Abstract
Background:
Tumor-induced coagulation is widely observed in cancer patients. Moreover, it is associated with tumorigenesis, tumor progression and metastasis, by creating a proliferative and proangiogenic microenvironment. Therefore, D-dimer, a fibrin degradation product, correlates with tumor prognosis in several cancer types.
Objectives:
This study aims to investigate whether D-dimer levels can be a predictive and monitoring indicator for chemotherapy response in metastatic breast cancer (MBC) patients.
Design:
This was a prospective study.
Methods:
This study included two groups, 76 patients diagnosed with metastatic breast carcinoma and 25 patients with primary breast carcinoma. Plasma D-dimer levels were measured prospectively before chemotherapy initiation, and after the fourth treatment cycle in MBC patients. D-dimer levels before chemotherapy (D0) were analyzed using Receiver Operating Characteristic (ROC) curves to determine the optimal cut-off baseline values of D0, and to evaluate their discriminatory abilities in predicting response to chemotherapy.
Results:
In the preliminary response evaluation, the mean level of D-dimer significantly decreased by 0.65 μg/ml in patients with partial response patterns, and by 0.5 μg/ml in patients with stable disease. In the disease progression group, a marked increase was seen in D-dimer levels by 1.2 μg/ml. Analysis of ROC curves showed that D-dimer levels at D0 could discriminate the response to chemotherapy, whereas progressive disease rate correlated with higher levels of D-dimer.
Conclusion:
D-dimer level in plasma is a useful predictive and monitoring marker of response to chemotherapy in metastatic breast cancer.
Introduction
Breast cancer is still the most prevalent type of cancer in women according to the latest report GLOBOCAN 2020, and the primary cause of mortality for breast cancer patients is the occurrence of distant metastases. About 20% to 30% of breast cancers in the early stages are expected to progress into metastatic disease, while in the United States, 6% to 10% of all women suffering from breast cancer were found to have stage IV disease at initial diagnosis. 1 Breast cancer is a multi-stage, multi-complex disease that results from dysregulation of cell differentiation and apoptosis. It originates from undifferentiated terminal structures of the mammary gland followed by clonal growth of altered cells, eventually leading to epithelial hyperplasia before the final stromal invasion. The amplification and/or overexpression of transcription factors, growth factors, and their receptors, as well as the silencing of tumor suppressor genes, are some examples of molecular alterations in breast cancer development. 2
The invasion-metastasis cascade is a multistep cell-biological process that involves the migration of cancer cells to anatomically distant organ sites and their subsequent adaptation to alien tissue microenvironment producing metastases. 3
Chemotherapy is a crucial treatment option for metastatic breast cancer (MBC) in clinical practice. The need for chemotherapy is typically determined by a combination of factors, including the tumor’s subtype, features and metastatic site, the presence of visceral crisis, and any prior use of adjuvant or metastatic treatments. 4 The modern advancements in cancer biology and the rapid progress of experimental technologies in the past decade have led to an increasing number of indicators being proposed to predict treatment efficacy and clinical outcome for breast cancer patients. Recent developments in the understanding of breast cancer-related genes (bcl-2, ras, p53, etc.) have provided critical information on therapeutic effects and prognosis in addition to the general clinicopathologic aspects. However, genetic testing is not common in clinical routine practice due to the high cost and technical complexity involved with it. Therefore, new feasible and precise systemic markers must be discovered and employed as predictors of response and prognosis of breast cancer patients. 5
Cancer and the hemostatic system are associated, according to both experimental and clinical studies. 6 It has been demonstrated that coagulation and/or the fibrinolytic system are triggered either directly or indirectly in several forms of malignancies, including lung, colon, prostate, cervix, ovarian, and breast cancer, despite the fact that the precise mechanism is still unclear. Activation primarily occurs in 1 of 2 ways: either by thrombin production, which causes coagulation, or through plasminogen activators, which trigger the fibrinolytic system. 7 It has been demonstrated that tumor growth and dissemination, inflammatory cell response regulation, development of demarcation lines and tumor angiogenesis in cancer patients are all correlated with the activation of coagulation and the fibrinolytic system. 8
D-dimer is a sensitive indicator of fibrinolytic activity and is the smallest breakdown product produced by the proteolytic activation of plasmin. 7 The D-dimer level is frequently utilized as a diagnostic and therapeutic evaluation tool for thrombosis. 9 Patients with malignant tumors such as colorectal, prostate, lung, pancreatic, and ovarian cancer had poorer survival times when their D-dimer levels were elevated.10-14 Moreover, D-dimer is a biomarker for the effectiveness of chemotherapy in patients with advanced gastric, colorectal, lung, and serous ovarian cancers.15-18 Yet, the clinical significance of D-dimer levels in metastatic breast cancer is still unclear.
Therefore, we evaluated the role of D-dimer as a prognostic and predictive biomarker in patients with metastatic breast cancer who had received chemotherapy.
Materials and Methods
Study design
This prospective study (Clinical Trial ID: 59/011015/5, Syria on March 24, 2021) was conducted between September 2021 and May 2022 at the Breast cancer unit (Al Bairouni University Cancer Center).
All procedures performed in this study involving human participants were following the ethical standards of the Committee of Scientific Research Ethics at Al-Baath University, Syria (Ethics Approval Number. 2574, September 1, 2020). and carried out in accordance with the principles of the Declaration of Helsinki. A written informed consent was also obtained from all patients enrolled in the study before sample collection.
Patients
The eligibility criteria were as follows: 76 female patients attending the breast cancer unit at Al-Baironi Hospital, Damascus, Syria (from September 2021 to May 2022), with a confirmed diagnosis of metastases (stage IV, defined as stage IV disease according to the American Joint Committee on Cancer Staging System, AJCC) before commencing chemotherapy. 18 patients presented with hepatic metastasis, 14 patients with pulmonary metastasis, 8 patients with bone metastasis, 4 others with brain metastasis, and 32 patients with multiple metastases. Age varied between 30 and 82 years old (median year 53). The study also included 25 patients diagnosed with primary breast cancer (Stages I, II, and III of breast cancer) and they were used as a control group. All study participants presented adequate function of major organs (including cardiac, hepatic, and renal functions) and had no confirmed venous or arterial thromboembolism prior to chemotherapy initiation.
Exclusion criteria
Patients with venous or arterial thromboembolism, active infectious disease, cardiovascular infarction, stroke, serious wounds or fractures, previous cancer, or concomitant cancer of any origin, and pregnancy. Recent or current use of oral and parenteral anticoagulants or aspirin was not allowed.
Follow-up
The type of chemotherapy was established based on tumor histology, disease sites, previous chemotherapy administration, and presence of concomitant disease other than cancer.
Patients’ response was classified according to the response evaluation criteria in solid tumor (RECIST) (version 1.1) as follows: Complete response (CR), complete disappearance of clinical evidence of disease for a minimum of 8 weeks; partial response (PR), decrease in tumor burden ⩾ 30%; stable disease (SD); decreased in tumor burden by <30% or increased by <20%; progressive disease (PD), increase in tumor burden by ⩾20%; and nonvaluable response, due to specific reasons (eg, early death or toxicity). Disease Control (DC) was calculated based on CR + PR + SD. 19
Evaluation
Evaluation included conducting physical examination, laboratory tests, computed tomography (CT) scan of the chest/upper abdomen including adrenals, CT scan or magnetic resonance imaging (MRI) of the brain, and radionuclide bone scan or positron emission tomography/CT at initial diagnosis and after 4 therapeutic chemotherapy cycles. The related outcomes were estimated according to the Response Evaluation Criteria in Solid Tumors (RECIST) 1.1.
Methods
According to the study design scheme, D-dimer expression levels were detected before chemotherapy (D0) and at the first chemotherapy response evaluation (after the completion of the fourth cycle) (D1). A total of 3 ml whole blood was drawn from the antecubital vein of each subject and collected in sodium citrate vacutainer collection tubes. All samples were centrifuged at 2500 rpm for 15 minutes within 1 hour of blood collection. D-dimer levels were measured by a quantitative immunoenzymometric assay using AIA-900 Automated Immunoassay Analyzer by TOSOH (Japan). D-dimer Kit (ST AIA-PACK D-DIMER), catalog number 100A471001-097B.
Data collection
From these consecutive patients, different clinical and pathological variables were collected; age at diagnosis, menopausal state, type of prior systemic therapies including adjuvant treatment, histological grade, hormone receptor status, number and sites of metastases (liver, lung, bone, brain, and others), type of chemotherapy given to MBC patients.
Statistical analysis
All statistical analyses were performed using the IBM® SPSS statistical software (Version 24, USA). D-dimer levels were presented as mean ± standard deviation. Independent sample
Additionally, pretreatment plasma D-dimer levels (D0) were analyzed using ROC curve analysis to determine the optimal cut-off values for D0, and the Area Under the Curve (AUC) was calculated and compared to evaluate D0′s ability to discriminate patients’ response status to chemotherapy. The sensitivity and specificity of the plasma D-dimer level were determined. Patients were grouped into low and high D-dimer groups based on these cutoff values.
Associations of D-dimer levels with clinicopathological characteristics were analyzed by using the χ2 test. The relationships between plasma D-dimer levels and the response pattern were determined using Spearman’s correlation test and analysis of variance One Way ANOVA test. A
Results
The baseline characteristics of the included patients are shown in Table 1. The median age at diagnosis of the subjects was 53 years old (range, 30-82 years old). Of these 76 patients, 81.6% were diagnosed with invasive ductal carcinoma, 76.3% were diagnosed with estrogen receptor-positive cancer at initial diagnosis by core needle biopsy; 57.9% were progesterone receptor-positive (PR); 31.6% were Her2-positive cancer. Only 34.2% of patients were Synchronous Metastase Breast Cancer (SMBC). Table 1 includes chemotherapy protocols used as a single agent and as a combination. At the first imaging evaluation, 16 patients (21.1%) had PR, 20 patients (26.3%) had SD, 1 patient (1.3%) had CR, and 39 patients (51.3%) had PD according to RECIST criteria.
The plasma levels of D-dimer in a total of 76 MBC patients at D0 and 25 patients of the control group are shown in Table 2. D0 levels in MBC patients were significantly higher compared to the control group (1.49 vs 0.26 µg/ml;
The changes in pretreatment D-dimer levels and after 4 cycles of chemotherapy were assessed in patients and the results are shown in (Table 3 and Figure 1). At the first response evaluation, the mean D-dimer levels of PR patients and SD patients decreased by 0.65 and 0.5 µg/ml, respectively (
We compared D0 levels to evaluate chemotherapy response status for MBC patients using ROC curves (Figure 2). The mean absolute pretreatment D-dimer level was 1.49 ± 0.96 µg/ml (range, 0.44-5.4 µg/ml). The optimal cutoff value of the D-dimer level was 1.055 µg/ml, determined as the maximum point according to ROC curves with 76.9 sensitivity and 75.7 specificity and these results were statistically significant (
Patient characteristics.

Comparison of the D-dimer mean levels between the patients and control groups.

Changes in D-dimer levels in metastatic breast cancer patients before and after chemotherapy.
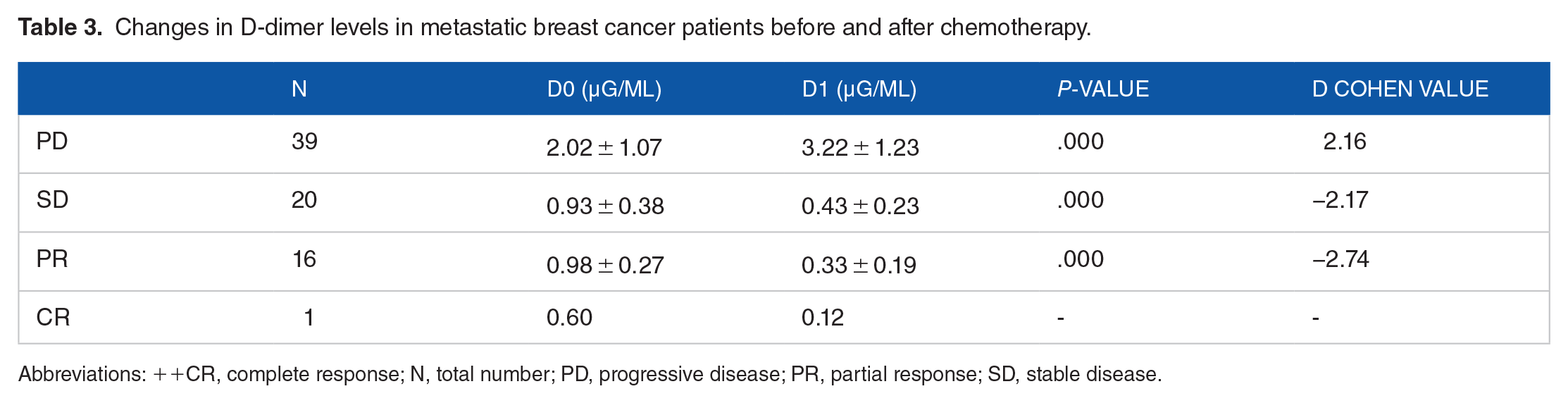
Abbreviations: ++CR, complete response; N, total number; PD, progressive disease; PR, partial response; SD, stable disease.
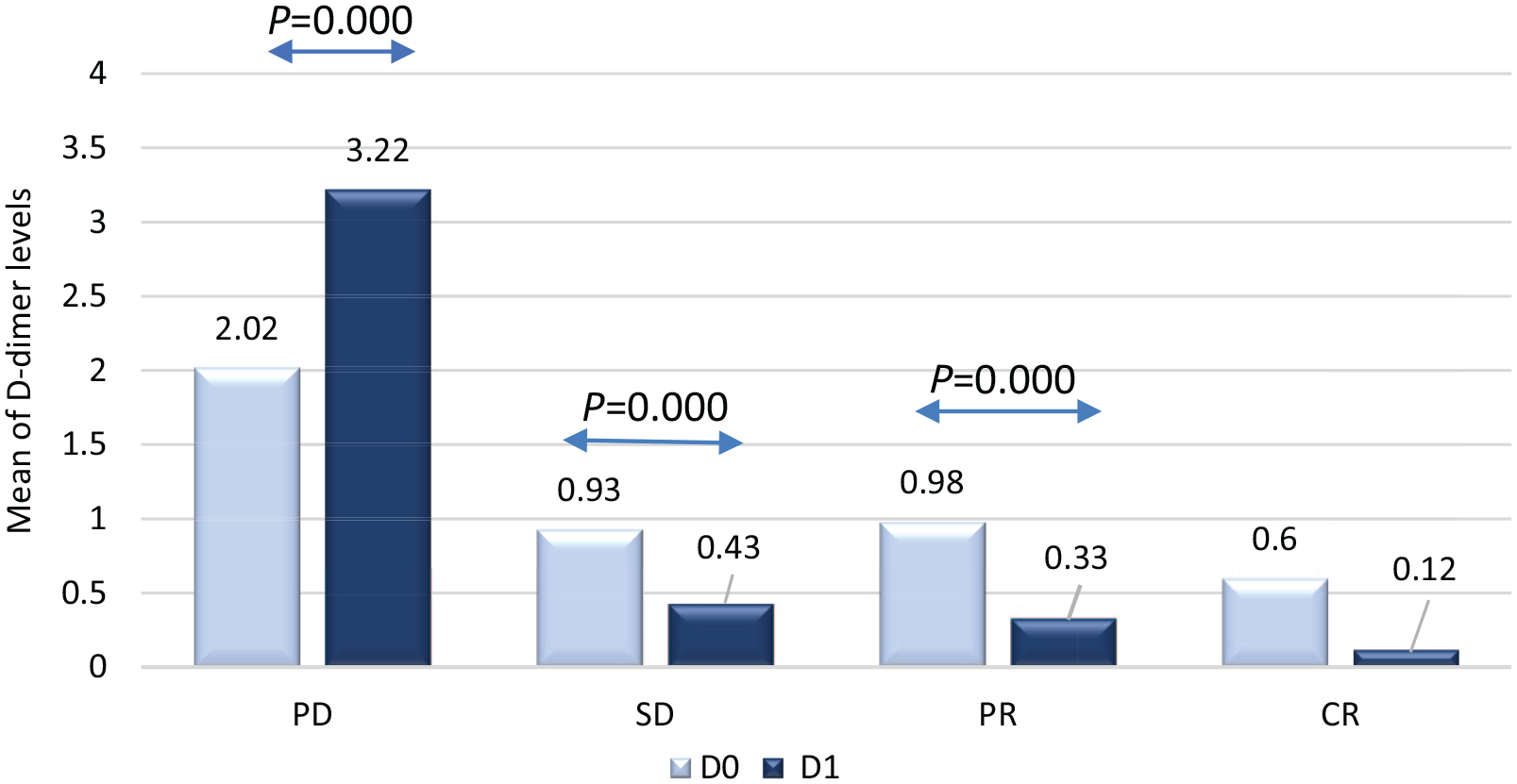
Changes in plasma D-dimer levels in MBC patients before and after chemotherapy.
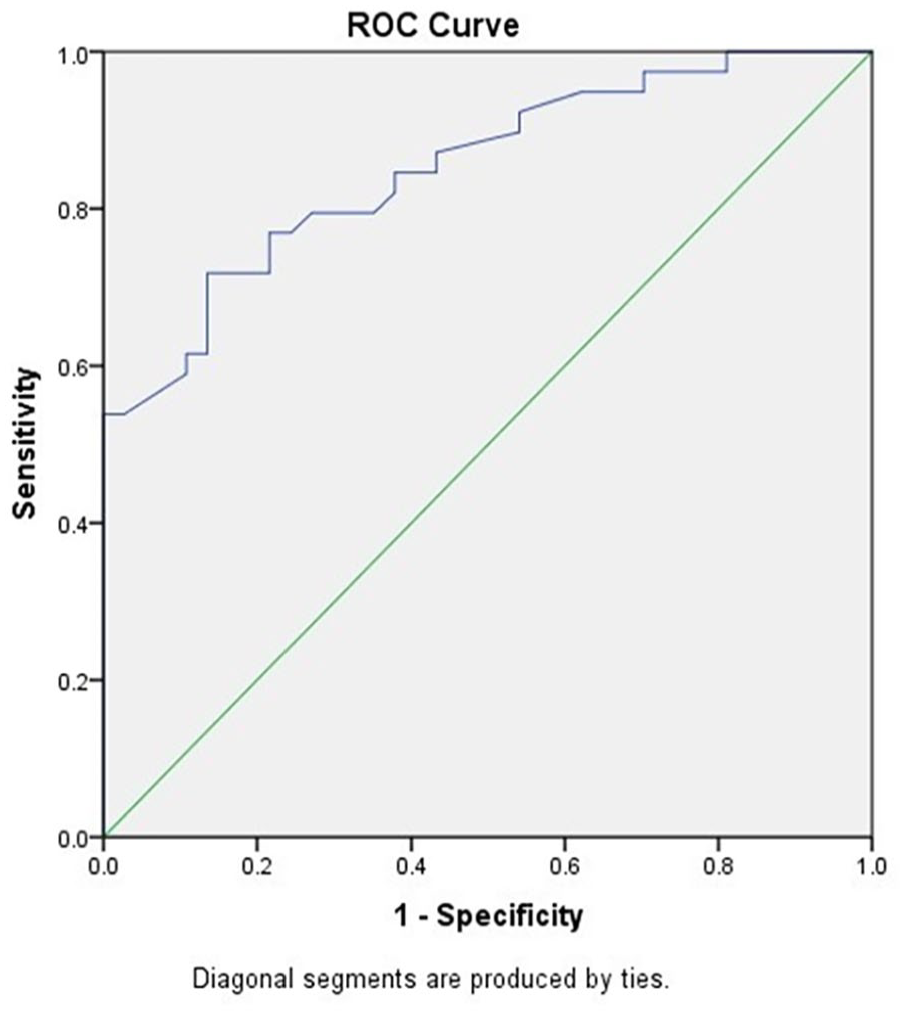
Comparison of the AUC/ROC curve in the assessment of initial D-dimer levels as a predictive value for chemotherapy response among MBC patients.
Based on Chi-square test results, there was a statistical difference (
According to ONE WAY ANOVA test results, patients with PD had considerably greater D0 and D1 than patients with SD, PR, and CR as shown in (Figure 4). These results indicated that D0 and D1 could serve as useful markers of tumor response to chemotherapy. As a result of Spearman’s test, we found a significant positive correlation between baseline D-dimer levels and chemotherapy response type (Responder, Non-Responder) (r = .610,
Turning to the correlation between tumor variables and therapeutic response type in MBC patient group and according to Chi-square test results. Firstly, for Estrogen receptor type, 18 patients (23.6%) had Estrogen receptor negative (ER-) disease, 13 patients of them experienced disease progression after treatment (
Clinical response to chemotherapy in MBC patients with pretreatment D-dimer levels greater or lower than the cut-off value.

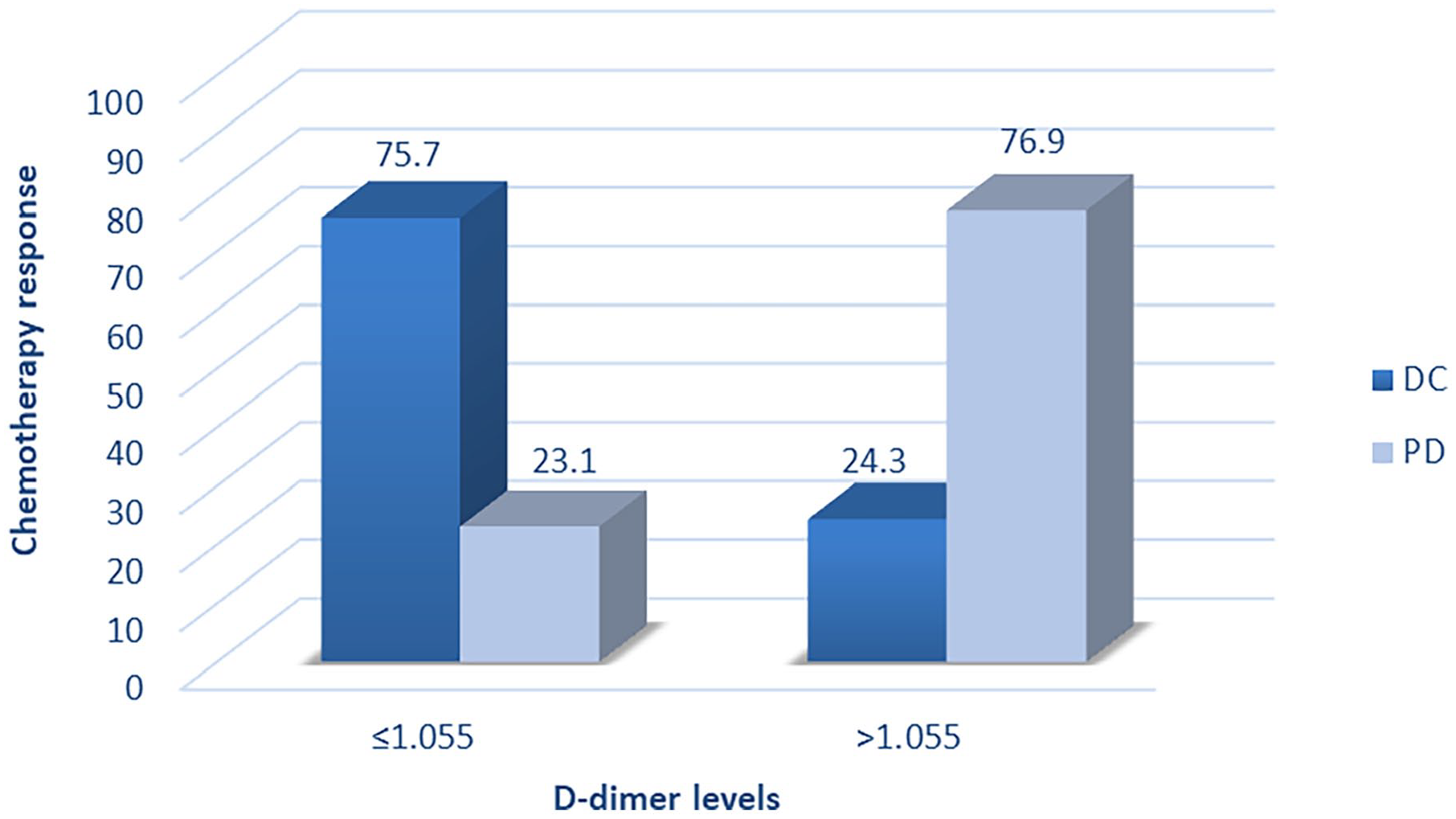
Correlation between D-dimer levels and patterns of response to chemotherapy.

Relation between D-dimer levels and chemotherapy response type.
Relation between D-dimer levels (D0 and D1) and chemotherapy response type by Spearman Test.
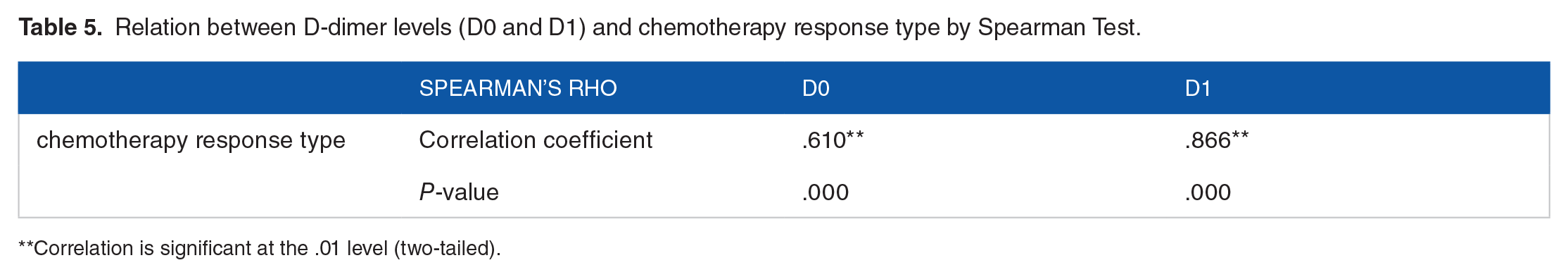
Correlation is significant at the .01 level (two-tailed).
On the other hand, regarding metastasis, 45 patients (59.2%) had 1 site of metastasis, 28 patients of them (62.22%) responded to chemotherapy (
Finally, in terms of menopausal status, 54 patients (71%) were postmenopausal and 32 patients of them (59.25%) suffered from progressive disease after treatment (
Discussion
Although mostly avoidable, thromboembolism in cancer patients continues to be a serious health concern and the second largest cause of death after cancer itself. A condition of hypercoagulability is characteristic of malignancy. Spontaneous venous thrombosis, thromboembolism following cancer surgery, thromboembolism during chemotherapy, and thrombosis of central venous access lines are clinical manifestations of thrombosis that occur in 10% to 15% of patients with clinically overt cancer. 20 through the production of procoagulant, fibrinolytic, proinflammatory, and pro-angiogenic cytokines, which result in the prothrombotic state and tumor metastasis in cancer patients. 21 among the past 20 years, the risk of Venous thromboembolism (VTE) has tripled among cancer patients and is now 9 times greater than in the general population. In the following 3 years, 10% to 20% of these patients will acquire cancer. The mortality rate of people with cancer with VTE is two- to threefold higher compared with those without VTE. 22
Furthermore, there is now a lot of data suggesting that the blood coagulation system is crucial to the biology of malignant tumors in addition to its involvement in cancer-associated thrombosis. The coagulation system interacts with cancer cells during angiogenesis, proliferation, and body-wide dissemination. In contrast, the risk of Cancer-associated thrombosis (CAT) increases many times when metastases are present.
Numerous studies have reported increased relative risks (RRs) of venous thromboembolic events in metastatic cancer patients compared with patients with localized disease for various types of cancer therefore it has been amply demonstrated that patients with metastatic breast cancer had significantly higher D-dimer levels than those with primary breast cancer.20,23
The present study confirmed that elevated plasma levels of D-dimer were associated with metastasis in patients with metastatic breast cancer. These results imply a potential role for D-dimer levels in predicting the propensity of malignancies to metastasize and progress in various cancers. Distant metastasis is the main cause contributing to cancer patients’ poor prognosis and treatment efficacy.
Moreover, the clinical importance of D-dimer level changes during chemotherapy was further supported through the analysis of the association between the therapeutic response and D-dimer level changes (D0 and D1) in this study which revealed that D-dimer levels in patients with PR and SD tended to decrease at the first response evaluation with a greater reduction degree in those with PR compared to SD patients while the levels increased in PD patients at the first response evaluation.
D-dimer’s function as a predictor of chemotherapy response has recently been reported in various cancer types. According to Inanc et al 17 analysis of colorectal cancer patients’ D-dimer levels before and after 3 cycles of CTx, patients with PR had significantly lower levels of D-dimer, but patients with PD had significantly higher levels. Moreover, Komurcuoglu et al 7 showed that lung cancer patients’ pretreatment D-dimer levels were considerably higher in nonresponders than in responders. Moreover, according to Antoniou et al 24 advanced lung cancer patients’ D-dimer levels can act as a predictive factor of the response to chemotherapy and tumor progression. These reports are in agreement with the results of this study which is the first report to suggest the clinical significance of D-dimer as a predictor for therapeutic response in patients with metastatic breast cancer.
Regarding the determination of the cut-off value in the present study, we evaluated whether D0 could be used to discriminate between responders and non-responders to chemotherapy using ROC curves. The AUC values indicated that the D0 had a stronger association with chemotherapy response. Whereas the progressive disease rate correlated with higher levels of D-dimer (>1.055 μg/ml;
These reports were in agreement with the results of Go et al 15 which reported that pretreatment D-dimer levels were higher in patients showing (PD) after CTx, but were statistically insignificant. According to Altiay et al 25 patients with locally advanced or metastatic lung cancer demonstrated a substantial correlation between plasma D-dimer levels and treatment response. Increases in D-dimer levels were linked to higher risks of progressive disease according to a research done by Khoury et al 26 on the variations in D-dimer levels in patients with refractory castration-resistant prostate cancer during treatment. Moreover, Antoniou et al 24 measured the D-dimer plasma levels in patients with advanced lung cancer before, during, and after chemotherapy, and found that a significant proportion of patients who had complete or partial therapeutic responses displayed a reduction in the D-dimer plasma values, whereas patients with progressive or recurrent disease displayed significantly increased values.
Tumor cells can change the balance between the coagulation, anticoagulation, and fibrinolytic systems through various mechanisms and eventually hypercoagulation may stem as a result of this. 27 Tissue factor (TF) contributes to cancer progression by initiating tumor growth and facilitating the spread of tumor cells. The TF/VIIa/Xa pathway can be activated downstream through the expression of TF in tumors resulting in fibrin and tumor stroma formation. Tumor cells become encapsulated with fibrin and platelet-rich thrombi and consequently are protected from the host immune defense in the microcirculation. 28 In vitro studies have shown that the TF-VIIa-Xa complex encourages breast cancer cell migration through PAR-2 signaling and by activating the mTOR (mammalian target of rapamycin) and MAPK (mitogen-activated protein kinase) pathways in the cancer cell. Additionally, TF/VIIa promotes migration by increasing the transcription of colony-stimulating factor 1 (CSF-1) and interleukin 8 (IL-8). 29
Thrombin contributes to forming a considerably more malignant phenotype by encouraging platelet-fibrin thrombin aggregation, tumor adherence to ECs and the ECM, tumor cell proliferation, and metastasis. Thrombin serves also a crucial role in angiogenesis by activating type IV collagenase, which is released by endothelial cells and therefore breaks down basement matrix proteins and collagen during neo-angiogenesis. 30
The activation of the fibrinolytic system is important for the maintenance of proper hemostasis after blood clotting activation. In the biology of cancer, activation of the uPA/uPAR system is necessary for effective localized proteolysis, adhesion, and migration as well as the invasion and metastasis of penetrating tumor cells.
Urokinase-type plasminogen activator binds to the urokinase-type plasminogen activator receptor (uPAR), which is found on tumor cells and monocytes, thus facilitating the alteration of plasminogen to plasmin. The extracellular matrix can be degraded by plasmin and this allows tumor cells and monocytes to invade the extracellular matrix and surrounding tissues which promotes invasion and metastasis. 20
In tissue biopsies taken from patients with breast cancer, Malinowsky and colleagues similarly showed a favorable connection between uPA/PAI-1 with PI3K/Akt and MAPK signaling pathway components. These mechanisms are hypothesized to stimulate tumor growth by suppressing apoptosis, hence promoting tumor proliferation. 28
Furthermore, to explain the association between changing D-dimer levels and chemotherapy resistance, it is clear that uPAR’s potential to enhance cell survival is especially significant to its action in cancer treatment. In cell culture model systems, uPA binding to uPAR prevents apoptosis by maintaining an elevated amount of phosphorylated ERK1/2. Similarly, uPAR-initiated cell signaling inhibits anoikis in vitro by activating the anti-apoptotic BCL-2 family member, BCL-xL. uPAR signaling controls BIM, a second BCL-2 family member that causes apoptosis. 31
In addition, several studies using cell culture systems have shown a significant role for the ECM in promoting cancer cell resistance to therapy. The serine protease plasmin stimulated by uPA/uPAR system helps ECM remodeling by destroying matrix proteins such as fibrin, fibronectin, and laminin. Cancer cell-derived ECM proteins (fibronectin, collagen, and laminin) protect cancer cells from chemotherapy-induced apoptosis via the activation of the PI3k/AKT pathway. 32
Matricellular proteins SPP1 and THBS1, respectively, cause resistance to apoptosis induced by cyclophosphamide and doxorubicin, 2 types of chemotherapy agents. Another ECM component, hyaluronan, activates PI3K signaling, which stimulates the multidrug transporter MDR1 and supports life via AKT. This enhances resistance to a range of chemotherapeutic treatments. 33
Plasma D-dimer is a pro-coagulation factor that may indicate the existence of circulating tumor cells or micro-metastases, which may be the cause of tumor recurrence. In patients with metastatic breast cancer, recent investigations have shown a favorable correlation between plasma D-dimer levels and circulating tumor cells. 34 Plasma D-dimer levels were a reliable predictor of prognosis in breast cancer, particularly in advanced cases. They may also be a reliable indicator of the cancer’s clinical stage progression, lymph vascular invasion, and metastasis.
Finally, based on the evidence available thus far, the inclusion of anticoagulants to traditional adjuvant treatment is particularly appealing and raises the possibility that they may have an antitumor impact. In vitro studies show that warfarin, heparin, fibrinolytic, and even antiplatelet agents inhibit tumor growth and metastasis. Since fibrin and thrombin have been linked to tumor cell adhesion and implantation, antifibrin or antithrombin medicines may work by preventing this implantation. Additionally, it has been shown that heparin inhibits factors that may aid in angiogenesis, such as tissue factor, platelet-activating factor, and vascular endothelial growth factor. Additionally, it has been proposed that fibrin deposits around tumors might provide defense against immune surveillance, as anticoagulants may help the immune system remove small amounts of cancerous cell deposits.
In order to verify the current (primarily preclinical) data, more research is required to ascertain whether vitamin K antagonists, heparin, low-molecular-weight heparin, or platelet aggregation inhibitors effectively prevent the development of metastatic tumors, taking into account various tumor types and treatment approaches.35,36
In addition, a number of published guidelines have been released regarding the prevention and treatment of VTE in cancer patients. These guidelines suggest the use of pharmacologic options, such as unfractionated heparin (UFH), low-molecular-weight heparins (LMWHs), fondaparinux, which is an indirect synthetic inhibitor of activated factor Xa, vitamin K antagonists (VKAs), and direct oral anticoagulants (DOACs), which include rivaroxaban and apixaban. Cancer patients’ treatment or prophylaxis of VTE must always strike a balance between the increased risk of bleeding caused by anticoagulants and the risk of recurrent VTE events. It must also take into account the patient’s values and preferences as well as the consequences of these outcomes, such as mortality and the financial burden and quality of life.21,22,37-39
Conclusion
D-dimer may be considered a reliable, practical, and accessible cancer biomarker. The significance of high plasma D-dimer levels as a predictor of tumor progression and suboptimal response to chemotherapy is thus extended and demonstrated by our findings. Such a conclusion should be validated in the next perspective research including larger cohorts.
Footnotes
Acknowledgements
The authors would like to thank Al Bairouni University Cancer Center staff for their assistance in conducting the research.
