Abstract
Background:
Measurement of chemokine CXCL10 is a novel approach to assess the transplant rejection risk and to detect the rejection. While CXCL10 has shown itself to be useful in renal transplants, there are only a few studies on the association between pancreas transplantation and CXCL10. Furthermore, the importance of autoantibodies associated with type I diabetes in pancreas transplantation are relatively poorly understood.
Objectives:
To determine whether there is an association between CXCL10 plasma levels and graft rejection in simultaneous pancreas and kidney transplant recipients, and to assess the effects of pancreas autoantibodies on pancreatic transplant rejection.
Design:
A retrospective case-control study was conducted.
Methods:
In total of 23 individuals (11 male and 12 female, mean age 39.5 (SD ± 7.6) years) who underwent simultaneous pancreas and kidney transplantation, and in 8 healthy controls (evenly distributed in terms of gender, mean age 26.1 (SD ± 1.5) years) plasma samples were analyzed for CXCL10 and autoantibody levels using ELISA. The data was categorized into preoperative, perioperative and postoperative samples and were further juxtaposed in relation to rejection episodes.
Results:
Preoperative CXCL10 levels did not differ from those for healthy individuals, but they rose postoperatively (P = .02). The median preoperative plasma concentration of CXCL10 was 68 pg/ml and increased to 123 pg/ml postoperatively. A postoperative plasma CXCL10 cut-off value of 297 pg/ml was indicative of rejection. Postoperative autoantibodies were detected in 10 recipients, 4 of whom were positive for anti-ZnT8 autoantibodies, and 2 were positive for more than 1 type autoantibody. All recipients who were positive for anti-ZnT8 autoantibodies or for more than 1 type autoantibody experienced rejection.
Conclusion:
Elevated CXCL10 levels during the first 3 months after transplantation seem to be a risk factor for rejection. The post transplant presence of anti-ZnT8 autoantibodies was associated with transplant rejection.
Introduction
Diabetes mellitus type I (T1D) is an autoimmune disorder characterized by pancreatic beta cell destruction leading to insulin deficiency and hyperglycemia. 1 Diabetic nephropathy occurs in 25% to 40% of people with T1D. 2 This diabetic kidney disease may lead to kidney failure. 2
One of the treatment options for complicated T1D is a simultaneous pancreas and kidney transplant (SPKTx) whereby transplanting a new pancreas an endogenous insulin supply is provided and hence the risk for hypo- and hyperglycemic episodes is reduced. 3 This also leads to a better kidney transplant prognosis.
Moreover, diabetes has increasingly displayed itself as a clearly intertwined group of diseases or, even more, as a spectrum. It has been demonstrated that autoimmunity together with beta cell dysfunction occurs also in type II diabetes (T2D). 4 Consequently, beyond treating T1D, a growing body of evidence suggests that SPKTx may also be a viable therapeutic option for certain individuals diagnosed with T2D. 5 Given the rising global prevalence of T2D, 6 the incidence of SPKTx may also significantly increase worldwide.
Although immunosuppression therapies required for SPKTx have evolved over time, rejection rates for simultaneous pancreas and kidney transplants are still estimated to range between 5% and 25%. 7 Additionally, there is a need for noninvasive biomarkers tailored to specifically evaluate rejection. Presently, the monitoring of transplant function relies on pancreatic enzymes such as amylase, lipase and measurement of blood glucose levels, while rejection episodes are still confirmed by biopsy.
One possible biomarker for assessing pancreatic transplant’s function is C-X-C motif chemokine 10 (CXCL10), also known as the interferon-γ-inducible protein-10 (IP-10). CXCL10 is a chemokine secreted both by immune cells and tissue cells such as endothelial cells or even fibroblasts and it mainly recruits type 1 T-helper lymphocytes, which in turn produce interferon-γ and tumor necrosis factor-α which once again stimulate the secretion of CXCL10 thereby amplifying the reaction. 8 CXCL10 interacts with the CXCR3 receptor on immune cells and drives cell activation and proliferation. The secretion of CXCL10 is more prominent in environments with memory T-cells compared to naïve T-cells, 9 and therefore, we hypothesized that CXCL10 plasma levels must be higher in patients with acute cellular rejection.
CXCL10 has been described as a novel biomarker for assessing the impact of transplant rejection on the heart, 10 lung, 11 and most importantly, kidney function. 12 It has also been shown that for renal transplants, it can be used as a kidney-specific biomarker when analyzing urine. 13 Furthermore, CXCL10 has also proven itself as a preoperative risk factor for acute renal transplant rejection.12,14
Although T1D is also a condition mediated by T-cells, it has been reported 8 that CXCL10 plasma concentrations are lower in individuals with long-term T1D than in individuals with newly diagnosed T1D. Therefore we also hypothesized that the preoperative CXCL10 values from our recipients should not differ from the CXCL10 values in the control group.
The correlation between pancreatic transplants and plasma CXCL10 levels is yet to be investigated. To the best of the authors’ knowledge, no research has been published on the relationship between human allogenic pancreatic transplant rejection and CXCL10 levels.
Another option for assessing the rejection risk is to analyze pancreas-specific autoantibodies. It has been shown that postoperative anti-GAD autoantibody presence is a risk factor for pancreatic transplant rejection,15,16 but it has also been shown that preoperative presence of autoantibodies does not seem to influence rejection. 17
Our objectives were: to measure CXCL10 levels in SPKTx recipients before and after transplantation and to find out whether there is association with graft rejection; additionally, to detect the levels of autoantibodies against tyrosine phosphatase-related islet antigen 2 (anti-IA2), zinc transporter 8 (anti-ZnT8) and glutamic acid decarboxylase 65 kDa isoform (anti-GAD65) and to assess their effect on pancreatic transplant rejection.
Research Design and Methods
Study Population
Plasma samples were obtained from a cohort of 23 consecutive individuals diagnosed with type I diabetes (T1D) who underwent SPKTx at Tartu University Hospital between 2015 and 2022 and who agreed to participate in the study. Samples were collected at various time points before, during, and after transplantation, with no fixed intervals, and the samples were grouped as such. Of the participants 11 were male and 12 female, with a mean age of 39.5 (SD ± 7.6) years.
The indication for transplantation was terminal renal failure associated with T1D and the main criteria for recipient selection were: C-peptide levels not detectable in plasma, age under 50 years, body mass index below 27, and patient’s ability to withstand the procedure (without significant cardiovascular risks). The diagnosis of rejection was based on the latest guidelines outlined in the UpToDate’s article “Pancreas Allograft Rejection,” 18 in accordance with the UpToDate guidelines; the type of graft rejection was diagnosed histologically on the basis of the criteria of the Banff classification of pancreas allograft rejection. 19
The control group comprised 8 healthy students, distributed evenly by gender, with a mean age of 26.1 (SD ± 1.5) years.
Plasma was separated from whole blood on the day of collection and stored at −80°C until analysis. Prior to analysis, all samples were allowed to thaw at room temperature for 1 hour and then mixed onto the ELISA plate before application.
This study was approved by the Ethics Review Committee on Human Research of the University of Tartu (163/T-6, 24.09.2007; 287/M-10, 19.11.2018; 384/M-15, 20.11.2023 and 396/M-21, 16.12.2024) and was conducted in accordance with the Declaration of Helsinki. Written informed consent was obtained from each participant before participation in the study.
Measurement of CXCL10
For measurement of CXCL10 plasma levels, a LEGEND MAX Human CXCL10 (IP-10) ELISA Kit (lot no.: B381449, cat no.: 439907, Bio Legend) was used. All but 1 sample were measured in duplicate, and the average of the 2 measurements was used in the statistical analysis. The remaining sample was measured 4 times; 2 average values were calculated from these measurements, and both were included in the analysis (both of those values are presented in Figure 1). In total, 2 ELISA kits were used and the manufacturer’s protocol for using the ELISA kits was followed. The results are expressed in pg/ml.

Measured CXCL10 levels categorized by SPKTx recipients. The figure shows a timeline for each SPKTx recipient on a logarithmic time scale. Time points at which blood samples were drawn are marked with a dot, and corresponding CXCL10 values are provided. Recipients who experienced rejection are marked in red, whereas those without rejection are marked in green. Black vertical lines mark the days when rejection was diagnosed. One sample was measured twice (duplicate), indicated by an asterisk.
Measurement of Diabetes- Associated Autoantibodies
SPKTx recipients were analyzed for presence of autoantibodies against IA2, ZnT8 and GAD65. The SPKTx recipients were deemed to be autoantibody-positive in the case of concentrations of no less than 7.5 U/ml anti-IA2, 15 U/ml anti-ZnT8, or 5 U/ml anti-GAD65. All diabetes-associated autoantibodies were measured using commercial ELISA kits from RSR Limited (Cardiff, United Kingdom). The catalog numbers were following: anti-IA2 ELISA: IAE/96/2, anti-ZnT8 ELISA: ZnT8/96, anti-GAD65 ELISA: GDE/96. All ELISA kits were used according to the test instructions provided by the producer.
Statistical Analysis
Analyses were performed using the RStudio version 2023.12.0 + 369. All recipients did not provide samples at every time period. For these missing samples, no imputation or substitution was performed; the recipients were simply excluded from the analysis for those specific time periods.
Statistical significance was calculated using the Wilcoxon and Fisher tests. For calculating cutoff values, a receiver operating characteristic curve was employed. All statistically significant results are presented with a 95% confidence interval. The results were considered statistically significant at P < .05.
A potential bias arising from the fact that not all SPKTx recipients provided the same number of samples or followed identical time intervals was acknowledged during the design of the analysis. To minimize this effect, no tests were conducted that would have included only 1 recipient from either the rejection or no-rejection group.
All data collection and statistical analysis was performed by the research team and no scientific data was generated nor modified by artifical intelligence tools. For presenting the data we used the STROBE case-control reporting guidelines (see Supplemental File 1). 20
Results
Alltogether 70 samples were included in the study: 62 from the SPKTx recipients and 8 from the controls. Among the 62 samples from the SPKTx recipients, 21 were taken preoperatively, 16 on the day of transplantation, and 25 postoperatively. No significant changes were observed between the 2 genders within the same time groups. The main characteristics of these samples are provided in Table 1.
CXCL10 Plasma Levels Categorized by Time and Rejection.

Abbreviations: Tx, transplantation; R*, group where rejection occurred.
Statistical significance (P-value) was calculated for recipients with and without rejection within the same time frame. The results were considered statistically significant at P < .05. The shading was added to enhance readability (to distinguish the samples from recipients in the rejection group (R*) or no rejection group). The shading has no statistical nor numerical meaning.
Nine SPKTx recipients from whom a total of 27 samples was collected were diagnosed with isolated pancreas transplant rejection; there was no suspition of kidney transplant rejection. Among the recipients who experienced rejection, a histological sample from the transplanted pancreas was obtained and acute T cell-mediated rejection was confirmed in all samples according to criteria of the Banff classification of pancreas allograft rejection. 19 Based on C-peptide levels, in 8 recipients out of 9 the transplant’s function was preserved; recipients with rejection and their corresponding biomarker levels are shown in Supplemental Table S1.
Post-Transplant Changes in CXCL10 Values
In the preoperative samples (n = 21) the median value of plasma CXCL10 levels was 68 pg/ml, with a minimum value of 41 pg/ml and a maximum value of 311 pg/ml. In most of the samples (n = 14; 67%) CXCL10 levels were below 100 pg/ml. For the controls, we found a median value of 70 pg/ml, with a minimum of 27 pg/ml and a maximum of 100 pg/ml. The statistical difference between preoperative samples and samples from the control group was found to be not significant (P = .21). Using the 95% reference interval, based on the control group the normal range of CXCL10 plasma values was calculated to be from 28 to 98 pg/ml.
In contrast, postoperative samples showed a median value of 123 pg/ml, with a minimum and maximum being 30 and 632 pg/ml, respectively. The dynamics of CXCL10 values for each recipient is represented in Figure 1.
Regardless of the occurrence of rejection, a significant difference in CXCL10 values occurred between the preoperative samples and those from the first 90 days of the postoperative period (P = .02; median difference 77.6, 95% CI 7.8-357.2). Comparison of the preoperative samples and all postoperative samples revealed no significant difference (P = .07). However, when the control group was included in analysis, a significant difference was noted in CXCL10 values compared to the values of all postoperative samples (P = .02; median difference 56.9, 95% CI 10.2-142.1).
Dependence of Pre- and Postoperative CXCL10 Levels on Rejection
No statistically significant result was noted when juxtaposing preoperative CXCL10 levels and rejection. Among all individuals whose samples were taken before the date of transplantation, the median value for those who experienced rejection was 41 pg/ml higher compared to those who did not experience rejection (101 pg/ml vs 60 pg/ml; P = .11).
When no correlation was detected between CXCL10 value and number of days from transplantation (rejection group: rho = −.45, P = .14; group with no rejection: rho = −.03, P = .91), then higher CXCL10 post transplant plasma values were associated with transplant rejection episodes (Figure 2). The postoperative cutoff value was calculated to be 297 pg/ml (AUC = 0.67). Interestingly, while showing low sensitivity for rejection episodes, this value was highly specific: all individuals for whom the CXCL10 values exceeded 297 pg/ml at least once in the postoperative period experienced a rejection episode.

Postoperative CXCL10 blood plasma levels in SPKTx recipients. The SPKTx recipients who experienced pancreatic transplant rejection at any time are indicated in red, while those with no noted rejection episodes are shown in black. A distinct threshold can be identified at 297 pg/ml, with values above this level being as a risk factor for transplant rejection.
Changes in CXCL10 levels during and after rejection revealed no significant correlation (P = .67). For the 12 samples taken during or after the date of rejection diagnosis, the CXCL10 baseline value at the time of diagnosis to be 272 pg/ml was calculated (P = .10). For each day after diagnosis, CXCL10 values decreased by 1.9 pg/ml (P = .48).
Presence of Anti-ZnT8 Autoantibodies as an Important Marker for Rejection
Out of the 14 recipients from whom plasma samples were taken after transplantation, the presence of autoantibodies was determined in 10. Among these 10 recipients, 4 tested positive for the anti-ZnT8 autoantibody at some point after transplantation, and all 4 experienced a rejection episode. In contrast, none of the 6 recipients who tested negative for anti-ZnT8 autoantibodies experienced a rejection episode. Our analysis indicated that the presence of anti-ZnT8 autoantibodies in plasma after transplantation is a significant risk factor for rejection (P = .001; OR = 3.01).
Similar analyses were conducted for anti-IA2 and anti-GAD65 autoantibodies however, these revealed no significant results related to transplant rejection (postoperative P-values .17 and .18, respectively). The number of recipients positive for autoantibodies is presented in Table 2 and in Supplemental Table S1.
Autoantibody Presence in Recipients Categorized by the Time of Sample Collection.

Abbreviation: aAb, autoantibody.
In total, we analyzed autoantibody presence in at least 1 sample in 22 individuals preoperatively and in 10 individuals postoperatively. Statistical significance (P-value) was calculated for the presence of autoantibodies and the prevalence of rejection in the recipients with the above autoantibodies. The results were considered statistically significant at P < .05.
No association with transplant rejection was observed when only 1 autoantibody regardless of the type was present (P = .38). Regarding the presence of the different autoantibodies preoperatively, no significant results were found: anti-ZnT8 (P = 1), anti-IA2 (P = .60), and anti-GAD65 (P = .67).
Although it was found that the presence of 2 or more types of autoantibody postoperatively was associated with rejection (P = .018), this group consisted only of 2 recipients and therefore this result must be interpreted with caution. Postoperative autoantibody presence and rejection prevalence are also shown in Figure 3.
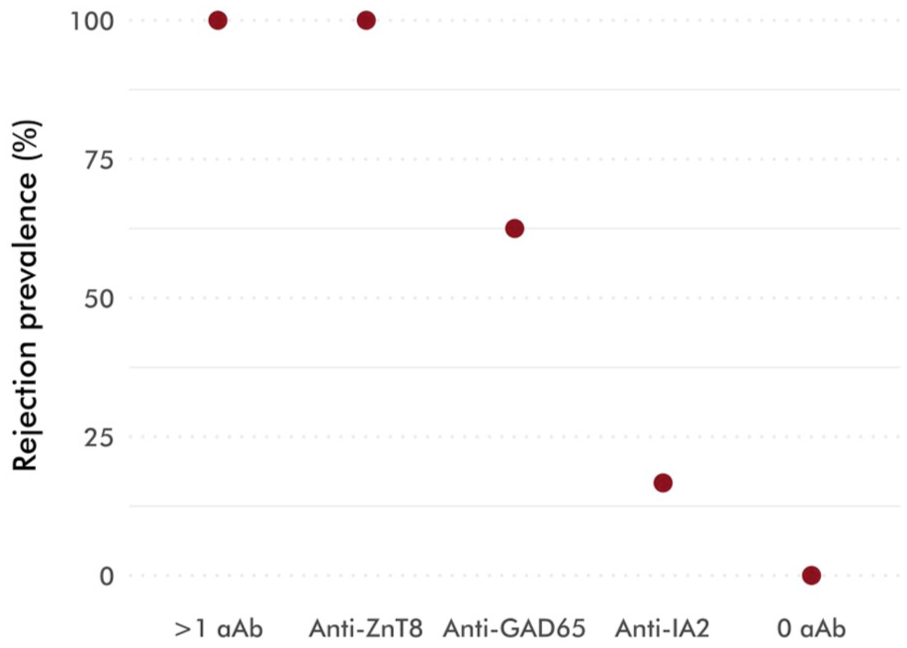
Postoperative autoantibody samples and prevalence of rejection episodes associated with these samples. The most significant risk factors for rejection are presence of 2 or more autoantibodies or presence of anti-ZnT8 autoantibodies. Absence of autoantibodies can be regarded as a protective factor, but the result is not statistically significant.
Discussion
Simultaneous pancreas and kidney transplantation is a viable method for regaining blood glucose control in persons with T1D. The longevity of the transplant is usually determined by the period during which the SPKTx recipient can live without the need for exogenous insulin. Therefore, a rise in blood glucose values is usually the first sign of pancreatic transplant rejection and there are no other convenient point-of-care or home-based tests.
Our findings suggest that CXCL10 plasma values for persons with uncontrolled T1D who are yet to undergo a transplantation procedure are not different compared to healthy individuals. Based on our small control group, we calculated the normal range of CXCL10 plasma values, using this ELISA kit, to be from 28 to 98 pg/ml. This finding is in line with Nicoletti et al 21 and suggests that CXCL10 levels can be used to identify inflammatory processes in people with T1D.
CXCL10 plasma values rise for a short time after transplantation and we have shown in this study that marked CXCL10 rise can be associated with higher risk for transplant rejection. Although for the lack of samples, we were unable to determine the time during which CXCL10 is elevated as a consequence of surgical transplantation trauma, Rydenfelt et al 22 showed that CXCL10 levels start to fall within only a few postoperative days. Our data suggest similar results and it is evident that at further time points rise in CXCL10 values is associated with other complications and even rejection.
Although not all recipients showed similar spikes in CXCL10 values immediately after transplantation, those who did were at a higher risk for experiencing a rejection episode. Interestingly, our study identified only isolated cases of pancreas transplant rejection. This may be due to the possibility that early immunosuppressive therapy aimed at treating pancreas transplant rejection could have mitigated renal transplant rejections. In our study, the cutoff value was calculated to be 297 pg/ml. Although the value was highly specific—every recipient whose postoperative CXCL10 levels exceeded 297 pg/ml experienced rejection—only 3 recipients (4 samples) exceeded this threshold, and therefore no definitive conclusions can be drawn at this stage. Still 1 possible pathophysiological mechanism behind this finding is that CXCL10 expression in pancreatic tissue attracts T cells and macrophages via CXCR3 receptor, 23 promoting inflammation and potentially leading to graft rejection.
For making use of CXCL10 values in diagnosing rejection, no clear correlation was found. Although the calculations only involved the samples taken on the day of or after transplant rejection diagnosis, unfortunately, our data included only 1 recipient for whom postoperative samples were taken before the date of rejection diagnosis. For that recipient, the last sample was taken 800 days before the diagnosis of rejection. Therefore, we were unable to calculate changes in CXCL10 values leading to rejection.
According to our findings, the presence of T1D associated autoantibodies before transplantation do not seem to play a role in enhancing transplant rejection. However, the postoperative presence of anti-ZnT8 autoantibodies was significantly associated with rejection episodes. These findings align with reports from other authors, indicating that the preoperative presence of any type of autoantibody 17 and the postoperative presence of autoantibodies against glutamic acid decarboxylase24,25 do not appear to influence rejection.
The presence of anti-ZnT8 autoantibodies suggests that pancreatic transplant rejection could be closely associated with the recurrence of autoimmunity against donor organ beta cells. According to Burke et al, 26 anti-ZnT8 autoantibodies are a strong risk factor for T1D recurrence. No recipient who tested negative for anti-ZnT8 autoantibodies in the postoperative period experienced a rejection episode. Hence, for recipients who were positive for anti-ZnT8 autoantibodies in the preoperative period, the disappearance of these autoantibodies after transplantation can be considered a protective factor against rejection.
For recipients from whom histological samples were obtained, acute cellular rejection was determined and the presence of anti-ZnT8 autoantibodies was retrospectively detected. It cannot be ruled out that these recipients could have experienced mixed acute T cell-mediated and antibody-mediated rejection without being histologically proven. Although T cell-mediated and antibody-mediated rejections require different treatments, 18 there is limited evidence-based information about the combined use of these treatments specifically for pancreatic grafts. Some indications suggest that in the case of T cell-mediated rejection, treatment should initially target only T cell-mediated rejection, 27 as the impact of mixed rejection on graft survival appears to be comparable to that associated with isolated T cell-mediated rejection. 28 Moreover, it has been shown that immunosuppressive therapy targeting T cells can slow the decline in C-peptide levels. 16
While our work aimed to provide insight into the principles of SPKTx, our findings must be interpreted with caution due to the relatively small sample size and no clinical conclusions can be made at this point. Nevertheless, our data gives atleast preliminary knowledge that CXCL10 and anti-ZnT8 autoantibodies can be used in managing SPKTx recipients and therefore further research into the immunological factors of SPKTx and the impact of mixed rejection on graft outcomes remains justified.
Conclusion
CXCL10 levels in individuals with long-term T1D do not significantly differ from those seen in healthy individuals. Based on our small study sample we have shown that measuring CXCL10 plasma levels could be a potential method for assessing the risk of pancreatic transplant rejection and is worth further research. Elevated CXCL10 levels during the first 3 months after transplantation seem to be a risk factor for rejection, although this elevation is not always present. In our study the post-transplant presence of anti-ZnT8 autoantibodies was significantly associated with transplant rejection. Currently, we did not find any association between pretransplant CXCL10 values or pretransplant autoantibody presence and rejection.
Supplemental Material
sj-docx-1-bmi-10.1177_11772719251381963 – Supplemental material for CXCL10 (IP-10) and anti-ZnT8 autoantibodies: Exploratory indicators of risk in pancreatic transplant rejection
Supplemental material, sj-docx-1-bmi-10.1177_11772719251381963 for CXCL10 (IP-10) and anti-ZnT8 autoantibodies: Exploratory indicators of risk in pancreatic transplant rejection by Mikk Jäätma, Marko Murruste, Karri Kase, Kaja Metsküla, Raivo Uibo and Tamara Vorobjova in Biomarker Insights
Footnotes
Acknowledgements
We thank the members of Tartu University Immunology Lab Helis Janson, Kristi Alnek and Maire Mandel for the preservation and handling of the samples used in the study and for providing us with the control group. Additionally, we extend our gratitude to the Tartu University Hospital general surgery and transplantation teams for providing the simultaneous pancreas and kidney transplants and for their continuous commitment to provide a better quality of life for our SPKTx recipients.
Ethical Considerations
This study was approved by the Ethics Review Committee on Human Research of the University of Tartu (229/M-16, 23.09.2013, 254/M-16, 21.12.2015, 298/M-21, 18.11.2019, 315/M18, 15.05.2020, 384/M-15, 20.11.2023 and 396/M-21, 16.12.2024) and was conducted in accordance with the Declaration of Helsinki.
Consent to Participate
Written informed consent was obtained from each participant before participation in the study.
Consent for Publication
All the patients and participants included in the study give their consent of using the scientific results of the study (incl. publication).
Author Contributions
M.J., T.V. and R.U. participated in the study design. M.J. and T.V. carried out data analysis and wrote the first draft and revised the final version of the manuscript; M.M. and K.K. participated in patient selection, sample collection and revision of the clinical part of the paper; K.M. participated in autoantibody analysis and in revising of the literature on autoantibodies; R.U. analyzed results in connection with other studies and revised the final version of manuscript. All authors agreed with the final version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Estonian Science Foundation grant (PRG712).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data of this study are available from the corresponding author upon reasonable request.
Limitations
The authors admit that this study has some limitations. Namely, the absence of participants undergoing SPKTx and the lack of regularity in the intervals of sample collection.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

