Abstract
Background:
Dyspnea is a common and diagnostically challenging symptom in the emergency department (ED), particularly among older adults with multimorbidity. Traditional risk stratification tools often perform poorly in this population. Galectin-3 (Gal-3), a biomarker of inflammation and fibrosis, may reflect biological aging and resilience, potentially improving early mortality risk assessment.
Objectives:
To evaluate the independent association between Gal-3 and 30-day mortality in patients presenting with acute dyspnea; to assess its utility for identifying patients at low risk of short-term mortality; and to determine its incremental value in improving prediction of 30-day mortality when added to clinical risk models.
Design:
Retrospective observational study based on the Acute Dyspnea Study (ADYS).
Methods:
The study included 763 adult ED patients with acute dyspnea. Gal-3 was measured as NPX (Normalized Protein Expression) values using the Olink proximity extension assay, and NT-proBNP was measured using standard laboratory methods. The primary outcome was 30-day all-cause mortality. Multivariable logistic regression and Cox regression analyses were performed, with internal validation by bootstrap resampling. Predictive performance was evaluated using ROC curves, AUC, and the Youden index, and incremental value was assessed by AUC comparison and net reclassification improvement (NRI).
Results:
Among the 763 patients, 49 (6.4%) died within 30 days. Gal-3 NPX was independently associated with 30-day mortality (OR 1.97; 95% CI: 1.16–3.36; p = 0.013). Although Gal-3 NPX alone demonstrated moderate discrimination (AUC 0.69), relatively low Gal-3 NPX levels within the cohort effectively ruled out short-term mortality, with a negative predictive value of 96%. Adding Gal-3 NPX to the clinical model modestly improved predictive performance (AUC increased from 0.803 to 0.819; NRI 0.028), primarily by enhancing identification of low-risk patients.
Conclusion:
Gal-3 NPX was independently associated with 30-day mortality in patients with acute dyspnea. Although its addition yielded only a modest, non-significant improvement in overall risk prediction, Gal-3 may be useful for ruling out short-term mortality, supporting its potential role as a negative prognostic marker in the ED setting.
Introduction
Shortness of breath, dyspnea, is one of the most common reasons for Emergency Department (ED) visits worldwide,1 -3 with an estimated prevalence of 7% to 15% among patients seeking urgent care. 4 Dyspnea is not only a distressing symptom but also a clinical challenge, with congestive heart failure (CHF) and chronic obstructive pulmonary disease (COPD) being among the most frequent underlying causes.5 -7 The prevalence of dyspnea-related diseases increases with age, and as the global population continues to age, these conditions are expected to place an increasing burden on healthcare systems. Age-related physiological changes can further complicate the presentation of dyspnea,1,4 and importantly, dyspnea is a significant predictor of mortality. 8 Consequently, accurate risk stratification in patients with acute dyspnea remains a challenge, emphasizing the need for robust biomarkers that can enhance mortality prediction, deepen the understanding of dyspnea’s complex pathophysiology, and improve clinical decision-making.
Traditional risk assessment models rely on clinical parameters (vital signs, basic blood test) and comorbidities but often fail to capture the biological processes driving disease progression and mortality. The biological age, shaped by factors such as inflammation, oxidative stress, and fibrosis, may be more informative than the chronological age or comorbidity burden alone. Biomarkers reflecting these mechanisms could improve risk stratification in acute care.
Galectins are a family of evolutionarily conserved lectins that bind β-galactosides through specific carbohydrate recognition domains. Among them, galectin-3 (Gal-3), a multifunctional protein, has received particular attention due to its central role in immune regulation, cell adhesion, proliferation, apoptosis, and angiogenesis.9,10 Through its interactions with glycoproteins in both extra- and intracellular environments, Gal-3 contributes to immune homeostasis, the development of inflammatory diseases and organ fibrosis. 11 As a potent pro-inflammatory mediator, apart from directly interacting with pathogens, Gal-3 activates multiple immune cell types such as neutrophils, monocytes/macrophages, and dendritic cells. 12 It drives cytokine responses, and has been linked to inflammatory diseases such as IBD 13 and increased mortality across a range of conditions and diseases, including among patients with chronic kidney disease (CKD). 14 Many studies show an association with heart failure and cardiovascular diseases,15 -20 although findings are partly conflicting, as in the study by Bellos on patients with CKD, 14 elevated serum galectin-3 levels were not consistently linked to heart failure, cardiovascular events, or cardiovascular mortality, although the same meta-analysis demonstrated a significant association between higher galectin-3 concentrations and increased all-cause mortality, supporting its role as a biomarker of adverse systemic outcomes. Although Gal-3 has been linked to morbidity and mortality in many respiratory diseases, one study has provided important insights into a potential protective role of galectin-3 in the development of bronchiectasis in neutrophilic obstructive airway diseases, although large-scale longitudinal studies are still required.21 -23
Even so these observations underscore galectin-3 as a marker of global biological vulnerability and systemic inflammation, aligning with its proposed relevance for short-term mortality risk in patients presenting with acute dyspnea. This study hypothesized that Gal-3 is independently associated with 30-day mortality in patients presenting with acute dyspnea. The aim was to assess whether Gal-3 provides prognostic information and whether its inclusion enhances mortality risk stratification when added to a prediction model based on conventional clinical variables.
Method
Study Population
This study includes 763 subjects from the Acute Dyspnea Study (ADYS). The original ADYS cohort comprises 1745 adult patients who presented to the ED at Skåne University Hospital in Malmö, Sweden, due to acute dyspnea of unknown origin, during weekdays between March 2013 and January 2019.24 -30 Critically ill patients and those with impaired consciousness were excluded, as they were either transferred directly to an intensive care unit (ICU) or too ill to provide consent.
After being informed about the study, patients were enrolled by research nurses, and written consent was obtained. The ADYS has received ethical approval (see Declaration section) and complies with the Declaration of Helsinki. 31 The reporting of this study conforms to the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) statement, which is part of the EQUATOR Network for transparent reporting of observational research (Supplemental Table 1).
Patients were interviewed using a standardized, study-specific questionnaire (Supplemental Table 2) developed by the ADYS research group under the supervision and final approval of Professor Olle Melander, Department of Clinical Sciences Malmö, Lund University Sweden. The questionnaire, together with an extensive variable list (Supplemental Table 3), was uniquely designed for the ADYS study to capture a broad range of clinical, demographic, and symptom-related variables relevant to patients presenting with acute dyspnea. These instruments were not based on previously validated questionnaires but were created specifically for this study. This has been acknowledged in the Discussion section as a limitation. A range of additional variables, including clinical examination findings, were collected. Blood samples were obtained within 1 hour after admission to the ED and were partly analyzed using standardized, accredited methods at the hospital laboratory. Additional samples were immediately frozen and stored at −80 °C for later analyses of specific biomarkers. The ADYS data register has since been supplemented with data from Statistics Sweden (SCB) regarding dates of death.
Gal-3 was analyzed in only the first 830 patients of the original ADYS cohort, using frozen biobank samples. As of the latest data registry update in 2021, with follow-up data through December 31, 2020, 820 of these individuals were still residing in Sweden and available for mortality follow-up. Due to missing data concerning sex, priority according to Medical Emergency Triage and Treatment System (METTS), 32 dyspnea level according to New York Heart Association Classification (NYHA classification) 33 and N-terminal pro-B-type Natriuretic Peptide (NT-proBNP) for 57 patients, the final cohort consisted of 763 patients (Figure 1).
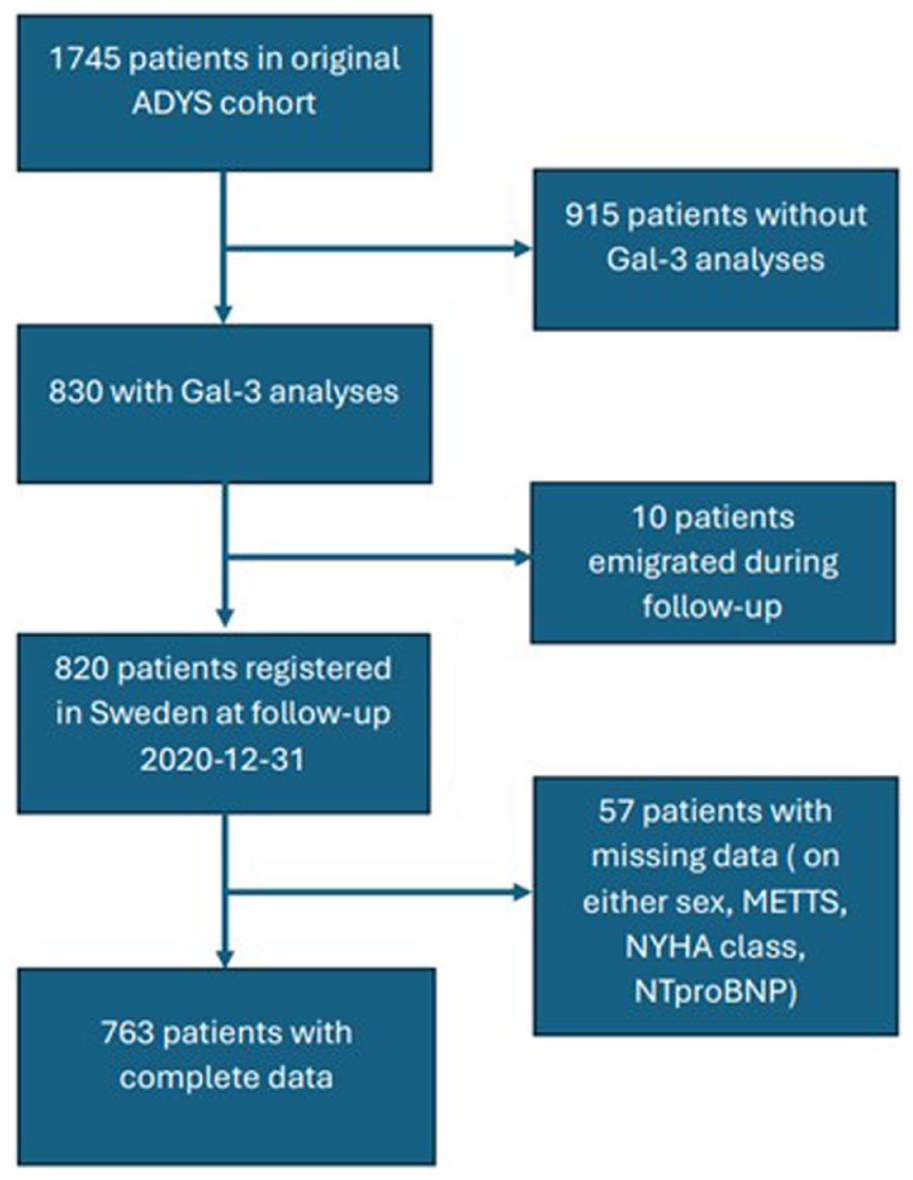
Flow-chart of cohort.
Variables
For this study the following variables were retrieved from the ADYS data register: age, sex, Body Mass Index (BMI), creatinine, C-reactive protein (CRP), METTS priority and dyspnea level according to the NYHA classification. To characterize the study population, the following past or ongoing conditions were recorded: Coronary Artery Disease (CAD), Congestive Heart Failure (CHF), Atrial Flutter/Fibrillation (AFIB), Diabetes, Hypertension, Chronic Renal Disease, and Chronic Obstructive Pulmonary Disease (COPD). These comorbidities were selected based on their strong association with aging or their established relevance in the development and clinical presentation of acute dyspnea.34 -36 All are common among elderly, tend to coexist, and contribute to physiological decline, frailty, and increased short-term mortality.
CRP and creatinine were analyzed directly at the hospital’s clinical laboratory. High sensitivity plasma CRP was analyzed using a particle enhanced turbidimetric assay (PETIA), and creatinine was analyzed by an IDMS calibrated enzymatic creatinine assay. Using creatinine, the estimated Glomerular Filtration Rate was calculated using the LM model. 37
Gal-3 and NT-proBNP were analyzed from frozen biobank samples. Galectin-3 was quantified from frozen biobank samples using the Olink Proseek® Multiplex Cardiovascular I 96X96 assay at Olink Laboratories, Uppsala, Sweden. The assay measures a broader panel of cardiovascular-related proteins; although all panel proteins were available in the dataset, the present study focused exclusively on Galectin-3. The Olink Proseek® Multiplex Cardiovascular I 96X96 kit is a Proximity Extension Assay (PEA) and results are reported in NPX units (Normalized Protein Expression). NPX is a relative, normalized, and log2-transformed scale. The values therefore do not represent absolute concentration (e.g., pg/mL) but rather reflect relative differences in protein expression between samples within the same analytical run and panel. Because the NPX scale is based on log2 transformation, an increase of 1 NPX unit corresponds to a doubling of the measured signal, whereas a decrease of 1 NPX unit corresponds to a halving. Differences in NPX values can thus be interpreted as fold changes between groups or time points (e.g., a difference of 1 NPX ≈ a two-fold change). According to Olink Proteomics AB, each analyte in the panel has been assessed in terms of sample material, specificity, precision, sensitivity, dynamic range, matrix effects and interference. NT-proBNP levels were measured on a Roche Cobase analyzer, following the manufacturer’s instructions.
METTS priorities 32 were initially registered by the emergency department nurses as part of routine clinical practice and subsequently documented in the research protocols by research nurses. Priority 1 (red priority) consists of the most critically ill patients in need of immediate medical attention by physician and nurses; priority 2 (orange priority) is classified as potentially life threatening and in need of medical attention within 20 minutes; priority 3 (yellow priority) is classified as not life threatening but in need of medical attention within 120 minutes; priority 4 (green priority) is classified as not life threatening and not in need of immediate. Priority 5 (blue priority) is also a category in METTS, classified as not in need of the ED facilities, but was not used in this study. A new binary METTS category was created, grouping priorities 1 and 2 as high priority, and priorities 3 and 4 as low priority.
Dyspnea severity was assessed according to the NYHA classification, 33 originally developed for patients with heart failure, but here it is applied to describe dyspnea level in all ADYS patients. Class 1 refers to patients with no limitation of physical activity due to dyspnea but still had dyspnea as the cause of chief complaint. without shortness of breath, Class 2 refers to patients with shortness of breath during heavy activity, class 3 during light activity, and class 4 refers to patients with shortness of breath at rest. A new binary dyspnea variable was created, categorizing NYHA classes I and II as low-grade dyspnea, and classes III and IV as high-grade dyspnea.
The cohort was supplemented with data from Statistics Sweden (www.scb.se) on whether patients were still residing in Sweden, and with mortality data up to December 31, 2020, obtained from the Swedish National Death Registry.
Model Building Strategy
To identify background variables most strongly associated with 30-day mortality, we performed a binary logistic regression analysis using forward selection based on the Wald statistic, incorporating all variables listed in Table 1. The final model retained 4 predictors: age, Gal-3, NT-proBNP, and the binarized METTS triage priority. A bootstrap analysis based on 1000 samples confirmed that these variables remained significant and stable, with no evidence of model instability or overfitting. Although sex was not retained in the final step of forward selection, it was included in the multivariable models due to its established clinical relevance and potential role as a confounder in outcome studies. Including sex also allowed for appropriate adjustment for baseline differences and improved the generalizability of the findings. Although NYHA functional class was not retained in the final selection, it was included in the model given its relevance as a proxy for dyspnea severity. The retention of NT-proBNP through forward selection, and its stability under bootstrap validation, was considered clinically reassuring, given its well-established role as a biomarker for congestive heart failure (CHF), a leading cause of acute dyspnea in this cohort. The inclusion of METTS triage priority was both statistically and clinically justified. In addition to being selected by the model, METTS served as a general indicator of acute illness severity and a relevant predictor of short-term mortality. Given the limited number of outcome events (n = 49), the number of covariates in the multivariable logistic regression models was restricted to 5 clinical variables in addition to Gal-3, in accordance with established recommendations to minimize the risk of overfitting and to ensure model stability.
Background Characteristics for Survivors and Non-Survivors Within 30 days (n = 763).

Abbreviations: AFIB, atrial fibrillation/flutter; BMI, body mass index; CAD, coronary artery disease; CHF, congestive heart failure; COPD, chronic obstructive pulmonary disease; CRP, C-reactive protein; eGFR, estimated glomerular filtration rate; Gal-3, Galectin-3; IQR, interquartile range; METTS, medical emergency treatment and triage system; NPX, normalized protein expression; a relative log2-scaled unit derived from Olink Proximity Extension Assay; NTproBNP, N-terminal pro-B-type natriuretic peptide; NYHA, New York Heart Association; SD, standard deviation.
Two-tails independent
Mann Whitney
Chi2 test.
Statistical Analysis
Baseline characteristics were presented as mean ± standard deviation (SD) for normally distributed continuous variables and as median with interquartile range (IQR) for non-normally distributed variables. Normality was assessed through histogram inspection and evaluation of skewness and kurtosis statistics. Categorical variables were reported as frequencies and percentages. Between-group comparisons were performed using the independent-samples
Kaplan–Meier survival curves were generated to visualize 30-day survival, and differences between groups were assessed using the log-rank test. The curves were stratified by Gal-3 NPX quartiles to explore survival differences across increasing biomarker levels. Multicollinearity among independent variables was evaluated using collinearity diagnostics within a linear regression framework, and no multicollinearity was detected in the models. The association between Gal-3 and 30-day mortality was assessed using stepwise binary logistic regression models, constructed as follows:
Model A: continuous Gal-3 NPX, age, and sex
Model B: Model A plus METTS and NYHA
Model C: Model B plus NT-proBNP
Odds ratios for Gal-3 are reported per 1-unit increase in continuous NPX (Normalized Protein Expression; log2-scaled relative units). Internal validation of all 3 models was performed using nonparametric bootstrapping with 1000 resamples to assess the robustness of parameter estimates and to evaluate the risk of model overfitting. Model discrimination and calibration were assessed using the optimism-corrected C-index and calibration slope/intercept derived from bootstrap resampling.
Receiver operating characteristic (ROC) curve analysis was performed for 2 purposes. First, to evaluate Gal-3 as an individual predictor of 30-day mortality and to explore an optimal cut-off value within this cohort using the Youden index. For this cut-off, sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) were calculated. Second, to assess the incremental predictive value of Gal-3 beyond established clinical variables, 2 models were constructed:
Baseline model: age, sex, METTS triage priority, NYHA class, and NT-proBNP
Extended model: baseline model plus Gal-3 (entered as a continuous NPX variable)
ROC curves and area under the curve (AUC) values were calculated for both models. AUCs were compared descriptively and, where applicable, statistically to evaluate whether the inclusion of Gal-3 improved the model’s discriminative ability. The statistical significance of differences in AUCs between models was assessed using DeLong’s test for correlated ROC curves. Net reclassification improvement (NRI) was calculated using pre-specified clinically relevant risk categories (5% and 10% predicted 30-day mortality) to evaluate whether the addition of Gal-3 improved the classification of patients as compared to the baseline model.
All statistical tests were 2-sided, and a
Results
Background Characteristics
The mean follow-up time concerning time to death or end-follow-up was 3.9 years (SD ± 2.7), ranging from 1 day to 7.8 years. In total, the cohort contributed approximately 2940 person-years of follow-up. The first patient was enrolled in March 2013 and the last in January 2019. There were significant differences in background characteristics between survivors and non-survivors (Table 1).
Non-survivors were generally older and had lower body weight. They showed higher levels of Gal-3, CRP, and NT-proBNP, as well as reduced kidney function as indicated by eGFR. Furthermore, non-survivors had a higher prevalence of prior heart failure, hypertension, and renal disease. A greater proportion were also triaged as more acute according to METTS priority, and more patients were classified in the higher NYHA classes, indicating worse functional status. In contrast, no significant differences were found in sex distribution, diabetes, atrial fibrillation, COPD, or coronary artery disease between the groups.
Survival Analysis by Galectin-3 NPX Quartiles
Within 30 days, 49 patients (6.4%) had died, while 714 survived. Women accounted for 54.1% of survivors and 59.2% of non-survivors. Kaplan–Meier analysis demonstrated that patients in the highest Gal-3 quartile (Q4) experienced significantly higher 30-day mortality compared with those in the lower 3 quartiles (log-rank

Kaplan–Meier curves illustrating 30-day mortality stratified by Galectin-3 NPX quartiles.
The log-rank test for trend across Gal-3 NPX quartiles showed a statistically significant difference in 30-day survival (χ2 = 16.54, df = 1,
Binary Logistic Regression for 30-Day Mortality and Gal-3; Models A–C.

Abbreviations: CI, confidence interval; Gal-3, galectin-3; METTS, medical emergency triage and treatment system; NTproBNP, N-terminal pro-B-type natriuretic peptide; NYHA, New York Heart Association; OR, odds ratio.
In Model A, higher Gal-3 NPX levels (per 1 unit increase; OR 2.35, 95% CI 1.40-3.94,
In Model B, adjusting additionally for clinical severity indicators (METTS triage priority and NYHA functional class), Gal-3 NPX (per 1 unit increase; OR 2.18, 95% CI 1.30-3.66,
The fully adjusted Model C, incorporating NT-proBNP, showed Gal-3 NPX remained independently predictive of mortality (per 1 unit increase; OR 1.97, 95% CI 1.16-3.36,
Internal validation was performed using 1000 bootstrap resamples, suggesting acceptable calibration and good agreement between predicted and observed probabilities (Supplemental Table 4).
Galectin-3′s Clinical Utility
To explore a Gal-3 threshold for risk stratification within this cohort, we performed a ROC analysis using Gal-3 NPX as a single predictor of 30-day mortality (Figure 3).
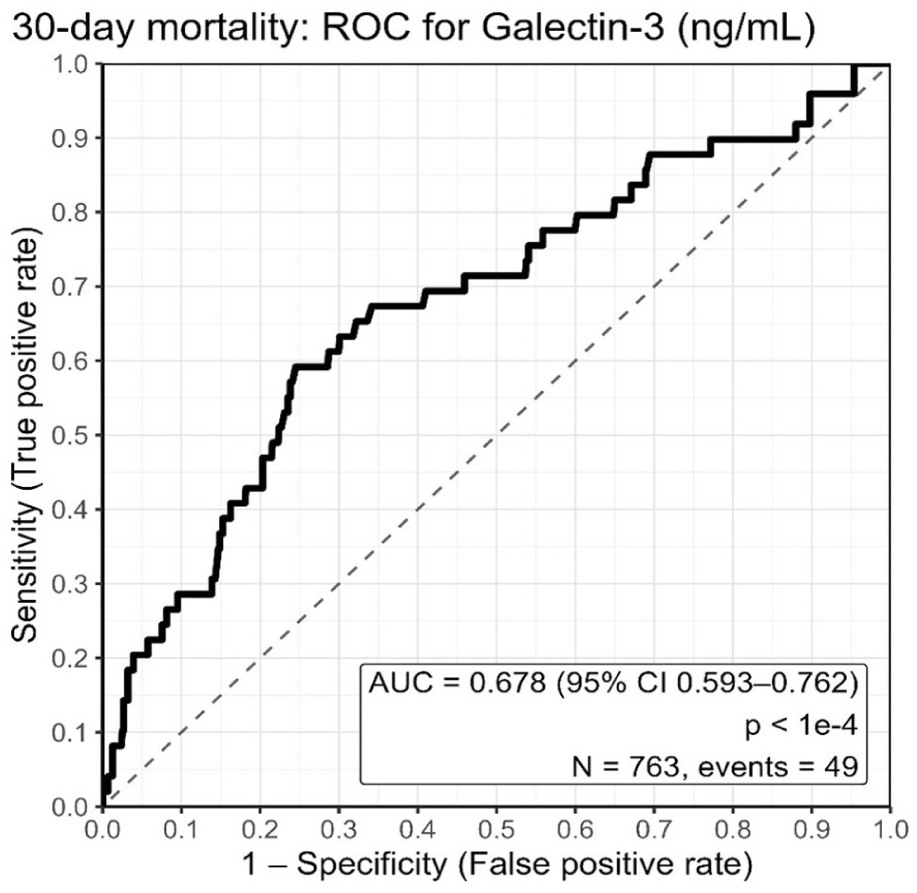
Receiver operating characteristic (ROC) curves for 30-day mortality prediction for Gal-3 NPX with AUC.
The area under the curve (AUC) for Gal-3 NPX as a single predictor of 30-day mortality was 0.678, indicating moderate discriminative ability. This curve was used to identify the optimal cut-off for predicting 30-day mortality using Youden index. The optimal cut-off (Youden index) was derived from NPX values and should be regarded as cohort- and platform-specific, and therefore not directly transferable to clinical decision-making without external validation (Table 3).
Youden Index Cutoff with Corresponding Sensitivity, Specificity, PPV and NPV.

Abbreviations: Gal-3, Galectin-3; NPX, Normalized Protein Expression; a relative log2-scaled unit derived from Olink Proximity Extension Assay; NPV, negative predictive value; PPV, positive predictive value.
The analysis identified a cut-off value of 5.68 NPX, which yielded a sensitivity of 59% and a specificity of 75%. The corresponding positive predictive value (PPV) was 15%, and the negative predictive value (NPV) was 96%. While this cut-off demonstrates that higher relative Gal-3 expression is associated with increased 30-day mortality, the low PPV indicates limited ability to correctly identify patients who will die within 30 days. In contrast, the high NPV suggests that lower Gal-3 expression is associated with a low short-term mortality risk. As Gal-3 was measured as relative NPX values, this cut-off should be regarded as population- and platform-specific and is not intended for direct clinical application, but rather for illustrating the prognostic gradient within this cohort.
Incremental Predictive Value of Galectin-3
Secondly, to evaluate the incremental predictive value of Gal-3, ROC curves and AUCs were calculated for 2 models: 1 including clinical variables only (age, sex, METTS triage priority, NYHA class, and NT-proBNP), and a second model including the same variables plus Gal-3 NPX entered as a continuous variable (Figure 4).

Receiver operating characteristic (ROC) curves comparing a clinical model including Galectin-3 NPX (Model A) with a clinical model without Galectin-3 NPX (Model B) for prediction of 30-day mortality.
The baseline model demonstrated good discriminatory ability, with an AUC of 0.803. The addition of Gal-3 NPX resulted in a modest improvement, increasing the AUC to 0.819, suggesting potential incremental prognostic information beyond established clinical and biomarker predictors. However, this improvement did not reach statistical significance (DeLong’s test; ΔAUC = 0.016; 95% CI –0.044 to 0.011, p = 0.25). Using the Youden-optimal threshold, adding Gal-3 increased the Youden index from 0.511 to 0.547 (ΔJ = 0.037), primarily by improving sensitivity (87.8% → 91.8%) with little change in specificity (62.9% → 63.3%). The optimal predicted risk thresholds were 5.36% and 4.74% for Models 1 and 2, respectively (Table 4).
Youden Index Cutoff with Corresponding Sensitivity, and Specificity.

Model 1: age, sex, METTS triage priority, NYHA class, and NT-proBNP. Model 2: Model 1 plus Gal-3 NPX.Cutoffs represent predicted 30-day mortality risk derived from the respective logistic regression models.
Net Reclassification Improvement (NRI)
To further assess the incremental prognostic value of Galectin-3, we calculated the NRI using clinically relevant 30-day mortality risk thresholds of 5% and 10% (Supplemental Table 5). Adding Galectin-3 (entered as a continuous NPX variable) to the baseline model (age, sex, NYHA class, METTS triage priority, and NT-proBNP) yielded an overall NRI of 0.028 (95% CI: −0.12 to 0.17;
Discussion
The main finding of this study was that Gal-3 was independently associated with 30-day mortality in patients presenting with acute dyspnea. Although its addition to a clinical risk model did not significantly improve discriminatory performance, the consistent association across all models supports its role as a complementary prognostic marker. Notably, relatively low Gal-3 NPX levels within this cohort were particularly effective in identifying patients at low risk of short-term mortality, highlighting its potential utility as a “rule-out” biomarker in the emergency setting. This was further supported by the high NPV observed for Gal-3–based risk stratification, as well as NRI, which demonstrated that the addition of Gal-3 primarily enhanced risk classification among survivors.
Relative Gal-3 expression reflects chronic processes such as fibrosis, inflammation, and tissue remodeling, mechanisms usually linked to long-term outcomes. Our findings suggest these also influence short-term mortality, likely by reducing physiological reserve. Elevated Gal-3 may capture biological aging and vulnerability beyond chronological age, consistent with prior studies showing correlations with age, senescence, and inflammation.38 -41 In patients with dyspnea, particularly with CHF, higher Gal-3 may indicate myocardial fibrosis and adverse remodeling, aligning with its established prognostic role in heart failure and the effects of pharmacological Gal-3 inhibition.18,42 -44 Although frailty was not directly assessed, the association between Gal-3 and adverse outcomes supports its potential as a biomarker of biological vulnerability, complementing clinical frailty indices by detecting subclinical decline even in relatively young patients.
The relatively modest incremental predictive value of Gal-3 in our multivariable models may be partly explained by the limited number of deaths within the 30-day follow-up period, which may reduce statistical power. Additionally, Gal-3′s association with mortality may be attenuated by stronger short-term prognostic markers such as NT-proBNP or clinical severity scores, which more directly capture acute pathophysiology. While these markers reflect immediate disease burden, Gal-3 may instead signal chronic biological frailty or, conversely, biological resilience when levels are low. As a result, the unique contribution of Gal-3 may be underrepresented when evaluated alongside variables that dominate short-term outcomes.
These findings may have potential relevance in acute care, where rapid and reliable risk stratification is important. While identifying high-risk patients is critical, recognizing those at lower risk can also be valuable. Gal-3 could potentially provide complementary information to established clinical tools, possibly aiding discharge decisions and resource allocation by reflecting underlying biological resilience. At present, Gal-3 testing is laboratory-based, and its role in emergency workflows remains to be defined. Future technological advances, such as point-of-care platforms, may facilitate broader clinical application.
Taken together, our findings suggest that Gal-3 captures dimensions of biological vulnerability not fully reflected by conventional clinical variables or acute-phase biomarkers. While higher Gal-3 levels were associated with increased 30-day mortality, this association may be more effectively captured by other, more immediate prognostic indicators such as NT-proBNP. In contrast, relatively low Gal-3 levels within this cohort appear to reflect a state of biological resilience, indicating preserved physiological integrity and reduced vulnerability to acute stressors, which was strongly associated with reduced short-term mortality risk. This highlights the potential value of Gal-3 particularly as a “rule-out” biomarker in early risk stratification. Future studies should explore its integration into real-time decision-making frameworks, ideally in combination with other clinical and biomarker data. Machine learning based models may further enhance predictive precision and enable more individualized care in patients presenting with acute dyspnea.
Strengths and Limitations
This study has several strengths. It is based on a relatively well-characterized cohort of patients with acute dyspnea presenting to the ED, enhancing its clinical relevance. The inclusion of a diverse and unselected dyspnea patient population from Malmö strengthens the external validity and may enhance generalizability to similar urban healthcare settings. The inclusion of both clinical variables and biomarkers allowed for a comprehensive evaluation of Gal-3 as an additive prognostic tool beyond established measures such as NT-proBNP, METTS triage priority, and NYHA classification. A key strength of our study is the use of modern statistical techniques, including internal validation through bootstrap resampling, which enhances methodological rigor and supports the robustness of our findings. Moreover, we report on both traditional performance metrics (AUC) and clinically relevant measures such as net reclassification improvement (NRI). This comprehensive and relatively uncommon approach in biomarker research strengthens both the reliability and the clinical applicability of our results. Additionally, the focus on 30-day mortality provides meaningful insight into early risk, which is highly relevant in acute care settings.
However, the study also has limitations. As it was conducted at a single center within the Swedish healthcare system, caution is warranted when extrapolating the results to other regions or healthcare systems with different patient demographics or clinical practices. Patient inclusion was restricted to weekday daytime hours, and critically ill patients unable to provide informed consent or admitted directly to the intensive care unit were excluded. These factors may have introduced selection bias and affected the representativeness of the cohort. In addition, dyspnea severity was assessed using the NYHA classification, originally developed for heart failure, which may have led to some misclassification in this broader dyspnea population. Only 49 patients (6%) in the cohort died, which is a relatively small number and may have limited the statistical power of mortality analyses. Gal-3 was analyzed retrospectively in a subset of patients from stored plasma samples, which may not fully reflect conditions for real-time laboratory testing and carries potential risks related to storage. Although multivariable adjustment was performed, residual confounding cannot be ruled out. Another limitation of the present study is that the questionnaire and variable list used for data collection in the ADYS study were not based on previously validated instruments. Instead, they were specifically developed by the research group to comprehensively capture clinical and demographic characteristics relevant to patients with acute dyspnea. Although the tools were carefully designed and tested during the initial phase of the study, the absence of prior external validation may introduce a degree of measurement bias or limit comparability with other cohorts. Another limitation concerns potential confounding related to pre-existing comorbidities. Although COPD and congestive heart failure (CHF) were included as candidate variables in the initial forward Wald selection aimed at identifying predictors of 30-day mortality, they were not retained in the final regression model. Given the limited number of outcome events, the number of covariates had to be restricted to ensure model stability and to avoid overfitting. It is therefore possible that elevated Gal-3 levels, at least in part, reflect underlying chronic conditions such as CHF or COPD. This should be considered when interpreting the observed associations between Gal-3 and short-term mortality. An additional limitation is that Gal-3 was measured as NPX (Normalized Protein Expression), a relative and platform-dependent unit derived from the Olink assay. As such, the observed cut-offs and effect sizes may not be directly transferable to other assays or clinical settings without external validation. Finally, Gal-3 testing is currently limited to specific laboratory settings, which restricts its immediate clinical applicability.
Implications
Our findings suggest that Gal-3 could potentially be a useful addition to early risk assessment frameworks for patients with acute dyspnea in the ED requiring external validation. While the incremental predictive value was modest, relatively low Gal-3 NPX levels within this cohort proved effective in identifying patients at low risk of short-term mortality. This may assist clinicians in ruling out high-risk status and prioritizing resources more efficiently. Incorporating biomarkers that reflect underlying biological processes, such as inflammation and organ vulnerability, could support the shift toward more individualized, pathophysiology-based models of acute care. Such approaches align with broader healthcare goals of improving prognostic precision and optimizing resource use in high-pressure clinical settings.
Conclusion
In this study of patients presenting with acute, unsorted dyspnea, Gal-3 was independently associated with 30-day mortality and modestly increased the performance of a clinical prediction model although without reaching statistical significance. Importantly, relatively low Gal-3 NPX levels within this cohort were strongly linked to reduced short-term mortality risk. These results support the biomarker’s potential as a tool for early risk stratification. Further validations in external cohorts and prospective evaluation in real-time clinical settings are needed. Future work should prioritize the development of point-of-care testing solutions to enable rapid integration of Gal-3 into acute care workflows.
Supplemental Material
sj-docx-1-bmi-10.1177_11772719251412983 – Supplemental material for Galectin-3 Is Independently Associated With Short-Term Mortality and Identifies Low-Risk Patients With Acute Dyspnea in the Emergency Department: A Retrospective Cohort Study
Supplemental material, sj-docx-1-bmi-10.1177_11772719251412983 for Galectin-3 Is Independently Associated With Short-Term Mortality and Identifies Low-Risk Patients With Acute Dyspnea in the Emergency Department: A Retrospective Cohort Study by Ahmad Zwawi, Ardavan M. Khoshnood, Ulf Ekelund and Torgny Wessman in Biomarker Insights
Supplemental Material
sj-docx-2-bmi-10.1177_11772719251412983 – Supplemental material for Galectin-3 Is Independently Associated With Short-Term Mortality and Identifies Low-Risk Patients With Acute Dyspnea in the Emergency Department: A Retrospective Cohort Study
Supplemental material, sj-docx-2-bmi-10.1177_11772719251412983 for Galectin-3 Is Independently Associated With Short-Term Mortality and Identifies Low-Risk Patients With Acute Dyspnea in the Emergency Department: A Retrospective Cohort Study by Ahmad Zwawi, Ardavan M. Khoshnood, Ulf Ekelund and Torgny Wessman in Biomarker Insights
Footnotes
Acknowledgements
We thank Professor Olle Melander, Department of Clinical Sciences, Lund University, Sweden, for initiating the ADYS study and providing access to the ADYS database, thereby making this study possible. The authors used ChatGPT (OpenAI, San Francisco, CA, USA) as a language support tool to refine the English text and to assist in identifying relevant literature. No scientific data were generated, modified, or analyzed using AI tools.
List of abbreviations
ADYS Acute Dyspnea Study
AUC Area Under Curve
BMI Body Mass Index
CAD Coronary Artery Disease
CHF Congestive Heart Failure
CI Confidence Interval
CRP C-Reactive Protein
ED Emergency Department
Gal-3 Galectin-3
ICD-10 International Classification of Diseases version 10
METTS Medical Emergency Treatment and Triage System
NPV Negative Predictive Value
NPX Normalized Protein Expression
NT-proBNP N-terminal pro-B-type Natriuretic Peptide
NYHA New York Heart Association
OR Odd ratio
PPV Positive Predictive Value
ROC Receiver Operating Characteristic
SD Standard Deviation
Ethical Considerations
This study has ethical approvals from Regionala Etikprövningsnämnden EPN, Lund, Sweden: Dnr 2011/537, 2012/762, 2016/138, 2017/301, 2018/781, and from Etikprövningsmyndigheten, Sweden: Dnr 2020-04301.
Consent to Participate
Participant’s informed and written consent was asked for and obtained.
Consent for Publication
This manuscript contains no person’s data and therefor can be published without consent from the participants.
Author Contributions
AZ and TW contributed and participated in overall scientific coordination and participated in study design, data acquisition, data analysis, and manuscript. UE and AK contributed with methodological input and critically revised the manuscript for important intellectual content. All authors read and approved of the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Code Availability
Not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
