Abstract
The aim of our study is to provide clinicopathologic and imaging features of breast primary angiosarcoma. We retrospectively analyzed cases of primary angiosarcoma diagnosed at the Pathological Laboratory of Lomé over a period of 20 years (2000-2019). Eight cases of primary angiosarcoma of the breast were collected, including 6 from women and 2 from men. The median age was 41.63 years (range from 17 to 66 years). Depending on the location, there were 4 of 8 cases in the left breast. Ultrasound classifications were BI-RADS 4 and 5. Histology revealed a malignant vascular proliferation composed of small lumens lined by atypical endothelial cells, evident mitoses, and foci of necrosis. On immunohistochemistry, the lesional cells expressed CD31, CD34, and Factor VIII. Based on Federation Nationale des Centers de Lutte Contre Le Cancer (FNCLCC) grading, the cases were grade II and III. Overall survival at 6 months was estimated to be 25% in a woman.
Introduction
Angiosarcomas are rare vascular cancers, 2% to 3% of soft tissue sarcomas but with a high aggressive and metastatic potential. 1 Angiosarcoma of the breast is an extremely rare tumor and accounts for less than 1% of all breast cancer cases. 2 A distinction is made between primary angiosarcoma of the breast and secondary angiosarcoma which occurs a few years after radiation therapy to the breast for invasive carcinoma. 3 Primary angiosarcomas are extremely rare and represent 0.04% of all malignant breast tumors. 4 Unlike secondary, the natural history of primary angiosarcoma is only partially understood. 1 Primary angiosarcoma is thought to occur in the breast parenchyma and then infiltrate the skin and surrounding subcutaneous tissue. 5 It is most often seen in younger patients, in their 30s and 40s.1,6 Clinically, women with primary angiosarcoma typically present with swelling, and exponential growth in the breast. 7 Treatment recommendations based on solid medical evidence are lacking. Nevertheless, surgery remains the cornerstone of treatment and may be combined with chemotherapy or radiotherapy. 8 The prognosis of the diagnosis is generally poor due to the very aggressive course of the disease and its tendency to have local recurrences and distant metastases.2-5 Most of the data available in the literature on primary breast angiosarcoma come from case reports.9,10 We initiated this study which reports a series of cases of primary angiosarcoma of the breast diagnosed in Togo. The objective was to study the epidemiological, clinical, diagnostic, therapeutic, and prognostic characteristics of primary angiosarcomas in the breast of our patients.
Methods
It is a retrospective review of the clinical and pathologic features of primary mammary angiosarcoma diagnosed histologically at the Pathological Anatomy Laboratory of Lomé University Hospital over a period of 20 years (2000-2019). Togo is a small country of 56 600 km2, with an estimated population of 7 200 000 inhabitants, located between Ghana in the west and Benin in the east. Specific data such as age at diagnosis, gender, symptoms at presentation, clinical history, tumor size, imaging data, treatment performed presence of local recurrence and distant metastasis, and survival of the patient were assessed on the basis of the available medical records. The study material consisted of biopsies and surgical specimens fixed in 10% buffered formalin. Histological analysis was carried out on samples coated with paraffin and fixed with formalin stained with hematoxylin–eosin–saffron. Immunohistochemical analysis was also performed on paraffin coated and formalin fixed tissue.
Results
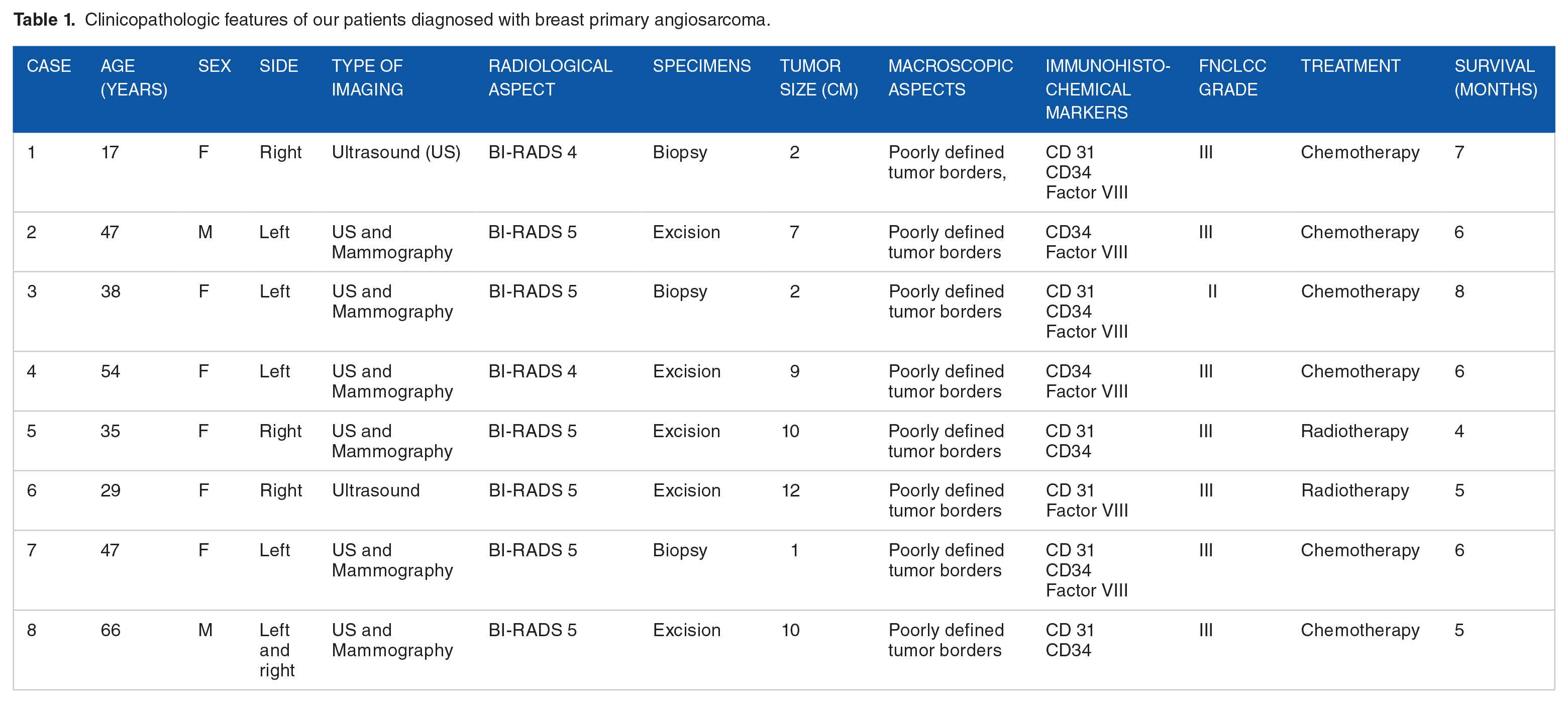
We recorded eight cases of primary angiosarcoma of the breast. These are two men and six women. Their median age was 41.63 years (ages ranging from 17 to 66 years). The lesions were right sided (4/8), left sided (3/8), or bilateral (1/8). Both men were farmers and of the 6 women, 4 were housewives and 2 were traders. They were 5/8 living in rural areas and 3/8 in urban areas. The family history of breast cancer was known in 3 patients. There was no history of irradiation in all patients. Table 1 summarizes the clinicopathologic features of our 8 patients diagnosed with angiosarcoma of the breast. Clinically, it was a large mass with irregular contours, associated with ulceration and lymphadenopathy (Figure 1). The imaging data made it possible to find lesions classified BI-RADS 4 and 5 (BI-RADS 4 and 5) on mammography and ultrasound and lumpectomy was performed in five patients (4 women and 1 man) (Figure 2). All lesions were solid masses. The size of the tumor, as evaluated radiologically or on resected samples, ranged from 2 to 12 cm, with a mean of 7 cm. Macroscopically, the lumpectomy specimens had size ranging from 5 to 10 cm in greatest dimension with firm cut surfaces including foci of hemorrhage, and poorly defined borders. Histology revealed a malignant vascular proliferation composed of small lumens lined by atypical endothelial cells, evident mitoses, and foci of necrosis (Figure 3). On immunohistochemistry, the lesional cells expressed CD31, CD34, and Factor VIII (Figures 4 to 6). The cells did not express AML, desmin, melan-A, and S-100 protein. Based on Federation Nationale des Centers de Lutte Contre Le Cancer (FNCLCC) grading, the grade was III for 7 tumors and II for 1 tumor. Radiotherapy was delivered to two patients (31 to 50 Gy). Sequential chemotherapy was administered as a neoadjuvant in 4 patients. The lungs were invaded by cancer in all patients (8/8), followed by the liver (3/8) and bone (1/8). Overall survival at 6 months was estimated to be 25% in a woman.
Clinicopathologic features of our patients diagnosed with breast primary angiosarcoma.

Macroscopic image of a breast tumor.

Mammogram of the left breast, showing an opaque mass of the upper quadrants with blurred contours classified as BIRADS 4.

Histological image showing vascular proliferation made of vessels lined with atypical cells (hematoxylin & eosin; GX20).

Positive immunostaining of tumor cells with Factor VIII.

Positive immunostaining of tumor cells with CD31.

Positive immunostaining of tumor cells with CD34.
Discussion
Primary angiosarcoma of the breast is a rare and aggressive disease, accounting for 0.04% of all malignant breast tumors.2,11 We found 8 cases in 20 years, reflecting the rarity of this cancer. Abdou et al 12 found similar results with 22 cases of mammary angiosarcoma over a period of 25 years. It is a cancer occurring mainly in adult women and young between 30 and 50 years old.7,8 Clinically, the tumor presented as a large mass with irregular contours, associated with ulceration and lymphadenopathy. Although most commonly mimicking adenocarcinoma of the breast, angiosarcoma usually presents as a large mass located in the breast parenchyma.13,14 These were radiologically suspicious lesions in all patients (BI-RADS 4 and 5). Reports on the imaging features of these tumors have been occasional until a recent review by Liberman et al. 15 Diagnostic imaging of these masses includes mammographic and ultrasound examinations; the imaging characteristics of the tumor may be nonspecific and sometimes heterogeneous.8,15 Macroscopically, the tumor has a size that varies between 1 and 20 cm (average of 5 cm). 16 It is poorly defined, spongy when cut and hemorrhagic. Histologically, tumor proliferation is composed of irregular anastomotic vascular cavities, lined with one or more layers of endothelial cells. 17 The diagnostic difficulty arises for the pathologist at both ends of the spectrum. Grade I angiosarcoma can be mistaken for a hemangioma. Grade III angiosarcoma poses the problem of differential diagnosis with poorly differentiated tumors. 18 Therefore, immunohistochemistry is mandatory to establish the definitive diagnosis of angiosarcoma; breast angiosarcomas have the same immunohistochemical profile as those of soft tissues. Endothelial expression markers CD31, CD34, von Willebrand factor (vWF), Ulex europaeus agglutinin 1 (UEA-1), Friend integration 1 leukemia (Fli-1), endothelin-1, vascular endothelial growth factor (VEGFR), and specific erythroblast gene associated (ERG) transformations can help identify angiosarcomas, each with different sensitivity and specificity.4,5,18 All the tumors presented by our patients expressed either focally or diffusely anti CD34, CD31, and anti-factor XVIII antibodies.
Of all breast cancers, angiosarcoma has the worst prognosis. The progression is more or less rapid and death occurs in a table of several metastases after a median survival of 24 months. 19 Mastectomy is the surgical modality of choice for the treatment of this tumor. 20 Lumpectomy should be supplemented by radiotherapy. The postoperative radiotherapy recommended by some authors does not seem to prevent local recurrence in others.3,4 Chemotherapy appears to provide a gain for survival and a decrease in local recurrence and metastasis. 21 In contrast, for hormone therapy, no study has supported its use in treatment.8,21 A new therapy may be offered by trials of biological treatments using anti-growth factor antibodies, in particular anti-vascular endothelial growth factor (VEGF) and anti-fibroblast growth factor (FGF).21,22
Conclusion
Primary mammary angiosarcomas are exceptionally rare malignant tumors with a very poor prognosis. Its clinical, radiological, and histological diagnosis is often difficult. Its prognosis is very poor, and only early surgery can hope for longer survival. The treatment differs in slightly from that of a classic breast carcinoma, and involves mastectomy surgery, without axillary dissection. The role of chemotherapy and radiotherapy remains to be demonstrated and should especially be considered in large tumors of grade III.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors Contributions
T.D. was responsible for the study design, undertook the field study, performed data collection, analysis and interpretation, and wrote the article. T.D.J., B.T., P.S., and M.T. were involved in the treatment and follow-up of the patient; they have reviewed the article. G.N.K. was responsible for the overall scientific management of the study and the preparation of the final article. All the authors have read and approved the final article to be submitted for publication.
Availability of Data and Materials
Extracted data are with the authors and available for sharing on request.
Ethics Approval and Consent to Participate
This study received approval from the head of the laboratory department to be conducted. Since it was counting records, patient consent was not required. However, during the counting and data collection patient names were not collected to preserve confidentiality. This study was approved by the “Comité de Bioéthique pour la Recherche en Santé (CBRS)” (Bioethics Committee for Health Research) from the Togo Ministry of Health, Ref N0: 0101/2016/MS/CAB/DGS/DPLET/CBRS). The study has been carried out in accordance with relevant guidelines and regulations.
