Abstract
Jasmonates (JAs) regulate plant growth and defense responses. On perception of bioactive JAs, the JA receptor CORONATINE INSENSITIVE1 (COI1) recruits JA ZIM-domain (JAZ) proteins for degradation, and JAZ-targeted transcription factors are released to regulate JA responses. The subgroup IIId bHLH transcriptional factors, including bHLH17, bHLH13, bHLH3, and bHLH14, interact with JAZs and repress JA responses. In this study, we show that IIId bHLH factors form dimers via the C-terminus in yeast. N-terminus of bHLH13 is essential for its transcriptional repression function. bHLH13 overexpression inhibits Arabidopsis resistance to the necrotrophic fungi Botrytis cinerea and defense against the insect Spodoptera exigua. COI1 mutation disrupts the oversensitivity of the quadruple mutant bhlh3 bhlh13 bhlh14 bhlh17 in various JA responses, including anthocyanin accumulation, root growth inhibition, and defense against B cinerea and S exigua. Disruption of the TTG1/bHLH/MYB complex blocks anthocyanin accumulation of bhlh3 bhlh13 bhlh14 bhlh17, whereas abolishment of MYC2 attenuates JA-inhibitory root growth of bhlh3 bhlh13 bhlh14 bhlh17. These results genetically demonstrate that IIId bHLH factors function downstream of COI1 to inhibit distinctive JA responses via antagonizing different transcriptional activators.
Introduction
Jasmonates (JAs), including jasmonic acid and its derivatives,1,2 regulate plant development and growth,3,4 including root growth,5,6 flowering, 7 stamen development, 8 trichome formation, 9 and leaf senescence. 10 Moreover, they control the secondary metabolism of plants, 11 including anthocyanin and glucosinolate biosynthesis. 12 They also function as defensive hormones, promoting tolerance to abiotic stresses and defensive responses to biotic stresses,13–15 including necrotrophic fungi and insect attacks. 16
In response to bioactive JA signals, the JA receptor CORONATINE INSENSITIVE 1 (COI1) 17 recruits JA ZIM-domain (JAZ) proteins for ubiquitination and subsequent degradation. In this way, JAZ-inhibited transcription factors are activated to regulate various JA responses.18–20
bHLH subgroup IIIe transcription factors, including MYC2, MYC3, MYC4, and MYC55,8,21–23; the TTG1/bHLH/MYB complex 9 ; the R2R3-type MYB factors MYB21 and MYB24 24 ; and ethylene-activated ETHYLENE INSENSITIVE3 (EIN3) and EIN3-LIKE1 (EIL1), 25 interact with JAZs and function as transcription activators to positively mediate distinct JA responses, including root growth inhibition, stamen development, trichome formation, anthocyanin accumulation, and pathogen resistance.26,27
In comparison, bHLH subgroup IIId transcription factors such as bHLH17/JASMONATE-ASSOCIATED MYC2-LIKE1 (JAM1), bHLH13/JAM2, bHLH3/JAM3, and bHLH14 function as transcriptional repressors to negatively regulate JA responses, including root growth, anthocyanin accumulation, male fertility, leaf senescence, and defense against insect or pathogen attacks.10,28–32 In this study, we found that the C-terminus of bHLH13 is necessary for its ability to dimerize, and that the N-terminus of bHLH13 is required for the transcriptional repression function. Meanwhile, bHLH13 overexpression attenuated plant resistance to the necrotrophic fungus Botrytis cinerea and defense against the insect Spodoptera exigua. Genetic and physiological analyses revealed that introduction of the coi1-1 mutation into the quadruple mutant bhlh3 bhlh13 bhlh14 bhlh17 disrupted its JA hypersensitivity and that disruption of the TTG1/bHLH/MYB complex abolished anthocyanin accumulation in bhlh3 bhlh13 bhlh14 bhlh17 plants. Moreover, the myc2 mutation attenuated JA-inhibited root growth in bhlh3 bhlh13 bhlh14 bhlh17 plants. These results provide genetic evidence that bHLH subgroup IIId transcription factors act downstream of COI1 to repress distinct JA responses by antagonizing diverse transcriptional activators.
Materials and Methods
Plant materials and growth conditions
The Arabidopsis thaliana mutants coi1-1, bhlh3 bhlh13 bhlh14 bhlh17, bhlh3 bhlh13 bhlh14 bhlh17 coi1-1, bhlh3 bhlh13 bhlh17, ttg1, myc2, and gl3 egl3, and the bHLH13-overexpressing plants used in this study were described previously.9,10,28 Mutants bhlh3 bhlh13 bhlh17 gl3 egl3 tt8, bhlh3 bhlh13 bhlh14 bhlh17 ttg1, and bhlh3 bhlh13 bhlh14 bhlh17 myc2 were generated by crossing. Arabidopsis seeds were surface-sterilized with 20% bleach, plated on Murashige and Skoog (MS) medium, chilled at 4°C for 3 days, grown in a greenhouse under a 16-hour (22°C-24°C)/8-hour (17°C-19°C) light/dark photoperiod for 7 days, and then transferred to soil.
Yeast 2-hybrid assays
The coding sequences of select bHLH3, bHLH13, bHLH14, bHLH17, and bHLH13 fragments were cloned into pLexA and pB42AD for fusion with the DNA-binding domain (BD) and activation domain (AD), respectively. The primers used for vector construction are presented in Supplemental Table 1. The indicated plasmid pairs were cotransformed into EGY48 using the Matchmaker LexA Two-Hybrid System (Clontech Laboratories, Mountain View, CA, USA) and selected on SD-Ura/-His/-Trp solid medium. Positive clones were grown in liquid SD-Ura/-His/-Trp medium for 24 hours and then tested for protein-protein interactions on SD/Gal/Raf/X-gal (-Ura/-His/-Trp/-Leu) induction medium. Images were taken after 3 days of incubation at 30°C.
Protoplast transient expression assays
bHLH13 and its derivatives were fused to the GAL4 DNA-BD (GAL4DB) under the control of the 35S promoter. The primers used for vector construction are listed in Supplemental Table 1. GAL4(4X)-D1-3(4X), which contains 4 tandem copies of the GAL4 DNA-binding site, was used as a reporter; binding of the GAL4DB induced GUS expression. The firefly luciferase gene (LUC) driven by the 35S promoter was used as an internal control. The GUS signal was normalized to the internal LUC activity level. The preparation and transfection of Arabidopsis protoplasts were done as described previously. 28
Anthocyanin content and root length measurements
Arabidopsis seeds were plated on MS medium supplied with different concentrations of methyl-JA (MeJA), chilled at 4°C in the dark for 3 days, and then transferred to a greenhouse. The 11-day-old seedlings were measured for root length and anthocyanin content as described previously. 28 Our anthocyanin content data are presented as the (A535-A650) per gram of fresh weight.
Botrytis cinerea infection
Ten rosette leaves were detached from 4-week-old plants of each genotype, placed in plastic Petri dishes (150 mm in diameter) containing 1% agar and wet filter paper, inoculated with 5 μL of B cinerea (106 spores/mL) in potato dextrose broth, covered with lids, and kept in the dark under high humidity at 22°C. Lesion sizes were determined 48 hours postinoculation.
Insect defense assays with S exigua
Spodoptera exigua eggs were treated at 27°C for 2 days, and the newly hatched larvae were fed with detached rosette leaves of a similar size in a plastic Petri dish. After 8 days of feeding, the weights of the surviving larvae were measured.
Quantitative real-time polymerase chain reaction
To produce the data shown in Figures 3E and 6E, seedlings grown on MS medium with or without 5 μM MeJA for 11 days were collected for RNA extraction. Quantitative real-time polymerase chain reaction (PCR) was performed with Real Master Mix (SYBR Green I; Takara Bio Inc., Kusatsu, Japan) using the ABI7500 Real-Time PCR System (Applied Biosystems, Foster City, CA, USA). The primers used are listed in Supplemental Table 1. ACTIN8 was used as an internal control.
Accession numbers
The Arabidopsis Genome Initiative numbers for the genes mentioned in this article are as follows: bHLH3 (AT4G16430), bHLH13 (AT1G01260), bHLH14 (AT4G00870), bHLH17 (AT2G46510), MYC2 (AT1G32640), TT8 (AT4G09820), GL3 (AT5G41315), EGL3 (AT1G63650), TTG1 (AT5G24520), COI1 (AT2G39940), VSP1 (AT5G24780), LOX2 (AT3G45140), PDF1.2 (AT5G44420), TAT1 (AT4G23600), and ACTIN8 (AT1G49240).
Results
bHLH13 forms dimers and functions in transcriptional repression
To investigate whether bHLH subgroup IIId transcription factors can form dimers, we performed yeast 2-hybrid (Y2H) assays using fusion proteins in which bHLH17, bHLH13, bHLH3, and bHLH14 were combined with the DNA-BD or AD encoded by pLexA and pB42AD, respectively. Our results indicated that BD-fused bHLH17, bHLH13, and bHLH3 interacted with AD-fused bHLH17, bHLH13, and bHLH3, but not with bHLH14 (Figure 1A), suggesting that bHLH17, bHLH13, and bHLH3 form homodimers and heterodimers.
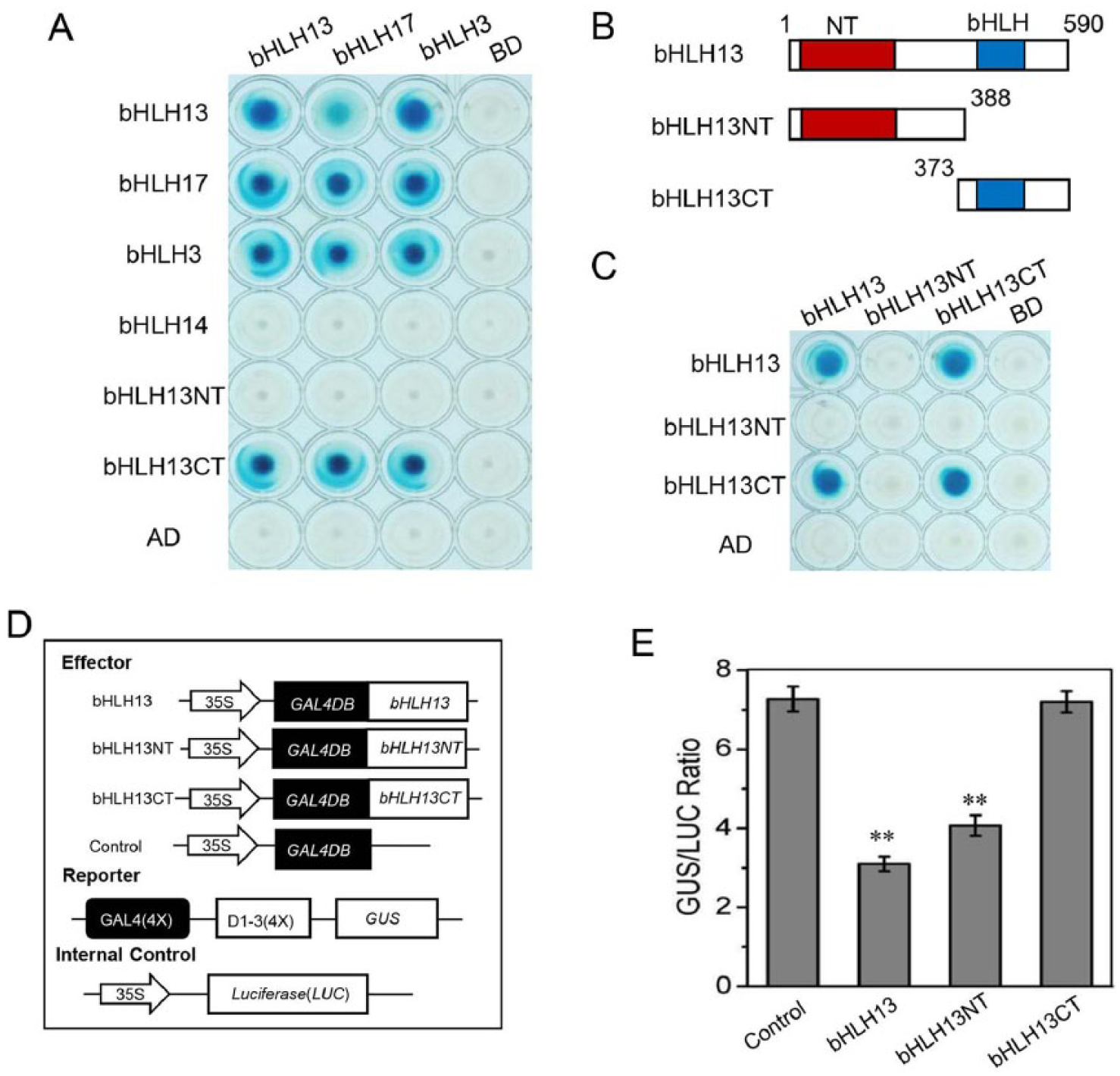
bHLH13 forms dimers and functions in transcriptional repression. (A) Results of Y2H assay for interactions between bHLH3, bHLH13, or bHLH17 and bHLH3, bHLH13, bHLH14, bHLH17, bHLH13NT, or bHLH13CT. bHLH3, bHLH13, and bHLH17 were fused individually with the DNA-BD or AD. bHLH14, bHLH13NT, and bHLH13CT were fused individually with the AD domain. (B) Schematic diagram showing the bHLH13 domain constructs. The conserved N-terminal (NT) and bHLH domains are indicated by red and blue boxes, respectively. Numbers indicate the amino acid positions. (C) Results of Y2H assay for interactions among bHLH13, bHLH13NT, and bHLH13CT fused with the BD or AD domain, respectively. (D) Schematic diagram showing the constructs used in our transient expression assays. (E) Transient expression assays revealed that bHLH13 and bHLH13NT could perform transcriptional repression, whereas bHLH13CT could not. The GUS/LUC ratio represents GUS activity relative to the internal control, LUC. The values are the means (± SE) from 3 biological replicates. Asterisks represent Student t test significance compared with the control (**P < .01).
To test which domain of bHLH subgroup IIId transcription factors is responsible for this dimerization, we divided bHLH13 into 2 parts, its N-terminal region (bHLH13NT) and its C-terminal region (bHLH13CT) containing the bHLH domain (Figure 1B), for fusion with the BD or AD. The Y2H assays revealed that bHLH17, bHLH3, bHLH13, and bHLH13CT formed dimers with bHLH13CT, but not with bHLH13NT (Figure 1A to C), suggesting that bHLH17, bHLH13, and bHLH3 form homodimers and heterodimers via their C-terminal bHLH domains.
Our previous transient expression data showed that bHLH subgroup IIId transcription factors, including bHLH13, are transcriptional repressors. 28 In this study, we fused bHLH13NT and bHLH13CT with the GAL4DB to examine which domain is essential for transcriptional repression using the GAL4DB and a reporter gene containing 4 DNA-binding sites (GAL4(4X)-D1-3(4X)-GUS) in a protoplast transient expression assay (Figure 1D). As shown in Figure 1E, bHLH13NT and bHLH13 repressed the expression of the GUS reporter, whereas bHLH13CT did not, suggesting that the N-terminus of bHLH13 is necessary for its transcriptional repression activity.
bHLH13 overexpression represses plant defense responses
The mutation of bHLH subgroup IIId transcription factors can enhance plant resistance to the necrotrophic fungus B cinerea and defense against the insect S exigua. 28 We thus used our previously generated bHLH13 overexpression transgenic plants (an approximately 16-fold increase over the wild-type level) 28 and tested whether that overexpression would attenuate plant defense responses. As shown in Figure 2A, bHLH13-overexpressing plants were more susceptible to infection by B cinerea. Larvae gained more weight when fed with leaves from bHLH13-overexpressing plants than when they were fed with wild-type Col-0 leaves (Figure 2B). These results suggest that bHLH13 overexpression inhibits plant defense against necrotrophic fungi and insect attacks.

bHLH13 overexpression represses defense responses in Arabidopsis. (A) The lesion sizes on detached rosette leaves from 4-week-old wild-type Col-0 and bHLH13-overexpressing (bHLH13OE-7) plants at 2 days postinoculation with Botrytis cinerea spores. The values are the means (± SE) from 3 biological replicates. Asterisks represent Student t test significance compared with Col-0 plants (**P < .01). (B) The weight of Spodoptera exigua larvae 8 days after feeding with rosette leaves from Col-0 and bHLH13OE-7 plants. The values are the means (± SE) from 3 biological replicates. Asterisks represent Student t test significance compared with feeding with Col-0 plants (*P < .05).
The mutation of COI1 blocks the JA hypersensitivity of bhlh3 bhlh13 bhlh14 bhlh17 mutant
The mutation of COI1 leads to JA insensitivity, whereas the abolishment of bHLH subgroup IIId transcription factors results in JA hypersensitivity. 28 We thus next investigated whether the mutation of bHLH subgroup IIId transcription factors could suppress the phenotypes of coi1-1.
As shown in Figure 3, compared with wild-type, bhlh3 bhlh13 bhlh14 bhlh17 plants were much more sensitive to JA in terms of root growth inhibition, anthocyanin accumulation, and the expression of JA-responsive genes, including VEGETATIVE STORAGE PROTEIN1 (VSP1) and LIPOXYGENASE2 (LOX2). The bhlh3 bhlh13 bhlh14 bhlh17 coi1-1 quintuple mutant lacked the JA hypersensitivity of the bhlh3 bhlh13 bhlh14 bhlh17 mutant in terms of root growth inhibition, anthocyanin biosynthesis, and JA-induced gene expression. These data indicated that coi1-1 abolished the responses of bhlh3 bhlh13 bhlh14 bhlh17 plants to exogenous JA.

coi1-1 blocked root growth and anthocyanin accumulation in the quadruple mutant of IIId bHLH factors. (A-C) Root phenotypes of (A) 7-day-old seedlings, and (B) root lengths and (C) anthocyanin contents of 11-day-old wild-type (WT) Col-0, bhlh3 bhlh13 bhlh14 bhlh17 (Q4), coi1-1, and bhlh3 bhlh13 bhlh14 bhlh17 coi1-1 (Q4 coi1-1) seedlings grown on MS medium supplied without (Mock) or with the indicated concentrations (µM) of MeJA (JA). FW indicates fresh weight. The values are the means (± SE) from 3 biological replicates. Lowercase letters indicate significant differences by a one-way analysis of variance (ANOVA; P < .05). (D) Phenotypes of 7-day-old seedlings (upper panel) and 11-day-old seedlings (lower panel) of the indicated genotypes grown on MS medium supplied without (Mock) or with the indicated concentrations (µM) of MeJA (JA). (E) Quantitative real-time PCR analysis of VSP1 and LOX2 in 11-day-old seedlings of the indicated genotypes grown on MS medium without (Mock) or with 5 μM MeJA (JA). ACTIN8 was used as an internal control. The values are the means (± SE) from 3 biological replicates. Lowercase letters indicate significant differences by a one-way ANOVA (P < .05).
In addition, in contrast to the elevated defense of bhlh3 bhlh13 bhlh14 bhlh17 plants, both coi1-1 and the quintuple mutant bhlh3 bhlh13 bhlh14 bhlh17 coi1-1 were susceptible to B cinerea and S exigua (Figure 4), indicating that the mutation of COI1 blocked the increased defense of bhlh3 bhlh13 bhlh14 bhlh17 against B cinerea and S exigua.

coi1-1 blocked the defense responses of the quadruple mutant of IIId bHLH factors. (A) The lesion sizes on detached leaves from 4-week-old wild-type Col-0, bhlh3 bhlh13 bhlh14 bhlh17 (Q4), coi1-1 and bhlh3 bhlh13 bhlh14 bhlh17 coi1-1 (Q4 coi1-1) plants at 2 days postinfection with Botrytis cinerea spores. The values are the means (± SE) from 3 biological replicates. Lowercase letters indicate significant differences by a one-way analysis of variance (ANOVA) (P < .05). (B) Larval weight after feeding with rosette leaves from wild-type Col-0, Q4, coi1-1, and Q4 coi1-1 plants for 8 days. The values are the means (± SE) from 3 biological replicates. Lowercase letters indicate significant differences by a one-way ANOVA (P < .05).
Taken together (Figures 3 and 4), our results showed that bhlh3 bhlh13 bhlh14 bhlh17 was unable to restore the phenotype of coi1-1, and that coi1-1 suppressed the JA hypersensitivity of bhlh3 bhlh13 bhlh14 bhlh17 plants.
Disruption of TTG1/bHLH/MYB complex abolishes the anthocyanin accumulation in the mutants of IIId bHLH factors
JA-induced anthocyanin accumulation is enhanced by mutations in bHLH subgroup IIId transcription factors, and it is blocked by abolishment of the TTG1/bHLH/MYB complex.9,28 bHLH17 antagonizes the TTG1/bHLH/MYB complex to repress expression of the anthocyanin biosynthetic gene DIHYDROFLAVONOL REDUCTAS (DFR). 28 We therefore tested whether mutations in bHLH subgroup IIId transcription factors could restore anthocyanin accumulation in plants carrying mutations in the TTG1/bHLH/MYB complex, or whether anthocyanin accumulation in plants with mutated bHLH subgroup IIId transcription factors would be blocked by the mutation of the TTG1/bHLH/MYB complex.
Compared with wild-type plants, bhlh3 bhlh13 bhlh14 bhlh17 and bhlh3 bhlh13 bhlh17 plants accumulated more anthocyanin in response to JA treatment, whereas neither ttg1 nor gl3 egl3 tt8, a triple mutant of the TTG1/bHLH/MYB complex, was responsive to JA in terms of anthocyanin accumulation (Figure 5). JA exposure did not induce anthocyanin accumulation in bhlh3 bhlh13 bhlh14 bhlh17 ttg1 and bhlh3 bhlh13 bhlh17 gl3 egl3 tt8 plants (Figure 5), suggesting that the mutation of the TTG1/bHLH/MYB complex blocked JA-induced anthocyanin accumulation in bHLH subgroup IIId transcription factor mutants.

Mutation of the TTG1/bHLH/MYB complex disrupted anthocyanin accumulation in the mutants of IIId bHLH factors. The anthocyanin contents in 11-day-old wild-type Col-0, bhlh3 bhlh13 bhlh14 bhlh17 (Q4), ttg1, bhlh3 bhlh13 bhlh14 bhlh17 ttg1 (Q4 ttg1), bhlh3 bhlh13 bhlh17 (bhlh3/13/17), gl3 egl3 tt8, and bhlh3 bhlh13 bhlh17 gl3 egl3 tt8 (bhlh3/13/17 gl3 egl3 tt8) plants treated without (Mock) or with 15 μM MeJA are shown. The values are the means (± SE) from 3 biological replicates. Lowercase letters indicate significant differences by a one-way analysis of variance (P < .05).
myc2 suppresses JA-inhibited root growth in bhlh3 bhlh13 bhlh14 bhlh17
We previously showed that bHLH17 inhibited the MYC2-activated expression of TYROSINE AMINOTRANSFERASE1 (TAT1). 28 Thus, we investigated the phenotypes of bhlh3 bhlh13 bhlh14 bhlh17 myc2 plants to study the genetic relationship between bHLH subgroup IIId transcription factors and MYC2. As shown in Figure 6, myc2 partially suppressed the hypersensitivity of bhlh3 bhlh13 bhlh14 bhlh17 plants in terms of JA-inhibited root growth, JA-induced anthocyanin accumulation, and JA-induced expression of VSP1, TAT1, and LOX2; however, it enhanced the JA-responsive expression of PLANT DEFENSIN1.2 (PDF1.2), suggesting that bHLH subgroup IIId transcription factors regulate JA responses in part by antagonizing MYC2.
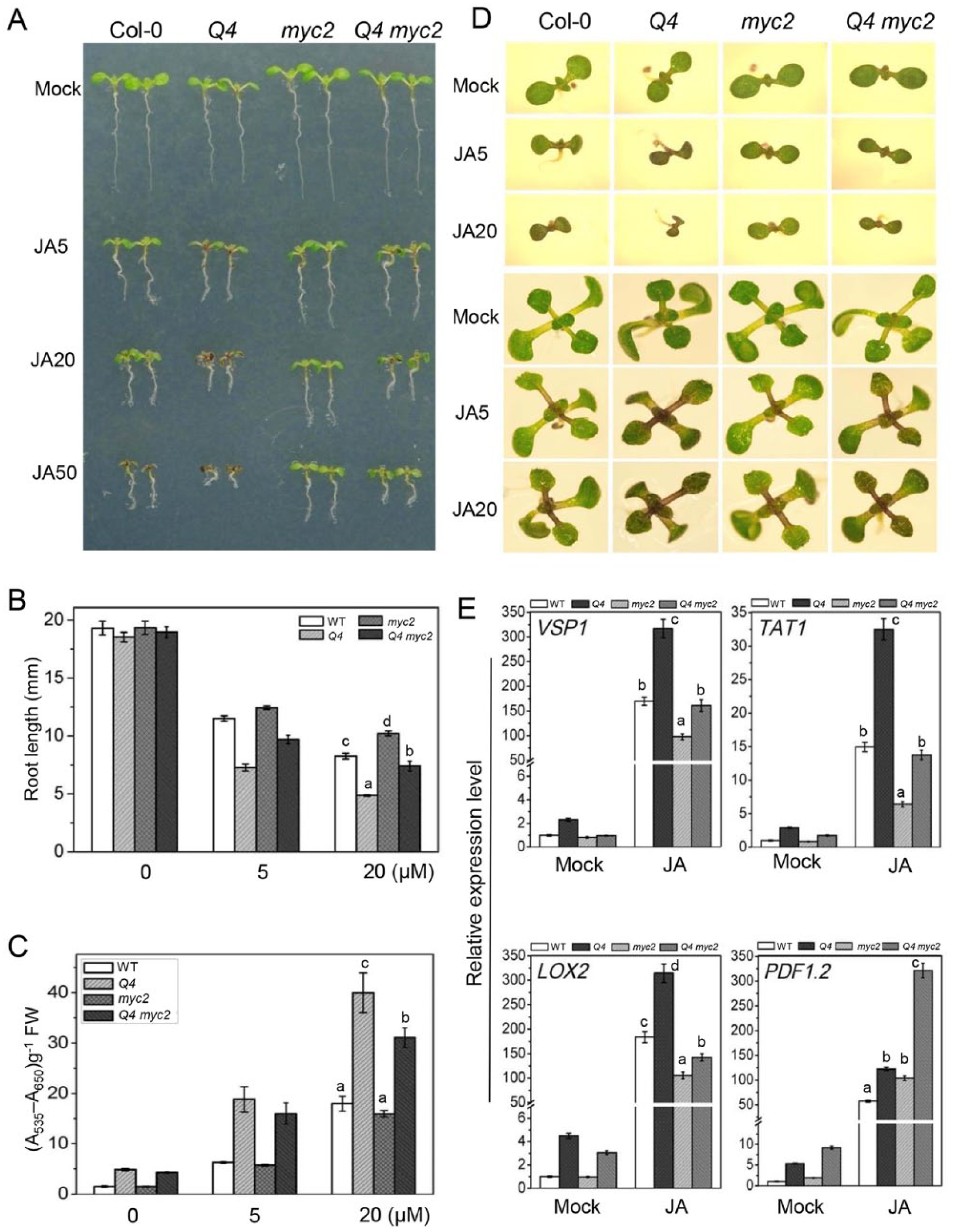
myc2 suppressed root growth and anthocyanin accumulation in the quadruple mutant of IIId bHLH factors. (A-C) The root phenotypes of (A) 7-day-old seedlings and (B) the root lengths and (C) anthocyanin contents of 11-day-old seedlings of wild-type (WT) Col-0, bhlh3 bhlh13 bhlh14 bhlh17 (Q4), myc2, and bhlh3 bhlh13 bhlh14 bhlh17 myc2 (Q4 myc2) grown on MS medium and supplied without (Mock) or with the indicated concentrations (µM) of MeJA (JA) are shown. FW indicates fresh weight. The values are the means (± SE) from 3 biological replicates. Lowercase letters indicate significant differences by a one-way analysis of variance (ANOVA) (P < .05). (D) Phenotypes of 7-day-old seedlings (upper panel) and 11-day-old seedlings (lower panel) of the indicated genotypes grown on MS medium supplied without (Mock) or with the indicated concentrations (µM) of MeJA (JA). (E) Quantitative real-time PCR analysis of the expression levels of VSP1, TAT1, LOX2, and PDF1.2 in 11-day-old seedlings of the indicated genotypes supplied without (Mock) or with 5 μM MeJA (JA). ACTIN8 was used as an internal control. The values are the means (± SE) from 3 biological replicates. Lowercase letters indicate significant differences by a one-way ANOVA (P < .05).
Discussion
The results of this (Figure 1A) and previous studies30,31 indicate that the bHLH subgroup IIId transcription factors bHLH17, bHLH13, and bHLH3 form homodimers and heterodimers. We also found that the bHLH domain at the C-terminus mediates these dimeric interactions (Figure 1A to C). A recent study showed that tetrameric MYC2 is more efficient than dimeric MYC3 in activating gene expression. 33 It will be interesting to investigate whether bHLH subgroup IIId transcription factors form tetramers to repress JA responses.
Previous studies have shown that bHLH subgroup IIId transcription factors such as bHLH17 function as transcriptional repressors; they bind to the promoters of target genes (eg, DFR and TAT1), antagonize transcriptional activators (eg, MYC2 and the TTG1/bHLH/MYB complex), and repress JA responses. 28 In this study, we found that the N-terminus of bHLH13 is required for its transcriptional repression activity (Figure 1D and E), and that the overexpression of bHLH13 inhibits plant defense against B cinerea and S exigua (Figure 2). Consistent with this, the JA-induced expression of defense-related genes PDF1.2 and VSP1 was attenuated in bHLH13-overexpressing plants. 28 It remains to be determined how bHLH13 functions as a transcriptional repressor through its N-terminus without dimerization.
Our bHLH subgroup IIId transcription factor mutants exhibited JA hypersensitivity due to hyperactivation of their downstream target genes by transcriptional activators. The bhlh3 bhlh13 bhlh14 bhlh17 coi1-1 mutant exhibited similar JA insensitivity to coi1-1, including JA-inhibited root growth (Figure 3), anthocyanin accumulation (Figure 3), defense responses (Figure 4), and a stay-green phenotype, 10 demonstrating that even though bHLH subgroup IIId transcription factors were mutated in bhlh3 bhlh13 bhlh14 bhlh17 coi1-1, JA responsiveness and JA-related defense could not be rescued because transcriptional activators (eg, MYC2/3/4 and the TTG1/bHLH/MYB complex) were still inhibited by JAZ accumulation.
We also used loss-of-function mutations to investigate the genetic relationship between bHLH subgroup IIId transcription factors and transcriptional activators (MYC2/3/4 and the TTG1/bHLH/MYB complex). The myc2 myc3 myc4 mutant disrupted the JA-induced leaf senescence of the quadruple mutant bhlh3 bhlh13 bhlh14 bhlh17. 10 myc2 attenuated the JA-inhibited root growth and JA-induced anthocyanin accumulation in bhlh3 bhlh13 bhlh17 32 and bhlh3 bhlh13 bhlh14 bhlh17 plants (Figure 6), respectively, suggesting that bHLH3, bHLH13, bHLH14, and bHLH17 genetically antagonize MYC2 in JA signaling. Furthermore, our genetic and physiological data show that disruption of the TTG1/bHLH/MYB complex blocked anthocyanin overaccumulation in bHLH subgroup IIId transcription factor mutants (Figure 5). These findings suggest that bHLH subgroup IIId transcription factors function as general repressors of JA responses, and that they antagonize different transcriptional activators to repress distinct JA responses.
Conclusions
In this study, we found that the C- and N-terminal regions of bHLH13 are required for the dimerization and transcriptional repression function, respectively. bHLH13 overexpression inhibited Arabidopsis against B cinerea and S exigua. Meanwhile, the abolishment of COI1, the TTG1/bHLH/MYB complex, and MYC2, respectively, disrupted JA hypersensitivity, anthocyanin accumulation, and JA-inhibited root growth in the quadruple mutant bhlh3 bhlh13 bhlh14 bhlh17. Our findings provide a new insight into the antagonism between IIId bHLH factors and different transcriptional activators in JA pathway.
Supplemental Material
Supplemental_Table_1 – Supplemental material for bHLH13 Regulates Jasmonate-Mediated Defense Responses and Growth
Supplemental material, Supplemental_Table_1 for bHLH13 Regulates Jasmonate-Mediated Defense Responses and Growth by Huang Huang, Hua Gao, Bei Liu, Meng Fan, Jiaojiao Wang, Cuili Wang, Haixia Tian, Lanxiang Wang, Chengyuan Xie, Dewei Wu, Liangyu Liu, Jianbin Yan, Tiancong Qi and Susheng Song in Evolutionary Bioinformatics
Footnotes
Acknowledgements
The authors thank Dr Kang Chong for providing the vectors used in our protoplast transient expression assays.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Natural Science Foundation of China (31670315), the Support Project of High-Level Teachers in Beijing Municipal Universities in the Period of 13th Five-Year Plan (CIT&TCD201704086), Da Beinong Young Teachers’ Scientific Research Foundation of Beijing University of Agriculture (17ZK001), the Research Fund for Young Scientists of BUA (SXQN201803), and the Beijing Nova Program (Z171100001117037).
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
SS and HH designed the study. HH, HG, BL, MF, JW, CW, HT, LW, CX, DW, TQ, and SS performed the experiments. HH, HG, LL, JY, and SS analyzed the data. HH, GH, and SS wrote the manuscript. HH, HG, BL, and MF contributed equally to this work.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
