Abstract
Background:
Colorectal cancer (CRC) has a high rate of recurrence, in particular for advanced disease, but prognosis based on staging and pathology at surgery can have limited efficacy. The presence of circulating tumor DNA (ctDNA) at diagnosis could be used to improve the prediction for disease recurrence.
Objectives:
To assess the impact of detecting methylated BCAT1/IKZF1 ctDNA at diagnosis in combination with demographic, lifestyle, clinical factors and tumor pathology, to assess predictive value for recurrence.
Design:
A retrospective cohort study.
Methods:
The cohort included 180 patients (36 with recurrent CRC), who had undergone complete treatment and surveillance for a minimum of 3 years. Participant clinical details and ctDNA methylated BCAT1/IKZF1 results were compared between those with and without recurrence, and cox regression analysis assessed each factor on disease-free survival.
Results:
Clinical factors independently associated with reduced disease-free survival included nodal involvement (HR = 3.83, 95% CI 1.56-9.43, P = .003), M1 stage (HR = 4.41, 95% CI 1.18-16.45, P = .027), a resection margin less than 2 mm (HR = 4.60, 95% CI 1.19-17.76, P = .027), perineural involvement (HR = 2.50, 95% CI 1.01-6.17, P = .047) and distal tumors (HR = 3.13, 95% CI 1.07-9.18, P = .037). Methylated BCAT1/IKZF1 was detected in 51.7% (93/180) of pre-treatment plasma samples. When a positive ctDNA finding was considered in combination with these clinical prognostic factors, there was improved predictive power of recurrence for patients with perineural involvement (HR = 4.44, 95% CI 1.92-10.26, P < .001), and it marginally improved the predictive factor for M1 stage (HR = 7.59, 95% CI 2.30-25.07, P = .001) and distal tumors (HR = 5.04, 95% CI 1.88-13.49, P = .001).
Conclusions:
Nodal invasion, metastatic disease, distal tumor site, low resection margins and perineural invasion were associated with disease recurrence. Pre-treatment methylated ctDNA measurement can improve the predictive value for recurrence in a subset of patients, particularly those with perineural involvement.
Registration:
Australian and New Zealand Clinical Trials Registry #12611000318987.
Introduction
Colorectal cancer (CRC) is currently the third leading cancer diagnosed worldwide with a high incidence and mortality rate. It remains a prevalent disease contributing to 2 million new cases in 2020 and was the second most common cause of cancer death worldwide with almost 1 million deaths. 1 Adenocarcinomas that arise in the colon or rectum are heterogeneous, arising from the accumulation of genetic and epigenetic alterations that develop over many years. 2 Over the last two decades, a deeper understanding of the molecular pathway of CRC, improved pathological staging, advances in chemotherapy treatments and surgical techniques have improved CRC outcomes. However, there remains a high risk of CRC recurrence particularly for patients with stage II-IV disease, with 30% to 50% of patients experiencing disease recurrence after a curative resection. 3 Most recurrence is observed within the first 3 years of resection, 4 and 90% are identified within 4 years. 5 Delayed detection of recurrence is associated with poor survival, 6 reduced quality of life, and a greater burden of cost on public health system. Consequently, there has been increased interest in developing more sensitive methods for post-treatment surveillance of CRC patients, with the view that early detection and treatment of recurrence can improve patient outcomes and survival. Improving identification of those patients who are at greatest risk for recurrence, and those who would benefit from more intensive surveillance, will potentially lead to better patient outcomes.
There are a range of clinicopathological factors at diagnosis that are significantly associated with risk of CRC recurrence, such as presence of metastatic disease, 7 lymphovascular invasion, nodal involvement 8 and perineural involvement. 9 Furthermore, other demographic factors have also been identified as predictive of recurrence. Steele et al 10 showed that patients diagnosed with CRC at a younger age (<50 years) were more likely to develop a recurrence compared to patients aged over 50 years. However, the impact of other patient factors including lifestyle factors on the risk recurrence of CRC, remains largely unexplored. 11
The current approach to post-operative surveillance involves regular blood testing for carcinoembryonic antigen (CEA), CT imaging, and colonoscopies, 12 particularly for those at greater risk of recurrence with stage II or more advanced CRC. However, these measures can be invasive, costly, and have failed to show a definitive benefit,13,14 with survival remaining low. 15 Importantly, CEA, the guideline recommended blood biomarker, has low specificity and sensitivity at predicting recurrence of CRC,16-18 particularly as it is influenced by poor renal function, hypothyroidism, obstructive pulmonary disease, obesity, aging, and smoking. 19 Emerging research on circulating tumor DNA (ctDNA) is evolving as a promising surveillance tool for CRC. Several studies have found that ctDNA can be used to monitor tumor burden and identify residual disease, and that persistently elevated levels post-surgery may be indictive of greater risk of CRC recurrence.20-22 One such example is through detecting ctDNA methylated in branched chain amino-acid transaminase 1 (BCAT1) and/or IKAROS family zinc finger 1, (IKZF1). These 2 genes are extensively hypermethylated in adenoma and CRC tissue. 23 IKZF1 is a hematopoietic DNA-binding transcription factor that has a crucial role in regulating lymphocyte and myeloid differentiation. 24 Moreover, it regulates cell-cell interactions by activating the Notch-signaling pathway.25,26 In comparison, the cytosolic isoform of BCAT1, regulates the degradation of amino acids that is pivotal for cellular metabolism and growth. 27 Importantly, there is growing evidence indicating that BCAT1 promotes cell proliferation, interferes with cell cycle progression, differentiation and apoptosis.28-30 Hence, the disordered regulation of BCAT1 and IKZF1 has perpetuated the growth of a several solid cancers including endometrial, 28 CRC, 31 liver, and lung cancer. 32 In CRC, these biomarkers can be detected in up to 95% of tumor tissues 33 and in approximately 62% (ranging from 49% to 74%) of plasma samples from patients with CRC. 34
Identifying ctDNA post-operatively is associated with greater risk for recurrence of CRC and has greater sensitivity than CEA, 17 especially with more advanced TNM and AJCC staging. However, these studies relied on prolonged monitoring of ctDNA post-treatment to determine relapse and little is known about the impact of pre-treatment ctDNA in predicting recurrence.
Thus, despite the advancement identifying predictive factors for CRC recurrence, a robust predictive multifactorial model that can be used in routine clinical practice is lacking. For example, nodal involvement has previously been shown to be a risk factor for future CRC recurrence, but despite some patients having nodal involvement at diagnosis only 41% of them will develop recurrence. 35 Thus, clinical care of CRC patients post-operatively could benefit from improvement of recurrence risk prediction. This could be achieved with a model that includes new and known risk factors, or through the addition of ctDNA blood biomarkers. As the levels of pre-treatment methylated BCAT1 and IKZF1 ctDNA have been shown to be significantly associated with stage of cancer34,36 and tumor burden, 21 it is possible that the presence of these ctDNA biomarkers can also be used for prognosis assessment to supplement the information gained from known risk factors. Therefore, the first aim of this study was to explore a range of patient factors such as demographics, lifestyle factors, medications, medical conditions, as well as tumor pathologies and blood markers, to assess their efficacy in predicting recurrence. Following this assessment, the second aim was to determine if detection of ctDNA methylated in BCAT1/IKZF1 at diagnosis further improved the prediction of those individuals at greatest risk for CRC recurrence.
Methodology
Overview
In this retrospective study, the cohort was selected from cases diagnosed with CRC who were undergoing surveillance for disease recurrence. The manuscript was prepared and revised according the Strengthening the Reporting of Observational studies in Epidemiology (STROBE) guidelines. A period of 3 years surveillance was required as most disease recurrences will be detected within this timeframe. 4 Cases were monitored according to institutional practice and current national guidelines using clinical information from physicians, CT imaging, colonoscopy results and blood samples. Blood samples were analyzed for methylated BCAT1 and/or IKZF1 (ctDNA) and routine clinical blood tests, including CEA. Blood test results prior to treatment commencement, patient demographics and clinical factors were included in the analysis to identify factors that were prognostic for future disease recurrence.
Part of this cohort has been previously reported in a study that assessed post-treatment ctDNA of disease free survival. 21
Demographics of study population
The study cohort comprised of 180 patients undergoing treatment for primary CRC (adenocarcinoma) at Flinders Medical Centre (South Australia) from September 2011 to December 2018. Each patient had undergone at least 3 years of regular surveillance after initial treatment, including regular clinical and blood assessment, colonoscopies, and CT scans, supplemented by additional imaging as required.
Cases were eligible for inclusion if they had invasive CRC, adequate staging, and ctDNA blood samples collected prior to treatment for methylated BCAT1 and IKZF1 analysis. Cases excluded were those with stage IV disease who did not have curative resection. In addition, patients who did not undergo surgery to verify that they were in remission and those with less than 3 years of surveillance, or deceased prior to surgery, were excluded from the analyses.
Data collection
Clinical and pathological parameters such as medications, symptoms, histopathology, and imaging, as well as routine blood analysis (CEA, electrolyte, urea, creatinine, and complete blood profile) and BCAT1/IKZF1 ctDNA methylation results were collected. Demographic and lifestyle data collected included age, sex, smoking status, alcohol intake, body mass index (BMI), and any concurrent comorbidities. These details were collected through patient interview or retrieved from electronic hospital databases.
Staging of the tumor was defined based on the TNM staging and AJCC stage (AJCC guidelines version 8) 37 which was verified with clinicopathological findings obtained during surgery. However, for patients with rectal tumors where neoadjuvant therapy was given, staging was determined from pre-treatment staging MRI scans. For synchronous cancers, designated stage was defined as the most advanced lesion. Proximal CRC comprised tumors proximal to the splenic flexure, with the remainder classified as distal colon or rectum.
Medical history for cardiovascular conditions (including ischemic heart disease, arrythmia, heart failure and peripheral vascular disease), respiratory conditions (including asthma, chronic obstructive pulmonary disease, and lung fibrosis), and renal conditions (including acute and chronic kidney failure, glomerulonephritis, and renal artery stenosis) were collected. The frequency and quantity of alcohol consumption (self-reported) were classified according to the national standard of alcohol intake in Australia, 38 which included non-drinker, light (below the national standard) moderate (equivalent to the national standard) and heavy (exceeding the national standard). Smoking status was also self-reported and was classified as no smoking history, previous smoker, or active smoker. BMI (kg/m2) was classified as normal (BMI < 25 kg/m2), overweight (BMI ⩾ 25 and <30 kg/m2) and obese (BMI ⩾ 30 kg/m2)
Analysis of plasma for ctDNA
Venous blood was collected before initiating therapy and cell-free circulating DNA (ccfDNA) was extracted from 3.9 to 4.5 mL plasma for analysis of ctDNA methylated for BCAT1 and IKZF1, as previously reported. 34 Briefly, following ccfDNA extraction and bisulfite conversion, a real-time quantitative multiplex PCR assay was used to determine the amount of methylated BCAT1 and IKZF1 DNA, with measurement of ACTB used to ensure adequate ccfDNA in the sample. All samples were analyzed in triplicate. Detection of methylation in either BCAT1 or IKZF1 was recorded as positive for ctDNA.
Statistical analyses
Continuous variables are presented as mean ± standard deviation (SD) if normally distributed, whereas the non-normally distributed variables are median with interquartile range (IQR). Pearson’s chi-square was used to compare categorical variables.
Univariate cox regression analysis was used to assess the effect of clinical and demographic variables on the time to recurrence, with hazard ratio (HR) indicating the degree of associations and 95% confidence intervals (95% CI) indicating the variation of the association. A multivariable survival analysis using a cox regression was performed to compare the disease-free survival (DFS) for factors harboring a P < .05 in the univariate analysis, or those of clinical interest (eg, sex, age), with recurrence as the failure event. Cases that were missing variables that were commonly associated with risk for recurrence (eg, resection margins) were excluded from the multivariable analysis. Survival plots were presented as Kaplan-Meier survival plots. Significant predictive factors identified in the multivariable analysis were then evaluated in combination with the methylated ctDNA result using univariate cox regression and the survival was plotted using a Kaplan-Meier curve.
All statistical data were analyzed using with STATA (v16.0, StataCorp LLC, TX, USA). Tests with P values less than .05 were deemed significant.
Results
Patient population
Following exclusions, there were 180 patients eligible for inclusion, including 102 (57%) males and 78 (43%) females with invasive CRC (Figure 1). The median age was 68.2 years (IQR 58.6-76.6 years). Most patients were diagnosed with AJCC stage III (n = 70, 39%), with the remaining patients having stage I (n = 50, 28%), stage II (n = 55, 30%), and stage IV (n = 5, 3%) CRC with curative resection. More patients had distal colon or rectal tumors (n = 106, 59%), compared to 41% of the cohort with tumors in the proximal colon (n = 74).

Study inclusion and exclusion criteria.
Of this cohort, 36/180 (20%) of participants developed recurrence within the surveillance period, with the remaining 144/180 (80%) disease-free and alive during that period. The median disease-free period in the recurrence group was 19.5 months (IQR 15.7-30.5 months) and the range was from 2.8 to 90.5 months. Most patients developed distant recurrence (n = 31, 86%), with only 5 cases (14%) developing a loco-regional recurrence. The distant metastatic disease occurred mainly in the lung with distant nodal involvement (n = 13, 42%) and liver with distant nodal involvement (n = 9, 29%) and some patients had concurrent liver and lung recurrence (n = 3, 10%). The remainder of metastatic disease spread to other peritoneum (n = 2, 6%), distant nodes only (n = 2, 6%), liver and brain (n = 1, 3%) or liver and peritoneum (n = 1, 3%) concurrently. With the non-recurrence cohort, the median period of follow up was 81.7 months (IQR 62.6-98.3 months) and the range was 38.2 to 127.1 months. There were no significant differences between the age or sex of the patients with and without recurrence during the follow-up period (P > .05, Table 1).
Demographic factors in individuals who developed recurrence compared to those that remained disease free.

Abbreviations: BMI, body mass index; SD, standard deviation.
Alcohol intake classification as described in methodology.
Association of patient and clinical factors with recurrence of CRC
Patient demographic and lifestyle factors were assessed by univariate analysis to determine if there was a significant association with CRC recurrence. Recurrence was not associated with any of the measured lifestyle factors, including elevated BMI, smoking status or alcohol intake (P > .05) (Table 1).
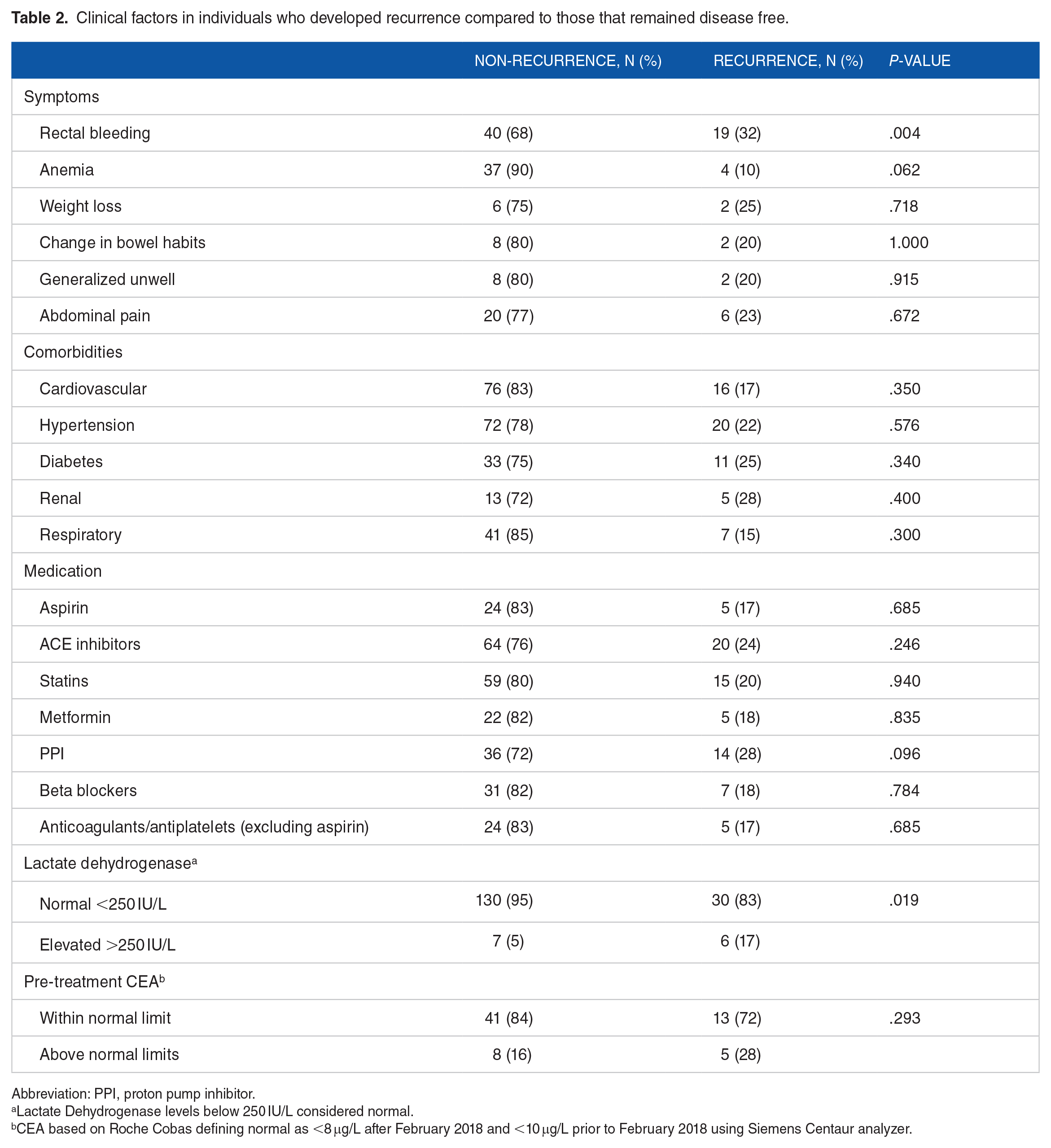
The patient clinical parameters were also reviewed for associations with future recurrence, including the indications at diagnosis, concurrent medical conditions, and medications (Table 2). Per-rectal bleeding was significantly associated with recurrence (P = .004); however, no other symptoms, comorbidity or medication were associated with CRC recurrence (P > .05).
Clinical factors in individuals who developed recurrence compared to those that remained disease free.
Abbreviation: PPI, proton pump inhibitor.
Lactate Dehydrogenase levels below 250 IU/L considered normal.
CEA based on Roche Cobas defining normal as <8 μg/L after February 2018 and <10 μg/L prior to February 2018 using Siemens Centaur analyzer.
It was observed that patients with future recurrence were more likely to have an elevated lactate dehydrogenase at diagnosis (LDH > 250 IU/L; an enzyme produced during anaerobic metabolic pathway and frequently elevated in cancer cells 39 ) compared to those without recurrence (P = .019), however, there were no differences in pre-treatment CEA positivity rate between the patients with and without recurrence (P > .05, Table 2).
Association of tumor pathology and risk of recurrence
Several tumor pathology markers were more prevalent in individuals who later developed CRC recurrence. Individuals with recurrence were more likely to have been diagnosed with a primary distal colonic or rectal cancer (31/36, 86%) compared to those without recurrence (75/144, 53%, P < .001) (Table 3). In addition, those with diagnosis at an advanced stage of disease (stage III and IV) were more likely to later develop recurrence compared to those with a diagnosis at early stage (9% vs 36% respectively).
Tumor pathologies in individuals who developed recurrence compared to those that remained disease free.

Abbreviations: EMVI, extramural vascular invasion; IMVI, intramural vascular invasion.
Overall, in the cohort, predominantly moderately differentiated tumors were observed (n = 134, 77%), with the remaining having well differentiated (n = 15, 9%) and poorly differentiated (n = 25, 14%); with most having nil mucinous features (n = 145, 81%). However, grade of tumor and mucinous features were not linked with recurrence (P > .05; Table 3).
Most patients had more than 12 lymph nodes examined during surgery (n = 141, 81%) with a mean of 18 lymph nodes assessed. There was no recurrence in patients with T1-stage, but overall T stage was linked with recurrence (P = .012; Table 3). Nodal involvement (N1 stage, 27/36, 75%) and metastatic disease (M1 stage, 4/36, 11%) were significantly associated with diagnosis of recurrence (P < .01).
Most patients in our cohort had a surgery to remove the tumor with majority having a right hemicolectomy (n = 74, 41%), high anterior resection (n = 36, 20%) or a low anterior resection (n = 32, 18%). The remainder of patient either did not have any resection of the tumor (n = 7, 4%) or had other procedures including; abdominoperineal resection (n = 9, 5%), ultra-low anterior resection (n = 7, 4%), extended right hemicolectomy (n = 6, 3%), left hemicolectomy (n = 4, 2%) subtotal colectomy (n = 4, 2%) or ileocecal resection (n = 1, <1%). However, the type of surgery was not a predicator for recurrence when adjusted for tumor location (P > .05).
Other significant pathological markers assessed included extramural vascular invasion (EMVI), which was more prevalent in the recurrence cohort (11/36, 33%) compared to the those without recurrence (16/144, 12%; P = .004) (Table 3). Likewise, a greater proportion of the patients with perineural involvement developed recurrence (10/36, 28%) compared those that did not have recurrence (12/144, 8%; P = .002) (Table 3). Similarly, a significantly greater proportion of patients with lymphovascular invasion and colorectal tumor deposits developed recurrence (n = 17, 50% and n = 8, 27% respectively) compared to those that did not develop recurrence (n = 31, 22% and n = 7, 5% respectively) (P < .01).
Association of methylated ctDNA and risk of recurrence
The presence of methylated ctDNA at diagnosis (prior to any treatment) was higher in patients with a future recurrence (n = 22, 61%) compared to those without a recurrence (n = 71, 49%), however, this did not reach statistical significance (P = .205). When each gene in the methylated ctDNA test was assessed individually, BCAT1 was present in 47% (n = 17) of patients with recurrence, compared to 40% (n = 57) of patients without recurrence (P = .405). Similarly, IKZF1 was present in the plasma of 42% (n = 15) of patients that had recurrence compared to 36% (n = 52) of those without recurrence (P = .537).
Predictive variables for recurrence identified via multivariable analysis
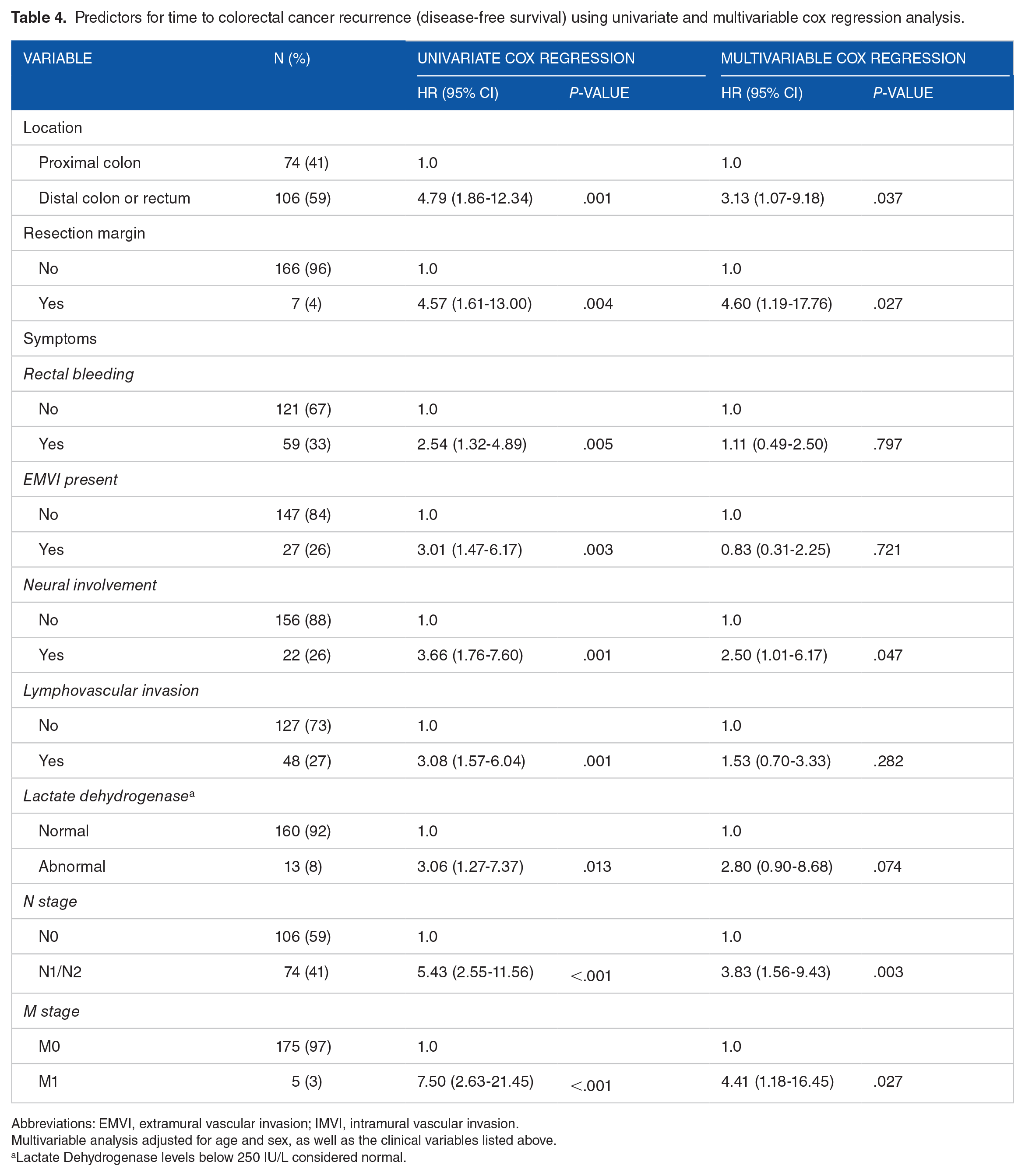
Analysis for DFS (P > .05; Table 4) with univariate Cox regression analysis showed that variables including tumors located in the recto-sigmoid colon (HR 4.79, P = .001), a resection margin less than 2 mm (HR = 4.57, P = .004), rectal bleeding (HR = 2.54, P = .005), EMVI presence (HR = 3.01, P = .003), perineural involvement (HR = 3.66, P = .001), lymphovascular invasion (HR = 3.08, P = .001), nodal involvement (HR = 5.43, P < .001), metastatic disease (HR = 7.50, P < .001) and LDH (HR = 3.06, P = .013) were all statistically associated with reduced DFS (Table 4). T stage was not significantly associated with DFS (P > .05).
Predictors for time to colorectal cancer recurrence (disease-free survival) using univariate and multivariable cox regression analysis.
Abbreviations: EMVI, extramural vascular invasion; IMVI, intramural vascular invasion.
Multivariable analysis adjusted for age and sex, as well as the clinical variables listed above.
Lactate Dehydrogenase levels below 250 IU/L considered normal.
Following adjustment for age and sex, multivariable cox regression analysis for DFS revealed that patients with nodal involvement and metastatic disease (HR = 3.83, 95% CI 1.56-9.43, P = .003; and HR = 4.41, 95% CI 1.18-16.45, P = .027, respectively; Table 4), had a reduced DFS. Likewise, location of tumors in the recto-sigmoid (HR = 3.13, 95% CI 1.07-9.18, P = .037), a resection margin less than 2 mm (HR = 4.60, 95% CI 1.19-17.76, P = .027) and perineural involvement (HR = 2.50, 95% CI 1.01-6.17, P = .047) remained statistically significant and thus concomitant to a shorter DFS. Kaplan-Meier survival curves are shown for these risk factors in Figure 2.

Kaplan-Meier survival curves for disease-free survival in association with: location of primary tumor (A), resection margins (<2 mm) (B), presence of nodal disease (N stage) (C), presence of metastatic disease (M stage) (D), and existence of perineural invasion (E).
Evaluation of significant prognostic factors in combination with methylated ctDNA
The diagnostic ctDNA result was assessed to examine whether it could improve the prognostic factors deemed significant in the multivariable analysis (Figure 3). Distal colon and rectal tumors with positive ctDNA (HR = 5.04, 95% CI 1.88-13.49, P = .001) showed a marginal increase in the hazard ratio compared to negative ctDNA in individuals that developed recurrence (HR = 4.45, 95% CI 1.57-12.64, P = .005).

Kaplan-Meier survival curves for disease-free survival in association with: distal colon and rectal tumors combined with circulating tumor DNA (ctDNA) compared to the proximal colon tumor location (A), presence of metastatic disease taking into consideration ctDNA (B), compared to no metastatic disease (M0); presence of nodal disease (N1/2 stage) taking into consideration ctDNA compared to no nodal involvement (N0 stage) (C), resection margin <2 mm compared to margin > 2mm considering ctDNA (D), and perineural invasion taking into consideration ctDNA compared to no perineural invasion (E).
Likewise, the addition of positive ctDNA with metastatic disease was significantly associated with recurrence (HR = 7.59, 95% CI 2.30-25.07, P = .001) whilst combination with negative ctDNA was not significantly associated with recurrence (HR = 7.24, 95% CI 0.98-53.65, P > .05). Additionally, perineural involvement with a positive ctDNA result at diagnosis was significantly associated with recurrence (HR 4.44, 95% CI 1.92-10.26, P < .001) compared to neural involvement with negative ctDNA result (HR = 2.59, 95% CI 0.79-8.57, P > .05). The ctDNA result was unable to differentiate between patients that had nodal involvement, as patients with N1 stage with negative and positive peri-diagnostic ctDNA were no different in their DFS (HR = 5.63, 95% CI 2.23-14.19, P < .001 vs HR = 5.34, 95% CI 2.39-11.89, P < .001, respectively). There were no patients with a resection margin less than 2 mm and a negative ctDNA result, however, a resection margin <2 mm and a positive diagnostic ctDNA result was significantly associated with reduced DFS compared to patients with resection margins greater than 2 mm (HR = 6.07, 95% CI 1.92-10.26, P < .001).
Discussion
While the diagnostic and management approach to CRC has improved, there remains a high rate of recurrence. CRC recurrence is associated with increased risk of mortality and reduced quality of life. Identification of prognostic factors for recurrence might improve survival rates in patients with CRC after curative interventions, as this will enable earlier detection and treatment. Prior studies have identified tumor stage, depth of invasion, and the degree of vascular or perineural invasion as having a statistically significant association with CRC recurrence.40,41 Current guidelines also recommend the use of post-operative CEA, despite it having a low sensitivity and specificity. Thus, this retrospective observational study analyzed the impact of a range of multifactorial variables and diagnostic ctDNA independently and examined the association of each variable on the risk of recurrence following resection of CRC. To our knowledge there has been no study that reviewed the impact of pre-treatment methylated ctDNA in combination with prognostic factors on predicting the risk for future recurrence of CRC.
Our study further reinforced existing findings in the current literature that patients diagnosed initially with nodal involvement (N stage 1 and 2) and metastatic disease (M1 stage) are at greater risk of recurrence following treatment for CRC 42 and have reduced DFS. This is likely due to greater disease burden and a higher risk of undetectable residual disease despite curative interventions. Furthermore, tumors with a resection margin less than 2 mm were associated with a higher risk of CRC recurrence and reduced DFS. This is likely from incomplete surgical excision contributing to greater risk of recurrence. The multivariable analysis supported this deduction (HR = 4.60), independent of the N stage and M stage. Our finding supports previous literature that showed a resection margin <2 mm is associated with poorer prognosis because of increased probability of recurrence.43,44
There has been ongoing debate in the literature regarding the overall impact of the location of CRC lesions on recurrence. Some of the data suggest that proximal colon cancers have poorer overall survival and reduced DFS and increased risk of recurrence,45,46 while another study failed to show this. 47 Interestingly, this study found that distal tumors were associated with a reduced DFS compared to proximal colon tumors, with hazard ratio 3.13 in the multivariable analysis (P = .037). This can be a consequence of distal tumors depositing in peri-anastomotic space leading to residual mesenteric disease such as extramural venous invasion or tumor deposits that subsequently lead to recurrence. 48
The rise of the global burden of chronic disease has resulted in the ongoing use of multiple medications to treat these illnesses, but there is limited literature about the impact of these medications on recurrence of CRC. While there is growing evidence to suggest that medications such as aspirin can reduce the development of precancerous colorectal adenomas,49,50 or that protein pump inhibitors (PPI) can reduce DFS, 51 the influence of these medications on recurrence of CRC is largely unknown. Importantly, to our knowledge, there have been very few studies that have assessed the effect of common cardiovascular medications such as angiotensinogen receptor inhibitors (ACEi), beta blockers, HMG-CoA reductase inhibitors (statins), and antidiabetic medications such as metformin on the recurrence of CRC. Our analysis did not find any significant association between CRC recurrence and the use of aspirin, PPI, ACEi, beta blockers, or metformin.
Post-operative ctDNA has shown a promising prognostic effect on the prediction of recurrence, 52 with clinical trials now using post-treatment ctDNA to guide treatment decisions. 53 However, the capacity for ctDNA assays to detect recurrence after an intervention such as surgery relies on the sensitive detection of very low levels of tumor DNA. Small amounts of cancer cells or residual disease after radical intervention may have undetectable ctDNA remaining. Risk of future recurrence is also associated with initial tumor burden, which we have previously shown to be correlated with presence of methylated BCAT1 and IKZF1. 21 Thus, utilizing pre-treatment diagnostic ctDNA may offer an alternative and more robust predictor for recurrence. Beagan et al 54 found that ctDNA can be detected in patients with concurrent peritoneal metastasis. However, within our cohort, pre-treatment ctDNA did not have a significant association with recurrence of CRC when assessed alone. This is likely explained by the fact that most of our cohort had non-metastatic disease and as such the amount of detectable ctDNA tends to be low.
The presence of pre-treatment methylated ctDNA, when combined with significant prognostic tumor pathology factors, showed a higher predictive power for disease recurrence of CRC, specifically for perineural involvement and metastatic disease, and then marginally improved for tumors located in the distal colon or rectum. There were no differences found in patients with nodal involvement, but this analysis may have been limited by a low sample size. Thus, our analysis found that while ctDNA had limited reliability as a predictive biomarker when used alone in the pre-treatment setting, utilizing it in combination with other prognostic factors can assist in identifying patients at greater risk of recurrence. Such a combination of risk factors may indicate which patients may benefit from stringent follow up including serial ctDNA and functional imaging, to allow for earlier detection of recurrence and better patient outcome.
The strengths of this study include a well described cohort who had a long follow up period minimum of 3 years, enabling sufficient time for recurrence to occur. Also, a great range of demographic, lifestyle, medical conditions, and tumor markers were simultaneously assessed. As the clinical variables that were available for analysis were routinely collected, this makes this study generalizable for other settings. However, there were some limitations. Firstly, while all eligible enrolled individuals were included in the analysis, the number of participants that developed CRC recurrence was relatively small, and a power calculation was not performed prior to analysis. In addition, since patients had multiple comorbidities that were managed by several medications, we were unable to confidently assess the patient’s compliance with their regular medications, which may impact the veracity of the results exploring the association between medication use and recurrence of CRC.
Conclusion
The early detection of CRC recurrence is believed to have a significant impact on survival and can improve patient outcomes. However, there is an ongoing need to find significant predictive variables that can better predict risk for CRC recurrence in routine clinical practice. This study found that high nodal staging, metastatic disease, distal colon and rectal tumors, and a small resection margin (<2 mm) was associated with a poorer prognosis and reduced DFS. Although pre-treatment ctDNA showed a limited benefit for predicting recurrence in isolation, when combined with other significant prognostic factors it improved prediction, enabling better identification of those patients at greater risk of CRC recurrence.
Supplemental Material
sj-doc-1-bmi-10.1177_11772719241232870 – Supplemental material for Evaluating the Role of Methylated Circulating Tumor DNA in Combination With Pathological Prognostic Factors for Predicting Recurrence of Colorectal Cancer
Supplemental material, sj-doc-1-bmi-10.1177_11772719241232870 for Evaluating the Role of Methylated Circulating Tumor DNA in Combination With Pathological Prognostic Factors for Predicting Recurrence of Colorectal Cancer by Hiba Al Naji, Jean M Winter, Susanne K Pedersen, Amitesh Roy, Susan E Byrne, Graeme P Young and Erin L Symonds in Biomarker Insights
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
