Abstract
Objectives:
Percutaneous core-needle biopsy (PCNB) is the standard of care to biopsy and diagnose suspicious breast lesions. Dependent on histology, many patients require additional open procedures for definitive diagnosis and excision. This study estimated the payer and patient out-of-pocket (OOP) costs, and complication risk, among those requiring at least 1 open procedure following PCNB.
Methods:
This retrospective study used the Truven Commercial database (2009-2014). Women who underwent PCNB, with continuous insurance, and no history of cancer, chemotherapy, radiation, or breast surgery in the prior year were included. Open procedures were defined as open biopsy or lumpectomy. Study follow-up ended at chemotherapy, radiation, mastectomy, or 90 days—whichever occurred first.
Results:
In total, 143 771 patients (mean age 48) met selection criteria; 85.1% underwent isolated PCNB, 12.4% one open procedure, and 2.5% re-excision. Incidence of complications was significantly lower among those with PCNB alone (9.2%) vs 1 open procedure (15.6%) or re-excision (25.3%, P < .001). Mean incremental commercial payments were US $13 190 greater among patients with 1 open procedure vs PCNB alone (US $17 125 vs US $3935, P < .001), and US $4767 greater with re-excision (US $21 892) relative to 1 procedure. Mean patient OOP cost was US $858 greater for 1 open procedure vs PCNB alone (US $1527 vs US $669), and US $247 greater for re-excision vs 1 procedure.
Conclusions:
A meaningful proportion of patients underwent open procedure(s) following PCNB which was associated with increased complication risk and costs to both the payer and the patient. These results suggest a need for technologies to reduce the proportion of cases requiring open surgery and, in some cases, re-excision.
Introduction
Breast cancer is the most common malignancy among women, with nearly 1 in 8 women developing invasive breast cancer during their lifetime. 1 Approximately 252 710 new cases of invasive breast cancer and 63 410 in situ lesions will be diagnosed in the United States in 2017 alone. 1 Conservatively, this incidence of disease translates to more than 183 350 lumpectomy surgeries and represents more than US $455 million in annual procedural spend.2,3
Dependent on the degree of suspicion determined from imaging, family, social, and oncologic risk factors, the workup of imaging abnormalities most often results in diagnostic biopsy. Historically, open surgical biopsy was viewed as the “gold standard” for definitive diagnosis of a suspicious lesion; however, given its invasive nature and the risks associated with missed diagnosis and general anesthesia, percutaneous core-needle biopsy (PCNB) has become the recommended standard of care for pre-malignant and malignant lesions.4–6 Accordingly, the American Society of Breast Surgeons has defined the incidence of PCNB use for first biopsy (vs open biopsy) as a Breast Quality measure. 5
However, despite society endorsement and widespread adoption of PCNB, a significant number of patients still undergo invasive open procedures for definitive diagnosis of high-risk lesions (HRLs) and malignancies.7,8 This is principally due to the partial lesion sampling methodology inherent to the PCNB method, where sequential, incongruent, partially representative cores (akin to “strips”) of tissue are obtained. 9 These cores may or may not include tissue from the region of greatest oncologic concern and have been observed to often result in an upgrade of the diagnosis on subsequent open surgical excision—occurring in 2% to 40% of procedures dependent on lesion type.10–16
Further increasing the procedure and cost burden of diagnosis, prior research has demonstrated re-excision rates of 10% to 60% following initial lumpectomy.17–20 Excluding purely therapeutic indications, repeat surgeries represent an added burden to the patient and health care system and have been shown to result in significant incremental medical expenditures. 15 The burden to commercial payers associated with patients undergoing multiple breast conserving surgery (BCS) procedures has been studied once previously. 15 Metcalfe and colleagues estimated that 23% of patients diagnosed with breast cancer required multiple open BCS procedures, incurring mean incremental all-cause medical payments US $10 821 higher than patients who did not undergo repeat BCS. 15 Although the study provided reasonable and useful estimates of the burden of multiple open procedures, it excluded the large population of patients who undergo PCNB and BCS after diagnosis of an HRL or suspicious lesion.
In the present study, we examined the complications, commercial payer expenditures, and patient out-of-pocket (OOP) cost associated with open biopsy or lumpectomy following PCNB as well as the burden associated with open re-excisions.
Materials and Methods
Dataset
This was a retrospective health care-claims database analysis using 2011-2014 data from the MarketScan Commercial Research Database (Truven Health Analytics, Ann Arbor, MI, USA). This database includes nationally representative information for more than 180 million unique patients covered with private insurance. The insurance claims-based structure of this database does not contain specific information on lesion histology, cancer stage, or estrogen/progesterone receptor status. The database is fully de-identified; therefore, this study did not require Institutional Review Board review.
Patients
Patients were selected based on claims with procedure codes for PCNB, including Current Procedural Terminology (CPT) codes from both before and after 2014—when bundled breast biopsy codes 19081, 19083, and 19085 were introduced. 21 All patients were women and had continuous commercial health insurance through the baseline, hospitalization, and follow-up periods. A 30-day gap in coverage was allowed for plan enrollment/re-enrollment to maximize the sample size for analysis.
The “index date” for analysis was defined as the date of a PCNB procedure performed in an outpatient setting (clinic, office, outpatient hospital, “other” outpatient, or surgical center location). The baseline period was defined as 1 year prior to index and the follow-up period as the day after PCNB through 90 days. Follow-up ended the day before the first identification of chemotherapy, radiation, or mastectomy, or, if none of the previous encounters were identified, at the end of 90 days following PCNB—whichever occurred first. This time-variable follow-up was designed to exclude medical resource use no longer related to diagnosis or BCS.
Patients were excluded from the analysis if there was evidence of previous diagnosis of any cancer, chemotherapy or radiation treatment, any invasive breast surgery, or participation in Medicare Advantage (to ensure completeness of claims data).
We defined an open procedure as either open biopsy or lumpectomy. Similar to methodology by Friese et al, 22 we considered multiple open procedures listed on the same day as a single procedure. In addition, we defined instances where lumpectomy was coded twice during the same visit as 1 procedure, with the assumption that these procedures were bilateral, or an open biopsy upgraded to a lumpectomy.
Three study cohorts were selected for analysis: (1) PCNB alone with no subsequent open procedure during follow-up, (2) PCNB followed by 1 open procedure during follow-up, and (3) PCNB followed by 2 or more open procedures during follow-up (re-excision).
Study measures
Patient clinical history was evaluated with International Classification of Diseases, Ninth Revision (ICD-9) diagnosis codes listed during the baseline, including family history of malignant neoplasm, history of smoking, and diagnosis of obesity. In addition, the Charlson Comorbidity Index (CCI) score was calculated for each patient.
Complications evaluated included fat necrosis (defined as diagnosis of fat necrosis of the breast and/or a procedure for debridement); breast cellulitis; mastodynia; hemorrhage, hematoma, or seroma; wound complications (defined as aspiration procedures, incision with drainage, treatment of wound dehiscence, postoperative fistula, or disruption of non-healing surgical wound); postoperative pain; lymphedema; infection (defined as infection and inflammatory reaction due to prosthetic device, implant, or graft; infected postoperative seroma; or other postoperative infection); venous embolism; and pulmonary embolism.
The total breast-related commercial insurer payments for each study cohort were summarized for the initial visit and over the 90-day follow-up. Payments were defined as the amount paid by insurance to medical facilities, excluding pharmaceutical costs. Records with a commercial payment listed as zero or above the 99th percentile were excluded from analyses to address potential coding errors and outliers. In addition, patient OOP costs were summarized. These costs were defined as the sum of patient copayments, coinsurance, and deductibles paid during the initial visit, plus any health care visits with a breast-related diagnosis or procedure through the end of follow-up. Records with an OOP payment listed as zero were included, as some plans may not require patient OOP expenses. Similar to commercial payments, records with an OOP cost above the 99th percentile were excluded from analyses to address potential coding errors and outliers. All payments were inflation-adjusted to 2015 US dollars using the Medical Care component of the US Consumer Price Index.
Data analyses
Sample selection and creation of analytic variables were performed using the Instant Health Data platform (Boston Health Economics, Inc., Waltham, MA, USA); statistical analyses were performed with R, version 3.2.1 (R Foundation for Statistical Computing, Vienna, Austria).
Descriptive analyses were conducted for all study measures and included mean, median, and standard deviations for continuous measures and proportions for binary measures. Statistical significance testing included the chi-square (χ2) test for comparison of categorical variables across all 3 cohorts, the one-way analysis of variance (ANOVA) test for comparison of continuous variables across all 3 cohorts, and the Wilcoxon-Mann-Whitney test for comparison of payments across 2 cohorts (PCNB alone vs 1 open procedure and 1 open procedure vs re-excision). A logistic regression model was used to evaluate factors associated with increased risk of multiple (>1) open procedures following PCNB.
Results
The dataset contained a total of 277 266 unique claims for PCNB recorded between January 1, 2009, and December 31, 2014. Most patients (85.1%) underwent PCNB alone, while 12.4% received lumpectomy or open biopsy following PCNB, and 2.5% underwent 2 or more open procedures following PCNB. Among those undergoing at least 1 lumpectomy procedure, we observed a re-excision rate of 16.9%.
Demographic and clinical characteristics are summarized in Table 1. The mean age of patients ranged from 47 to 54 years. Patients with PCNB alone were significantly younger than those with 1 or more open procedures. Family history of malignant neoplasm was greatest in the PCNB alone cohort (9.7%), followed by 8.3% in the 1 open and 7.9% in the re-excision cohorts (P < .001). History of smoking was similar across cohorts (approximately 4%), while diagnosis of obesity ranged from 6.6% to 7.5% of patients.
Patient demographics and clinical characteristics at index visit.
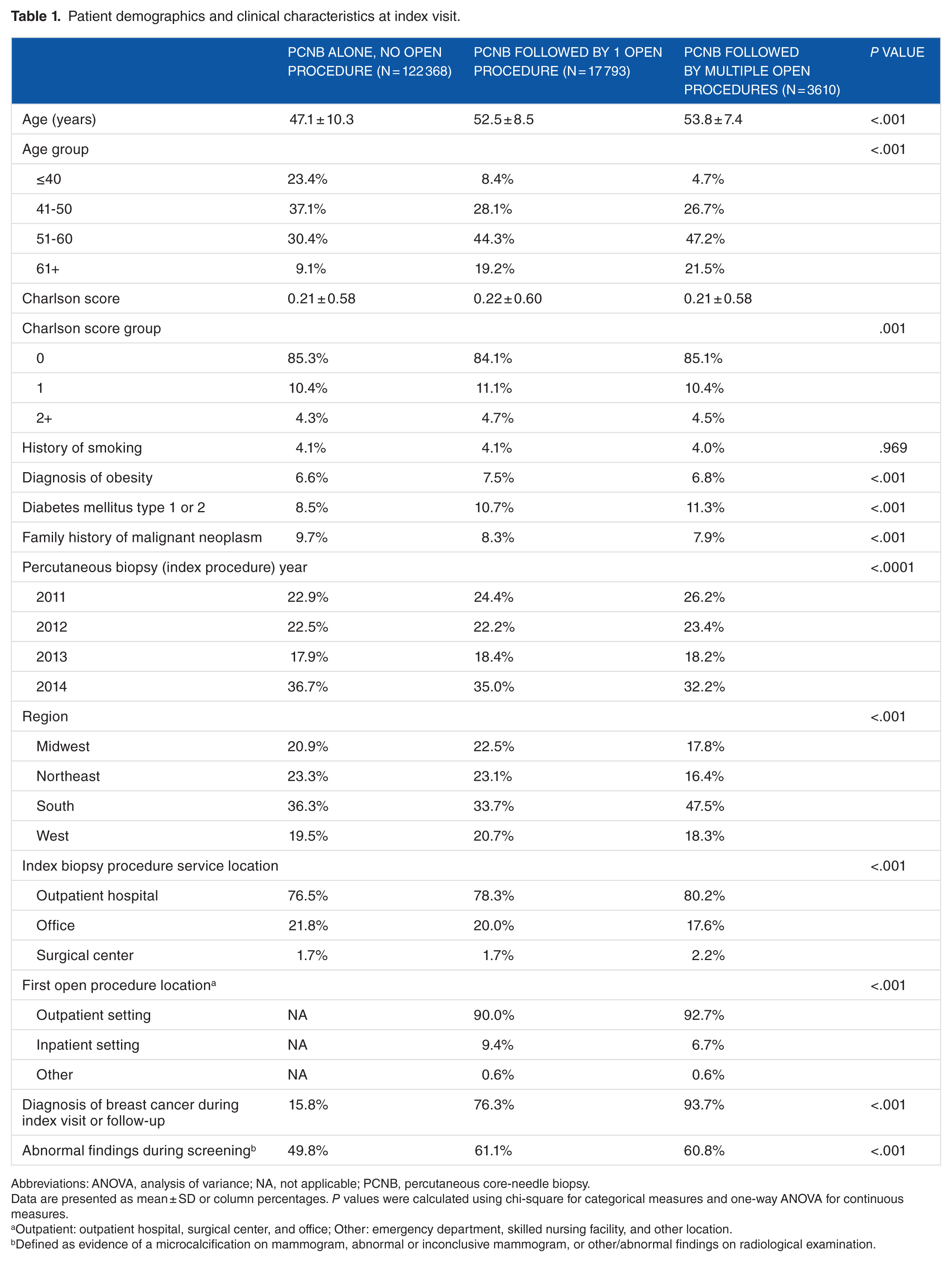
Abbreviations: ANOVA, analysis of variance; NA, not applicable; PCNB, percutaneous core-needle biopsy.
Data are presented as mean ± SD or column percentages. P values were calculated using chi-square for categorical measures and one-way ANOVA for continuous measures.
Outpatient: outpatient hospital, surgical center, and office; Other: emergency department, skilled nursing facility, and other location.
Defined as evidence of a microcalcification on mammogram, abnormal or inconclusive mammogram, or other/abnormal findings on radiological examination.
Most patients in this study underwent initial PCNB in 2014 (approximately one-third of each cohort). Nearly three-quarters of patients in each cohort had the initial PCNB performed in an outpatient hospital setting, while approximately one-fifth had their procedure in an office setting and approximately 2% in the inpatient setting (Table 1). Among patients with at least 1 open procedure following PCNB, the majority underwent their first open procedure in an outpatient setting (90.0%-92.7%), with 6.7% to 9.4% in an inpatient setting.
Diagnosis of breast cancer occurred in 15.8% of patients with biopsy alone, 76.3% with 1 open procedure, and 93.7% of those with a re-excision (P < .001). A claim with diagnosis of “evidence of abnormal findings during screening” ranged from 49.8% among patients with PCNB alone to 61.1% among patients with 1 open procedure.
Complications
Incidence of any complication was significantly lower among those with PCNB (9.2%) vs those with 1 open procedure (15.6%), or patients undergoing re-excision (25.3%; P < .001)—with significance remaining when comparing the incidence of complication among the 1 open vs re-excision cohorts (P < .001). Collectively, the most common complication was fat necrosis (Table 2); patients who underwent multiple open procedures had a greater incidence of fat necrosis compared with those who had 1 open procedure (10.6% vs 3.9%). The second and third most frequent complications were breast cellulitis (up to 4.8%) and hemorrhage, hematoma, or seroma (up to 8.8%), respectively.
Complications by diagnosis pathway: initial biopsy visit through 90-day follow-up.

Abbreviation: PCNB, percutaneous core-needle biopsy.
Any necrosis defined as diagnosis of fat necrosis of the breast and/or procedure for debridement.
Wound complications include aspiration procedure, incision with drainage, treatment of wound dehiscence, postoperative fistula, and disruption or non-healing surgical wound.
Infection defined as infection and inflammatory reaction due to other internal prosthetic device, implant, and graft; infected postoperative seroma; or other postoperative infection.
Payments
The incremental mean breast-related commercial insurance payment associated with undergoing 1 open procedure vs PCNB alone was US $13 190 (US $17 046 vs US $3935) and the incremental cost for re-excision vs 1 open procedure was US $4767 (US $21 892 vs US $17 046; Table 3). Overall, 87% of patients with PCNB alone incurred OOP costs, 97% among patients with 1 open procedure and 98% among those with re-excision. Mean patient OOP costs (including copayments, coinsurance, and deductible amounts) associated with undergoing PCNB were US $669, increasing to US $1527 among patients with 1 open procedure and US $1775 among those who underwent re-excision.
Medical payments by diagnosis pathway: initial biopsy visit through 90-day follow-up.

Abbreviations: OOP, out-of-pocket; PCNB, percutaneous core-needle biopsy.
Statistical significance between PCNB and 1 open procedure.
Statistical significance between 1 open procedure and multiple open procedures.
Total OOP cost includes copayments, coinsurance, and deductibles.
Logistic regression
Factors significantly associated with risk of undergoing re-excision included ages 41 to 50 (relative to aged 40 or younger), residence in South or West regions (relative to the Midwest), diagnosis of breast cancer during the study period, any complication occurring during the study period, and initial PCNB performed in a surgery center or outpatient hospital setting compared with a physician office setting (Table 4). Patients who underwent their first open procedure (either open biopsy or lumpectomy) in an outpatient setting (either hospital outpatient department, surgical center, or office) were significantly more likely to undergo re-excision compared with those who had their first open procedure in an inpatient setting.
Logistic regression model predicting risk of multiple open procedures (>1) compared with PCNB alone and 1 open procedure.

Abbreviations: CI, confidence interval; PCNB, percutaneous core-needle biopsy.
Defined as other, emergency department, or skilled nursing facility.
Discussion
The present results demonstrate that of the 14.9% patients advancing to an open procedure from PCNB, 16.9% underwent re-excision (defined as multiple open biopsy or lumpectomy procedures performed on separate days). Risk of re-excision varied substantially by age group, diagnosis, and first procedure location. Within this subpopulation undergoing re-excision, 7% of patients were not ultimately diagnosed with breast cancer. Overall, 15.8% of patients with PCNB alone, 76.3% with 1 open procedure, and 93.7% with re-excision were diagnosed with breast cancer. These findings suggest that a meaningful percentage of patients may undergo invasive procedures for diagnostic purposes rather than for treatment alone, which has patient and payer burden implications.
The mean incremental cost to a payer associated with a patient requiring a single open procedure (vs none) was US $13 190, with an additional incremental cost associated with re-excision of US $4767. These payments are diagnosis-related only and do not account for patients who advanced to mastectomy or additional treatment. Not surprisingly, the findings from Metcalfe et al 15 showed an incremental payment associated with repeat BCS of nearly double that observed in our study (US $10 821) due to the broader time period (2 years following initial BCS) and the study population (limited to patients with cancer diagnosis).
In addition, we examined the total OOP cost to the patient by treatment pathway. Patients who required 1 open procedure vs PCNB alone paid an average 2.3 times the OOP cost (US $1527 vs US $665); comparatively, patients who underwent re-excision paid 1.2 times the OOP cost relative to 1 open procedure (US $1775 vs US $1527). Historically, patient OOP costs have been well studied in breast care; however, prior research focused on costs related to initial genetic or mammography screening, or costs incurred following formal diagnosis of breast cancer.23–28 Unfortunately, patients are not consistently informed about the OOP cost associated with various treatment choices, as evidenced by a survey of more than 600 outpatient encounters for breast cancer management where only 16% of visits included a conversation about OOP cost. 29 When examining the indirect burden to patients of open lumpectomy procedures, an analysis of the Surveillance Epidemiology and End Results (SEER) registries found that among 608 women undergoing lumpectomy, nearly 1 in 5 (17%) missed greater than 1 month of work. 30 This finding was limited in that it did not control for receipt of chemotherapy or radiation; however, the results do suggest there is meaningful lost work time associated with a lumpectomy procedure.
Interestingly, an analysis of the Breast Cancer Surveillance Consortium (BCSC) registry showed that among lesions classified as BI-RADS 4 (Breast Imaging and Reporting and Data System), indicative of a suspicious abnormality, HRLs were identified on needle biopsy in only 5% of cases. 14 Although medical advances realized with the advent of PCNB allow more women to avoid unnecessary open biopsy or lumpectomy procedures, this improvement does not directly translate to women diagnosed with HRLs. Care for these patients often still includes open excision to definitively rule out malignancy, with nearly 53% of HRLs in the BCSC registry analysis undergoing open excision. 14 Comparing this result with our findings on the incremental cost to the payer of performing a lumpectomy vs biopsy alone (US $13 190), and the patient OOP cost (US $858) and missed work, suggests there are large cost savings to be realized if the initial lumpectomy could be avoided altogether. Such improvements may be possible with a technological advance enabling larger and more complete biopsy specimens. 31 This is not to say all re-excisions could be avoided (as this is dependent on clear margin status), or that all lumpectomies could convert to percutaneous excision, but rather there is an opportunity for cost savings via definitive lesion removal or improved biopsy samples at the time of first intervention.
Study limitations
This dataset was limited to medical claims and did not contain specific cancer-related details. First, we were unable to explore the influence of genetic mutations, receptor status, specific cancer type, palpability of the lesion, or stage of disease on the reason for undergoing PCNB alone vs 1 or multiple open procedures. Second, we did not evaluate the confounding factors of surgeon procedure volume (a rough surrogate measure of procedure skill and potential risk of negative margins), facility volume (potentially correlated with a population skewed toward cancerous diagnoses via referrals), use of adjunctive technology (such as ultrasound, specimen magnetic resonance imaging (MRI) or X-ray analysis, frozen cavity margins, etc), or specific facility type (academic, community, or private surgery center).
Conclusions
Although a majority of suspicious lesions identified on imaging are diagnosed by PCNB and do not require further surgical intervention, there continues to be a sizable percentage of women that require open biopsy or lumpectomy for diagnosis. Our results show that 23.7% of patients with 1 open procedure and 6.3% of those who underwent re-excision were not ultimately diagnosed with cancer. Although acknowledging that many of these patients may require surgical treatment irrespective of diagnosis due to medical history, family history, genetic factors, or other reasons, our results suggest there remains a population who may be better treated via percutaneous methods alone. Advances in biopsy diagnostic accuracy at the time of the first procedure could result in significant patient benefits and cost savings.
Footnotes
Acknowledgements
We thank Jeanne McAdara, PhD, for professional assistance with scientific editing and manuscript preparation.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
All authors are full-time employees of Medtronic Transformative Solutions.
Author Contributions
All authors gave substantial contributions to conception and design of the article. CK and CN performed the analysis of the data. All authors participated in drafting and revising the article for important intellectual content and approved the final draft.
