Abstract
Background:
Rheumatoid arthritis (RA) is complicated with interstitial lung disease (ILD). Gastroesophageal reflux disease is prevented by Helicobacter pylori infection and is a predisposing factor for idiopathic pulmonary fibrosis. However, the prevalence of H. pylori infection in RA patients with ILD has not been sufficiently investigated.
Objective:
In this study, we analyzed anti-H. pylori antibodies in RA patients with ILD.
Design:
Case-control observational study
Methods:
Anti-H. pylori antibodies were analyzed in the sera of RA patients using a commercially available enzyme-linked immunosorbent assay kit.
Results:
The positivity of anti-H. pylori antibodies in RA with ILD (n = 30 [18.0%], P = .0227), usual interstitial pneumonia (n = 10 [14.3%], P = .0212), and airway disease (n = 30 [18.0%], P = .0227) was significantly lower than that of RA without chronic lung disease (n = 78 [27.5%]). The positivity of anti-H. pylori antibodies was also lower in RA with chronic lung disease (n = 68 [18.2%], P = .0059). Multiple logistic regression analyses showed that the presence of anti-H. pylori antibodies was independently and protectively associated with chronic lung disease in RA.
Conclusion:
The seroprevalence of H. pylori was lower in RA with ILD. H. pylori infection prevented ILD in patients with RA by protecting them from gastroesophageal reflux disease.
Introduction
Rheumatoid arthritis (RA) is a chronic inflammatory disease characterized by the destruction of synovial joints; 1 although the pathogenesis of RA is unknown, it is believed to involve genetic and environmental factors. RA is complicated with extra-articular manifestations including chronic lung diseases (CLD), pleuritis, pericarditis, or vasculitis. CLD in RA mainly includes airway disease (AD), interstitial lung disease (ILD), and emphysema. 2 The complication of ILD or AD in RA confers a poor prognosis on the patient.3 -7 Thus, it is necessary to elucidate risk factors for ILD or AD in RA.
Helicobacter pylori is a gram-negative bacterium and childhood infections persist unless eradication is conducted. Chronic gastric infection by H. pylori causes peptic ulcers, 8 gastric adenocarcinoma, 9 and lymphoma. 10 However, H. pylori infection reduced the risk of asthma, 11 gastroesophageal reflux disease,12,13 and cardiovascular disease.14,15 H. pylori infection might also influence the risk of autoimmune diseases including RA, although this is still controversial.16 -23 It was suggested that the eradication of H. pylori might reduce the severity of RA.24,25 Although gastroesophageal reflux disease is a risk factor for idiopathic pulmonary fibrosis,26 -29 the prevalence of H. pylori in idiopathic pulmonary fibrosis was comparable.30,31 Furthermore, the prevalence of H. pylori in patients with AD or chronic obstructive pulmonary disease was increased.32 -34 This study aims to investigate the seroprevalence of H. pylori in RA with ILD.
Of note, the prevalence of H. pylori infection in the young is decreasing in industrialized countries, resulting in a reduced incidence of gastric cancer.35 -37 Higher anti-H. pylori antibody levels have been detected in sera from H. pylori-infected individuals, and anti-H. pylori antibody levels were decreased several months after eradication by antimicrobial therapy. 38 Thus, the measurement of serum anti-H. pylori antibodies can be used to reflect H. pylori infection. However, there are few reports on the seroprevalence of H. pylori in RA with ILD. In the present study, we investigated the seroprevalence of H. pylori in RA with ILD.
Material and Methods
Patients
A total of 657 patients with RA and 52 healthy controls (healthy individuals, mean age ± standard deviation: 35.4 ± 11.1, male number: 2 [3.8%], no matching or selection was performed.) were recruited at NHO Sagamihara National Hospital, NHO Tokyo National Hospital, NHO Miyakonojo Medical Center, NHO Himeji Medical Center, NHO Nagoya Medical Center, and NHO Nagasaki Medical Center. All the patients with RA fulfilled the Rheumatoid Arthritis Classification Criteria 39 or American College of Rheumatology criteria for RA. 40 RA patients with computed tomography images were recruited. A diagnosis of usual interstitial pneumonia (UIP, irregular linear opacities and honeycombing), nonspecific interstitial pneumonia (NSIP, bilateral ground-glass attenuation patterns predominantly in subpleural and basal regions), AD (centrilobular or peribronchial nodules, branching linear structures, bronchial dilatation, bronchial wall thickening, or atelectasis), emphysema (low attenuation area or bullae), or no CLD [CLD(−), no abnormalities in computed tomography images] was determined based on the predominant findings of chest computed tomography images of RA patients.41 -43 The CLD(+) group included patients with UIP, NSIP, AD, and emphysema. The ILD group included patients with UIP and NSIP. Steinbrocker stages of the RA patients were evaluated as previously described. 44 Because the collection of the sera was not conducted in the same year and the seroprevalence of H. pylori in the young generations is decreasing, birth year was used in the analyses.
This study was conducted in accordance with the principles expressed in the Declaration of Helsinki and was approved by the Research Ethics Committees of NHO Tokyo National Hospital (190010, 29 May 2019), NHO Sagamihara National Hospital, and the Central Institutional Review Board of the National Hospital Organization. Written informed consent was obtained from each participant.
Measurement of serum anti-H. pylori antibodies
Serum anti-H. pylori antibodies were detected by a commercially available enzyme-linked immunosorbent assay (ELISA) kit (E plate ‘Eiken’ H. pylori antibody II, Eiken Chemical Co., Ltd., Tokyo, Japan), according to the manufacturer’s instructions. The recommended cut-off level of the kit is 10 U/mL. The standard anti-H. pylori antibodies (3, 10, 30, 100 U/mL) and the horseradish peroxidase-labeled mouse anti-human IgG monoclonal antibody were used in the ELISA kit (https://www.info.pmda.go.jp/downfiles/ivd/PDF/170005_22200AMX00935000_A_01_03.pdf).
Statistical analysis
The presence of anti-H. pylori antibodies was compared with CLD(−) RA patients by Fisher’s exact test using 2 × 2 contingency tables. The clinical characteristics of RA patients with or without anti-H. pylori antibodies were compared by Student’s t-test or Fisher’s exact test using 2 × 2 contingency tables. Multiple logistic regression analysis was conducted to evaluate whether the presence of anti-H. pylori antibodies was independently associated with CLD or RA from other clinical information, including birth year, sex, Steinbrocker stage, and smoking status. Statistical significance was considered to be two-sided P-values less than .05.
Results
Detection of anti-H. pylori antibodies in RA patients and controls
Titers of anti-H. pylori antibodies were measured in the sera of RA patients (Figure 1). The presence of anti-H. pylori antibodies in each group was compared with those in CLD(−) RA patients (Table 1). The positivity of anti-H. pylori antibodies in RA with ILD (n = 30 [18.0%], P = .0227), UIP (n = 10 [14.3%], P = .0212), and AD (n = 30 [18.0%], P = .0227) was significantly lower than that in CLD(−) (n = 78 [27.5%]). The positivity of anti-H. pylori antibodies was also lower in RA with CLD (n = 68 [18.2%], P = .0059). Anti-H. pylori antibody levels in RA with CLD were also analyzed by Student’s t-test and were lower when compared with CLD(−) RA (mean ± SD, 7.3 ± 12.2 vs 10.6 ± 16.4 U/mL, P = .0048). Thus, the positivity of anti-H. pylori antibody was lower in RA with ILD, UIP, AD, or CLD.

Anti-H. pylori antibody levels in RA patients and controls.
The positivity of anti-H. pylori antibody in RA patients.
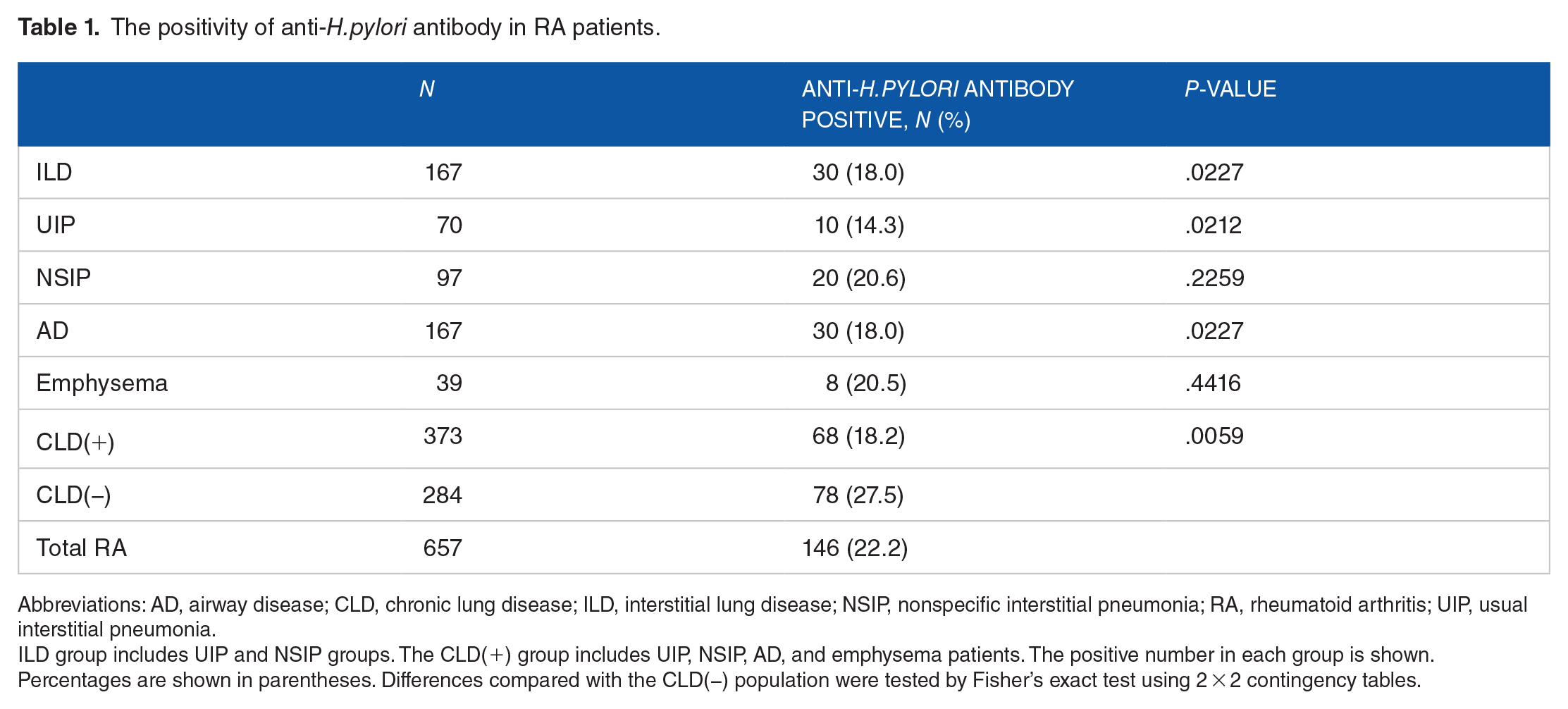
Abbreviations: AD, airway disease; CLD, chronic lung disease; ILD, interstitial lung disease; NSIP, nonspecific interstitial pneumonia; RA, rheumatoid arthritis; UIP, usual interstitial pneumonia.
ILD group includes UIP and NSIP groups. The CLD(+) group includes UIP, NSIP, AD, and emphysema patients. The positive number in each group is shown. Percentages are shown in parentheses. Differences compared with the CLD(−) population were tested by Fisher’s exact test using 2 × 2 contingency tables.
Clinical characteristics of RA patients with or without anti-H. pylori antibodies
A potential association of the clinical characteristics of RA patients with the presence of anti-H. pylori antibodies was analyzed (Table 2). CLD was associated with the absence of anti-H. pylori antibodies (P = .0059) whereas other clinical manifestations were not associated with the presence or absence of anti-H. pylori antibodies.
Association of anti-H. pylori antibody positivity with the clinical characteristics of RA.

Abbreviations: CLD, chronic lung disease; RA, rheumatoid arthritis; SD, standard deviation.
The CLD(+) group includes usual interstitial pneumonia, nonspecific interstitial pneumonia, airway disease, and emphysema patients. The number or mean of each group is shown. Percentages or SDs are shown in parentheses. Differences between anti-H. pylori antibody(+) and (−) RA populations were tested with Student’s t-test or Fisher’s exact test using 2 × 2 contingency tables.
Student’s t-test was used.
Multiple logistic regression analysis of anti-H. pylori antibodies
Multiple logistic regression analysis was conducted to exclude the influence of clinical characteristics on the protective association of the presence of anti-H. pylori antibodies with CLD (Table 3). In univariate analysis, the presence of anti-H. pylori antibodies was significantly associated with CLD (P = .0050, odds ratio [OR] 0.59, 95% confidence interval [CI] 0.41, 0.85). The association of the presence of anti-H. pylori antibodies remained significant (Padjusted = .0022, ORadjusted 0.53, 95% CI 0.35, 0.79), when conditioned on the other clinical characteristics. Thus, this suggested that the presence of anti-H. pylori antibodies was independently and protectively associated with CLD in RA.
Multiple logistic regression analysis of anti-H. pylori antibody positivity and clinical manifestations of CLD in RA.
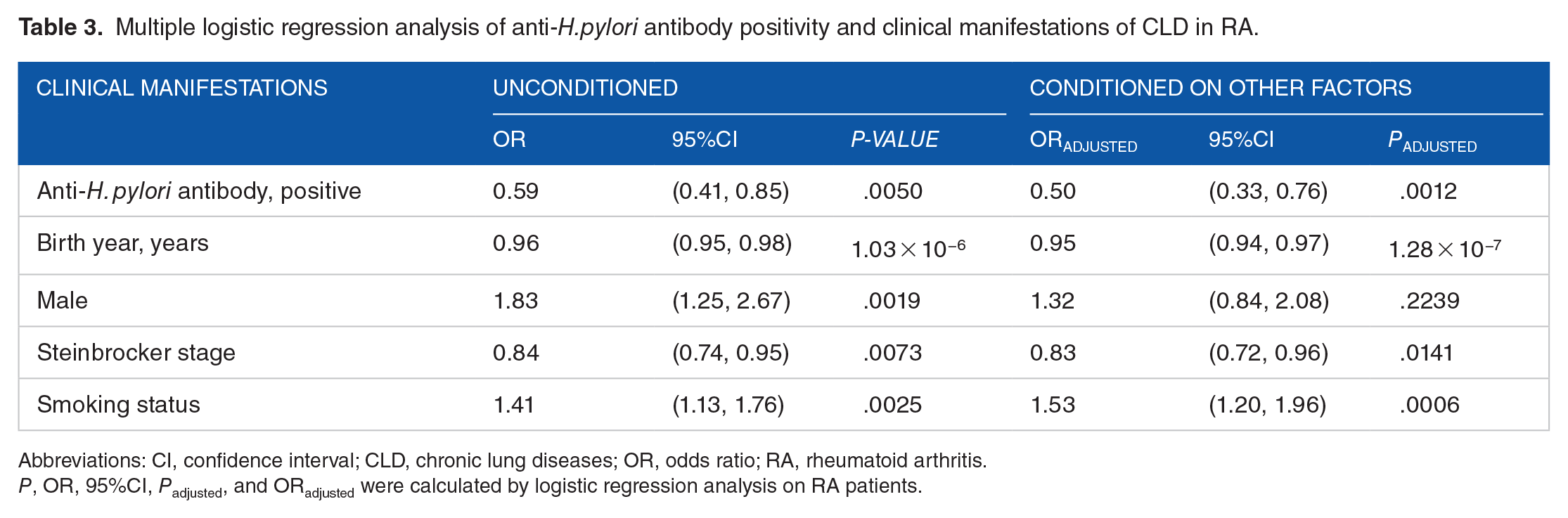
Abbreviations: CI, confidence interval; CLD, chronic lung diseases; OR, odds ratio; RA, rheumatoid arthritis.
P, OR, 95%CI, Padjusted, and ORadjusted were calculated by logistic regression analysis on RA patients.
Comparison of the presence of anti-H. pylori antibodies between RA patients and controls
The presence of anti-H. pylori antibodies was compared between RA patients and healthy controls (Supplementary Table S1), and no difference was revealed. In order to exclude the effects of other clinical features including birth year and sex on the association between the presence of anti-H. pylori antibodies and RA, multiple logistic regression analysis was conducted, and no association was detected. Thus, no association was observed between the presence of anti-H. pylori antibodies and RA.
Discussion
In this study, the positivity of anti-H. pylori antibodies in RA with ILD, UIP, and AD was lower compared with RA without CLD. We also revealed that the presence of anti-H. pylori antibodies was independently and protectively associated with CLD in RA. However, the presence of anti-H. pylori antibodies was not associated with RA, per se.
In some previous studies, the prevalence of H. pylori infection was comparable between RA patients and healthy individuals.16 -18,45 However, it was decreased in another study. 22 Furthermore, H. pylori infection increased the risk of RA in women less than 30 years old. 23 In the present study, the seroprevalence of H. pylori in RA was similar to that in healthy individuals, suggesting no association between H. pylori infection and RA, per se.
It is known that H. pylori infection prevents gastroesophageal reflux disease by decreasing gastric acid secretion.12,13 Gastroesophageal reflux disease with microaspiration was revealed to be a predisposing factor for idiopathic pulmonary fibrosis.26,27 Thus, the prevalence of H. pylori in idiopathic pulmonary fibrosis was estimated to be lower; however, it was not significantly lower than that in healthy controls, though there was a lower trend.30,31 In our study, the seroprevalence of H. pylori in RA patients with ILD was significantly lower compared with RA without CLD. Thus, H. pylori infection might prevent the development of ILD in RA probably by protecting patients from gastroesophageal reflux disease.
The prevalence of H. pylori in patients with AD was increased in previous studies.32,33 However, in this study, the seroprevalence of H. pylori in RA patients with AD was significantly lower compared with RA without CLD. These data suggested that the pathogenesis of AD in RA is different from that in AD without RA.
It was reported that the prevalence of H. pylori infection was influenced by birth year36,37 suggesting the associations between the presence of anti-H. pylori antibodies and CLD in our study could be influenced by birth year. After the effects of birth year were adjusted for in multiple logistic regression analyses, the associations between the presence of anti-H. pylori antibodies and CLD remained significant. Thus, the presence of anti-H. pylori antibody was independently and protectively associated with CLD in RA.
To the best of our knowledge, this is the first study to report the seroprevalence of H. pylori in RA patients with ILD. The results of the study indicated the lower seroprevalence of H. pylori in RA patients with ILD, UIP, AD, or CLD. This study includes some limitations. The sample size of the study was modest and this study was performed in Japanese populations. Larger-scale studies must be performed in different populations to replicate the results. Because information on gastroesophageal reflux disease in RA patients was not obtained in this study, the potential confounding effects of gastroesophageal reflux disease could not be excluded. The potential correlations between H. pylori infection, gastroesophageal reflux disease, and ILD in RA patients should be investigated in future studies with sufficient clinical information.
Conclusion
The seroprevalence of H. pylori was lower in RA with ILD. H. pylori infection prevented ILD in patients with RA by protecting them from gastroesophageal reflux disease.
Abbreviations
AD: airway disease
CLD: chronic lung diseases
ELISA : enzyme-linked immunosorbent assay
ILD: interstitial lung disease
NSIP: nonspecific interstitial pneumonia
RA: rheumatoid arthritis
UIP: usual interstitial pneumonia
Supplemental Material
sj-docx-1-bmi-10.1177_11772719241297171 – Supplemental material for Helicobacter pylori Seroprevalence in Rheumatoid Arthritis Patients with Interstitial Lung Disease
Supplemental material, sj-docx-1-bmi-10.1177_11772719241297171 for Helicobacter pylori Seroprevalence in Rheumatoid Arthritis Patients with Interstitial Lung Disease by Shomi Oka, Takashi Higuchi, Hiroshi Furukawa, Kota Shimada, Akira Okamoto, Misuzu Fujimori, Atsushi Hashimoto, Akiko Komiya, Koichiro Saisho, Norie Yoshikawa, Masao Katayama, Toshihiro Matsui, Naoshi Fukui, Kiyoshi Migita and Shigeto Tohma in Biomarker Insights
Footnotes
Acknowledgements
None.
