Abstract
Background:
Polycystic ovary syndrome (PCOS) is a common endocrinological condition affecting women of reproductive age, associated with insulin resistance and obesity. PCOS pathogenesis is complex and multifactorial, involving genetic and environmental factors.
Objectives:
This study aimed to determine and compare genotype and allele frequencies of single nucleotide polymorphisms (SNPs) in the apolipoprotein A5 (
Design and methods:
This was a case-control study conducted on women with (
Results:
Significant and direct associations were detected between PCOS susceptibility and
Conclusion:
We identified novel alleles and genotypes contributing to the genetic risk of PCOS in the Western Saudi population.
Introduction
Polycystic ovary syndrome (PCOS) is the most common endocrinological condition in women of reproductive age. 1 The prevalence of PCOS globally is approximately 4% to 6.6% following the 1990 National Institutes of Health (NIH) criteria and approximately 4% to 21% following the 2003 Rotterdam criteria. 1 In the Middle East, the prevalence was reported as 6.1% under the 1990 NIH criteria and 16.0% under the 2003 Rotterdam criteria. 2 PCOS is characterised by excessive androgen secretion, persistent anovulation and polycystic ovarian morphology (PCOM).3 -5 The Rotterdam criteria define PCOS by the presence of at least 2 out of the following 3 criteria: irregular period, clinical or biochemical hyperandrogenism (HA), and PCOM. 6 PCOS pathogenesis is complex and multifactorial, involving genetic and environmental factors. PCOS results from unbalanced hypothalamus–pituitary–ovarian axis signalling, promoting ovarian and adrenal HA. It is complicated by insulin resistance exacerbated by HA-related adipose tissue accumulation and dysfunction. Therefore, it is accompanied by lipotoxicity and oxidative stress. 7 Moreover, a strong association exists between PCOS and increased body weight, with PCOS affecting up to 14% of women with a body mass index (BMI) of at least 30 kg/m.2,8
Multiple studies have associated PCOS with a myriad of comorbidities, including glucose intolerance, dyslipidaemia, type 2 diabetes mellitus (T2DM), hypertension, endometrial cancer, obesity, depression, anxiety, non-alcoholic fatty liver disease, sleep apnoea, eating disorders and infertility.9,10 Among these, dyslipidaemia is among the most common in patients with PCOS. 5 PCOS patients show altered lipid levels, including decreased high-density lipoprotein-cholesterol (HDL-C) levels and elevated triglyceride (TG), total cholesterol (TC) and low-density lipoprotein-cholesterol (LDL-C) levels.11,12 One recent study found that mild hypercholesterolaemia was frequently observed in patients with PCOS. 13 Dyslipidaemia in PCOS is associated with frequent insulin resistance, although not all patients with PCOS have insulin resistance. 14 The association between dyslipidaemia and insulin resistance is explained by hyperinsulinaemia inhibiting lipolysis and increasing esterified acids. Therefore, increased levels of non-esterified fatty acids may increase TGs and decrease HDL-C levels. 15
Apolipoprotein A5 (
Perilipins (
SNPs in the
Materials and Methods
Study design
This case-control observational study was conducted between 2016 and 2018 in the Obstetrics and Gynaecology Clinics at the King Abdulaziz University (KAU) Hospital and Center of Innovation in Personalised Medicine (CIPM), KAU, in Jeddah, Saudi Arabia. It included 104 women of reproductive age, between 18 and 38 years old, with PCOS diagnosed according to the Rotterdam criteria, as well as 87 women with normal ovulation (controls). Women with 1 or more of the following criteria were excluded:
A condition with reproductive symptoms similar to PCOS, such as congenital adrenal hyperplasia, Cushing syndrome, hyperprolactinemia, thyroid disease and androgen-secreting tumours.
Chronic diseases, such as diabetes and cardiovascular disease.
Any other female infertility issue.
The sample size was calculated using Raosoft (www.raosoft.com), as previously described. 25 The research was reported according to the STROBE guidelines.
All participants completed a questionnaire that collected their demographic data, family history and treatments. An abdominal ultrasound was performed on days 2 to 4 of the menstrual cycle, using a SonixTouch machine (Ultrasonix Medical Corporation; Richmond, BC, Canada). Serum levels of luteinising hormone (LH) and follicle-stimulating hormone (FSH) were obtained using an automated multi-analysis system with electrochemiluminescence immunoassay (ECLIA) kits (Roche, Basel, Switzerland). Serum anti-Müllerian hormone (AMH) levels were measured with an enzyme-linked immunosorbent assay using Ultra-Sensitive Anti-Müllerian Hormone/Müllerian Inhibiting Substance (US AMH/MIS Kit, AnshLabs, Webster, TX, USA) according to the manufacturer’s guidelines. Non-fasting serum TC and TG levels were measured using a Beckman Coulter Kit (Beckman Coulter, Inc., CA, USA) since previous studies showed no significant differences between lipid profiles obtained with fasting and non-fasting blood samples.26 -30
PCOS patients were diagnosed by the presence of 2 out of the following 3 criteria: PCOM (⩾12 follicles with a diameter of 2-9 mm in 1 ovary), irregular period (⩽8 menstrual cycles in 12 months or a menstrual interval of more than 35 days) or HA (hirsutism, acne or androgenic alopecia). BMI was calculated as the individual’s weight (in kilograms) divided by their squared height (in m2).
The study was approved by the Biomedical Ethics Unit of the Faculty of Medicine at KAU (approval number 407-15). Written informed consent was obtained from participants before sample collection. The study was conducted following the Declaration of Helsinki.
Genotyping
DNA was isolated from peripheral blood using the QIAamp DNA Mini Blood Kit (Qiagen, Hilden, Germany) according to the manufacturer’s instructions. The SNPs were genotyped using TaqMan SNP genotyping assays (Thermo Fisher Scientific, Waltham, MA, USA) with 1 to 20 ng/μL of DNA:
Statistical analyses
A normality test using the Shapiro–Wilk test showed that the data did not follow a normal distribution. Therefore, quantitative data are presented as the median (interquartile range [IQR]), and qualitative data are presented as the frequency (percentage). Associations between the study groups (PCOS and control) and qualitative variables (acne, hirsutism, hair loss, HA, regular period, PCOM and the examined SNP genotypes) were assessed using Chi-square tests. Similarly, associations between the study groups and the examined SNP alleles were assessed using Chi-square tests, with the odds ratio (OR) and relative risk (RR) values used to determine the risk alleles for each SNP. Differences in quantitative variables (age, BMI, and serum LH, FSH, LH/FSH ratio, AMH, TC and TG levels) between the study groups were assessed using Mann–Whitney
Associations between the genotypes of the examined SNPs and the qualitative variables were assessed using Chi-square tests. Because
Data analyses were performed using IBM SPSS software (version 25; SPSS Inc., Armonk, NY, USA).
Results
Associations of APOA5 and PLIN1 SNPs with PCOS
The clinical characteristics of PCOS patients and controls are summarised in Table 1. The genotype frequencies of
Clinical characteristics of PCOS patients and controls.
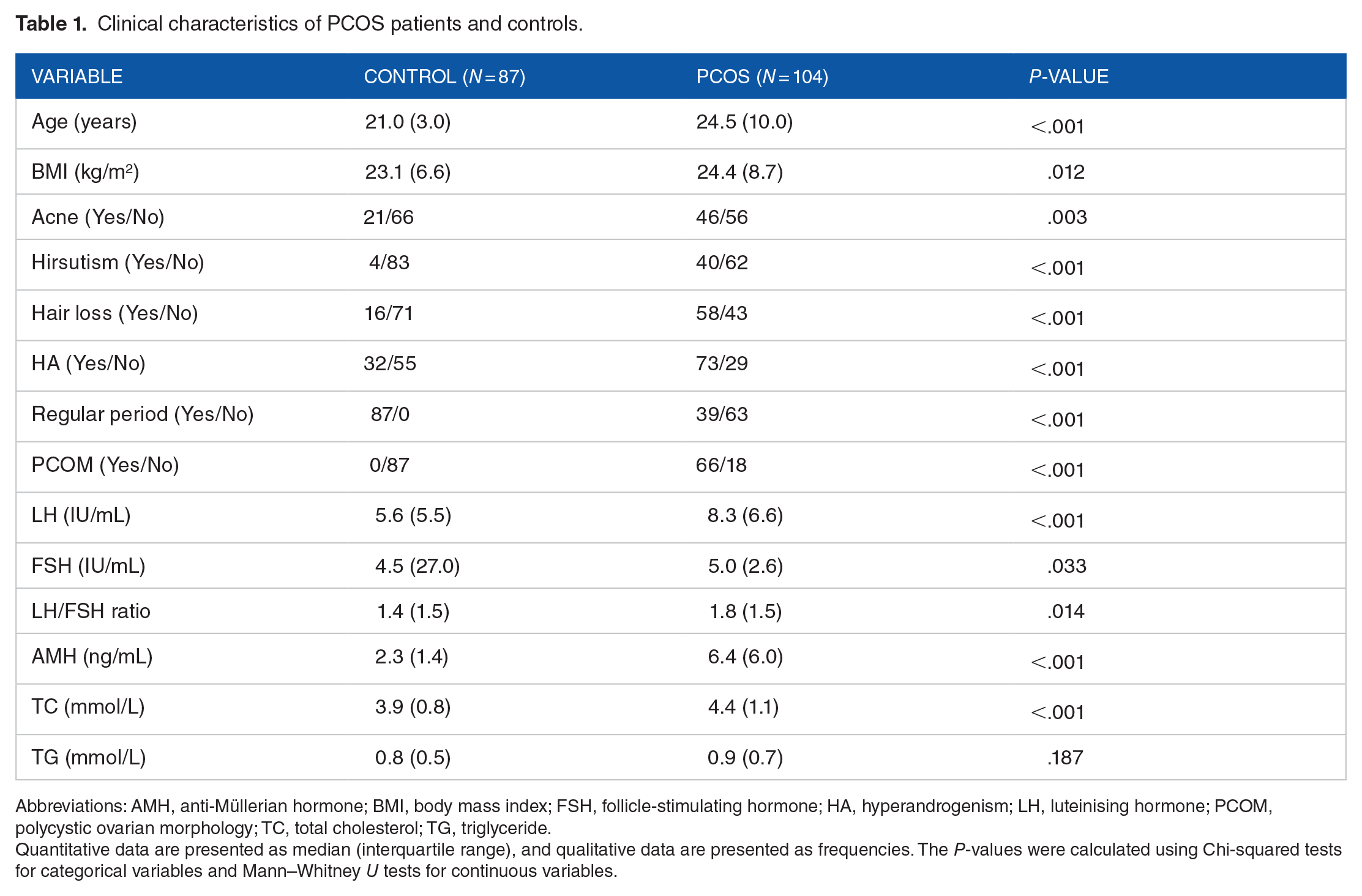
Abbreviations: AMH, anti-Müllerian hormone; BMI, body mass index; FSH, follicle-stimulating hormone; HA, hyperandrogenism; LH, luteinising hormone; PCOM, polycystic ovarian morphology; TC, total cholesterol; TG, triglyceride.
Quantitative data are presented as median (interquartile range), and qualitative data are presented as frequencies. The
Genotype frequencies for
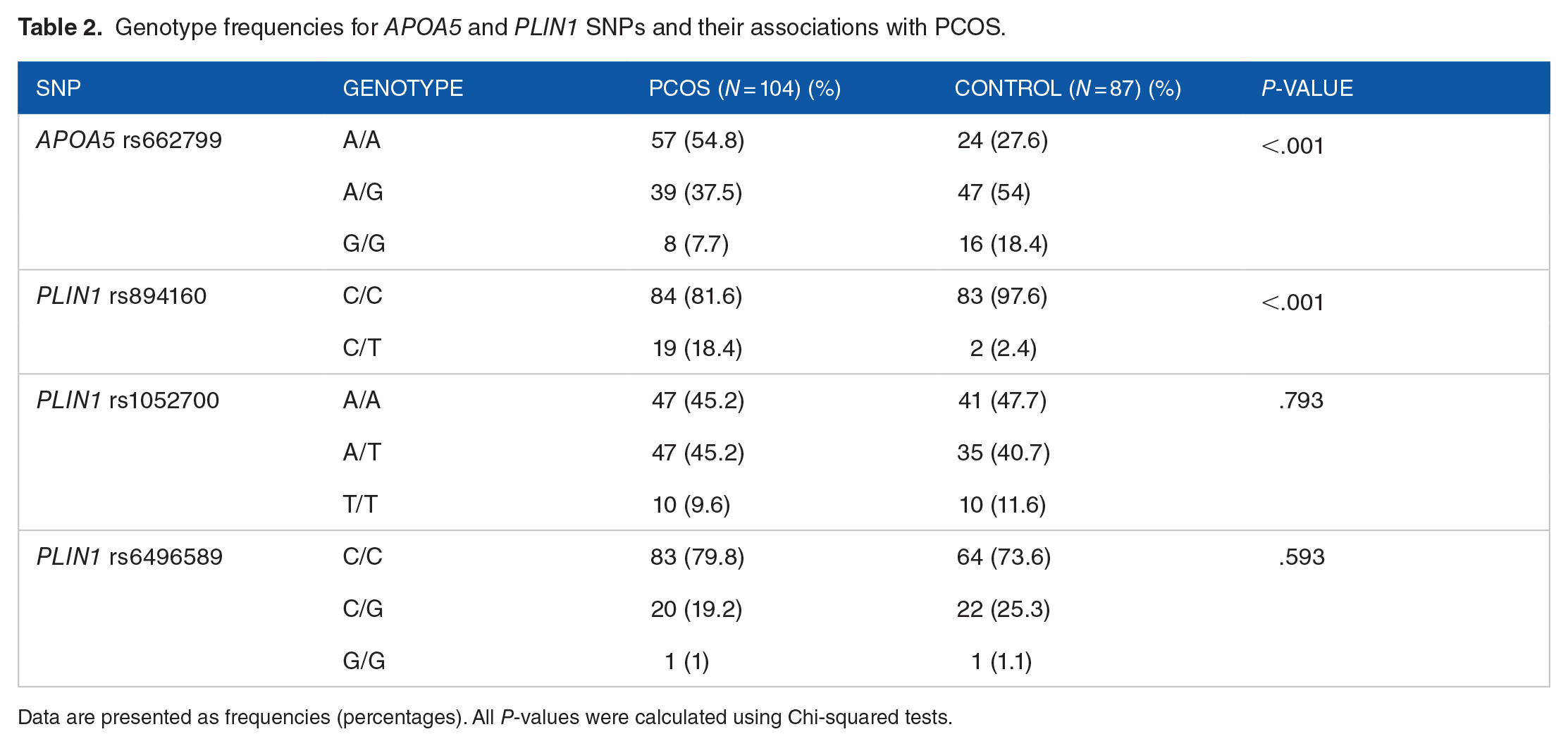
Data are presented as frequencies (percentages). All
Relative risks of PCOS for the APOA5 and PLIN1 SNP alleles
The allele frequencies of
Relative risk of PCOS for each

Abbreviations: OR, odds ratio; RR, relative risk.
Data are presented as frequencies (percentages). The
Associations between PCOS clinical characteristics and APOA5 SNP rs662799 and PLIN1 SNPs rs894160, rs1052700 and rs6496589
A significant association existed between irregular periods and
Associations between PCOS clinical characteristics and

Abbreviations: AMH, anti-Müllerian hormone; BMI, body mass index; FSH, follicle-stimulating hormone; HA, hyperandrogenism; LH, luteinising hormone; PCOM, polycystic ovarian morphology; TC, total cholesterol; TG, triglyceride.
Qualitative data are presented as frequencies and compared using Chi-squared tests. Quantitative data are presented as median (interquartile range) and compared using Kruskal–Wallis or Mann–Whitney tests.
A significant association existed between
Patients with PCOS carrying the AA genotype of the
Multinomial logistic regressions between PCOS clinical characteristics and genotypes of the examined SNPs.
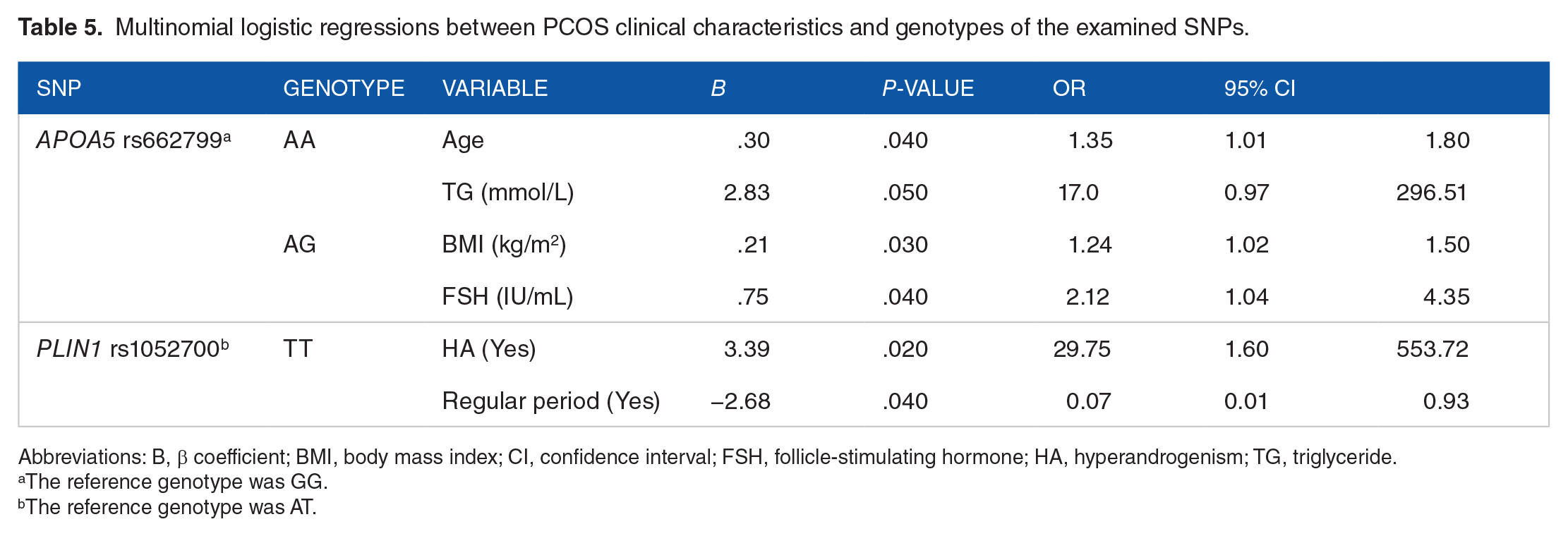
Abbreviations: B, β coefficient; BMI, body mass index; CI, confidence interval; FSH, follicle-stimulating hormone; HA, hyperandrogenism; TG, triglyceride.
The reference genotype was GG.
The reference genotype was AT.
Patients with PCOS carrying the TT genotype for
Discussion
In this study, we assessed the impact of SNPs in the
In our cohort, we found direct associations between
The second studied gene,
Our study revealed no direct association between
Our study found no significant association between
Conclusion
Our case-control study demonstrated that the frequencies of the A allele of
