Abstract
Background and objectives:
Peripherally inserted central catheters (PICC), and peripherally inserted midlines (MC) represent a possible less invasive alternative option to totally implantable vascular access devices (TIVAD) in cancer patients. This study aims to investigate the outcomes of PICC, and MC devices in patients undergoing chemotherapy.
Methods:
A prospective non-concurrent observational study was conducted at the IRCCS Ospedale Policlinico San Martino, Genova (Italy) on 559 cancer patients candidate to chemotherapy bearing PICC, and MC during the period 2019–2022. The primary endpoint was the comparative analysis for failure of the different types of peripheral insertion accesses requiring removal. Secondary outcome measures were age at diagnosis, gender, period of diagnosis, cancer site and type of chemotherapy. The negative binomial regression analysis was applied to estimate catheter removal rate ratios (RR), along with 95% confidence limits (95% CL), in each category of the risk factors.
Results:
The median follow-up time was 2.6 months (IQR = 1.4–4.6; min-max = 0.03–12.7), and at the end of the study period, a total of 45 catheter failures were detected. A risk reduction of approximately 75% (RR = 0.24; 95% CL = 0.11–0.51) was emphasized for patients with PICC when compared with those with MLC. Type of chemotherapy and cancer site appeared to be important predictive factors for catheter removal risk.
Conclusions:
For cancer patients undergoing chemotherapy by a non TIVAD, PICC must be preferred to MC because of higher safety and efficacy.
Keywords
Introduction
The choice of a safe and effective vascular access represents a key aspect in the therapeutic planning of oncohematological patients. Totally implantable vascular accesses (TIVAD), although safer compared to external hub devices, appears somehow more invasive and with potential patient’s compliance limitations.1–4 They may not always represent the first choice, dictated by issues related to the type of chemotherapy, duration of the treatment schedule and logistic aspects that may not be always easy to overcome.5–12
Peripherally inserted central catheters (PICCs) and midline catheters (MCs) represents a possible less invasive alternative option for oncohematological patients candidate to chemotherapy.
Easy insertion, moderate invasiveness and lower costs have made them popular in the recent decades. However, with the steady increase in their use there has been a consequent increase of complications resulting in an increased need of resources for their management.6,7
The most frequent complications of external devices are represented by local exit site infections, catheter-related bloodstream infections (CRBSI), catheter-related thrombosis (CRT), catheter occlusion, inadvertent dislodgment and other mechanical complications. They may result in a device failure and the subsequent need for replacement, additional costs and an unavoidable delay in the treatment programmes with a negative impact on patient’s management and quality of life.8–12
At the IRCCS Ospedale Policlinico San Martino, Genoa (Italy), more than 3000 procedures for not totally implantable devices are yearly performed on average and there is a perception among healthcare professionals that, based on the increase of use of external PICC and Midlines catheters, there is an increase on the rates of catheter complications in particular whenever used in cancer patients under chemotherapy.
A prospective non-concurrent observational study was designed to analyse safety and efficacy outcomes of PICC, and MC catheters used for the administration of chemotherapy treatments over the period from 01/01/2019 to 31/12/2022.
Materials and methods
Patients and data
All patients were identified through medical records and a complete medical history was retrieved along with demographic data. Information on the types of central and not central venous catheters (i.e. PICC, and MC), failures with removals, and catheter replacement during the study period were carefully evaluated.
The collected data, stored in an ad-hoc created database, were: type and indwelling time of the catheter, tumour site and histology, therapeutic protocol, catheter related thrombosis (CRT), CRBSI, local exit-site infections, dislocations, accidental removals and other complications.
The study followed the principles of the Helsinki Declaration and was approved by the IRB of the IRCCS Ospedale Policlinico San Martino (No.302/2022DB id 12384) and written consent was obtained from all participants.
The catheters were always inserted by a well-trained team of doctors and nurses of the hospital according to specific institutional protocols for a safe procedure of insertion. Vascular accesses were always performed under echography-guided vein visualization and a correct tip location for central catheters by using ECG intracavitary technique. Inserted PICCs and MCs were polyurethane 4F and 5F catheters (Synergy CT PICC Health Line, Power PICC BD-Bard and Health PICC Plan 1 Health). Chemotherapeutic regimens were classified according to their local toxicity, namely neutral (N: non-irritating or vesicant drugs), irritating (I: irritating and vesicant drugs) and hybrid (H: neutral and/or irritating drugs).
PICC catheters were used for any type of chemotherapy. Opposite, MCs were only used to infuse peripherally compatible N, and H drugs. If, following a failure of a MC, a new catheter was still necessary, a PICC was always positioned as a replacement catheter.
Baseline patients’ demographics and characteristics considered for analyses, in addition to the types of chemotherapies and catheters, were gender, age at diagnosis (four categories: 16–50, 51–60, 61–70 and 71–91), year of diagnosis (two categories: 2018–2020 and 2021–2022) and cancer sites which were aggregated as hemo-lymphopoietic system, digestive tract, breast, lung, genitourinary tract and other malignancies (i.e. thymus gland, head-neck, sarcoma, glioma and melanoma).
Study endpoints
The primary endpoint was to estimate the risk of peripheral device failure and removal because of complications.
The secondary endpoint was the potential association between the risk of device failure and other individual and clinical characteristics, with particular reference to age at diagnosis, type of tumour and type of infused chemotherapy.
Statistical analysis
Baseline patients’ demographics and clinical data considered for analyses in addition to the types of catheters (PICC, and MC) were gender, age at diagnosis (four categories: 16–50, 51–60, 61–70 and 71–91), year of diagnosis (two categories: 2018–2020 and 2021–2022) and cancer sites which were aggregated as hemo-lymphopoietic system, digestive tract, breast, lung, genitourinary tract and other malignancies (i.e. thymus gland, head-neck, sarcoma, glioma and melanoma). Chemotherapeutic regimens were classified according to their local toxicity, namely neutral (N: non-irritating or non-vesicant drugs), irritating-vesicant drugs (I) and hybrid drugs (H, neutral and irritating drugs).
Metric variables were summarized using the median value and the interquartile range (IQR) while categorical factors were described through absolute and relative (percentages) frequencies. In each category of all factors a catheter failure rate per month per 100 patients (CFR) was computed as a ratio between removal counts and observation time (person-months) accumulated over the follow-up period.
A multivariable negative binomial regression analysis was also applied to evaluate the predictive significance of patients’ baseline features, in particular the type of catheter used, on catheter failure rate. Such a method is able to estimate a rate ratio (RR) as an index of association adjusted for confounding and to allow for overdispersion in count data due to repeated catheter failures experienced by patients over the observation period. All statistical indexes were also accompanied by 95% confidence limits (95% CL) as measures of sampling variation. 13 A p-value ⩽0.05 was considered as statistically significant.
Statistical analyses were performed using Stata software (Stata Corp. Stata: Release 17. Statistical Software. College Station, TX. 2021).
Results
Five hundred and fifty-five oncology patients admitted to the day-hospital of the oncohematological department during the period 01/01/2019–31/12/2022 were enrolled in the study and followed prospectively from the date of the first catheter insertion until the end of 2022.
Table 1 shows the baseline characteristics of the patients considered for analysis. Notably, the median age was 65.6 years (IQR = 56.6–73.0), which was quite high due to the prevalence of elderly patients (32.2% in 16–60 vs 68.8% in 61–91). Women were more frequent than men (55.6% vs 44.4%) while most patients (75.1%) had been diagnosed with cancer in 2021–2022. Nearly a third (32.7%) of patients had hemo-lymphopoietic neoplasms and 4% had thymic and head-neck cancers, sarcomas, gliomas and melanomas. All other tumours shared fairly similar percentages ranging from 13% (breast) to 18% (digestive tract; Table 1).
Patients’ characteristics.

No.%: absolute/relative frequency; IQR: inter-quartile range.
Other malignancies: thymus gland (2; 0.4%), head-neck (16; 2.9%), sarcoma (1; 0.2%), glioma (1; 0.2%), melanoma (2; 0.4%).
At disease onset, hybrid chemotherapy was the most frequently administered treatment (82.1%) followed by irritating-vesicant (11.3%) and neutral therapies (6.6%). Inserted catheters were represented by PICCs (83%), and MCs (15%).
The median follow-up time was 2.6 months (IQR = 1.4–4.6; min-max = 0.03–12.7), and at the end of the study period, a total of 45 catheter failures were detected, 30 of which occurred after the first insertion, 13 after the second and 2 after the third. Therefore, regression analyses were performed on 609 observations at different follow-up times.
Table 2 shows the results of the negative binomial regression analysis in terms of both catheter failure rate and related RR. Regarding catheter type, a risk reduction of approximately 75% (RR = 0.24; 95% CL = 0.11–0.51) was emphasized for patients with PICC when compared with those with MC. This difference is also clearly depicted in Figure 1 where the cumulative catheter failure rate estimated in the two categories are compared. In addition, hybrid (RR = 1.29; 95% CL = 0.41–4.06) and irritant-vesicant (RR = 1.79; 95% CL = 0.33–9.72) therapies resulted to be associated with higher risks of catheter failure when compared to neutral therapy (CFR = 1.39; 95% CL = 0.43–4.46). The cumulative CFR estimated in each therapy category (N, H and I treatments) is shown in Figure 2.
Results of negative binomial regression modelling of catheter failure rates.

CF: catheter failures; PM: person-months at risk; CFR: catheter failure rate per month per 100 patients; RR: rate ratio; 95% CL: 95% confidence limits for CFR/RR; Ref.: reference category; p-value: probability level associated with the likelihood-ratio test; ML: midline catheter; PICC: peripherally inserted central catheter.
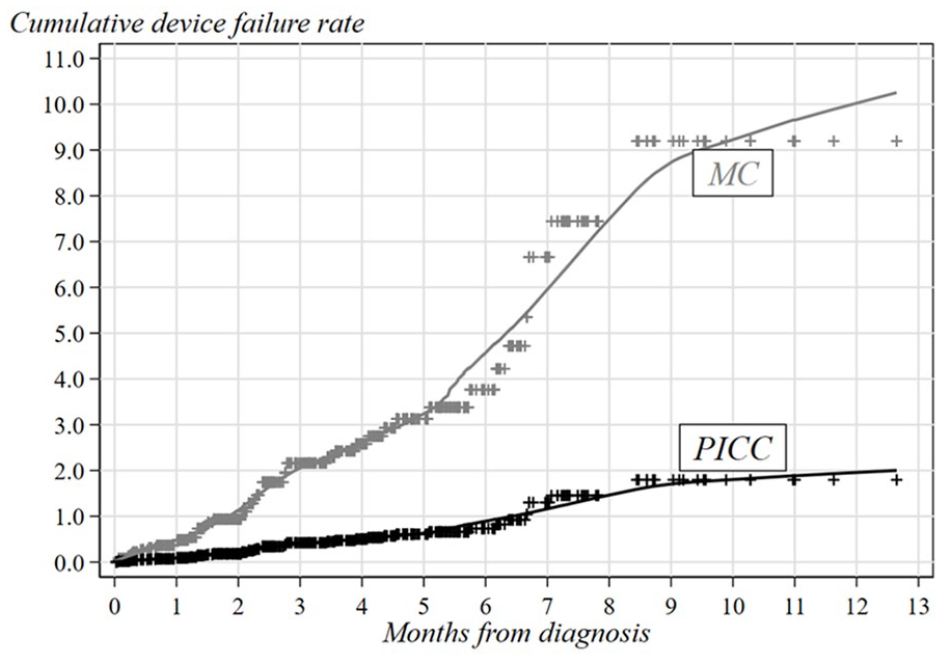
Smoothed cumulative catheter failure rates adjusted for confounding according to type of catheter.
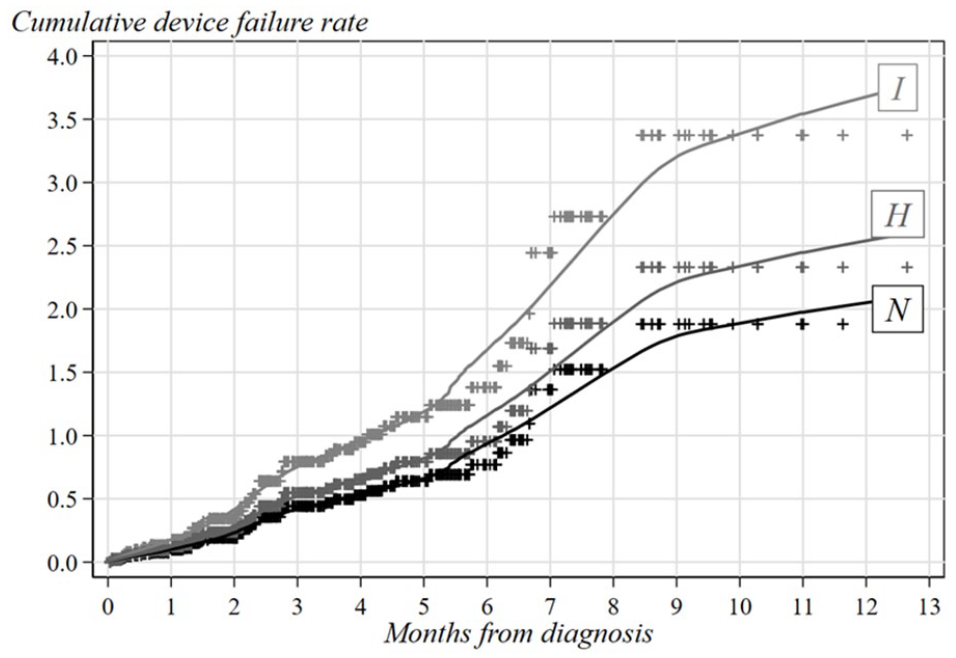
Smoothed cumulative catheter failure rates adjusted for confounding according to type of chemotherapeutic treatment.
Finally, it is worth noting that among the cancer sites considered in this analysis, hemo-lymphopoietic neoplasms were found to confer the highest risk of device removal (CFR = 3.47; 95% CL = 2.24–5.35) while patients with lung (CFR = 0.82; 95% CL = 0.25–2.75) and digestive tract (CFR = 0.59; 95% CL = 0.13–2.83) cancers showed the lowest rates corresponding to a risk reduction of about 75% (RR = 0.24; 95% CL = 0.07–0.83) and 85% (RR = 0.17; 95% CL = 0.03–0.90), respectively.
Table 3 shows the incidence of different reasons for failure reported for MC, and PICC catheters. CRT and CRBSI were shown to be the most frequent ones accounting for 28.9%, and 26.7% of cases, respectively. Differences between the incidence of various complications between MC and PICC were not statistically significant (p = 0.096).
Different failures reported for single type of vascular accesses.

MC: midline catheter; PICC: peripherally inserted central catheter; CRBSI: catheter-related bloodstream infection; CRT: catheter related thrombosis.
Chi-squared test for independence: 7.9; p-value: 0.096.
Discussion
The use of venous catheters for chemotherapy delivery in cancer patients represents a standard of practice in order to guarantee for patient’s safety and treatment efficacy. Based on their easy and non-invasive insertion, PICC and MC are emerging infusion devices in oncology considered a possible alternative to more invasive and costly TIVAD. PICCs can be used for any type of chemotherapy and treatment while MCs are indicated only for the infusion of peripherally compatible infusions. Nevertheless, the perception of a higher incidence of complications of external and not totally implantable devices represent a possible limiting factor to their use. Both PICC and MC can develop complications, but be different risk of catheter failure must be considered when planning their insertion. 14
The use of PICC for the administration of chemotherapy has been reported to be a safe and reliable alternative to totally implantable devices in cancer patients with a failure risk ranging from 8% to 15%. 15 Midline catheter has become popular in the last decades to facilitate infusions in difficult insertion venous access patients (DIVA) and in acute hospitalized patients’ settings. A multicentre study 16 highlighted possible advantages over PICC with lower risks of major complications but an elevated incidence of adverse events leading to an early removal of the devices. MC catheters were shown to be versatile venous access devices that appeared to be effective and safe for short-term treatments in patients requiring infusion of fluids peripherally compatible. Inasmuch, MCs have been claimed to somehow reduce the incidence of CRBSI compared to PICC. 17 Unlike PICC, scant literature exists on their safety and efficacy of use in the infusion of chemotherapy drugs.
Although devices that are not totally implantable may represent a not optimal choice for the administration of chemotherapy in cancer patients,18–20 the Authors aimed to compare the effectiveness of PICC and MC devices by analysing the frequencies of failure leading to a catheter removal. Catheter failure rate per month per 100 patients showed that the use of a PICC device (CFR = 1.40; 95% CL = 0.90–2.18) was safer compared to that of MC (CFR = 5.89; 95% CL = 2.98–11.6). Noteworthy results were also for type of chemotherapeutic treatments. With respect to patients that undergone only neutral N treatments, all other patients (i.e. hybrid and irritating-vesicant treatments) had a higher occurrence of complications which gave rise to risk excesses of about 30% and 80% in the hybrid (CFR = 1.79; 95% CL = 1.15–2.77) and irritating-vesicant (CFR = 2.48; 95% CL = 0.70–9.72) subgroups, respectively. Although the heterogeneity in CFR observed for the different cancer diagnoses was not statically significant, hemo-lymphopoietic non-solid tumours had a somehow increased risk of removal when compared to solid tumours. The catheter failure rate resulted to be independent from age at diagnosis and gender. MCs had a shorter time to failure when compared to PICC ones (Figure 1) suggesting a lower efficiency over the dwell-time and a PICC greater effectiveness in particular for prolonged treatments.
Our study has some relevant strengths. To our knowledge this is the first study to date to compare non-totally implantable central and not central vascular accesses for chemotherapy delivery. Secondly, it analyses relevant risk factors for failure such as type of cancer and composition of the infused chemotherapy. On the other hand, some limitations should be also mentioned. It is a single-centre observational study and therefore the possibility of selection and/or residual confounding biases brought about by unknown or unmeasured factors cannot be ruled out. In addition, the limited number of catheter failures and the relatively high number of predictive factors considered in regression modelling prevented us from analysing each cause of failure and accordingly comparing for single type of inserted vascular accesses. Nevertheless, the cumulative incidence of failures was not statistically significant for the type of vascular access (p = 0.096). This observation is partially in disagreement with the literature data where MC catheters are reported to have a somehow greater incidence of CRT.10,16,17 However, these observations refer to patients not under active chemotherapy and, in our opinion, this factor could justify the differences with our data results. We believe that further future studies on the selective use of MCs to administer peripherally compatible chemotherapy could be decisive in clarifying this aspect.
Conclusion
Whenever a chemotherapy is planned by using a not totally implantable vascular access an adequate selection of the device has to be considered. Given lower risk of complications and failure of the device and a major longevity, PICC must be preferred over MC. For the infusion of the majority of chemotherapy types, an expanded use of MCs appears to be unsustainable.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Sergio Bertoglio is Chief Medical Officer of Plan1 Health s.r.l., Amaro (UD), Italy.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
