Abstract
Introduction:
The CELT ACD (Vasorum Ltd, Dublin, Ireland) is an access closure device achieving hemostasis with a steel radiopaque structure deployed into the arterial wall without sheath exchange. There is limited data regarding its use in neurointerventional procedures, particularly with larger arteriotomies.
Methods:
A prospective registry of neurointerventional procedures at a single center between July 2024 and February 2025 was retrospectively reviewed. Consecutive transfemoral cases with 6, 7, and 8F sheaths undergoing closure with a newly-deployed 6 or 7F CELT were included. The primary outcomes were rates of device failure and access site complications.
Results:
Of 116 patients, there were 119 CELT closures. Successful deployment occurred in 98.3% (117/119). Other than a small groin hematoma (0.8%; 1/119), there were no other access site-related complications reported.
Conclusion:
This early experience suggests favorable performance and minimal complications with CELT in neurointerventional procedures with larger arteriotomies.
Introduction
Compared to manual compression, vascular closure devices (VCDs) shorten the time needed to achieve hemostasis, decreased the time of immobilization before ambulation, and reduce complication rates, particularly in patients on anticoagulants.1–3 Increasing utilization of neuroendovascular procedures over recent years, including the use of larger arteriotomies to introduce a newer generation of neurovascular devices, has underscored the need for safe and effective VCDs. 4 VCD complications still remain prevalent and display varying degrees of effectiveness when compared to manual compression.5,6 Recent literature has focused on providing clarity on the effectiveness of VCD products in cardiovascular intervention.7,8 However, delayed adoption in neurointervention has limited available data on novel VCD effectiveness. Novel VCDs have demonstrated promise for safe and effective closure optimized for the current landscape of neurointerventional procedures.4,9,10
The CELT (Vasorum Ltd, Dulbin, Ireland) closure device has predominantly been used in cardiovascular intervention but has recently garnered attention in the neurointerventional space. CELT’s mechanism to achieve hemostasis involves deployment of a biocompatible stainless steel radiopaque structure into the arterial wall without requiring sheath exchange; it is easily visible on fluoroscopy and allows for future reaccess. Studies in vascular intervention demonstrate the CELT’s ability to reduce time to hemostasis, ambulation, and discharge, while maintaining a low complication rate. 11 However there is limited literature of the real-world experience with this newer device, especially regarding its use in neurointerventional procedures. 12 The objective of this study is to determine the safety and performance of CELT in diagnostic and interventional neurovascular procedures in 6–8F arteriotomy closures.
Methods
De-identified data may be made available upon reasonable request to the corresponding author. This study was approved by the institutional review board with waiver of informed consent. The observational analysis was reported in accordance with the STROBE guidelines. This retrospective study was conducted in accordance with the principles of the Declaration of Helsinki. 13
Patient selection
A prospectively maintained registry of consecutive patients undergoing neurointerventional procedures between July 2024 and February 2025 at a single center was retrospectively reviewed. Starting in July 2024, institutional protocol for 6F or larger transfemoral access was to utilize the appropriately-size CELT. Patients undergoing transradial neurointervention were excluded from this analysis per protocol. For 6F arteriotomies, the 6F device was selected; for 7–8F arteriotomies, the 7F device was utilized. Patients were included in the analysis if a new CELT was deployed for femoral access closure of 6–8F arteriotomies. Patients were not excluded for prior transfemoral access. A flow diagram of patient selection is shown in Figure 1.
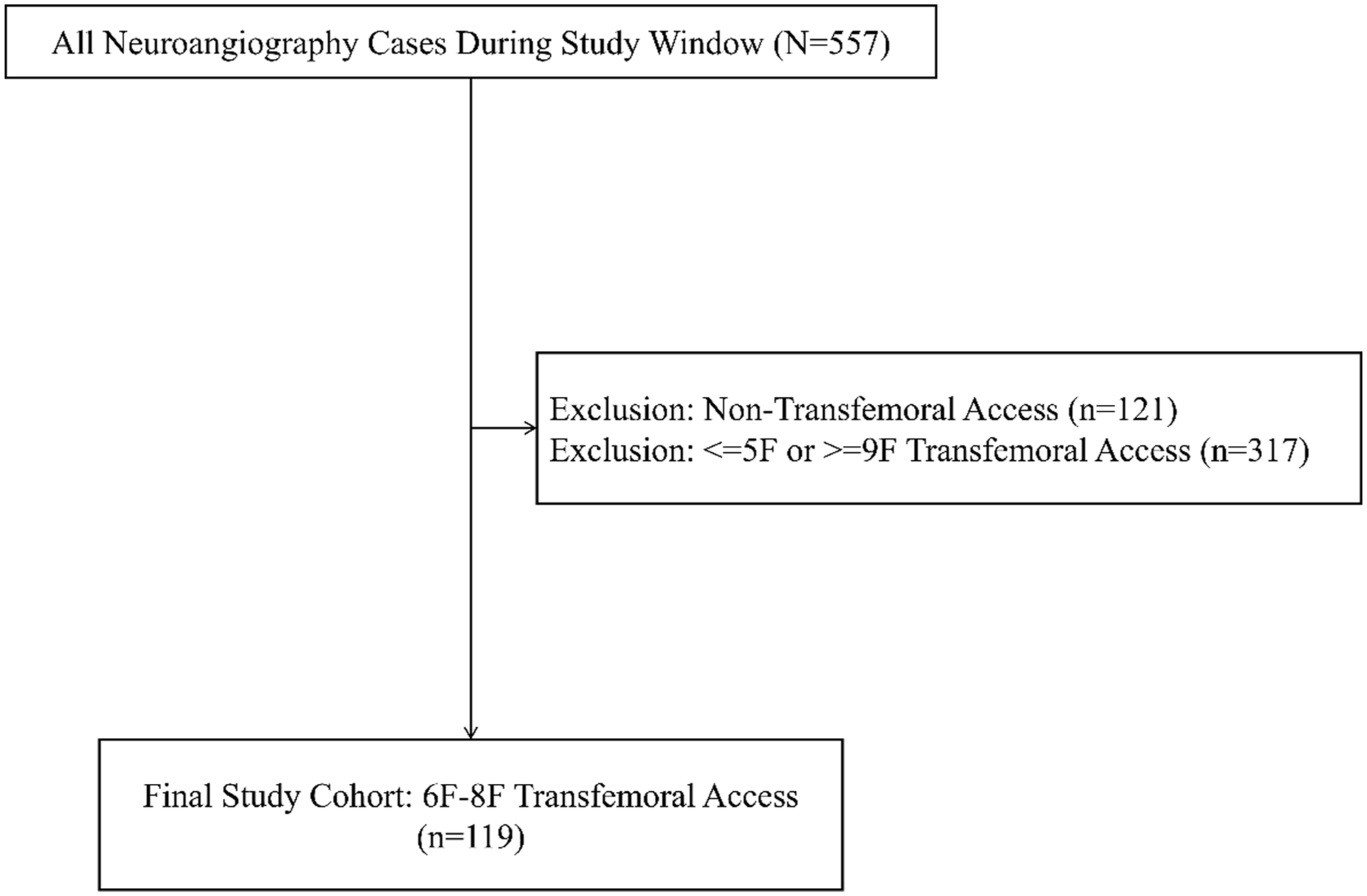
Depiction of case selection. Cohort included only transfemoral 6–8F CELT ACD deployments; non-transfemoral cases were not captured. Final cohort size is 119.
CELT technique
The CELT vascular closure (CELT) is an FDA approved metallic implant that is used to close arterial puncture sites in endovascular procedures allowing hemostasis without manual compression. Although primarily used in femoral artery closures, CELT has also been applied in radial and carotid arteries. 14 Sizes of CELT range from 5 to 7F. Additionally, the 7F CELT can be utilized to close to 8 and 9F arteriotomies.
The CELT was deployed according to the Instructions for Use. Our institutional protocol includes ultrasound guidance to confirm anterior wall puncture in the common femoral artery using a micropuncture arterial access kit. Further, fluoroscopy is utilized at the time of device deployment to confirm adequate intraluminal placement above the site of arteriotomy using AP fluoroscopy prior to deployment. After deployment but immediately prior to detachment, fluoroscopy was used to push and pull the device to ensure the device was stably attached to the common femoral artery wall. After detachment and deployment of the device, our local protocol included performing single-shot fluoroscopy to confirm deployment without unintended device migration or embolization. No routine manual compression was held after CELT deployment. After closure, a post-operative groin ultrasound is performed per institutional protocol for closures of all 8F arteriotomy sizes or larger. Patients were uniformly ordered to lay flat with the access extremity straight for 2 h post-procedure, prior to ambulation. All patients were scheduled to receive follow-up in-person office visits at 2–3 weeks post-procedure.
Outcomes
Baseline demographic data were collected, including: age, sex, comorbidities (i.e. hypertension, hyperlipidemia, diabetes mellitus, coagulopathy, and atrial fibrillation), any antiplatelet/anticoagulant use, body mass index (BMI), serum laboratory values related to hemostasis (i.e. international normalized ratio (INR), platelet count), and length of hospitalization. Procedural variables collected include: the indication for neurointerventional procedure, laterality of access site, and arteriotomy size.
Success of CELT deployed, defined as placement of the CELT at the target site without dislodgement on DSA and with no or minimal compression required for achieving hemostasis, was ascertained. Complications of the VCD during hospitalization were collected, such as access site infection, femoral artery dissection, groin hematoma, femoral artery pseudoaneurysm, lower limb ischemia, and need for vascular surgical intervention for device retrieval/removal. If a groin ultrasound was obtained, the frequency of an abnormal finding identified on imaging was collected.
The primary objective of this study was the overall rate of successful CELT deployment in neuroendovascular procedures. Secondary outcome was the rate of specific complications associated with the device and proportion of patients requiring any additional manual compression.
Statistical analysis
Descriptive analyses were performed. Continuous measures were reported as medians with interquartile ranges (IQR), and categorical measures were reported as frequencies. Missing data was not imputed. All analyses were performed in JMP v 18 (SAS Institute, Cary, NC, USA).
Results
Of 116 patients, there were 119 closures with a CELT during the study window. The average age was 65.5 years (IQR 57–78.8), and 48.6% (56/117) were female (Table 1). The most common comorbid condition was hypertension (76.7%; 89/116), and 39.7% (46/116) were on antiplatelets or anticoagulants prior to the procedure. Approximately one-third of the patients (36/116; 31.0%) had BMI ⩾30 kg/m2. Most procedures were either embolization/coiling (57/119; 47.9%) or thrombectomies (33/119; 27.7%; Table 2). Most procedures had 6F arteriotomies (73/119; 61.3%); 25.2% (30/119) had 7F arteriotomies and 13.4% (16/119) had 8F arteriotomies.
Demographics.
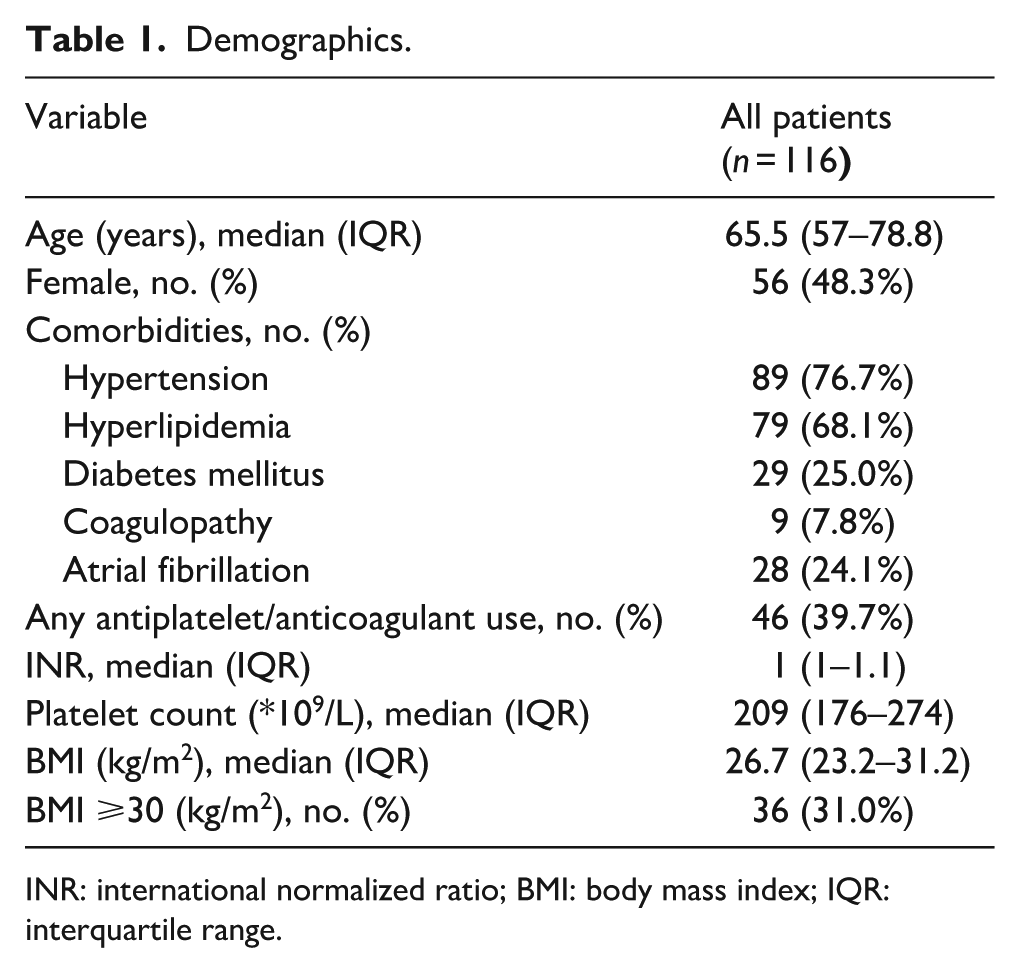
INR: international normalized ratio; BMI: body mass index; IQR: interquartile range.
Intraprocedural outcomes.
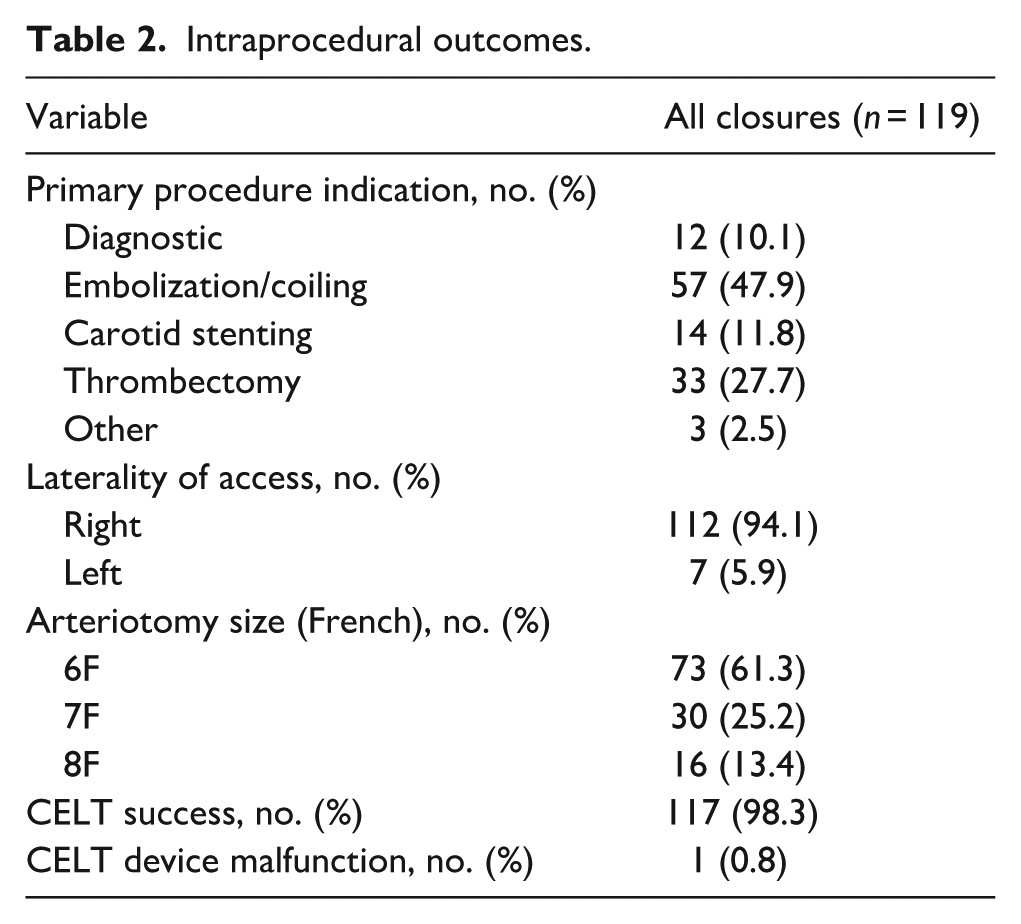
Successful deployment of CELT, without dislodgement or prolonged compression, occurred in 98.3% (117/119) of closures (Table 2). Two patients (1.7%, 2/119) required additional manual compression for hemostasis; one closure required prolonged compression after CELT deployment to achieve adequate hemostasis; in this case, the bleeding was felt to be cutaneous and the patient was both fully anticoagulated and was under P2Y12 inhibition. In the second case, the CELT device failed to deploy; the device was withdrawn and hemostasis was achieved using manual pressure.
One post-procedural access site complication included a small groin hematoma (1/119; 0.8%; Table 3). No other access site-related complications (i.e. infection, dissection, lower limb ischemia, need for surgical intervention/embolectomy for device retrieval) were reported.
Postprocedural outcomes and complications.
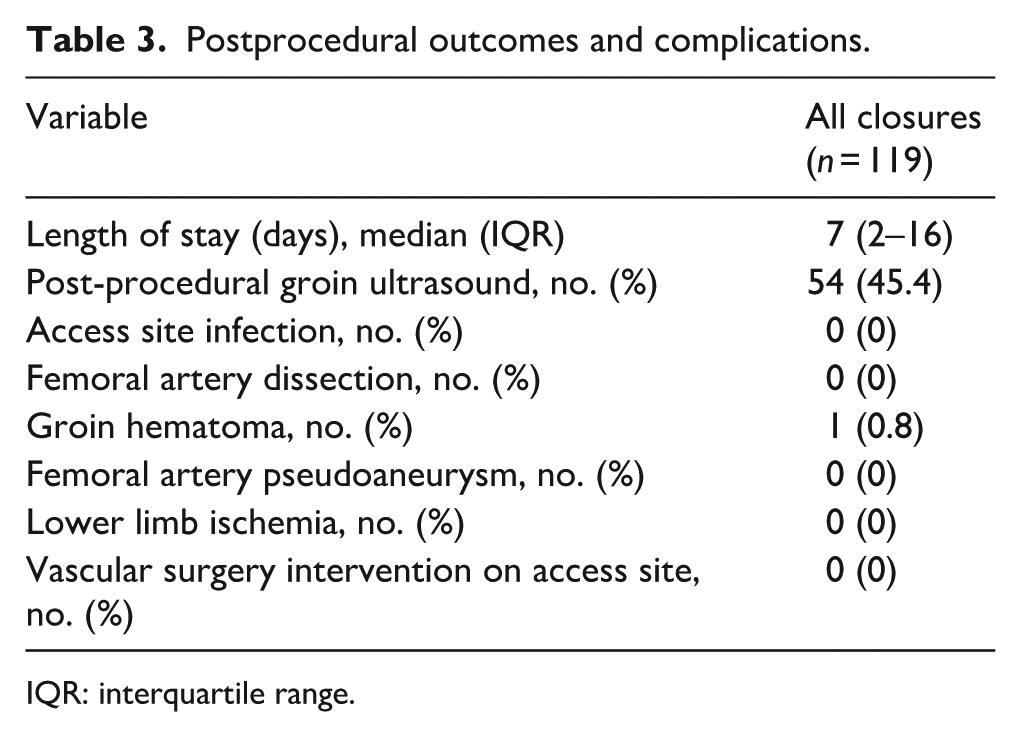
IQR: interquartile range.
Discussion
Our study focuses on the neurointerventional usage of CELT in 119 femoral artery closures for 6, 7, and 8F arteriotomies. The CELT device demonstrated a 98.3% deployment success with 1 device malfunction (0.8%) and 1 post procedure complication (0.8%), with no major adverse events. The consistency of success with different sized CELT closure devices is pivotal for safe and efficient closure for patient outcomes.
Our 0.8% post procedure complication is nominally lower than the 2% complication rate reported by in a single interventional radiology setting. 15 A cardiovascular interventional multicenter analysis reports 4.1% complication rate for CELT 6F arteriotomies in cardiovascular intervention with 73.9% of all patients being on anticoagulants. 16 While a 4.1% complication rate for CELT 6F arteriotomies appears high; it is still lower than the 6.8% complication rate reported for manual compression closure supporting the claim that CELT has similar to lower complication rates than manual compression. 16 A recent series of 9F arteriotomy closures for complex neurointerventional procedures using the 7F CELT has concordantly demonstrated 95% of cases having no complications and 93.2% of cases achieving hemostasis with 3.75% requiring manual compression. 17 Device failure (1.5%, 6 cases) and subcutaneous hematoma (2.3%, 9 cases) made up a majority the 5% complication rate. 17 When comparing cases of CELT hemostasis failure requiring prolonged compression, we reported one case while Johnston et al. 17 reported 15 cases. The higher complication rate may be attributable to sample size and the usage of 9F arteriotomies.
Our initial institutional experience suggests that the CELT device offers a highly effective option for closing larger femoral arteriotomies utilized during neurointervention (6–8F), with complication rates comparable to or lower than those reported in broader literature for cardiovascular and neurointerventional closures. The CELT may offer a rapidly hemostatic, effective femoral artery closure for neurointerventional cases, even in the setting of anticoagulation and/or antiplatelet use. The patients in this cohort reflect a consecutive analysis of a real-world neurointervention practice inclusive of hemorrhagic and ischemic cerebrovascular disease; importantly, there were no ischemic complications experienced with the use of CELT in this cohort, suggesting that this device is safe for large arteriotomies in a population where both femoral atherosclerotic disease and antithrombotic use are common. 18 Lastly, unique to CELT is the fact that this closure device is radioopaque, allowing for direct visualization during deployment via fluoroscopy at the site of arteriotomy. This may allow for easier re-assess of the common femoral artery during subsequent procedures, with enhanced ability to directly visualize the closure device.
Limitations
This is a retrospective analysis from a single center utilizing consecutive cases as part of an institutional protocol. This series represents the early experience with the CELT device during the study period at our institution; thus, the data presented here likely captures the learning curve associated with the use of the CELT and likely expands the generalizability of this analysis, yet the results may improve with familiarity and time. However, as a single-institution analysis, the data presented here may lack generalizability in different demographic populations, across institutions, or with varied operator experiences. We have attempted to delineate our protocol with detail to mitigate this limitation. Most cases in this consecutive series were 6F arteriotomies, which limits the strength of conclusions that can be drawn for larger sheath sizes utilizing the 7F CELT. Further, it is possible that delayed complications may have been missed by our analysis, potentially underestimating complication rates. Mitigating this limitation is the length of stay in this cohort; the median inpatient admission was 7 days, and no patients had delayed major complications while inpatient. This data should be further supported by larger series in multicenter cohorts.
Conclusion
Overall, our study highlights the safety and effectiveness of CELT closure devices for 6, 7, and 8F arteriotomies in the neurointerventional setting.
Footnotes
Author contributions
Ahmet K Dalkilic: Data Collection, Writing—original draft; Manisha Koneru: Data Collection, Formal Analysis, Writing—original draft, Writing—review & editing, Supervision; Iris Charcos: Data Collection, Writing—review & editing; Joshua Santucci: Data Collection, Writing—review & editing; Ahmad A Ballout: Data Collection, Writing—review & editing; Jane Khalife: Data Collection, Writing—review & editing, Supervision; Hamza A Shaikh: Data Collection, Writing—review & editing, Supervision; Pratit D Patel: Data Collection, Writing—review & editing, Supervision; Ajith J Thomas: Data Collection, Writing—review & editing, Supervision; Tudor G Jovin: Data Collection, Writing—review & editing, Supervision; Daniel A Tonetti: Conceptualization, Investigation, Data Collection, Writing—original draft, Writing—review & editing, Supervision.
Data availability
De-identified data may be made available upon reasonable request to the corresponding author.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Daniel Tonetti is a consultant for Route 92 Medical. All other authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This retrospective cohort study was approved by the Cooper University Health Care Institutional Review Board (#22-096) with waiver of informed consent.
Consent to participate
This retrospective cohort study was approved by the Cooper University Health Care Institutional Review Board (#22-096) with waiver of informed consent.
Consent to publication
This retrospective cohort study was approved by the Cooper University Health Care Institutional Review Board (#22-096) with waiver of informed consent.
