Abstract
Companion diagnostics (CDx) hail promise of improving the drug development process and precision medicine. However, there are various challenges involved in the clinical development and regulation of CDx, which are considered high-risk in vitro diagnostic medical devices given the role they play in therapeutic decision-making and the complications they may introduce with respect to their sensitivity and specificity. The European Union (E.U.) is currently in the process of bringing into effect in vitro Diagnostic Medical Devices Regulation (IVDR). The new Regulation is introducing a wide range of stringent requirements for scientific validity, analytical and clinical performance, as well as on post-market surveillance activities throughout the lifetime of in vitro diagnostics (IVD). Compliance with General Safety and Performance Requirements (GSPRs) adopts a risk-based approach, which is also the case for the new classification system. This changing regulatory framework has an impact on all stakeholders involved in the IVD Industry, including Authorized Representatives, Distributors, Importers, Notified Bodies, and Reference Laboratories and is expected to have a significant effect on the development of new CDx.
Keywords
Introduction
Clinical practice today heavily relies on the in vitro analysis of biological specimens for information that will support and/or guide diagnosis and therapeutic efficacy monitoring. Such in vitro diagnostics (IVD) can be used alone or in combination with other devices and/or therapies.1-4 When used in combination with a therapeutic drug, they are referred to as Companion Diagnostics (CDx). 5 The development of CDx depends on companion biomarker(s), intending to stratify patients based on their predicted response to a drug and its potential toxicity levels.6-9
Precision Medicine (PM), or stratified medicine, has been the driving force of the shift from the so-called trial-and-error medicine to a concept of individualized prevention, diagnosis, and treatment.10-12 PM translates patient-specific clinical, genetic, and environmental data into patient-specific therapeutic strategies, improving response to therapy and potential remission.13,14 In many cases, the deriving treatment is based on a predictive biomarker, which can reveal “biological processes, pathogenic processes, or pharmacologic responses to a therapeutic intervention” 8 and requires a CDx. Although oncology drugs prevail among the therapeutics linked to FDA-approved CDx, 15 other medical conditions such as neuropathic, chronic musculoskeletal, cardiovascular, and metabolic disorders have also indirectly benefited from the ongoing research on biomarker-based therapies and efforts to develop CDx, mainly in terms of patients’ quality of life and prognostic potential and/or positive prognostic outcomes.6,16–23 In oncology, the growing understanding of cancer’s pathophysiology and the clinical complexity of its management tends to render non-specific cytotoxic drugs less attractive when compared to personalized chemotherapeutic agents and immunotherapeutic approaches. 13 This not only improves prognosis but allows for more informed prediction of response and tolerance to treatment.13,24,25
The exponential growth of PM is depicted by the fact that 25% of all new drugs approved by the FDA in 2019 were personalized medicines (42% in 2018). 26 On top of that, an average of 65% of drug approvals by the European Medicines Agency (EMA) and the FDA have at least one biomarker consideration in the drug development process between 2015 and 2019. 27 Therefore, considering the significant role that CDx plays in the clinical use of biomarkers, their development, validation, manufacturing, and distribution processes must be tightly regulated. This article sets the stage for the opportunity and challenges of in vitro diagnostics today and aims to discuss how the clinical development of CDx is impacted by ongoing regulatory reforms, especially within Europe, due to the imminent implementation of in vitro Diagnostic Medical Devices Regulation (IVDR).
State of the Art for CDx
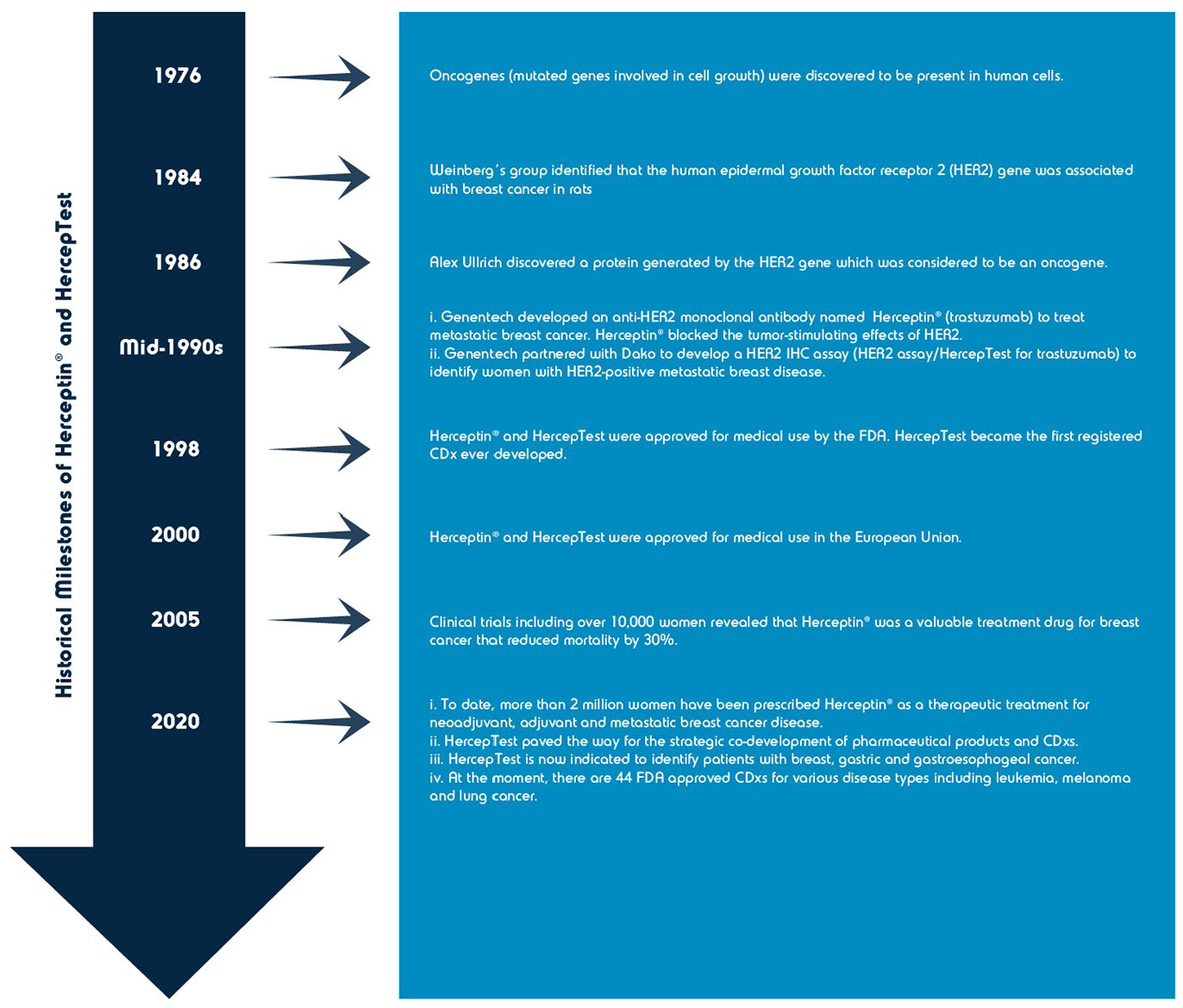
The first predictive biomarker associated with drug development was the HER2 protein, which resulted in the approval of trastuzumab (Herceptin) and HER2 immunohistochemical (IHC) assay, HercepTest (Dako) (see Figure 1) for the detection and treatment of metastatic HER2-positive breast cancer in 1998 by the FDA.2,11,28,29 Since then, the development and nomenclature of predictive biomarker assays linked to specific pharmaceutical agents has rapidly grown.1,3 At the dawn of the millennium, the term “companion diagnostic” was introduced and adopted by FDA (see Table 2 for current definition). 30
As of March 2021, the total number of FDA-approved CDx assays was 44 (see Figure 2). In fact, 43% (n = 19) of approvals have taken place between 1998 and 2015, while 57% (n = 25) of all FDA-approved CDx happened in the last 5 years. 15 Interestingly, this list only includes 2 non-cancer-related indications: Deferasirox (Exjade), a Fe-chelator indicated for the treatment of non-transfusion-dependent thalassemia which was approved in 2013 and in 2015, imatinib mesylate (Gleevec) was approved for the treatment of aggressive systemic mastocytosis. 15 The continuous expansion of indications for marketed CDx not only boosts personalized medicinal approaches but also enables basic and clinical research on both drug-response and mechanism level. This growth is also a reflection of the Regulators’ intention to adopt more innovative approaches for the regulation of these products in order to facilitate access to new therapeutic and diagnostic options.

CDx approved by FDA up to 22 March 2021 by cancer type.
With respect to the analytical platforms used, polymerase chain reaction (PCR) took over for the identification of specific known mutations after the 2011 approval of Roche’s 4800 BRAF V600 Mutation Test, which was intended to be used for melanoma patients potentially eligible for treatment with vemurafenib (Zelboraf). 34 Up until then, the preferred platforms were Immunohistochemistry (IHC) and in situ hybridization (ISH)28,29,35 but overall, 36% (n = 16) of all FDA-approved assays are PCR-based.
Technical limitations of these platforms and the restrictions imposed by the limited amount of material obtained from biopsies resulted in the development of parallel multi-genic DNA sequencing platforms, such as next-generation sequencing (NGS), which enable the simultaneous analysis of hundreds of genetic alterations in a single test.36-39 The first FDA-approved assay based on NGS was the FoundationFocus CDx BRCA Assay for the detection of BRCA1 and BRCA2 alterations in tissues from ovarian cancer patients potentially eligible for treatment with rucaparib (Rubraca). In 2017, the FDA approved FoundationOne CDx (Foundation Medicine) as the first FDA-approved comprehensive genomic profiling (CGP) assay for all solid tumors incorporating multiple companion diagnostics. The test detects substitutions, insertions, deletions, and copy number alterations in 324 genes with implications to inform on therapies covering 5 different cancers: non-small cell lung cancers (NSCLC), melanoma, breast, colorectal, and ovarian cancers (see Table 1). 15 This was followed by the approval of a liquid biopsy platform of the FoundationOne CDx test in 2020. As of February 2021, 7 NGS-based assays were approved by FDA. As we move into an era of big data analytics, more resources and better-defined criteria are being considered, particularly when moving to more automated platforms such as Artificial Intelligence (AI) and Machine Learning (ML) for predictive therapeutics and prognostics.40-42
FDA-approved FoundationOne CDx assays until 22 March 2021.

Basic Considerations for the Development of a CDx
The principal objectives of a CDx1-3,30,43 are to:
(a) Identify the appropriate patient group who are most likely to benefit from a therapeutic product;
(b) Identify the patient groups for which the therapeutic product has been adequately proven safe and effective, allowing for adjustment of treatment to achieve optimal safety;
(c) Predict serious adverse reactions that some patients may present as an outcome of the therapeutic drug used;
(d) Monitor the response to treatment to improve/adjust the dosage scheme and to ensure continued patient safety.
However, the practicality of achieving these objectives is far from ideal, as designing a validated CDx test does not guarantee accurate detection of the optimal patient population and the corresponding provision of treatment. The post-approval delay in clinical uptake of a CDx for a targeted treatment take as long as 5 years, 44 resulting in a delay in the access of patients to more effective therapies. Coordination of drug and CDx development is one of the main challenges that must be addressed, because a failure to co-develop and co-launch the drug and its CDx often results in substantial loss of revenue whilst stifling PM-focused decision-making and compromising on the clinical outcomes of some patients. A typical example of this can be seen in the case of Novartis’s Gleevec and its CDx produced by Dako Denmark over 20 years ago. The Danish diagnostic firm significantly delayed the development of the CDx trying to avoid the possibility of launching a CDx that would not be “linked” to an associated drug but this ultimately resulted in substantial revenue loss for the Swiss pharma giant which could not support the use of its approved drug with a CDx.45,46
Whilst CDx incorporation can take place at the beginning of a clinical trial, the advancement of genomic technologies and the adoption of genetic screening in clinical settings is a relatively recent addition to the traditional drug discovery process. This means that dedicated clinical trials are sometimes required to repurpose existing therapeutics in order to extract more diagnostic information in favor of more stratified CDx. Given that PM considers individual genetic variability and is an important factor of CDx design, it is worth highlighting that a disproportionate majority of data from Genome Wide Association Studies (GWAS) are extracted from individuals of European descent. In fact, 71.8% of the data was generated from only 3 countries, namely the U.S., Iceland, and the United Kingdom 47 resulting in a significant proportion of the world’s population not being represented in the genomics datasets available to pharmaceutical companies and clinicians. Additionally, most clinical trials take place in the U.S. and Europe. The combination of the lack of diversity in clinical trial participants and genomic data limits PM and CDx potential for underrepresented populations. A good example of this is seen in clopidogrel, an antiplatelet prodrug used to treat cardiovascular disease. Most drugs are metabolized by the Cytochrome P450 (CYP) enzyme family. CYP2C1, one of the principal enzymes involved in the bioactivation of clopidogrel, is highly polymorphic, with the most common loss-of-function variant, CYP2C19*2, having a frequency of 25% to 30% (European), 70% (Asian), and 14% to 20% (African). This genetic variation reduces activation of clopidogrel, increasing the risk of serious cardiovascular events in patients undergoing balloon angioplasty or stent placement, particularly in the Asian population. 16 Indeed, these findings have led FDA to subsequently issue a warning on the label for clopidogrel. 48 Whilst CYP2C19 is not a CDx to clopidogrel, using an example of how patients from different populations respond to medications metabolized by certain genes clearly illustrates the importance of improving CDx design and sensitivity in order to facilitate more accurate PM clinical decisions. Further improvements in CDx approvals should come from a better understanding of both the pathology in question and the companion drug’s mechanism of action.
The most common pitfalls related to CDx development pertain to the49-52:
❒ Determination of clinically-relevant and population-specific genetic markers;
❒ Identification of genetic variants linked to specific diseases and/or genetic variants correlating with response to treatment;
❒ Hesitance to analyze large molecule sets (i.e., analysis of proteins, lipids, metabolites, etc.) as biomarkers;
❒ Identification and stratification of the eligible patient groups;
❒ Adoption and establishment of solid regulatory measures that encourage innovation and patient safety;
❒ Reliability and consistency of the biomarker test quality;
❒ Timely availability of test results;
❒ Reimbursement options in different countries.
Changing Regulatory Framework
From a regulatory perspective, because the development of CDx combines pharmaceutical and medical devices, their regulatory requirements must reflect the complexity and costs associated with scientific, clinical, operational, and commercial decisions. This is why a straightforward regulatory and commercialization strategy is crucial and must be developed following the identification of user needs, definition of intended use, and implementation of a quality management system. 52 Up until 2017, there was no official consensus in the definition of CDx but the publication of IVDR 2017/746 has aligned the U.S. and E.U. definitions (see Table 2). 27
Major differences in the regulation of CDx in the U.S. and in Europe (IVDR).

In the U.S., the development of IVDs is guided by the Investigational Device Exemptions (IDE) regulation, Title 21, Code of Federal Regulations (21 CFR) Part 812, which sets specific requirements for studies of investigational devices. 54 However, many IVD devices are exempt from IDE regulations if the testing is non-invasive, does not pose a significant risk in the process of sampling, or is approved for use by another product of procedure. 55
IVDs that are under consideration, or those that are exempt from IDE regulations, have to adhere to labeling requirements under 12 CFR 809, stating either “For Research Use Only,” “Not for use in diagnostic procedures,” or “For Investigational Use Only.” The performance characteristics of this product have not been established.
Investigational IVDs are classified based on the level of risk that the study presents to subjects and can be of54,56,57:
❒ Significant risk (SR), regulated by CFR Part 812—full IDE requirements, application to FDA for IDE approval;
❒ Non-significant risk (NSR), regulated by CFR Part 812.2 (b)—abbreviated IDE requirements, approval of the investigation by an institutional review board (IRB) and compliance with informed consent requirements or;
❒ Excepted devices regulated by CFR Part 812.2(c)—investigations are exempt from most of the requirements of IDE regulation.
In the vast majority of cases, CDx are classified as Class III Medical Devices by FDA because the risk associated with a CDx is similar to the risk associated with the drug that will or will not be administered based on a CDx test.9,56
Typically, class III Medical Devices require a premarketing approval (PMA) procedure according to section 515 of the FD&C Act 58 but there have been 2 CDx tests (MRDx BCR-ABL Test and FerriScan) cleared by 510(k) and 2 cleared by a Humanitarian Device Exemption (HDE) utilizing Gleevec as a companion. 59
The 2018 FDA guidance on the “Principles for Co-development of an In-vitro Companion Diagnostic Device with a Therapeutic Product” 60 set a framework for the management of potential issues sponsors may face when developing a therapeutic product and an accompanying IVD companion diagnostic, referred to as co-development, such as:
❒ Different schedules and agency interactions for therapeutic agents and CDx development and corresponding managing issues;
❒ Alignment in the goals of the therapeutic product, and how they align with the IVD;
❒ Anticipation of the complexity raised by including an IVD in a therapeutic product clinical trial design process;
❒ Decision on what data are needed for a new drug application (NDA) or a biologics licensing application (BLA).
Across the Atlantic in Europe, the legal framework for IVDs and thus for CDx, is currently changing. Publication of the Regulation 2017/746 on IVDR, 53 which came into force in May 2017, is aimed to replace the IVDD 98/79/EC in a transitional period of 5 years (see Figure 3). The new Regulation applies to all IVDs and their accessories and introduces new definitions and rules not only for CDx but also for in-house tests, kits, and single-use IVDs. From May 2017 to May 2022, IVDs and associated CDx will transition from being CE-marked under IVDD, to being CE-marked under the new IVDR. During this period, IVDs can be placed under either directive and those certified by a Notified Body (NB) under IVDD may have an additional 2 years (until 2024) to place their product on the market. Those that were not supervised by a NB must adhere to the new Regulation. Along with more stringent clinical requirements for safety and performance (see Tables 3 and 4 for an overview of new requirements), and in order to ensure the active involvement of all stakeholders in the implementation of the new requirements, IVDR launched a new understanding of traceability and calls for revised, explicit roles for Authorized Representatives, Distributors, and Importers (refer to Art. 11-14 and Art. 16 of IVDR). It also mandates both Manufacturers and Authorized Representatives to have at least one person in their organization that shall hold the role of the Person Responsible for Regulatory Compliance (PRRC, refer to Art. 15 of IVDR).

Timeline for the implementation of IVDR Regulation in Europe.
Overview of EU-Regulation IVDR 2017/746.

Clinical evidence requirements for IVDR.

The Regulation stipulates that clinical evidence used for the approval of an IVD must represent the sum of evidence collected after co-evaluating scientific validity, analytical performance, and clinical performance.5,61,62
The IVDR establishes four risk classes A, B, C, and D in order of lowest to highest risk class. For example, Class D covers general life-threatening conditions or transmissible agents in blood, whilst class A covers laboratory devices and instrumentation. In terms of classification, 7 risk-based rules (see Table 5) are introduced, which result in the up-classification of 80% to 90% of IVDs currently marketed in Europe. This automatically translates into a need for Quality Management Systems (QMS) remediation and consequently to the extensive revision of Technical Documentation (TD). CDx, most software that is a part of IVD instruments (SaMD), single-use IVDs, and genetic tests will fall into class C. However, software for the interpretation of automated readings of line immunoassay for the confirmation and determination of antibodies to HIV-1, HIV-1 group O. and HIV-2 in human serum and plasma, shall fall be in class D per Rule 1.53,63
Classification of in-vitro diagnostic medical devices per IVDR.

The IVDR establishes four risk classes D, C, B, and A, with D being the highest risk class (highlighted in red) and A the lowest (highlighted in green).
The conformity assessment concept (see Figure 4) outlined in IVDR (Annex IX, section 5.2 53 ), which is not new but is largely affected by the imminent up-classification, inevitably results in the involvement of an NB, and the assessment of the manufacturer’s QMS. 61

IVDR conformity assessment routes.
The conformity assessment for CDx (see Figure 5) foresees a consultation procedure between the NB and a medical authority, depending on who is responsible for the authorization of the corresponding medicinal product. 62 Manufacturers are expected to provide a summary of safety and performance with Instructions For Use (IFU), and to evaluate the IVD for the associated medicinal product. However, because a CDx is dependent on a therapeutic agent, it is expected that new CDx consultations will primarily be performed in collaboration with the EMA. The Competent Authority must provide its opinion within 60 days, but this period may be extended once for a further 60 days if there is sufficient justification for this extension. This is the reason why manufacturers of CDx should allow for 120 days for the regulatory procedure to take place. Unlike reference laboratory testing, if the scientific and technical feedback is unfavorable, the certification process may continue based on the recommendation of the NB, provided there is a justification for this overruling.

Overview of conformity assessment of a CDx in Europe under IVDR.
Any potential clinical trial application, including those needed for biomarker and CDx establishment, is regulated by national agencies and the procedure differs in each country according to individual laws. The development process for investigational IVDs and CDx is thus required to be agreed by the sponsor, the National Competent Authority, and the chosen NB on a case-by-case basis.27,62
Another challenge introduced by IVDR revolves around Laboratory Developed Tests (LDT), which are regulated for the first time in the context of IVDs in Europe. Although many requirements are not introduced by IVDR, their incorporation into the regulation mandates quality management, accreditation, and conformity with GSPRs. A significant number of these tests will now require CE IVD marking regardless of the location of the analytical laboratory. 64 IVDR allows an in-house exemption, meaning that healthcare institutions may still manufacture, modify, and use LDTs on a small scale for targeted patient groups, as long as an equivalent device that is already on the market fails to adequately perform. Practically, this exemption will only be applicable to a very limited number of LDTs which come out of laboratories that do not require CE marking.65,66
Other Regulatory Considerations: Japan, China, Australia, and WHO’s IVD Prequalification Program
The U.S., E.U., and Japan are the largest markets for IVDs, respectively. 67 More recently, other markets have emerged, such as China, which has become of great interest due to the rapid growth in its oncology market. 67 Australia’s IVD market remains very small, accounting for approximately 1.35% of global IVD sales, however, imports of IVDs make up 95% of the Australian market, the majority of which are from the U.S. and E.U. 68
Japan, China, and Australia have their regulatory authorities that assess IVD applications, including CDx (see Table 6 for an overview). In 2013, the Japanese Pharmaceutical and Medical Devices Agency (PMDA) published guidance on the approval of CDx. 69 The guidance states that an application for an unapproved CDx and the corresponding therapeutic should be made simultaneously. This is to encourage entities developing CDx and those with the corresponding drugs to work together from an early stage, enabling the approval of the CDx before the therapeutic.69,70 This simultaneous application is also strongly recommended by Australia’s Therapeutic Goods Administration (TGA), 4 which recently introduced new regulations that will be fully enforced by 2022, including the use of a Unique Product Identifier (UPI) for every CDx.
CDx definitions and regulatory requirements around the world.
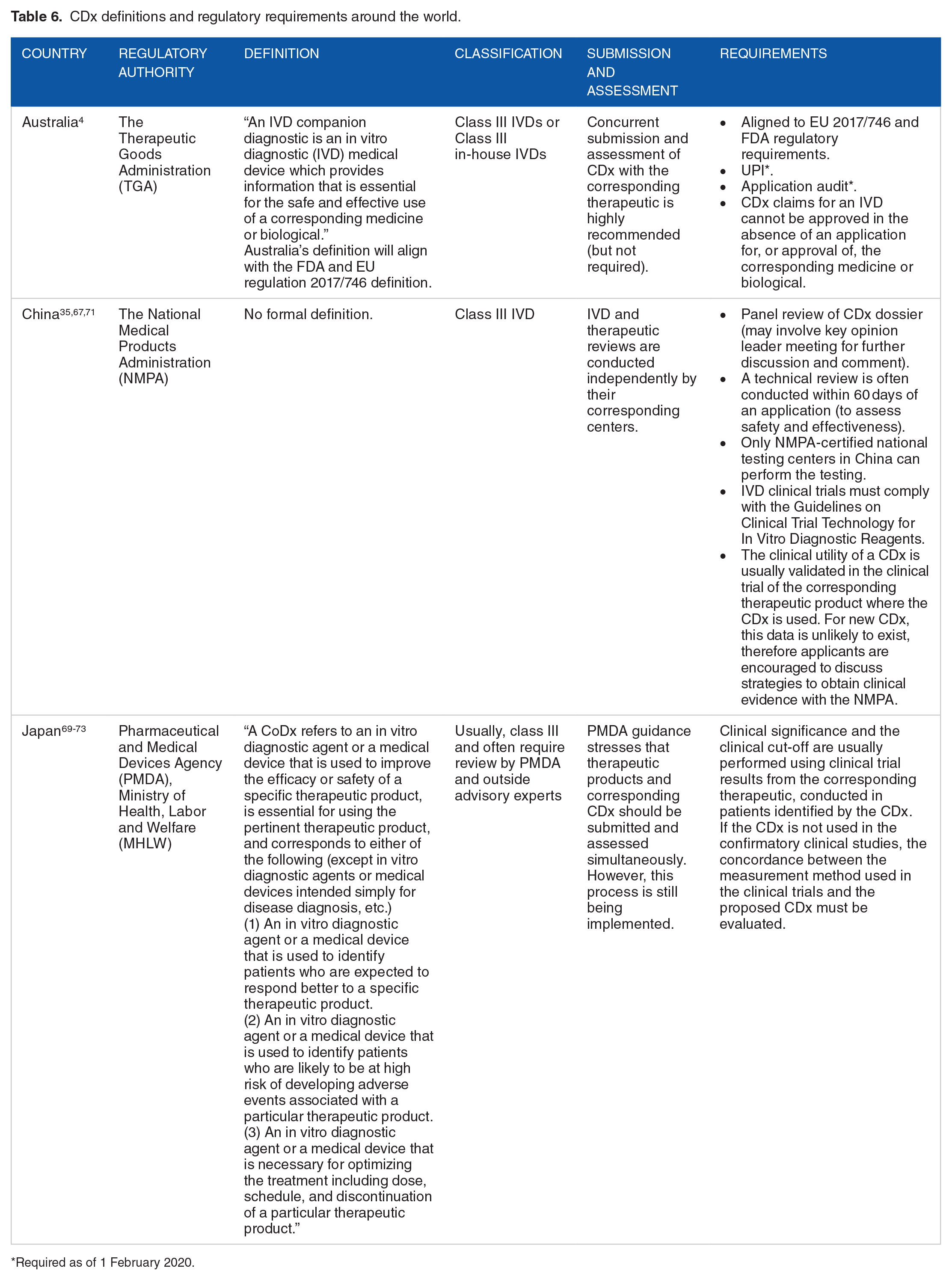
Required as of 1 February 2020.
The Chinese regulatory authority, the National Medical Products Administration (NMPA), has not yet published any guidance for CDx. 67 However, as CDx are classified as class III IVD products they follow IVD regulations, which stipulate that applications are to be supported by clinical trials conducted in China. 67 Currently, the regulatory assessment process for CDx is challenging, due to different regulations and independent review centers for IVDs and therapeutics, with limited interaction/collaboration between the two. However, in 2017 new procedures for approval were proposed to facilitate the development/approval of IVDs, including CDx. For instance, the NMPA may now accept clinical data from global trials outside of China (for other proposed changes see Xu et al. 67 ).
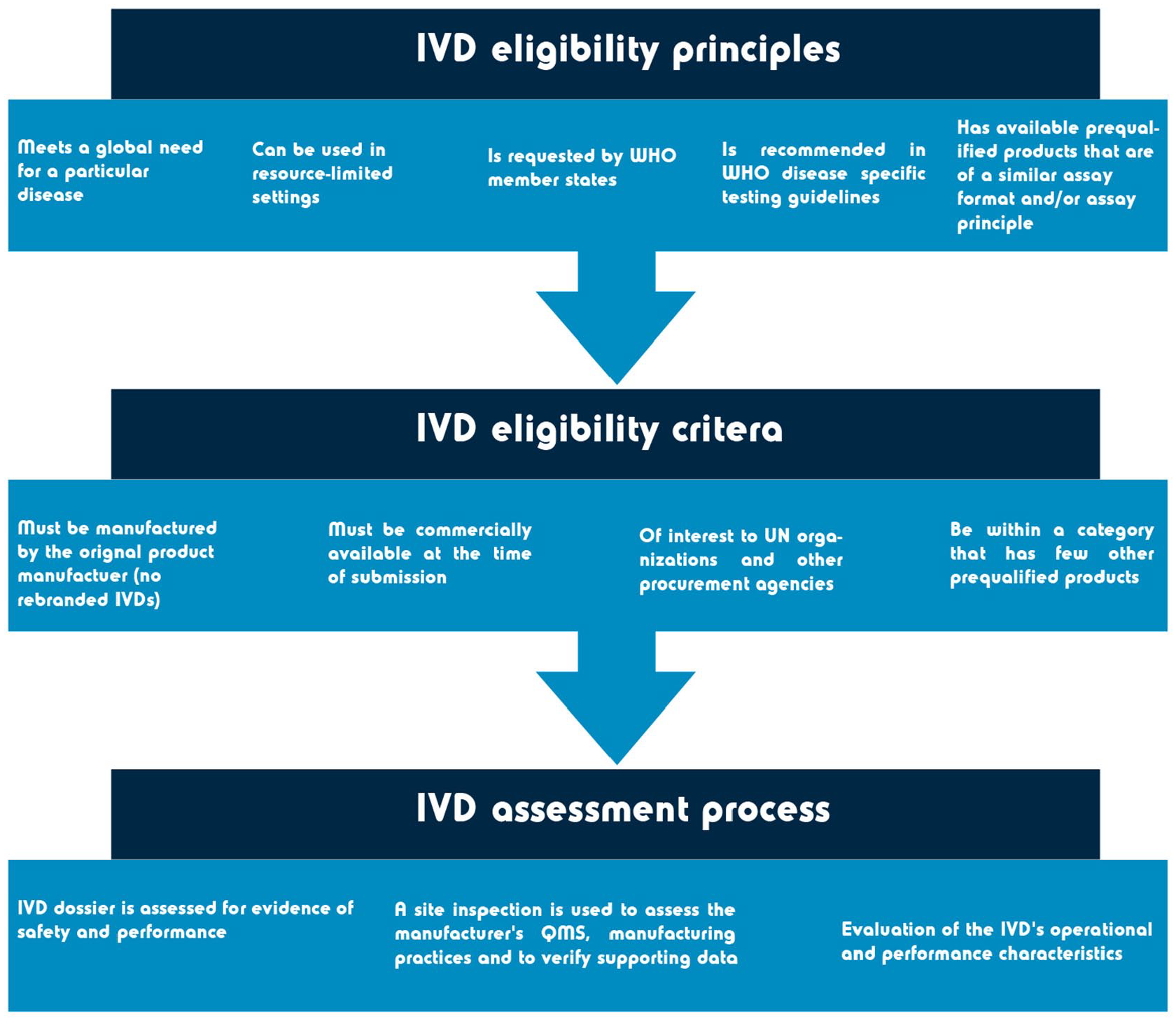
For regulatory authorities that do not have an established assessment procedure, and/or lack the experience and resources to assess IVDs, help is available from the World Health Organization (WHO) prequalification (WHO-PQ) assessment. United Nations agencies and WHO member states can use the WHO-PQ as a guide to determine whether IVDs meet the required safety and performance requirements. To be eligible for the WHO-PQ assessment, manufactures must submit a pre-assessment form and meet several requirements (see Figure 6).
Although the regulatory authorities differ slightly in their assessment of CDx, they all face a common issue: the lack of a global reimbursement scheme. Currently, reimbursement procedures differ by country, even within E.U. member states.75,76 This means that CDx that are funded in the U.S. may not be reimbursed in the E.U. and vice versa. For emerging and smaller markets, such as China and Australia, this may cause a barrier to the development of new CDx. 43
Concluding Remarks
Development, opportunity, and growth of the CDx market have the potential to further personalize therapeutic strategies and improve patient access, outcomes and their response to innovative pharmaceutical agents and/or diagnostic methods. To meet the complicated, growing regulatory demands of IVD technologies, an international consensus on the basic regulatory requirements for their approval and continuous monitoring is a sine qua non and should be seen as a step forward for public health. Currently, the lack of standardization and use of multiple platforms for the same biomarker raises concerns on reproducibility and sensitivity of the specific analytical assay the platform assesses. Therefore, approval of CDx must be aligned with the scrutinized assessment of their analytical validity. Implementation of IVDR 2017/746, although a challenging regulatory shift for CDx, opts to optimize the field through the alignment of European requirements with the American and Japanese ones and the continuous, dynamic monitoring of the real-world use of IVDs.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
