all patients referred for NME breast MRI-guided biopsy in screening settings. All patients had a negative second-look mammography or ultrasonography. We correlated the distribution and internal enhancement pattern (IEP) of the NME lesions with histology. Invasive ductal carcinomas (IDC) of no special type and ductal carcinoma in situ (DCIS) were considered malignant lesions.
Results:
From January-2018 to July-2021, we included 96 women with a total of 96 lesions in the study. There were 90 benign and 6 malignant lesions with DCIS prevalence (5/6 cancers). The most frequent benign lesion type was fibrocystic changes. There were no NME lesions with diffuse or multiple area distribution features referred to MRI-guided biopsy. The positive-predictive values (PPV) were respectively 0.0%, 2.5%, 9.0%, and 11.0% for linear, focal, regional, and segmental distribution describers, and 0.0, 3.0%, 7.9%, and 50% for homogenous, heterogeneous, clumped, and clustered-ring enhancement patterns.
Conclusion:
We observe the high potential risk for malignancy in the clustered-ring enhancement followed by the clumped pattern. Segmental distribution presented the highest predictive-positive values.
Introduction
Breast magnetic resonance (MRI) scan indications are increasing for diagnosis and screening in clinical practice. The main indications are staging of known cancer, screening for breast cancer in women at increased risk, and evaluation of response to neoadjuvant chemotherapy. Other indications for breast MRI are diagnosing carcinoma of unknown primary origin and problem-solving due to high negative predictive value. Uncertain benign findings at mammography or ultrasonography that cannot be sampled for biopsy could have a complementary evaluation by MRI.1-5
Despite high sensitivity for detecting lesions, one limitation of MRI is low specificity, as established and discussed in the literature. MRI evaluates morphology by high-resolution sequences and functionality by dynamic sequences. Some benign and malignant lesions could have the same expression in the images; since benign lesions are more common than malignant, we expect more false-positive results than true-positive ones. 3 Another factor influencing imaging interpretation is the background enhancement pattern (BEP). An asymmetrical BEP could result in a false-positive result in an asymptomatic patient. 6
The increasing number of false-positive results is related to the broad availability and reduced cost of MRI scans and implementation of FAST screening protocol.7,8 Some lesions newly diagnosed by MRI could be found in second-look mammography or ultrasonography and have ductal carcinoma in situ (DCIS) as the prevalent lesion at pathology. 9
Although breast MRI safety has been exhaustively discussed and validated in the literature, some concerns regarding gadolinium brain retention and systemic nephrogenic fibrosis persist.10,11 There are also some individual restrictions to performing an MRI scan with contrast because of reported allergies, renal failure, and MRI-unsafe implants. 12
Most of the published articles discuss the benefits of the MRI scan. 13 However, few studies reported the MRI harms and how we can manage and reduce the false-positive results.14-16 According to the Breast Imaging and Reporting Data System (BI-RADS) fifth edition lexicon, one of the main limitations of MRI lies in diagnosing the NME. 17 The NME lesion is challenging since it is unique for the MRI lexicon, and its management and follow-up lack standardization. (Figure 1)18,19
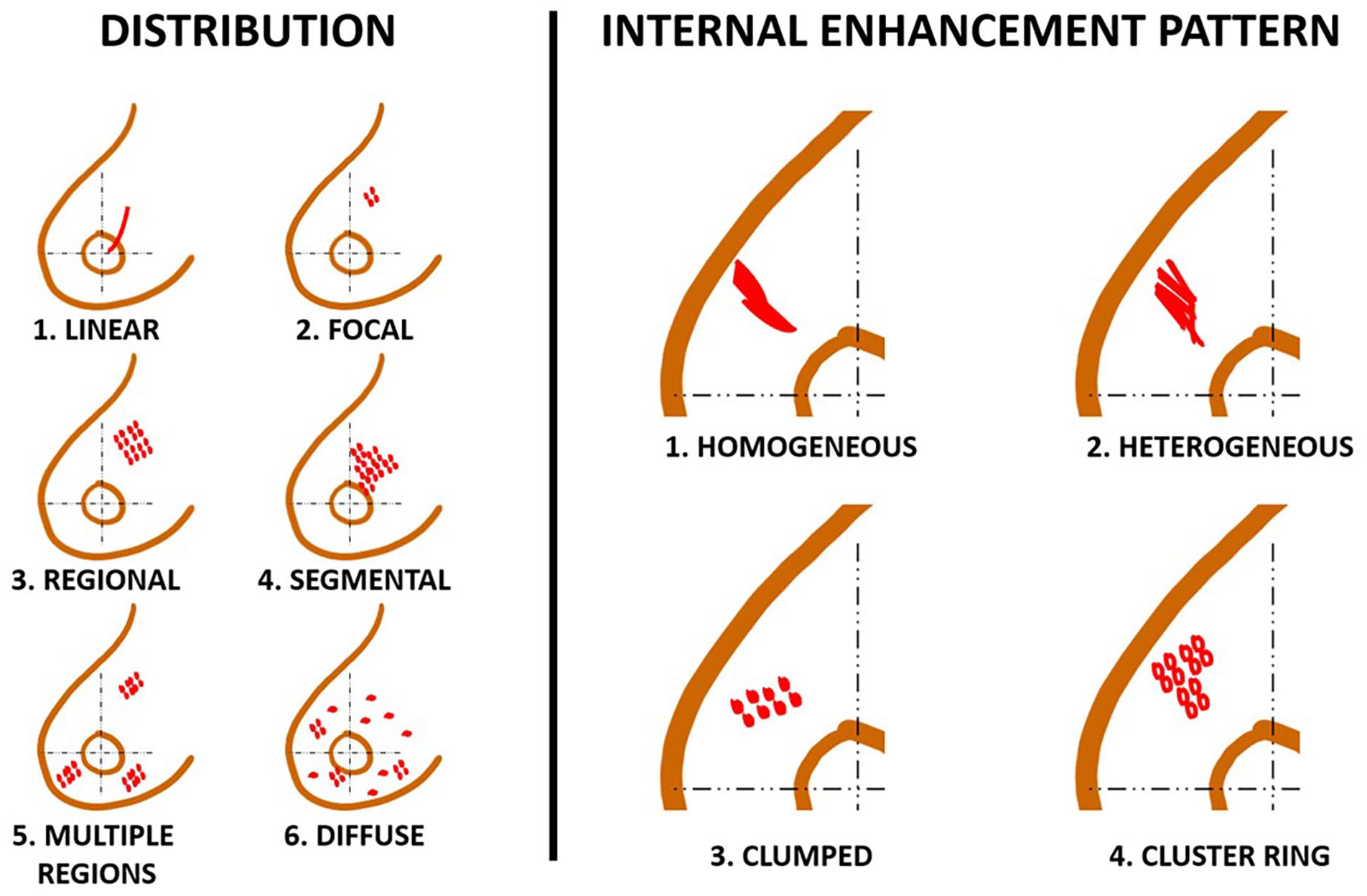
Examples of the BI-RADS classification of non-mass enhancement lesions by distribution and internal enhancement pattern.
This study aims to report our experience with NME lesions diagnosed by screening MRI referred for MRI-guided biopsies and discuss the management and follow-up of these lesions.
Material and Methods
We retrospectively evaluated all MRI-guided breast biopsies from January-2018 to July-2021 in a single institution dedicated to women’s health care. We included patients referred for NME MRI-guided breast biopsy in screening settings without correlation to second-look mammography or ultrasonography. Following the screening criteria, we excluded patients with masses, lesions, clinical complaints, or a previous breast cancer diagnosis within the last 5 years. We only included in the study incidental findings of NME lesions in asymptomatic patients referred for screening MRI.
Our institutional board waived the ethical committee approval. All patients signed an informed consent term for data availability for scientific purposes.
Not all patients referred for MRI biopsy in our institution had had their previous screening breast MRI performed in our practice. As the BI-RADS descriptors are subjective and have the issue of the interobserver agreement, we decided to re-evaluate these lesions. Two radiologists re-evaluated the previous exams with suspicious findings and classified the lesions according to the latest BI-RADS lexicon. The NME lesions were classified according to distribution and IEP. (Figure 1) 17 According to the lexicon, NME consists of lesions that are neither a mass nor a focus. NME extends over small or large regions, and the enhancement pattern is discrete from the normal surrounding glandular tissue. NME can be described according to distribution (focal, linear, segmental, regional, multiple regions, and diffuse) and internal enhancement patterns (homogeneous, heterogeneous, clumped, and clustered ring). 17
We performed a second-look ultrasound to find the NME lesion. We opted to perform the biopsy guided by ultrasound if the lesion was found.
We excluded from the study lesions other than NME, like masses and architectural distortions, and NME in diagnostic MRI.
Patients eligible for percutaneous biopsy were positioned in the MRI scan in the prone position, using a dedicated 8-channel breast coil. After breast compression, we performed a pre-contrast sequence, followed by a 4 minute dynamic series with fat suppression. The automatic subtraction reconstruction was calculated from the acquired dynamic series in the sagittal plane. All studies were performed on a 1.5 T MR scanner (Spree; Siemens. Malvern, PA). MRI-guided core needle biopsies were obtained using a 10-gauge vacuum-assisted needle (Vacora, Bard Biopsy Systems; Temple, AZ, USA). Multihance (0.1 mmol/kg, Bracco Diagnositics Inc., Monroe Township, NJ) was the IV contrast. A marker was placed at the time of biopsy for each procedure, and post-biopsy mammograms were performed.
Previously reported NME lesions, with negative findings at the biopsy scan, were followed-up after six months of the exam. These findings were considered a physiological enhancement. (Figure 2)

Figure (A, B, C, D). Screening MRI in a 56 year-old wonan. The pre biopsy MRI images, post contrast (A) and MIP reconstruction (B) shows a regional and clustered NME (orange arrow). The 6 month follow-up (C and D) shows an atenuation of the MRI findings. The biopsy was not performed.
The radiologists correlated the imaging and pathological reports when the pathological results were available. If there was agreement, we generated a final report. We indicated further lesion evaluation when there was disagreement, indicating a surgical biopsy.
All patients with negative biopsy were scheduled for a short-time follow-up breast MRI in the following six months to evaluate the lesion stability.
We correlated the distribution and IEP of the NME lesions with histology. Invasive ductal carcinomas (IDC) of no special type and DCIS were considered malignant lesions. We also performed the same test assessing pathological lesions of uncertain malignant potential (category 3) as positive findings for comparison purposes. (Figure 3)

Figure (A, B, C, D). Screening MRI in a 45 year-old wonan. The MRI images, post contarst (A) and subtraction (B) shows a linear and heterogeneous NME (orange arrow). The histology of the biopsy was a pappillary lesion, category 3 (C and D).
For the correlation between the distribution and internal enhancement pattern descriptors with the benign and malignant lesions, we used the chi-squared test (Medcalc, Acacialaan, Ostend, Belgium) software for statistical analysis, adopting a significance level for P < 0.05.
Results
From January-2018 to July-2021, we evaluated 122 lesions referred for MRI-guided biopsy to diagnose the NME lesions. All patients were asymptomatic, with a previous negative second-look mammography or ultrasonography.
We included 96 patients with 96 lesions in the study. The median age of the patients was 49.3 years, and the average lesion size was 2.4 cm. (Figure 2) There were six malignant lesions, with DCIS prevalence (5/ 6)). The most frequent benign lesion was fibrocystic changes (36/ 90). The lesions of uncertain malignant potential, category 3, included papillary lesion, atypical ductal hyperplasia, and radial scar, with 29 lesions (29/ 90). There were no NME lesions with the diffuse or multiple areas distributions features referred to MRI-guided biopsy. (Table 1)
Characteristics of non-mass enhancement lesions referred for MRI-guided biopsy.

When evaluating the correlation to NME exclusively with malignant histology, clustered ring pattern was the only describer associated with breast cancer with a significant p level. The result was confirmed when we associated the lesions of category 3. (Table 2)
Correlation of non-mass enhancement lesions to malignancy and to suspicious lesions.

The predictive-positive values for the distribution describer were respectively 0.0, 2.5%, 9.0%, and 11.0% for linear, focal, regional, and segmental, and for enhancement pattern respectively 0.0, 3.0%, 7.9%, and 50% for homogenous, heterogeneous, clumped, and clustered-ring.
Of the 20 lesions with negative findings at the MRI-guided biopsy scan, 13 patients performed follow-up scans confirming the false-positive results in the previous scan. We considered these NME lesions as physiological changes.
Of the 29 lesions of uncertain malignant potential (category 3), 12 have surgical excision biopsies with the same MRI-guided biopsy results. The other 17 were followed up by imaging as the histological results were in accordance with the images’ features.
The DCI lesion that appears as NME was a heterogeneous focal enhancement in the central localization of the left breast. The lesion occurred at the surgical site of a luminal A carcinoma diagnosed and adequately treated for eight years.
Discussion
Since breast MRI has been widely used in clinical practice for diagnostic and screening purposes, we observe a rise in lesions exclusively seen at MRI scans. Despite the high sensitivity of MRI to detect breast cancer, false-positive results are the main weakness of the method. Usually, the suspected lesions described in MRI have biopsy indications, and sometimes the diagnostic procedures are not trivial. Breast MRI-guided biopsy is expensive, has the limitation of contrast usage, and has toleration concerns for claustrophobic patients. Although the biopsy is simple to perform, the number of steps to get the specimens is time-consuming as it is not a real-time procedure.20-23
The best alternative for MRI-guided biopsy is when the lesion is present in a second-look evaluation by ultrasound scan or mammography. When there is an unequivocal equivalence of the findings, performing the percutaneous biopsy guided by the preferred modality is safe. (Figure 3) The MRI-guided biopsy is the only alternative for percutaneous biopsy when there is no correspondence. An option in these cases when we cannot complete the biopsy is a short-time follow-up (6 months).
The MRI’s unique findings are related to NME, a lesion that resembles calcifications in mammography. The most common tumors diagnosed as NME are DCIS, low-grade DCI, and lesions with an uncertain malignancy potential (category 3). 23 In contrast, the benign lesions usually related to this finding are fibrocystic changes and fibroadenosis. These lesions commonly pertain to low-aggressiveness tumors, the same observed in mammographic calcifications. The low-grade lesions have a higher duplication time and less potential for distant metastasis. For example, 50% of DCIS evolve to DCI, and a low-grade carcinoma has a median duplication time of 241 days, compared with 103 days for triple-negative cancer.18,24
Our study evaluated only NME lesions exclusively diagnosed at MRI in patients without clinical complaints from the MRI screening settings. We believe that understanding the relevance of the NME finding is imperative to manage this diagnosis. The primary malignant histology finding in our study was DCIS. At the same time, the invasive cancer lesion was a recurrence of a luminal-A carcinoma in the surgery site eight years after the treatment. This indolent characteristic of the NME concedes a secure 6-month follow-up for non-biopsied lesions. (Figure 4)

Flow-chart of the study design to evaluate non-mass enhancement in screening breast Magnetic Resonance.
Based on our findings, we introduced in our clinical practice a subclassification of the BI-RADS category 4 for NME in screening settings. 17 We classify NME with clustered-ring enhancement patterns and segmental distribution as BI-RADS 4b. We base this subclassification on the BI-RADS recommendation for calcifications, where PPV ranges > 10% to =< 50. All 4b lesions are referred for percutaneous biopsy even when the second-look evaluation was negative. (Figures 5, 6, 7 and 8)

Figure (A, B, C, D). Screening MRI in a 45 year-old wonan with breast implants. The MRI images, PD- proton density (A) and post-contrast (B) shows a focal and heterogeneous NME (orange arrow). The histology of the biopsy was an invasive ductal carcinoma (C). The biopsy was performed guided by a second-look ultrasound (D).
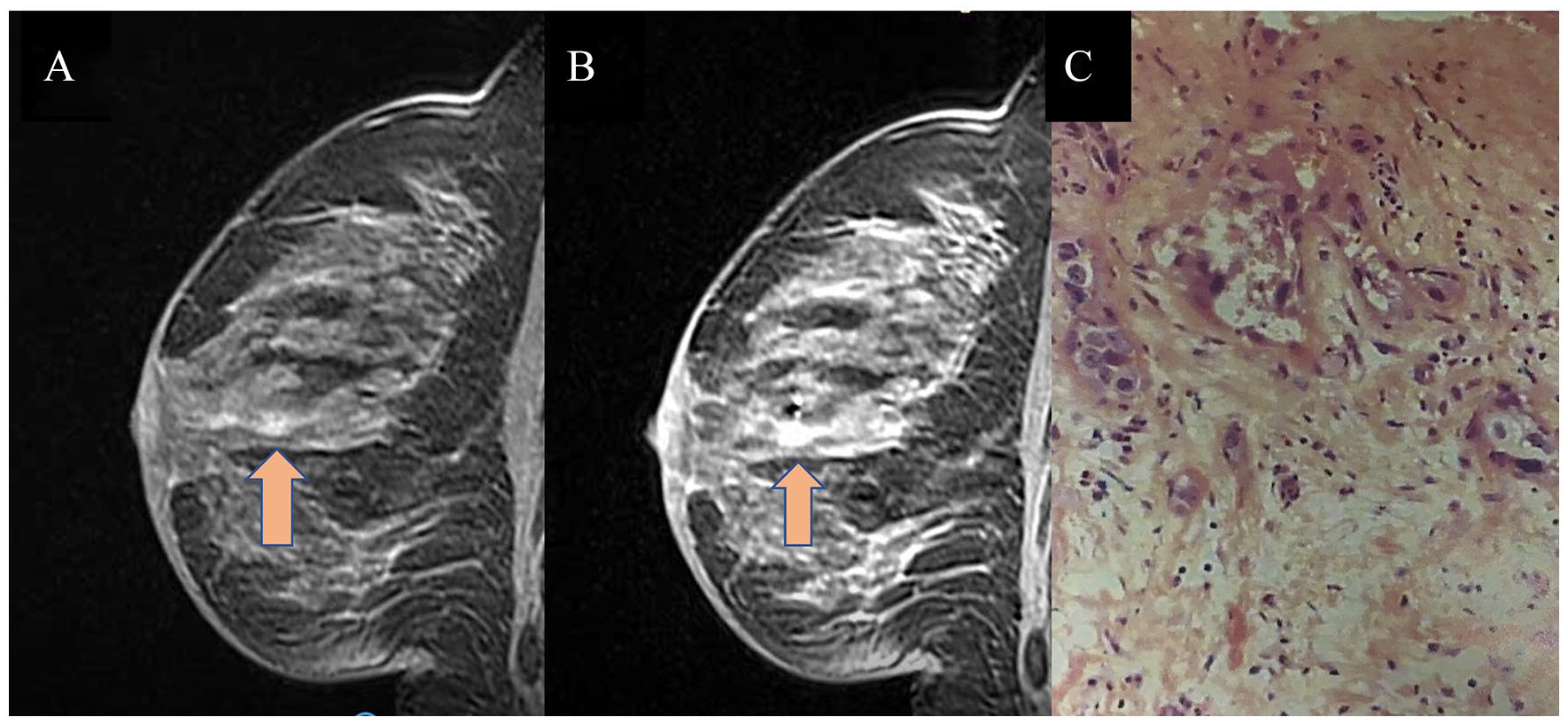
Figure (A, B, C). Screening MRI in a 56 year-old wonan with history of breast cancer in the right breast for 8 years. The MRI images, pre-biopsy (A) and post-biopsy (B) shows a focal and heterogeneous NME (orange arrow). Figure B shows the biopsy marker at the site of biopsy. The histology of the biopsy was an invasive ductal carcinoma (C).

Proposal for final classifications of non-mass enhancement lesions as primary finding in screening breast Magnetic Resonance imaging.

Figure (A, B, C, D, E, F). Screening MRI in a 43-year-old woman. The MRI images, pre-contrast (A), post-contrast (B), subtraction (C) and post-biopsy (D) shows a segmental and clustered rind NME in the upper quadrant. Figure D shows the biopsy marker at the biopsy site. The histology of the biopsy was a ductal carcinoma in-situ (E and F).
On the other hand, lesions classified with the other descriptors were classified as BI-RADS 4a and referred for a second-look evaluation by ultrasound scan and mammography. We suggest a 6-month follow-up as an alternative for the biopsy if the second look was negative. When the lesion is stable at follow-up, we recommend additional follow-ups of another 6 months and 1 year. We perform a new second-look evaluation and biopsy when the lesion progresses, regardless of the second-look result. (Figure 3)
Linear, multiple areas and diffuse distribution with homogeneous internal enhancement pattern and negative second-look evaluation are classified as BI-RADS 3 since we did not find any malignancy or lesions with uncertain malignancy potential. We believe the introduction of this protocol should reduce the number of biopsies in false-positive MRI results.
When we analyzed the NME distribution and enhancement pattern, we observed an association with malignancy only in the clustered-ring descriptor. We expected this finding since the most suspicious feature of NME is the clustered-ring pattern, in analogy to fine-linear branching calcifications in mammography. Although malignancy was prevalent in segmental distribution, with the highest PPV value for the distribution descriptor, the association with malignancy was not statistically significant. (Figure 6)
The study has some limitations. First is the retrospective nature of the study. Another limitation is the small number of malignancies; however, since we only evaluated asymptomatic patients in screening breast MRI settings with negative ultrasound and mammography, malignancy is exceptional. The NME lesions were incidental findings in screening MRI, and most were related to benignity. As a private practice, we did not have access to the follow-up data of all patients. We also excluded patients when the NME features were described as associated findings, which explains the PPV difference from other studies that included NME as primary and associated findings.25-27
Our results show the features of the breast NME lesions in asymptomatic patients referred for screening MRI. We observed the high potential risk for malignancy in the clustered-ring enhancement, followed by the clumped pattern. Regarding the distribution, segmental presented the highest PPVs.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Eduardo de Faria Castro Fleury: Conceptualization, Methodology, Data curation, Writing- Original draft preparation. Caio Castro: Visualization, Investigation. Mario Sergio Campos do Amaral: Supervision, Validation. Decio Roveda Junior: Reviewing and Editing,
