Abstract
Objectives:
People with substance use disorders (SUD) are suggested to have higher risk of hospitalization, intubation, or death from coronavirus disease 2019 (COVID-19), although data are mixed. Little is known about other COVID-19-related complications in this group. We compared morbidity and mortality among individuals with and without SUD who were admitted to an urban safety net hospital with COVID-19 early in the pandemic, contemporaneous to other published studies on this subject.
Methods:
We performed a retrospective study of patients ⩾18 years old admitted with COVID-19 from March 16th to April 8th, 2020. SUD included alcohol, opioid, cocaine, amphetamine, and benzodiazepine use disorders and was identified using diagnostic codes, free text clinical documentation, and urine drug screens. The primary outcome was inpatient mortality. Secondary outcomes included clinical complications (eg, secondary infections, venous thromboembolism) and resource utilization (eg, mechanical ventilation, length of stay). We used multivariable regression to assess the relationship between SUD and mortality.
Results:
Of 409 patients, the mean age was 56 years and 13.7% had SUD. Those with SUD were more likely to be male, have experienced homelessness, have pulmonary disease or hepatitis C, or use tobacco or cannabis. After multivariable analysis, SUD was not associated with mortality (aOR 1.03; 95% CI, 0.31-3.10). Secondary outcomes were also similar between groups.
Conclusions:
Our findings suggest that persons with and without SUD have similar COVID-19-related outcomes. Previously reported increased COVID-19 complications may be from medical comorbidities.
Introduction
The coronavirus disease 2019 (COVID-19) pandemic has highlighted the unique health disparities that have long impacted people who use drugs.1,2 Persons with substance use disorders (SUD) experience numerous barriers to care, such as difficulty engaging with traditional care models, financial insecurity, unstable housing, and social stigmas.3,4 These barriers were further compounded by the association between lockdowns and increased substance use, especially in those with a preexisting history of SUD.5,6
Multiple large cohort studies from early in the pandemic have shown higher rates of hospitalization, intubation, and death from COVID-19 in those with SUD.7-10 Alternatively, 2 large studies from the Veterans Health Administration found no associations between SUD and COVID-19-related mortality,11,12 and another cohort of 5107 people with SUD showed mixed results depending on substance use pattern. 13 Given these conflicting data, the Centers for Disease Control and Prevention has classified persons with SUD as suggestive of higher risk for severe COVID-19. 14
Moreover, little is known about the incidence of other COVID-associated clinical complications (eg, venous thromboembolism, bacterial superinfections, renal failure requiring dialysis) among persons with SUD, nor of the relationship between SUD and COVID-19-related resource utilization (eg, duration of hospitalization and intensive care unit [ICU] stay). Such factors could influence how clinicians anticipate the healthcare needs and likelihood of clinical decompensation of people with SUD.
It is critical to better understand how SUD impacts COVID-19 disease severity. Safety net hospitals like Boston Medical Center (BMC) treat patients regardless of ability to pay and therefore primarily serve under-resourced communities. They also treat a large proportion of patients with SUD and offer unique insights into the effects of the COVID-19 pandemic on these individuals.
The goal of the current study is to assess the association between SUD and inpatient COVID-19-related mortality, as data published thus far have been mixed. Additionally, we examined the clinical complications and healthcare utilization of hospitalized patients with COVID-19 and SUD as compared to those without SUD. Using clinical records from BMC, we built on prior studies by providing more granularity regarding COVID-19 presentation, such as clinical severity on admission as estimated by oxygenation status, as well as COVID-19-related complications. Given that prior studies have been limited by the reduced sensitivity of diagnostic codes to identify SUD,7-12,15,16 we conducted more detailed manual data abstraction to better identify patients with SUD. We used a cohort contemporaneous to prior studies that had been done early in the pandemic, which allows our findings to be interpreted in the same context as the Centers for Disease Control and Prevention risk stratification.
Materials and Methods
Setting, study population, and inclusion criteria
We created a retrospective cohort of patients with COVID-19 admitted to BMC during the first month of the initial surge in Massachusetts. With approximately 500 beds, BMC is the largest safety net hospital in New England and provides care to a racially and ethnically diverse and low-income patient population. 17 Moreover, BMC serves a large proportion of patients with SUD and associated morbidities, such as hepatitis C virus (HCV) and human immunodeficiency virus (HIV).18,19 In addition, BMC provides care to communities in Boston with the highest COVID-19 disease burden, including low-income areas and locations with predominantly Black and Latino individuals. 20
We included patients at least 18 years old and admitted between March 16th, 2020, and April 8th, 2020, for symptoms consistent with COVID-19 with a positive reverse-transcriptase polymerase chain reaction test (nasopharyngeal or tracheal aspirate) for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). We excluded patients who contracted COVID-19 during their hospital stay or who were admitted for reasons unrelated to COVID-19 and then were incidentally found to be SARS-CoV-2 positive. March 16th was the admission date of the first COVID-19 patient with positive nucleic acid testing at BMC. April 8th was used as an end date due to revision of BMC’s COVID-19 treatment protocol.
Data collection
We manually abstracted data on demographics, medical comorbidities, substance use histories, symptoms, and clinical course from the electronic medical record using International Classification of Disease, 10th Revision (ICD-10) codes and clinician documentation. We followed patients until discharge, transfer to another facility, or death.
We estimated baseline clinical severity by oxygenation status on admission. As routine blood gases were not collected on admission, we calculated oxygenation as the ratio of peripheral capillary oxygen saturation to fraction of inspired oxygen (SpO2/FiO2), which prior literature has established to approximate partial pressure of oxygen (PaO2) to FiO2 ratios. 21 FiO2 was estimated based on the EPIC II Study “Estimating FiO2” table for patients who were not mechanically ventilated or managed on non-invasive positive pressure ventilation. 22 The Boston University Medical Center Institutional Review Board approved this study.
Exposures
Evidence of lifetime SUD was the exposure of interest and was obtained via review of ICD-10 diagnosis codes listed on patient problem lists at the time of COVID-19 diagnosis (Supplemental Table 1). To increase the sensitivity of SUD identification, we additionally reviewed free text clinical documentation and urine drug screen to assess for SUD or heavy substance use suggestive of SUD that was not documented by ICD-10 codes. For these additional reviews, we included current or past alcohol intake consistent with heavy alcohol use or alcohol use disorder as defined by the National Institute on Alcohol Abuse and Alcoholism 23 ; cocaine use; or non-prescription opioid, benzodiazepine, or amphetamine use. Prescribed opioid, benzodiazepine, and amphetamine use was classified as SUD if there was documentation of concern for a use disorder in the electronic medical record. We did not include cannabis use disorder in our definition of SUD because of substantial under-detection of cannabis use disorder by ICD-10 codes24-26 and inconsistent language describing cannabis consumption which limits identification of use disorders with free text documentation.26,27 We also did not include other substances such as inhalants and hallucinogens as they were rarely reported in the charts.
Use of immunomodulatory agents as COVID-19 directed therapy was the secondary exposure. During the spring of 2020, data were limited on COVID-19 treatment options. BMC developed a standardized and closely monitored protocol for administering off-label immunomodulatory therapy to decompensating patients based on degree of hypoxemia and changes in blood inflammatory markers. Clinicians used either interleukin-6 receptor inhibitors (IL-6 receptor inhibitors; tocilizumab or sarilumab) or an interleukin-1 receptor inhibitor (IL-1 receptor inhibitor; anakinra). 28 IL-6 receptor inhibitors have since been added to national treatment guidelines for patients with severe progressive COVID-19 disease after multiple observational studies and clinical trials. 29 Most patients with COVID-19 were also prescribed hydroxychloroquine and azithromycin, although these were removed from the protocol in April 2020 shortly after the period we are evaluating, as newer studies pointed away from their efficacy. Details of the BMC treatment algorithm are discussed more extensively in a prior study. 28
Primary and secondary outcomes
Our primary outcome was inpatient mortality in patients with COVID-19. Secondary outcomes included inpatient clinical complications (secondary bacterial infections, renal failure requiring dialysis, venous thromboembolism, stroke, hepatitis, myocardial infarct, multisystem organ failure, and a composite outcome “any complication” including the presence of at least one of any of the clinical complications) and resource utilization (mechanical ventilation, ICU admission, hospital and ICU length of stay, and time to ICU admission and intubation). These outcomes were identified by manual chart abstraction. In addition, we evaluated factors associated with IL-6 receptor inhibitor administration given BMC’s frequent use of these medications that may have influenced likelihood of clinical complications.
Statistical analysis
We reported categorical variables as counts and percentages and continuous variables as means and standard deviations for variables that followed a normal distribution. Median and interquartile ranges were reported for continuous variables that did not follow a normal distribution. We evaluated differences between patients with and without SUD using chi-squared tests for categorical variables, Fisher’s exact test for categorical variables with small counts, 2-sided t-tests for continuous variables, and 2-sided Wilcoxon rank sum tests for continuous variables that were non-normal. We included in the multivariable models clinical and demographic variables associated with severe COVID-19 14 as well as variables significant at the .2 level in the bivariate analysis. We dichotomized race into non-White and White for the multivariable models given our sample size and because other studies have shown associations of non-White race with poorer COVID-19 outcomes. 14 Associations from multivariable analyses were expressed by odds ratio (OR) with corresponding confidence intervals and p-values with significance defined at the 0.05 level. Analyses were conducted with R v4.0.3 (R Core Team, 2020). 30
Results
Cohort characteristics
A total of 409 patients were included in the cohort (Figure 1) and 56 (13.7%) met criteria for SUD. Baseline characteristics of the study population are presented in Table 1. Mean age and racial/ethnic backgrounds of the SUD and non-SUD groups were similar. Patients with SUD were more likely to be male (83.9% vs 52.1%), experience homelessness (33.9% vs 4.8%), have chronic pulmonary disease (23.2% vs 5.7%), and have a positive HCV antibody (23.2% vs 1.7%). The most commonly used substances were alcohol (71.4%), cocaine (39.3%), and opioids (32.1%). People with SUD were also more likely to use tobacco (67.9% vs 24.9%) and cannabis (16.1% vs 4.0%). Patients in the SUD group presented to care 2 days earlier after symptom onset than people in the non-SUD group (4.0 vs 6.0 days, P = < .001) (Table 2). Patients without SUD were more likely to present with fever, cough, or diarrhea, but otherwise presenting symptoms were similar (Table 2). Both groups presented with similar baseline oxygenation as measured by SpO2/FiO2 (Table 2).

Study flow.
Characteristics of patients by substance use disorder (SUD) status.

Chi-squared tests for categorical variables, Fisher’s exact test for categorical variables with small cell counts, t-tests for continuous variables, and Wilcoxon rank-sum test for non-normal continuous variables.
Coronary artery disease, congenital heart disease, or heart failure.
Body mass index ⩾30 kg/m²
Chronic obstructive pulmonary disease or restrictive lung disease.
Chronic kidney disease stage 3 or higher, or on dialysis.
Human immunodeficiency virus.
Hepatitis B virus surface antigen.
Hepatitis C virus antibody.
Heavy alcohol use or alcohol use disorder as defined by the National Institute on Alcohol Abuse and Alcoholism.
Symptoms and oxygenation at presentation by substance use disorder (SUD) Status.

Chi-squared tests for categorical variables, Fisher’s exact test for categorical variables with small cell counts, t-tests for continuous variables, and Wilcoxon rank-sum test for non-normal continuous variables.
Ratio of peripheral capillary oxygen saturation to fraction of inspired oxygen, to measure oxygenation on hospital admission.
Outcomes
In the SUD group, 7 people (12.5%) died while admitted for COVID-19 infection, as compared to 30 (8.5%) in the non-SUD group (Table 3). We did not find an association between SUD and our primary outcome of inpatient mortality (aOR 1.03; 95% CI, 0.31-3.10) after multivariable analysis controlling for age, sex, non-White race, baseline oxygenation by SpO2/FiO2, administration of IL-6 receptor inhibitor therapy, cardiac disease, and tobacco use (Table 4). Factors significantly associated with mortality included age (aOR 1.04; 1.01-1.08), baseline oxygenation by SpO2/FiO2 (aOR 0.58; 0.41-0.84), and presence of cardiac disease (aOR 2.55; 1.00-6.54).
Patient outcomes by substance use disorder (SUD) Status.

Chi-squared tests for categorical variables, Fisher’s exact test for categorical variables with small cell counts, t-tests for continuous variables, and Wilcoxon rank-sum test for non-normal continuous variables.
Some patients received more than one immunomodulatory agent.
STEMI = ST segment elevation myocardial infarction and NSTEMI = non-ST segment elevation myocardial infarction.
Intensive care unit.
Multivariable logistic regression for the outcome of death.

Ratio of peripheral capillary oxygen saturation to fraction of inspired oxygen, to measure oxygenation on hospital admission in units of 1 standard deviation.
Interleukin-6 inhibitor: use of tocilizumab or sarilumab as COVID-19 directed therapy per hospital protocol.
Coronary artery disease, congenital heart disease, or heart failure.
We did not find any associations between SUD and clinical secondary outcomes in the bivariate analysis, including the composite index of “any complications” (Table 3). Resource utilization secondary outcomes were also similar in the bivariate analysis (Table 3).
COVID-19 directed therapies were similar between groups in the bivariate model, although there was a trend toward increased treatment with tocilizumab in the non-SUD group (P = .052; Table 3). We then evaluated factors associated with receiving an IL-6 receptor inhibitor (tocilizumab or sarilumab) for COVID-19 directed treatment per the BMC hospital protocol, using multivariable regression adjusting for age, sex, non-White race, baseline oxygenation by SpO2/FiO2, obesity, mechanical ventilation, fever, tobacco use, and hospital length of stay. In this model, there was no association between SUD and treatment with an IL-6 receptor inhibitor (aOR 0.45; 0.17-1.09; Table 5). Factors significantly associated with treatment with an IL-6 receptor inhibitor included male sex (aOR 1.78; 1.02-3.15), non-White race (aOR 2.71; 1.16-7.13), obesity (aOR 2.26; 1.26-4.13), fever (aOR 3.82; 1.87-8.50), tobacco use (aOR 0.51; 0.26-0.97), and hospital length of stay (aOR 1.06; 1.02-1.10).
Multivariable logistic regression for the outcome of treatment with IL-6 receptor inhibitor. a
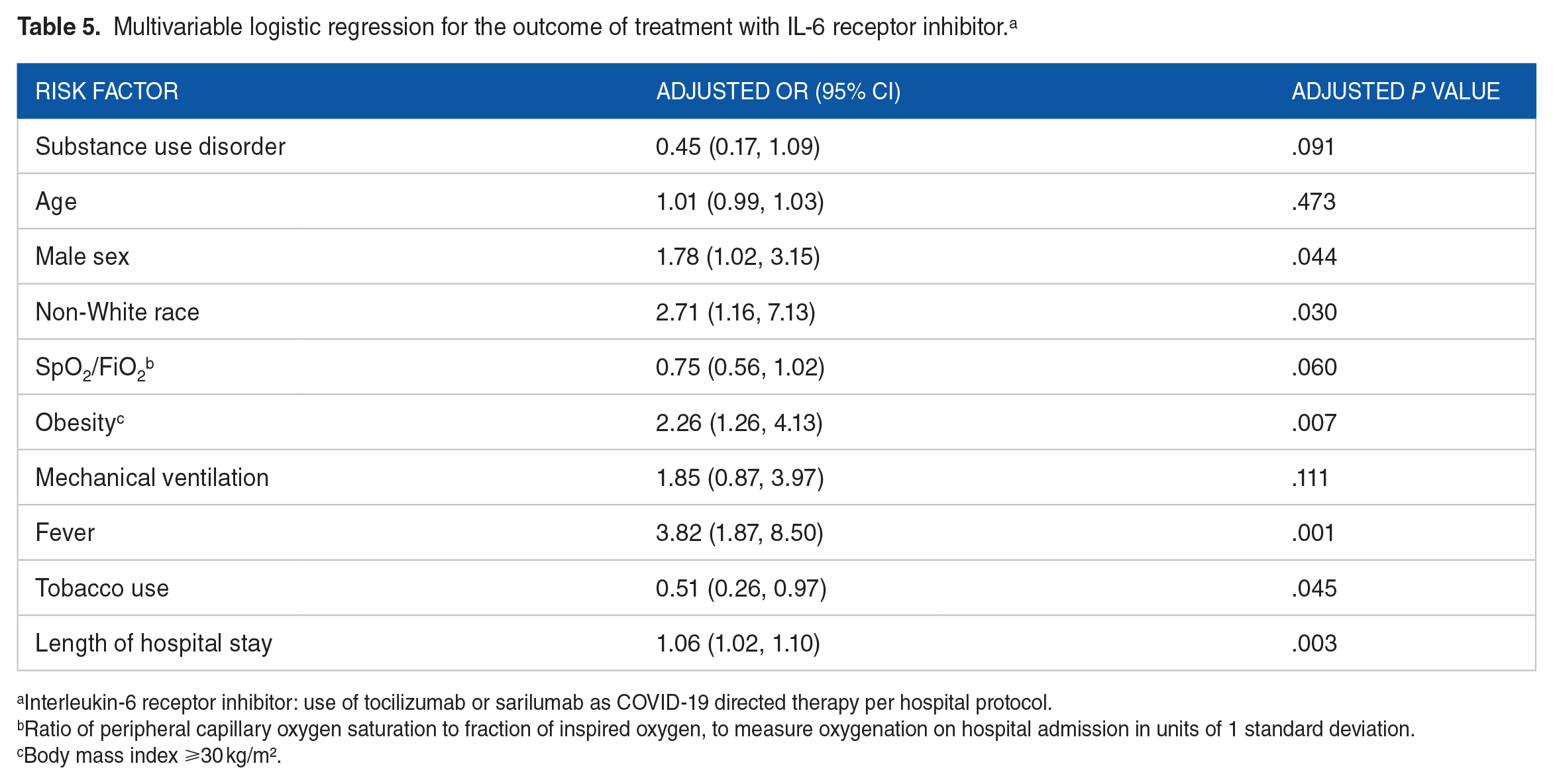
Interleukin-6 receptor inhibitor: use of tocilizumab or sarilumab as COVID-19 directed therapy per hospital protocol.
Ratio of peripheral capillary oxygen saturation to fraction of inspired oxygen, to measure oxygenation on hospital admission in units of 1 standard deviation.
Body mass index ⩾30 kg/m².
Discussion
In this retrospective cohort study of patients admitted to a safety net hospital during the early phase of the COVID-19 pandemic, SUD was not associated with the primary outcome of COVID-19-associated inpatient mortality. The secondary analysis showed that those with and without SUD had similar COVID-19-related clinical complications, including secondary infections, renal failure requiring dialysis, acute liver injury, venous thromboembolism, cardiac complications, and the composite “any complications.” Of note, some clinical outcomes such as stroke were very uncommon overall. Likewise, there was no difference in resource utilization secondary outcomes between the 2 groups. In contrast to other studies,7,10 we found similar likelihood of mechanical ventilation and ICU admission in patients with and without SUD. Although patients with SUD presented to the hospital earlier in their disease course, their total hospital length of stay was ultimately similar to patients without SUD. Insights such as these into the clinical complications and resource utilization patterns of patients with SUD and COVID-19 can help clinicians anticipate the trajectory of infection and healthcare needs in this vulnerable group.
As BMC sees a high proportion of patients who use substances, it was an apt location for the study: almost 14% of the study population had SUD, exceeding the national average of 10.8% in people ages 18 or older. 31 We also present findings on complications from COVID-19 in people with SUD beyond hospitalization, intubation, and death. The observed similarity in complication rates is particularly important given BMC’s diverse patient population that is largely composed of groups disproportionately impacted by COVID-19, namely Black and Latino populations. 32 This study also evaluated for duration of symptoms before hospital presentation, which is not often included as a variable in observational studies of COVID-19 despite the importance of early treatment for severe disease.28,33 The timing early in the pandemic provides a more complete picture of the natural history of the interaction of substance use and COVID-19, as many now-standard inpatient therapies like corticosteroids and monoclonal antibodies were not yet part of routine care. 29
The similarity in complications between those with and without SUD run counter to some published works that have shown worse outcomes in those with SUD7-10 and led to the classification of those with SUD as “suggestive higher risk for severe COVID-19 outcomes” by the Centers for Disease Control and Prevention. 14 Some hypotheses have been proposed to explain worse outcomes seen in other cohorts of people with SUD. One United States-based study found higher rates of cardiovascular, renal, liver, and pulmonary disease in patients with SUD, which may in part explain the higher rates of morbidity and mortality. 8 In line with these findings and other known COVID-19 risk factors, 14 underlying cardiac disease, increased age, and lower oxygenation on admission were associated with mortality in our cohort. Similarly, a database review of approximately 5550 people with COVID-19 showed increased rates of hospitalization, intubation, and mortality in those with a SUD diagnosis but also noted that the increased mortality in this group was attenuated after controlling for hypertension, chronic obstructive pulmonary disease, ischemic heart disease, and cerebrovascular disease. 7 In our cohort those with SUD had baseline higher rates of pulmonary disease but similar rates of other comorbidities known to contribute to COVID-19 mortality, such as obesity, diabetes, and cardiovascular disease.34,35 However, chronic pulmonary disease was not significantly associated with mortality in our cohort and therefore were not included as a covariate in the final model. As our cohort had otherwise similar baseline comorbid conditions and similar outcomes in those with and without SUD, the previously reported increased COVID-19 complications in patients with SUD may in fact be secondary to increased rates of underlying medical conditions in this population.
We considered that our outcomes may have differed from those in other studies because of BMC’s regular use of IL-6 receptor inhibitors as COVID-19-directed therapy, which was uncommon early in the pandemic but is now recommended for severe progressive COVID-19. 29 Thus we conducted a secondary analysis of factors associated with receipt of IL-6 receptor inhibitors. We found that having SUD was not associated with increased administration of IL-6 receptor inhibitors, suggesting that the similar outcomes in those with and without SUD were not strongly impacted by use of this therapy. Although IL-6 receptor inhibitor therapy may be most effective when given to severely but not critically ill patients with COVID-19,28,29 the SUD group’s earlier hospital presentation after symptom onset did not affect odds of IL-6 receptor inhibitor administration or of mortality.
Another important factor to note is that as a safety net hospital, Boston Medical Center has an established program to assess the social needs of patients and to link individuals with appropriate resources within the medical center and throughout the community. 36 There are also specific programs targeting individuals with SUD. These programs might explain some of the findings from the current study which shows that patients with SUD had similar outcomes when compared to individuals without SUD. The important role of social determinants of health has been demonstrated in the literature. Studies have shown that social determinants of health, such as education, employment, income, social support, and community safety, influence 47% of health outcomes while health behaviors, clinical care, and physical environment are estimated to account for 34%, 16%, and 3%, respectively. 37
There are some notable limitations to our study. We are presenting results from a single site, which might limit generalizability of our findings despite the racial and ethnic diversity of the BMC patient population. Second, the data presented are from the earliest phase of COVID-19 in the United States, so trends may have differed with subsequent waves and as COVID-19 management strategies have evolved over time. Because of this timing, however, we were able to evaluate more granular clinical outcomes within the same time period of the studies used to determine COVID-19 risk categories. 14 Third, we did not specifically control for socioeconomic factors like medical insurance status or income level, as over 75% of the BMC patient population has public payer insurance (Medicare, Medicaid, or Children’s Health Insurance Program) or no insurance.17,37 Lastly, we were not powered to detect differences in COVID-19 outcomes between current versus past SUD. This area would benefit from further research.
In conclusion, in this study of hospitalized individuals at an urban safety net hospital with a diverse patient population in the early days of the COVID-19 pandemic, inpatient mortality and morbidity between patients with and without SUD were similar. Our findings provide a detailed evaluation of outcomes in a unique patient population that has been disproportionately impacted by COVID-19 and may provide beneficial insights for similar settings across the country. These results point away from SUD as an independent risk factor for severe COVID-19 and further suggests a focus on medical comorbidities to mitigate the effects of COVID-19. Additional studies are needed to further evaluate for differential outcomes in this high-risk population, particularly in an era of newer COVID-19-directed therapies.
Supplemental Material
sj-docx-1-sat-10.1177_11782218231160014 – Supplemental material for Comparing COVID-19-related Morbidity and Mortality Between Patients With and Without Substance Use Disorders: A Retrospective Cohort Study
Supplemental material, sj-docx-1-sat-10.1177_11782218231160014 for Comparing COVID-19-related Morbidity and Mortality Between Patients With and Without Substance Use Disorders: A Retrospective Cohort Study by Angela McLaughlin, Rebecca Burns, Morgan Ryan, Wafaa Abbasi, Leah Harvey, Jacqueline Hicks, Pranay Sinha and Sabrina A Assoumou in Substance Abuse: Research and Treatment
Footnotes
Acknowledgements
The authors acknowledge the contributions of Megan Davis, Janhavee Deshpande, Kelly Drozdowicz, Lauren Dzera, Nirupa Gadi, Annie Heyman, David Leeds, Nivedita Mandal, Kelvin Noronha, Aneesha Pydi, Alina Razak, Tyler Ryan, Faizah Shareef, Erin Smith, Preetha Velu, Judy Wang, and Sarah Zambrano for participating in chart review and data abstraction.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institute on Drug Abuse under grant number K23DA044085 to S.A.A. and R25DA033211 to L.H.; the BU-CHART Fellowship, National Institute of Allergy and Infectious Disease under grant number T32AI052074) to A.M., L.H., and P.S.; the Boston University School of Medicine Department of Medicine Evans Career Investment and Evans Junior Faculty Merit Awards to S.A.A.; the Burroughs Wellcome/ASTMH Postdoctoral fellowship to P.S.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Conceptualization: AML, RB, SAA. Methodology: MR, AML, RB, SAA. Interpretation of study data: AML, RB, SAA, PS, LH, WA. Formal analysis: MR, JH. Funding acquisition: SAA. Supervision: SAA. Writing – original draft: AML, RB. All authors were involved in review & editing and had final approval of the submitted manuscript.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
