Abstract
Some systemic rheumatic diseases and disorders, especially fibrotic and vascular disorders, are often refractory to corticosteroid therapy. Recently, ever accumulating evidence suggests that platelet-derived growth factor (PDGF) is involved in those refractory diseases. Imatinib mesylate inhibits the activation of PDGF receptor as well as c-Abl, Bcr-Abl and c-Kit tyrosine kinases. It has therefore been widely used for the treatment of chronic myeloid leukemia and gastrointestinal stromal tumors. Imatinib effectively suppresses the activation and proliferation of fibroblasts, mesangial cells and smooth muscle cells both in vitro and in vivo. Additionally, it has recently been reported that some patients with rheumatoid arthritis or idiopathic pulmonary arterial hypertension demonstrated a good clinical response to imatinib therapy. Imatinib may therefore overcome the limitations of current therapeutic strategy with corticosteroids and immunosuppressive agents for refractory diseases, such as systemic sclerosis and interstitial lung diseases, without clinical intolerability.
Introduction
The developmental process of systemic autoimmune diseases has been an unsolved mystery for decades. Despite the identification of autoreactive T lymphocytes or autoantibodies, both of which may participate in the pathogenesis of the diseases, any precise insight into “immunodistortion” present in each patient is lacking. Therefore, overall immunosuppression, instead of a specific correction of “immunodistortion,” has been a common choice in clinical practice. Corticosteroids and immunosuppressive agents have been the mainstay of the therapeutic strategy of systemic rheumatic diseases. However, the presence of refractory diseases/patients to such approaches, and various adverse effects such as increased risk of severe infections, limits their efficacy.
Recent progress in our general understanding of molecular pathophysiology of diseases possibly downstream of the “immunodistortion” has led us to develop molecular targeted therapies in which the function of the key molecule(s) of the diseases is manipulated in order to control disease activity. We have then an excellent safety profile in terms of an eventual lack of lethal dosage enabled most of the biologic response modifiers (biologics) that can be applied at the dosage with which they sufficiently inhibit the function of the targeted molecule. For example, tumor necrosis factor a (TNFα) inhibitors dramatically improved the outcome of patients with rheumatic diseases such as rheumatoid arthritis (RA) and seronegative spondyloarthropathies [Takeuchi, 2005]. Those agents include a chimeric anti-TNFα monoclonal antibody (infliximab), a humanized anti-TNFá monoclonal antibody (adalimumab), and a p75 TNF-receptor/IgG-Fc fusion protein (etanercept). In addition, a chimeric anti-CD20 monoclonal antibody (rituximab), anti-interleukin-6 receptor monoclonal antibody (tocilizumab), and cytotoxic T-lymphocyte antigen 4 (CTLA-4; CD152)/IgG-Fc fusion protein (abatacept) have been also successful in RA. On the other hand, low molecular weight chemical agents developed as molecular targeted therapies do have some drawbacks compared to biologics in terms of selectivity and limitations in dosage. Nonetheless, their intracellular activity which large-molecular agents such as biologics do not possess, although slightly less selective, is still indispensable in the treatment of diseases in which various cytoplasmic/nuclear molecules play important roles.
In diseases refractory to glucocorticoids and conventional immunosuppressants such as cyclophosphamide—both of which are chiefly targeted to leukocytes—, cells of mesenchymal origin such as fibroblast and vascular cells play a pivotal role. Novel molecular targeted therapies should therefore include growth factors participating in the remodeling of mesenchymal tissues. Among various growth factors, platelet-derived growth factor (PDGF) is an excellent candidate due to its multipotent roles in many rheumatic diseases [östman and Heldin, 2001; Paniagua and Robinson, 2007]. It can be interfered with monoclonal antibodies against PDGF or its receptor (PDGF-R). Another approach in inhibiting PDGF signaling is the application of tyrosine kinase inhibitors of PDGF-R. In contrast to non-selective tyrosine kinase inhibitors such as genestein, imatinib mesylate (STI571; Gleevec® or Glivec®) has been widely used for the treatment of chronic myeloid leukemia (CML) [Druker et al. 2001a; Druker et al. 2001b] and gastrointestinal stromal tumors (GIST) [Demetri et al. 2002]. Therefore, in order to speculate on the clinical importance of PDGF-targeted therapy in refractory diseases and patients, this review article summarizes the involvement of PDGF in systemic autoimmune diseases, as well as preclinical or clinical results of imatinib or other PDGF-targeted therapies.
Signaling from PDGF-R and Its Inhibition by Imatinib
PDGF is a family of homo- or hetero-dimeric molecules of disulfide-bonded polypeptide chains with a conserved sequence of approximately 100 amino acid residues containing a characteristic motif of 8 cystein residues [östman and Heldin, 2001; Heldin and Westermark, 1999; Pietras et al. 2003]. Combinations of subunits AA, AB, BB, CC and DD have been identified to date. PDGF is synthesized by many cell types such as platelets, endothelial cells, fibroblasts, macrophages, and vascular smooth muscle cells. The PDGF-Rs occur as a (~170 kDa) and β (~180 kDa) homodimers or heterodimers. The receptor sequences have 5 immunoglobulin-like domains in their extracellular parts and tyrosine kinase domains intracellularly into which is inserted an interrupting sequence of about 100 amino acids. PDGF-AA binds to PDGF-Rαα, AB and CC to αα and αβ, BB binds to all 3 combinations of receptors, and DD binds to αβ and ββ [Pietras et al. 2003]. The dimerization and activation of receptor tyrosine kinases lead to cellular activation of targeted cells including fibroblasts, smooth muscle cells and mesangial cells. A large number of SH2 domain-containing protein kinases/phosphatases and adaptor proteins, such as phosphatidylinositol 3-kinase (PI3K), phospholipase Cγ (PLCγ), SH2-containing protein tyrosine phosphatase SHP-2, Grb2, Nck and Shc, link PDGF-R with downstream signaling molecules and lead to upregulated gene expression and proliferation, or transformation (Fig. 1) [östman and Heldin, 2001; Heldin and Westermark, 1999].
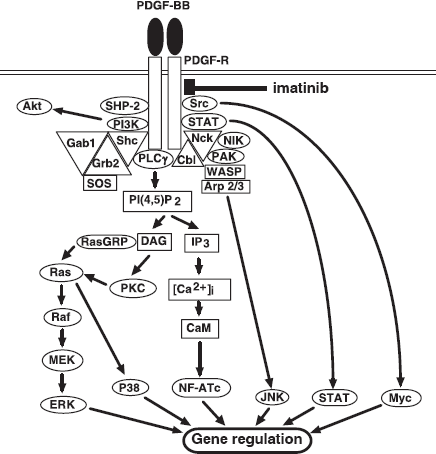
Signaling through the PDGF-R. The binding of PDGF dimer (PDGF-BB, for example) to PDGF-R results in the tyrosine phosphorylation of its cytoplasmic domain, where many adapter proteins and kinases/phosphatases assemble and augment the activation signaling leading to upregulation of gene expressions. Imatinib inhibits the activation of PDGF-R tyrosine kinase.
Originally, imatinib focused on attempts to inhibit PDGF-R kinase with an IC50 of 0.12–0.15 μM. However, it was soon noted that imatinib effectively inhibited c-Abl (IC50 0.1–0.3 μM), Bcr-Abl (IC50 0.25 μM) and c-Kit (IC50 < 1 μM) [Carroll et al. 1997; Druker et al. 1996; Capdeville et al. 2002]. Imatinib was also reported to be well tolerated and have significant antileukemic activity in patients with CML with whom treatment with interferon a had failed, as well as those with blast crisis of CML or Bcr-Abl-positive acute lymphoblastic leukemia [Druker et al. 2001a; Druker et al. 2001b]. Imatinib was later revealed to induce a sustained objective response in more than half of patients with an advanced unresectable or metastatic GIST [Demetri et al. 2002]. Imatinib has now been widely approved for the treatment of CML and unresectable and/or metastatic c-Kit (CD117)-positive GIST.
Systemic Sclerosis
Systemic sclerosis (SSc; scleroderma) is characterized by vascular damage and excessive fibrotic response on the basis of immunologic abnormalities (“immunodistortion”). The progression of organ damage is usually insidious in SSc, with the exception of scleroderma renal crisis which can be successfully treated with angiotensin-converting enzyme inhibitors. Therefore, overall disappointing response to corticosteroids has been attributed to the paucity of involvement of active inflammation, characterized by massive leukocyte infiltration. For this reason, disease-modifying drugs for SSc have not been available to date [Wollheim, 2007].
As for vascular damage, inhibitors targeting endothelin receptors, such as bosentan, represent a breakthrough in the treatment of pulmonary arterial hypertension (PAH), a complication with one of the worst prognosis in SSc, systemic lupus erythematosus (SLE) or mixed connective tissue disease (MCTD) [Denton et al. 2006; Kondo, 2001]. Bosentan may also reduce the fibrotic process via its inhibitory effect on fibroblasts [Braun-Moscovici et al. 2004], although these effects have not yet been proven in clinical trials. At the same time, a recent report describing a stimulatory autoantibody against PDGF-R is of great interest in view of the pathogenesis and management of fibrosis in SSc [Baroni et al. 2006]. Although the determination of autoantibodies recognizing nuclear components have been useful in defining clinical subgroup of SSc, antinuclear antibodies (ANAs) have not been shown to be directly pathogenic. In contrast to this, stimulatory anti-PDGF-R antibodies exclusively found in sera from SSc patients were demonstrated to activate PDGF-R and induce the upregulation of the Ha-Ras-ERK1/2 (external signal regulated kinases 1 and 2) cascade. This stimulated the expression of type I collagen gene expression and α-smooth muscle actin (α-SMA) [Baroni et al. 2006] which resembles the case of anti-thyroid stimulating hormone antibodies developing hyperthyroidism.
Early in 2007, Distler et al. reported that imatinib reduced basal and PDGF- or TGF-β-stimulated synthesis of extracellular matrix (ECM) proteins such as collagen and fibronectin; and that a 50–150 mg/kg/day intraperitoneal administration of imatinib effectively inhibited dermal thickness, the number of myofibroblasts and synthesis of ECM proteins in bleomycin-induced dermal fibrosis model—all without evidence of toxic adverse effects [Distler et al. 2007]. A significant increase in activated PDGF-Rβ expression in idiopathic PAH lungs compared with healthy donor lungs, and successful reversal of pulmonary vascular remodeling, in both monocrotaline-induced rat model and hypoxia-induced mouse model of PAH [Schermuly et al. 2005], led to the compassionate treatment of a patient with idiopathic PAH with a daily administration of 200 mg of oral imatinib [Ghofrani et al. 2005]. The patient's condition improved impressively over 3 months in pulmonary vascular resistance (from 1056 dyn·sec·cm-5 to 815 dyn·sec·cm-5), six-minute walk distance (from 260 m to 383 m), and New York Heart Association functional class (from IV to II) [Ghofrani et al. 2005].
These reports provide the possibility that imatinib may be a disease-modifying drug for SSc. On the other hand, recombinant human anti-TGFβ1 antibody (CAT-192) showed no evidence of efficacy in a multicenter, randomized, placebo-controlled phase I/II trial for early-stage diffuse cutaneous SSc [Denton et al. 2007]. The blockade of multiple isoforms of TGFβ may be required for an effective inhibition of fibrosis. In this context, the pan-isoform-specific anti-TGFβ antibody 1D11 could be potentially effective for fibrotic diseases. Nonetheless, imatinib has the advantage of inhibiting both PDGF and TGFβ signaling via PDGF-R and c-Abl, respectively. Interstitial lung disease (ILD) in SSc will be discussed in the following section.
ILD in Systemic Rheumatic Diseases
The lung is one of the vital organs involved most frequently in systemic rheumatic diseases, and the presence of ILD is a significant prognostic factor of polymyositis/dermatomyositis (PM/DM), rheumatoid arthritis (RA), systemic vasculitis syndromes and SSc. The clinical courses of patients with ILD associated with those diseases can be categorized into 4 groups: 1) A/SIP with rapid deterioration within a month (acute) or within 2–3 months (subacute); 2) chronic progression of pulmonary fibrosis causing non-productive coughing, breath-shortening upon exertion, and occasionally leading to respiratory failure after more than 6 months; 3) acute or subacute exacerbation of chronic ILD that is recurrent in some cases; and 4) asymptomatic ILD detected in a milder form by radiographic examination or pulmonary function tests in the absence of clinically apparent signs and symptoms throughout the observation period [Kameda and Takeuchi, 2006]. The American Thoracic Society/European Respiratory Society (ATS/ERS) international multidisciplinary consensus classification is usually used for the classification of idiopathic interstitial pneumonias (IIP) [American Thoracic Society; European Respiratory Society 2002]. Diffuse alveolar damage (DAD) and organizing pneumonia (OP) usually develop acutely or subacutely, while non-specific organizing pneumonia (NSIP) shows a subacute course, and usual interstitial pneumonia (UIP) represents a chronic form of ILD called pulmonary fibrosis. However, whether ILD associated with systemic rheumatic diseases resembles IIP remains debatable: the histological findings may consist of the overlapping features of two or more patterns, NSIP and OP, or NSIP and UIP. Moreover, patients having idiopathic UIP (clinically idiopathic pulmonary fibrosis) show poorer prognosis compared to patients with systemic rheumatic diseases, such as SSc demonstrating UIP in lung histology [Bouros et al. 2002].
Nevertheless, patients with ILD associated with systemic rheumatic diseases also tend to show a favorable response to corticosteroids when the lung histology reveals OP or cellular NSIP, while they demonstrate a relatively poor response when the lung histology shows DAD or UIP; fibrotic NSIP stands for an intermediate response. The addition of a single immunosuppressive agent or the combination of 2 or more improved the mortality and morbidity of rapidly or slowly progressive ILD associated with PM/DM or SSc [White et al. 2000; Kameda et al. 2005]. Despite the intensified immunosuppressive strategies, a significant number of ILD patients resulted in fatal outcomes. Because the pathological examination of ILD with poor prognosis, DAD and UIP, shares an excessive fibrosis, another therapeutic strategy such as anti-fibrotic agents is necessary. Pirfenidone, N-acetylcysteine and interferon-γ have been shown to be potentially effective for the prevention of lung fibrosis [American Thoracic Society 2000].
At the end of the last century, it was reported that the inhibition of the autophosporylation of PDGF-R by AG1296 effectively prevented V2O5-stimulated proliferation of alveolar epithelial/mesenchymal cells as well as hydroxyproline accumulation [Rice et al. 1999]. In that model, a specific inhibition of epidermal growth factor (EGF) receptor activation by AG1478 also showed a preventive efficacy, although slightly less compared to AG1296. Another approach using an expression plasmid of the extracellular domain of PDGF-Rβ for bleomycin-treated C57BL/6 mice also ameliorated the increases in the wet weight, hydroxyproline content and the histologic changes in the lung [Yoshida et al. 1999].
Daniel et al. proved that imatinib dramatically reduced hydroxyproline content and prevented histopathologic changes in bleomycin-treated lungs of 129tsvems mice [Daniel et al. 2004]. They also suggested that the efficacy of imatinib is mediated, in part, by the inhibition of serine/threonine TGF-β receptor kinase signaling through c-Abl tyrosine kinase. Aono et al. also showed that imatinib (50 mg/kg) significantly attenuated bleomycin-induced pulmonary fibrosis in terms of histology and collagen deposition in C57BL/6 mice. Interestingly, the early treatment with imatinib (from day 0 to 14), but not the late treatment (from day 15 to 28) significantly inhibited bleomycin-induced lung fibrosis in that model [Aono et al. 2005]. However, another bleomycin-induced lung fibrosis model using rats showed that imatinib (50 mg/kg) treatment commenced at Day 10 was still effective, while prednisolone was effective exclusively in the preventive model started at Day 1 [Chaudhary et al. 2006]. In the radiation (20 Gy)-induced lung fibrosis model using C57BL/6 mice, treatment with either of 3 PDGF-R inhibitors (SU9518, SU11657 and imatinib) markedly attenuated the development of pulmonary fibrosis in excellent correlation with clinical, histological, CT results, and life span. In addition, no decline in their efficacy was observed when they were started after thoracic irradiation compared to a prevention schedule [Abdollahi et al. 2005]. These results may provide a possibility that imatinib could be chosen for the treatment of corticosteroid(/immunosuppresants)-refractory ILD. In contrast to the above, a recent report failed to demonstrate the efficacy of imatinib (10 mg/kg, intraperitoneal or oral administration) in the protection against bleomycin-induced pulmonary fibrosis in C57BL/6 mice [Vittal et al. 2007].
Lupus Nephritis
Systemic lupus erythematosus (SLE) is a prototype of systemic autoimmune diseases and its renal involvement has been posing major challenges for physicians. Renal accumulation of immune complexes followed by complement activation, and antibody-effector-cell interactions through the Fc receptor have been implicated in the pathogenesis of lupus nephritis. High-dose corticosteroids combined with immunosuppressive agents such as cyclophosphamide or mycophenolate mofetil are the mainstay in current strategy for the induction of response in lupus nephritis. Some patients however do not respond favorably to the above regimens or show repeated relapses, and ultimately progress to end-stage renal disease.
Renal expression of PDGF, which is synthesized by mesangial cells, endothelial cells, macrophages, and smooth muscle cells in the kidney, and of PDGF-R was significantly increased in the glomeruli of patients with mesangial proliferative glomerulonephritis (IgA nephropathy, Henoch-Schönlein purpura nephritis, and lupus nephritis) compared with normal glomeruli [Matsuda et al. 1997]. Thus, PDGF/PDGF-R axis is considered a novel therapeutic target of lupus nephritis and other forms of glomerulonephritis. Indeed, administration of neutralizing anti-PDGF IgG [Johnson et al. 1992] or oligonucleotide aptamer [Floege et al. 1999], which specifically binds to the PDGF-B chain and inhibits its activity to anti-Thy-1 glomerulonephritis rats resulted in a significant reduction in mesangial cell proliferation and largely prevented the increased deposition of ECM.
Imatinib (0.013–2.0 μM) inhibited PDGF-stimulated, but not FGF-stimulated, mesangial cell proliferation in a dose-dependent fashion in vitro [Gilbert et al. 2001]. Moreover, the efficacy of imatinib in vivo has been proven in 3 different models of (lupus) nephritis. First, PDGF-R tyrosine kinase blockade by imatinib (50 mg/kg/day) was associated with a significant reduction in mesangial cell proliferation, the number of α-SMA-positive mesangial cells, and glomerular type IV collagen deposition in male Wistar rats with anti-Thy-1.1 glomerulonephritis [Gilbert et al. 2001]. Second, 50 mg/kg imatinib inhibited proliferation of glomerular cells and crescent formation, and also prolonged the life span of MRL/lpr female mice [Sadanaga et al. 2005]. Intriguingly, attenuation of lymphadenopathy and salivary gland inflammation, as well as reduction in serum anti-double-stranded DNA antibodies, was also observed in the imatinib-treated mice. Third, (NZB/W)F1 mice treated with imatinib (50 mg/kg b.i.d./day) showed ameliorated survival, delayed onset of proteinuria, and preserved renal function [Zoja et al. 2006]. Histologic examination provided evidence of reduced glomerular hypercellularity, deposits, tubulointerstitial damage, and accumulation of α-SMA-positive myofibroblasts.
Rheumatoid Arthritis
The synovial membrane in patients with RA is characterized by hyperplasia, angiogenesis, and an infiltrate of inflammatory cells including CD4+ T lymphocytes [Choy and Panayi, 2001]. Synovial fibroblast-like (SFL) cells from RA patients show transformed characteristics [Firestein and Zvaifler, 2002; Müller-Ladner et al. 2000; Yamanishi and Firestein, 2001]: altered morphology, anchorage-independent growth [Lafyatis et al. 1989], loss of contact inhibition, oncogene activation [Müller-Ladner et al. 1995], monoclonal or oligoclonal expansion [Imamura et al. 1998], cartilage invasion in severe combined immunodeficient mice [Müller-Ladner et al. 1996], etc.
PDGF-Rs are abundantly expressed on the surface of RA-SFL cells, and stimulation with PDGF enhances both the anchorage-dependent and -independent growth of RA-SFL cells and thus implicate PDGF in the activation and transformation of RA-SFL cells [Lafyatis et al. 1989; Rubin et al. 1998; Remmers et al. 1991; Sano et al. 1993; Watanabe et al.]. Indeed, PDGF immunostaining of RA synovia is more extensive and intense than that of osteoarthritis (OA) or normal synovia. Also, PDGF-R expression is elevated in RA synovia compared with OA and normal synovia [Remmers et al. 1991]. Moreover, PDGF, together with TNFα, were identified as the major growth factors of RA-SFL cells [Thornton et al. 1991]. Furthermore, thrombin activity in synovial fluid is significantly higher in the patients with RA than with OA, and the mitogenic activity of thrombin toward RA-SFL cells is associated with an increase in the expression of mRNA of PDGF-Rs [Ohba et al. 1996].
We demonstrated that 1 μM of imatinib effectively inhibited the PDGF-stimulated tyrosine-phosphorylation of PDGF-R in RA-SFL cells, as well as PDGF-enhanced anchorage-dependent and -independent RA-SFL cell proliferation in vitro [Kameda et al. 2006]. Inhibition of PDGF-R signaling by 1 L M of imatinib did not induce apoptosis in cultured RA-SFL cells [Sandler et al. 2006]. The efficacy of imatinib in vivo has been almost simultaneously reported. Imatinib (33–100 mg/kg) effectively prevented and treated collagen-induced arthritis model of DBA/1 mice in terms of synovitis, pannus formation and joint erosion, although preventive administration was more efficacious than therapeutic administration [Paniagua et al. 2006]. Notably, imatinib inhibited mast cell c-Kit activation, proinflammatory cytokine production, immunoglobulin production from B cells, and T cell response. However, collagen-induced arthritis in Lewis rats were resistant to imatinib treatment: only high-dose (150 mg/kg, not 50 mg/kg) imatinib showed a significant inhibition of osteoclast formation and joint destruction, and failed to reduce paw swelling [Ando et al. 2006].
Although the downstream signaling pathway from PDGF-R in RA-SFL cells has not been clarified, many adaptor proteins are likely to be involved. For example, Gab1 and Gab2 were expressed in RA-SFL cells, and both adaptor proteins were rapidly tyrosine-phosphorylated after the stimulation of RA-SFL cells with 10 ng/ml of PDGF [Kameda et al. 2006]. The fact that the expression of Gab1 lacking the pleckstrin homology domain is associated with the enhanced anchorage-independent growth of Syrian hamster embryo fibroblasts under growth factor stimulation suggests that similar alteration in signaling proteins might be involved in the acquisition of the transformed phenotype of RA-SFL cells [Kameda et al. 2001]. This is supported by the fact that PDGF stimulation enhanced anchorage-independent growth of RA-SFL cells [Kameda et al. 2006].
Recent reports on RA patients successfully treated with imatinib were more encouraging than above in vitro and in vivo results. Miyachi et al. reported a case with RA and chronic myeloid leukemia, both of which were successfully treated using imatinib [Miyachi et al. 2003]. Moreover, Eklund et al. described three patients with severe RA who were treated for 12 weeks with escalating daily doses (from 100 to 400 mg) of imatinib [Eklund and Joensuu, 2003]. All three patients had failed to respond to prior anti-rheumatic medications, including methotrexate and infliximab. All reported less pain and disease activity, and their health assessment questionnaire scores improved subsequent to the imatinib treatment. Therefore, the addition of, or the switching to, imatinib may be beneficial to patients with RA who failed in conventional therapies.
Perspective and Conclusion
Besides those described above, PDGF is a fascinating molecular target in various other systemic rheumatic diseases such as spondyloarthropathy and systemic vasculitis. The strategies of PDGF signaling inhibition include antibodies against PDGF or PDGF-R, soluble recombinant fusion proteins of PDGF-R and IgG, oligonucleotide aptamer interfering PDGF-R signaling, and inhibitors of PDGF-R tyrosine kinase or downstream signaling molecules. Among many candidates, imatinib possesses some advantages in: 1) clinical use with well acceptable tolerability; 2) additional inhibition of Abl and c-Kit, which leads to the suppression of TGF-β and mast cell activity, respectively. Thus, imatinib is likely to play a multipotent role in the treatment of various systemic rheumatic diseases (Table 1). And indeed, several clinical trials examining the efficacy of imatinib for skin sclerosis, ILD and PAH are ongoing.
Summary of preclinical findings positively supporting the application of imatinib for systemic rheumatic diseases.

To date, the principal of the treatment for systemic rheumatic diseases has been chiefly targeting leukocytes. Thus, future therapeutic strategies should focus on other cell types, including fibroblasts, epithelial cells and endothelial cells (Fig. 2). Combined therapy of antiinflammatory and anti-fibrotic agents may shed light on the treatment of refractory diseases described in this review and further improve the long-term survival of patients with systemic rheumatic diseases.
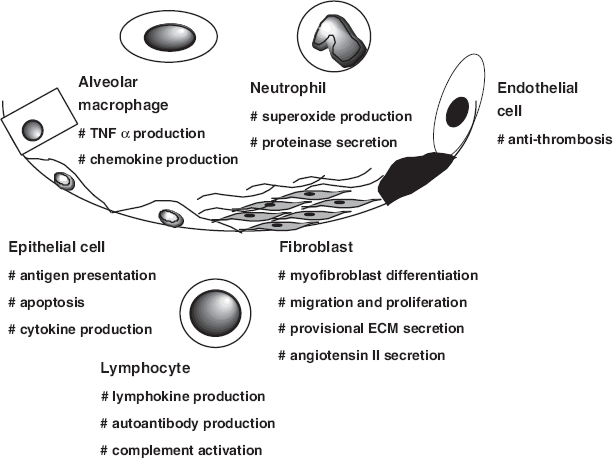
Predicted molecular mechanisms and possible therapeutic targets of rheumatic inflammation and fibrosis. Various cell types shown here are likely to be involved in the development and progression of rheumatic inflammation. Autoimmune responses and the subsequent infiltration of neutrophils and lymphocytes are known to occur. Resident macrophage activation, increased apoptosis of epithelial cells and endothelial dysfuction may result in the formation of fibrin clots and a provisional matrix, initiating the proliferation of fibroblasts. Any of those cells could serve as therapeutic targets in the treatment of systemic rheumatic diseases, and a simultaneous control of them seems to be essential in overwhelming the persistent inflammation.
