Abstract
Purpose:
Postischemic reperfusion injury may exacerbate cerebral damage and capillary dysfunction, leading to brain edema (BE), hemorrhagic transformation (HT), necrosis, and injury from free radicals with subsequent infarct growth (IG). Several plasmatic biomarkers involved in the ischemic cascade have been studied in relation to radiological and clinical outcomes of reperfusion injury in ischemic stroke with heterogeneous results. This article provides a brief overview of the contribution of circulating biomarkers to the pathophysiology of parenchymal damage in ischemic stroke patients treated with revascularization therapies.
Methods:
We included full reports with measurements of plasma markers in patients with acute ischemic stroke treated with revascularization therapies.
Findings:
Our research included a large number of observational studies investigating a possible role of circulating biomarkers in the development of parenchymal damage after acute stroke treatments. To make the results clearer, we divided the review in 4 sections, exploring the relation of different biomarkers with each of the indicators of parenchymal damage (HT, BE, IG, recanalization).
Discussion and conclusion:
Definite conclusions are difficult to draw because of heterogeneity across studies. However, our review seems to confirm an association between some circulating biomarkers (particularly matrix metalloproteinase-9) and occurrence of parenchymal damage in ischemic stroke patients treated with revascularization therapies.
Introduction
Ischemic stroke is one of the main causes of death and disability worldwide. 1 Administration of intravenous recombinant tissue-type plasminogen activator (rt-PA) and endovascular procedures is the mainstay of acute stroke treatment to obtain vessel recanalization and tissue reperfusion.2,3
However, despite successful recanalization, not all patients achieve favorable clinical outcome. For instance, we use the term “futile recanalization,” when ischemic tissue is not reperfused even in case recanalization is successful. This situation has been generally attributed to the occurrence of “no-reflow phenomenon,” caused by microvascular obstruction, and/or arterial reocclusion. 4
When attempting revascularization therapies in the first hours after stroke onset, vascular complications such as hemorrhagic transformation (HT) or brain edema (BE) arise as an obstacle. A critical role in the molecular mechanisms determining HT and BE is the disruption of the neurovascular unit, a dynamic terminal structure composed by a microvessel and its endothelial cells, basal lamina matrix, astrocyte end-feet, pericytes, astrocytes, neurons and their axons, and supporting cells (microglia and oligodendroglia).5,6
Restoration of blood flow (reperfusion) may further exacerbate ischemic damage and capillary dysfunction, leading to adverse functional, metabolic, or structural changes in ischemic tissues, the so-called “reperfusion injury” (RI). 7 This process is summarized in Figure 1.
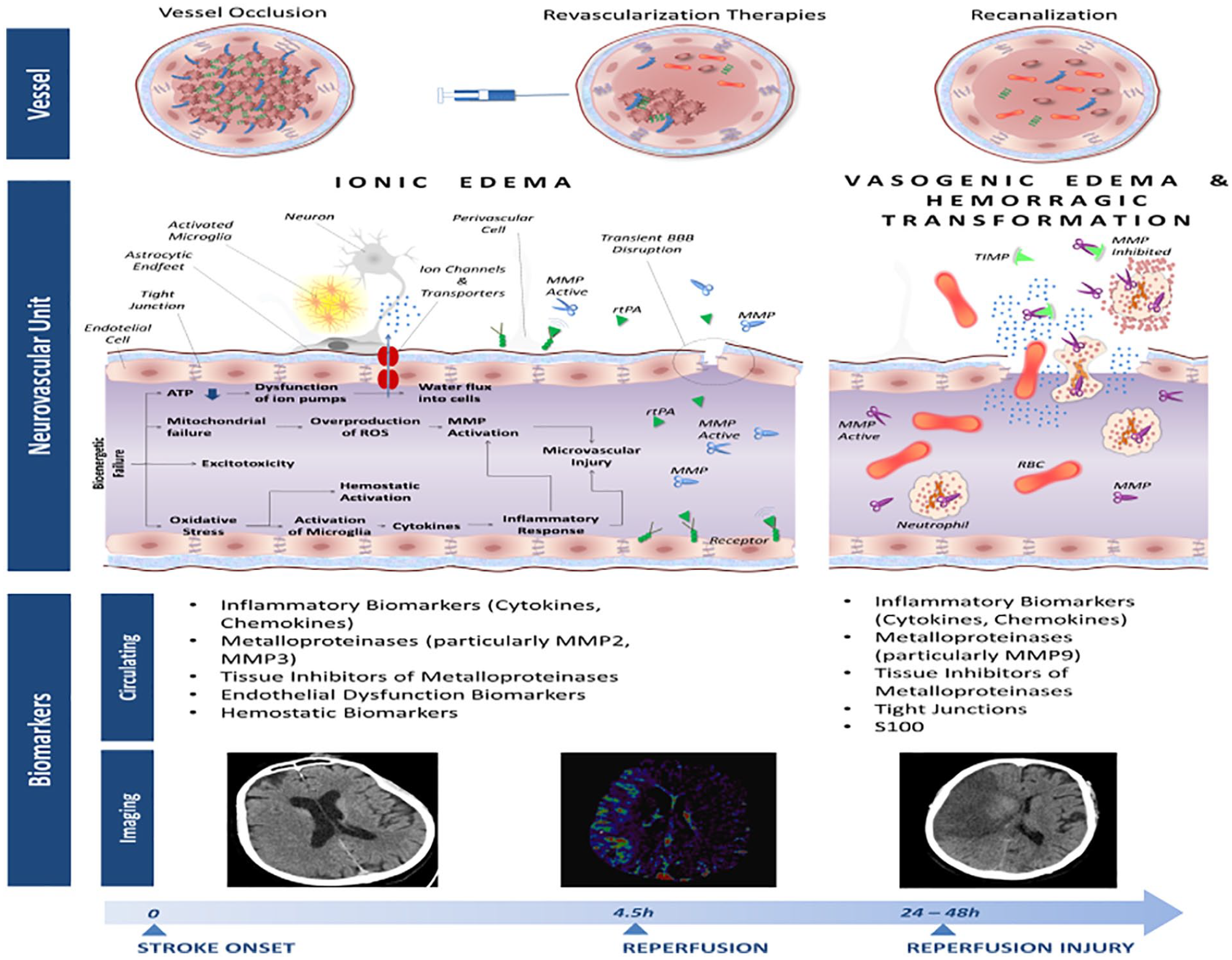
After vascular occlusion, a bioenergetic failure occurs with consequent decrease in adenosine triphosphate (ATP) production and loss of ionic homeostasis in neurons. Consequently there is an excessive sodium and water influx with concomitant cell swelling. Oxidative stress due to overproduction of reactive oxygen species (ROI) may trigger release of Matrix Membrane Metalloproteinases (MMPs) by neurons, glia, astrocytes, pericytes resulting in BBB damage through digestion of the endothelial basal lamina. In this early phase, the BBB opening could be reversible. After the early BBB opening, there is a second phase of severe BBB injury within 24–72 h after infarction that results in greater tissue damage through leukocyte infiltration and marked release of MMPs from neutrophils transmigrated to the ischemic brain. Unbalance between MMPs and their natural inhibitors (tissue inhibitors of metalloproteinases, TIMPs) may exacerbate BBB disruption.
The aim of this work is (1) to review potential circulating markers of parenchymal damage that might be used in the hyperacute phase of stroke to predict outcome, (2) to evaluate possible limits for clinical application of these biomarkers, and (3) to review the studies that integrate blood-brain barrier (BBB) assessment with advanced neuroimaging techniques and blood biomarker levels.
Methods
We included all published full reports involving patients with acute ischemic stroke (AIS) treated with revascularization therapies. The extensive MEDLINE search is reported in Supplementary Material.
We chose as indirect indicators of parenchymal damage the occurrence of HT, BE, and infarct growth (IG). Indeed, we used vascular recanalization as a surrogate of successful tissue reperfusion and we studied its association with circulating biomarkers.
Results
We divided our review in 4 paragraphs: (1) biomarkers and HT, (2) biomarkers and IG, (3) biomarkers and recanalization, (4) neuroimaging markers of BBB disruption.
Biomarkers and HT
We found 5 studies investigating the role of circulating matrix metalloproteinases (MMPs) in hemorrhagic transoformation (Castellanos 2004 number-1, Castellanos 2007 number-2, Montaner et al ex number 8, Inzitari et al ex number 9, Piccardi et al ex number 10). However only 2 studies measured MMPs levels before and after thrombolysis (Table 1). Montaner et al 10 examined baseline and 12- and 24-h blood samples of 41 patients with acute cardioembolic stroke, confirming an independent association between pretreatment MMP-9 and parenchymal hemorrhage. In 327 rt-PA-treated stroke patients, relative pre-post rt-PA variation of MMP-9 11 and relative increase of MMP-9/tissue inhibitor of metalloproteinases (TIMP)-1 and MMP-9/TIMP-2 ratios 12 were significantly associated with symptomatic HT while no association was found between baseline levels of MMP-9 and symptomatic HT. 11 In a small study of 23 stroke patients and 47 healthy controls, patients with thrombolytic failure (defined as absence of recanalization) had the highest levels of baseline MMP-9 and half of them developed HT. 26
Circulating biomarkers and HT in ischemic stroke patients.

Abbreviations: α2-AP, Alpha 2-antiplasmin; APC, activated protein C; A2M, alpha2 macroglobulin; c-Fn, cellular fibronectin; CRP, C-reactive protein; EVT, endovascular treatment; FMPPs, fluorescent molecular peroxidation products; HT, hemorrhagic transformation; IFN-gamma, interferon-gamma; IL, interleukin; IP-10, interferon gamma-induced protein 10; Lp, lipoprotein; MCP-1, monocyte chemoattractant protein 1; MIP, macrophage inflammatory protein; MMP, matrix metalloproteinases; PAI-1, plasminogen activator inhibitor-1; rt-PA, recombinant tissue plasminogen activator; SAP, serum amyloid P; S100B, S100 calcium-binding protein B; SSAOa, semicarbazide-sensitive amine oxidase activity; TAFI, thrombin-activatable fibrinolysis inhibitor; TIMPs, tissue inhibitors of metalloproteinases; TNF, tumor necrosis factor; VAP-1, vascular adhesion protein-1;
The role of another serine protease, activated protein C (APC), was studied in 117 rt-PA-treated patients. No correlation was observed between pretreatment levels of APC and parenchymal hemorrhage occurrence, whereas a significant increase in APC level 2 h after rt-PA was found among patients with HT. 13 Among biomarkers possibly involved in BBB disruption, vascular adhesion molecule-1 (VAP-1), a cell surface and circulating enzyme involved in recruitment of lymphocytes and neutrophils through its semicarbazide-sensitive amine oxidase (SSAO), has been shown to predict HT after rt-PA administration: patients with HT had higher baseline levels of VAP-1/SSAO, compared with patients without HT, with a graded response between baseline levels of VAP-1/SSAO activity and degree of bleeding. 17 The role of S100B protein, a calcium-binding protein released into the blood stream when BBB is disrupted, was studied in a retrospective cohort of 275 patients receiving rt-PA, resulting in significantly higher serum levels of S100B in patients with HT. 14
Concerning the predictive role of systemic markers of inflammation and cytokines on the development of HT, Gori et al 15 studied the effect of baseline and post-rt-PA circulating levels of a large panel of inflammatory markers in 327 rt-PA-treated patients. None of the pre-rt-PA biomarkers level was significantly related to HT and only pre-post rt-PA variation of interleukin (IL)-10 levels was significantly and independently associated with HT. 15 Oxidative stress might also be implied in early BBB disruption and subsequent RI: in 100 consecutive stroke patients, clinical deterioration at 48 h was significantly associated with higher baseline levels of fluorescent molecular peroxidation products (FMPPs). In this subgroup of patients, about a third had symptomatic HT and showed significantly higher levels of FMPP compared with patients without hemorrhage. 16 Finally, 2 studies explored the possible association between hemostatic markers and HT,18,19 with only one demonstrating a predictive role of baseline levels of plasminogen activator inhibitor-1 and thrombin activatable fibrinolysis inhibitor 19 in 76 patients receiving rt-PA treatment. Finally, 5 studies showed an association between high neutrophil-lymphocytes ratio (NLR) and HT in AIS patients. Particularly, 2 studies showed an association between NLR and HT in rt-PA-treated stroke patients20,21 and 3 studies showed an association between NLR and HT in endovascular-treated stroke patients.22-24 Only one study 25 showed no association between NLR and HT in AIS patients treated with rt-PA.
Biomarkers and cerebral edema/IG
We found 2 studies exploring the role of blood biomarkers in predicting the occurrence of cerebral swelling in stroke patients treated with rt-PA (Table 2). Moldes et al 27 studied 134 patients: in the 19 patients with severe BE (diagnosed as extensive swelling causing any shift of the midline structures at 24-36 h), higher levels of endothelin-1 (ET-1) and cellular fibronectin (c-Fn) were observed, compared with patients without BE, although only ET-1 levels > 5.5 fmol/mL remained an independent predictor of BE after multivariate analysis. In 76 patients with acute stroke treated with rt-PA, MMP-9, proMMP-10, and c-Fn levels were found to be independently associated with higher degree of BE and higher infarct volume on follow-up computed tomography (CT) scan. 28
Circulating biomarkers and brain edema/infarct growth in acute ischemic stroke patients.

Abbreviations: Asp, aspartate; c-Fn, cellular fibronectin; CPU, carboxypeptidase U; CRP, C-reactive protein; ET-1, endothelin-1; Glu, glutamate; ICAM-1, intercellular adhesion molecule 1; IL, interleukin; MMP, matrix metalloproteinases; NSE, neuron-specific enolase; pro-CPU, procarboxypeptidase U; rt-PA, recombinant tissue plasminogen activator; sCD40L, soluble CD40 ligand; S100B, S100 calcium-binding protein B; sTM, soluble thrombomodulin; TIMPs, tissue inhibitors of metalloproteinases; TNF, tumor necrosis factor; VCAM-1, vascular cell adhesion molecule.
Regarding the association between circulating biomarkers and IG, our search retrieved 7 studies. In a small cohort of 24 rt-PA-treated patients, baseline MMP-9 and MMP-13 correlated strongly with diffusion-weighted image (DWI) lesion expansion. 29 A predictive role of MMP-9 for final infarct volume measured on DWI-MRI (magnetic resonance imaging) was also observed in a series of 16 patients with acute stroke. 10 In 226 consecutive acute stroke patients, neuron-specific enolase (NSE) ⩾ 23 ng/mL, IL-6 ⩾ 10 pg/mL, and active MMP-9 ⩾ 21 ng/mL were associated with the absence of clinical-diffusion mismatch (and consequently with a large infarction) at baseline. 30 The increase of inflammation markers in the first 5 days from stroke onset correlated significantly with IG (evaluated either with CT or with MRI) 31 in the subgroup of 43 patients receiving rt-PA, those with a clinically significant improvement showed a decrease in C-reactive protein (CRP) and white blood cells levels compared with those without improvement. The role of IL-6 and tumor necrosis factor-alpha on final infarct volume after thrombolysis was also confirmed by Rodriguez et al. 28
IG appears to be correlated with higher levels of markers of neuronal and glial damage: in a secondary analysis of blood samples taken in the National Institute of Neurological Disorders and Stroke (NINDS) rt-PA Stroke Study at baseline, 2 and 24 h after thrombolysis, 36 higher peak of myelin basic protein (MBP) and S100B levels were associated with larger infarcts on follow-up scan. No differences were observed between rt-PA (178 patients) and placebo (181 patients) groups in 2- or 24-h marker concentration, peak marker concentration, or change in marker concentration. 32
In a series of 197 patients (46 receiving rt-PA), a correlation between DWI lesion enlargement and higher levels of glutamate was found; the association was observed both in treated and untreated patients. 33 Poststroke plasmatic levels of Caspase-3, a protein involved in apoptosis, measured at different time points, were positively correlated with IG assessed by DWI-MRI in 58 patients treated with systemic thrombolysis. 34
The decrease of plasmatic levels of the precursor of the antifibrinolytic enzyme carboxypeptidase U (pro-CPU) in the first 72 h from stroke onset was associated with larger final infarct volume measured as DWI lesion at 48 to 72 h in 12 patients (5 treated with intra-arterial urokinase, 7 receiving rt-PA). 35
Biomarkers and recanalization
In a small study enrolling 23 patients with acute stroke treated with rt-PA (7 patients), intra-arterial urokinase (5 patients) and combined intravenous rt-PA/intra-arterial urokinase (11 patients), Heo et al 26 found significantly higher baseline levels of MMP-9 in 8 patients with persistent occlusion after treatment (Table 3).
Circulating biomarkers and recanalization in ischemic stroke patients.

Abbreviations: α2-AP, α2-antiplasmin; ADAMTS13, a disintegrin and metalloproteinase with a thrombospondin type 1 motif, member 13; Ang-1, angiopoietin-1; APOE, apolipoprotein-E; BNP, brain natriuretic peptide; CD41 + MP, glycoprotein IIb expressing microparticles; CRP, C-reactive protein; G-CSF, granulocyte-colony stimulating factor; IGF-1, insulin growth factor-1; Lp-PLA2, lipoprotein-associated phospholipase A2; MMP, matrix metalloproteinases; PAI-1, plasminogen activator inhibitor-1; rt-PA, recombinant tissue plasminogen activator; S100B, S100 calcium-binding protein B; sEPCR, soluble endothelial protein C receptor; sTM, soluble thrombomodulin; TAFI, thrombin-activatable fibrinolysis inhibitor; TAT, thrombin-antithrombin complex; VEGF, vascular endothelial growth factor; vWF, von Willebrand factor.
Bustamante et al 46 evaluated the role of baseline levels of ADAMTS13 (a disintegrin and metalloproteinase with a thrombospondin type 1 motif, member 13) in relation to vessel patency among a cohort of 108 rt-PA-treated stroke patients; an additional cohort of 78 patients treated with endovascular thrombectomy was examined to explore the relation between ADAMTS13 and futile recanalization. Recanalization after rt-PA was associated with higher levels of ADAMTS13, while, in the group treated with endovascular approach, reduced serum levels of ADAMTS13 appeared to be an independent predictor of futile recanalization. 46 Despite these results, von Willebrand factor (vWF) levels do not seem to correlate with recanalization: in 64 rt-PA-treated stroke patients, vWF levels at baseline did not differ among subgroups of patients (31 with no occlusion at baseline, 19 with persistent occlusion, 14 with complete recanalization). 47 Conversely, patients with persistent occlusion had higher baseline levels of other endothelial biomarkers such as thrombomodulin and soluble endothelial protein C receptor.
Fernandez-Cadenas et al 42 sought to identify genetic variants of biological markers associated with recanalization in 497 acute stroke patients evaluated by transcranial Doppler monitoring after rt-PA; 3 single-nucleotide polymorphisms in IL-1B and vWF were associated with successful recanalization, supporting the hypothesis that variants of endothelial factors could modulate early recanalization by modification of coagulation factors activity. Considering coagulation factors, lower baseline levels of the complex thrombin-antithrombin were associated with higher recanalization rates at 1, 2 and 6 h after rt-PA infusion in 89 rt-PA-treated patients. 41 Considering a large panel of coagulation and thrombosis biomarkers, Martí-Fàbregas et al38,39 found that only baseline levels of α2-antiplasmin, but not their change before and after thrombolysis, were predictive of recanalization in a series of 63 rt-PA-treated stroke patients. In humans, small membrane-bound particles known as microvesicles or microparticles are shed from platelets in response to activation or apoptosis: Bivard et al 45 demonstrated a positive correlation between plasma levels of glycoprotein IIb (CD41+)-expressing microparticles and successful post-rt-PA recanalization; in contrast, C-reactive protein was negatively correlated with recanalization. Single studies have explored the role of different biomarkers in small series of rt-PA-treated patients reporting a possible association between persistent occlusion and higher mass and activity of lipoprotein-associated phospholipase A2 (Lp-PLA2) 44 and elevated levels of brain natriuretic peptide. 37 Conversely, high levels of growth factors 40 and a single S100B value <0.4 μg/L 43 show an association with successful clot lysis.
Neuroimaging markers of BBB leakage in AIS
BBB leakage following AIS can be visualized in vivo by fluid-attenuated inversion recovery (FLAIR) MRI or CT perfusion (CTP) permeability maps. However, the assessment of BBB integrity, so far, has not been incorporated into standard of care.
Regarding the role of circulating biomarkers as predictors of in vivo imaging of BBB leakage, there are few available studies in literature.
Barr et al 48 enrolled 33 acute stroke patients (9 of whom treated with rt-PA) and showed a potential predictive role of baseline MMP-9 and MMP-9/TIMP-1 ratio for the presence of Hyperintense Acute Reperfusion Injury Marker, a gadolinium-enhancement of the subarachnoid cerebrospinal fluid space in the hemispheric sulci on 24-h FLAIR-MRI, which is considered expression of BBB damage.
In a series of 180 AIS patients (both rt-PA treated and untreated), an independent association between increased baseline MMP-9 levels and FLAIR-ratio (FR) was demonstrated. FR was estimated by comparing relative FLAIR-hyperintensity with the contralateral unaffected hemisphere on acute MRI images. Authors used FR as a surrogate of BBB breakdown and demonstrated an independent association between FR and HT. 49
Discussion
Our research included a large number of observational studies investigating a possible role of a wide panel of circulating biomarkers (located at different levels of the ischemic cascade) in parenchymal damage after reperfusion therapies.
The most consistent association between plasmatic levels of biomarkers and HT regards MMPs (particularly MMP-9). However, the heterogeneity across study designs limits the transferability of results into clinical setting. First of all, sample sizes appear largely variable. Furthermore, some studies include both untreated and treated patients without specifying the number of patients who underwent different treatment strategies. Another crucial point is in the variability in blood sample withdrawal. Finally, the definition of HT was heterogeneous across studies, ranging from inclusion of radiological characteristics alone to a combination of clinical and radiological features. Only if confirmed in future well-designed studies, the association between plasmatic levels of MMP-9 and HT might have important therapeutic implications: the available literature suggests that combination therapies of MMP-9 inhibitor (administration of minocycline, new chemical entities like DP-b99, and finally other approaches like hypothermia) along with rt-PA could be potentially beneficial for stroke patients. 50
An ideal blood biomarker for stroke should be reliable, rapidly measurable, and readily available; unfortunately, in most clinical settings, most of the above-mentioned circulating biomarkers have no viable tests for point-of-care determination.
For this reason, NLR, an easily available marker of subclinical inflammation, seems to have a promising transferability into clinical setting. A recent meta-analysis showed that high NLR might predict HT and 3-month mortality in patients with AIS. 51
Considering the association between circulating biomarkers and IG or BE, only one study showed a significant association between MMP-9 and severe BE. 28
The role of circulating biomarkers in IG has been investigated only in 8 studies that found possible associations with MMPs (MMP-9, MMP-13, pro-MMP-10),10,27-30 inflammatory markers (CRP, NSE, IL-6),28,30-33 and markers of cytotoxic stress and glial-neural damage (MBP, S100B, Glutamate).32,33 Interestingly, the predictive role of MMP-9 was demonstrated in 5 studies.10,27-30 Similar to HT, the definition of IG is very heterogeneous across studies, with differences in timing of IG assessment and in the neuroimaging methodology (6 studies used DWI-MRI while 4 studies used plain CT).
Current treatments against BE (osmotic agents or decompressive craniectomy) and IG are exclusively targeted to manage end-stage events without modifying the underlying molecular mechanisms. 52 Some pivotal evidence suggests that sulfonylurea receptor-1 inhibitors such as glyburide may attenuate BE in stroke patients.53-55
Recanalization failure is commonly considered one of the main factors of poor prognosis after revascularization therapies. Both size and pathological composition of clots have been reported to be major determinants of recanalization failure. 56 Different circulating biomarkers have been studied in prospective cohorts with conflicting results. Current more robust evidence regards endothelial biomarkers with only one study showing a possible involvement of high MMP-9 baseline levels on futile recanalization. 26 In the future, the measure of plasmatic levels of circulating biomarkers might integrate clinical and neuroimaging data to improve treatment selection and individualization of therapy in the acute setting of stroke. In particular, it may be desirable to identify those patients at high risk to develop BE, HT, and IG or those who have poor chance of recanalization after acute treatments. As far as we know, only 2 studies integrated circulating biomarkers and advanced neuroimaging techniques48,49 for the identification of BBB leakage. However, these studies included a small number of patients with heterogeneous characteristics, including different final diagnoses (treated and untreated stroke patients, stroke mimics).
Definitive conclusions are difficult to be drawn because of heterogeneity across studies. However, our review confirms that some circulating serum biomarkers involved in the ischemic cascade (particularly MMP-9) might be helpful to predict futile recanalization and feared complications of reperfusion therapies. For this reason, large prospective studies exploring the role of blood biomarkers in well-phenotyped stroke patients are needed to confirm the above-mentioned observations.
To be noted is that recent endovascular trials challenged the concept of ischemia-RI, showing that, unlike animal models, there is no significant lesion growth after complete reperfusion in most patients benefiting from endovascular thrombectomy. 57 While by a side this observation might impair the interest in studying predictors of RI, by another point of view, the investigation of circulating biomarkers, integrated with in vivo imaging of BBB disruption, might represent an important element to understand the pathophysiology of postischemic parenchymal damage in the contest of everyday clinical practice, with possible implications for the design of acute neuroprotection studies.
Supplemental Material
Supplementar_Material__xyz24045f860e6ee_1 – Supplemental material for Blood Biomarkers of Parenchymal Damage in Ischemic Stroke Patients Treated With Revascularization Therapies
Supplemental material, Supplementar_Material__xyz24045f860e6ee_1 for Blood Biomarkers of Parenchymal Damage in Ischemic Stroke Patients Treated With Revascularization Therapies by Benedetta Piccardi, Silvia Biagini, Veronica Iovene and Vanessa Palumbo in Biomarker Insights
Footnotes
Acknowledgements
The authors thank Professor Domenico Inzitari (University of Florence) for his inspirational and intellectual guidance, leading to constant generation of new ideas, and Maria Elena Della Santa (CNR, Neuroscience Institute) for her contribution to language editing and proof reading.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Review conception and design: BP; VP. Acquisition of data: BP; VP; SB; VI. Interpretation of data: BP; VP. Drafting the manuscript: BP; VP; SB; VI. Critical revision: BP; VP
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
