Abstract
Background:
Serious complications can develop from erroneous insertion of a peripheral intravenous catheter (PIVC). Successful PIVC insertion has a great place in the continuation of IV treatment without causing complications that may require the removal of the PIVC.
Methods:
The population for this prospective observational study included patients who had undergone PIVC insertion in the surgical, intensive care, and inpatient units of a large teaching and research hospital in Istanbul. Data were collected via a patient information form, a PIVC and treatment information form, the Visual Infusion Phlebitis (VIP) scale, and a PIVC nurse observation form. The SPSS 24 software package was used to analyze the data.
Results:
Of the 168 patients enrolled, 64.9% (n = 109) were male, and the mean age was 56.71 ± 17.97 years. The mean PIVC dwell time was 73.46 ± 21.57 h, and 32.7% (n = 55) of the catheters were removed due to discharge from hospital. Overall, 29.2% (n = 49) of the cases developed phlebitis, with half of the cases (51%, n = 25) developing grade 2 phlebitis and developed within 48 h in 38.8% of cases (n = 19). The mean PIVC dwell time was significantly lower in patients with phlebitis than in those with no phlebitis (p < 0.05).
Conclusions:
It was determined that PIVC dwell time could be increased by routine surveillance to monitor for phlebitis regularly. It is recommended that education programs for nurses be developed to prevent PIVC-related complications.
Introduction
Peripheral intravenous catheters (PIVCs) are used to administer patient treatment that improve recovery times. However, they can lead to complications such as phlebitis, infiltration, thrombosis, infection, extravasation, subcutaneous ecchymosis, hematoma, and nerve damage.1–8 This usually requires the removal of the PIVC.1,7,9 The Infusion Nursing Society Standards of Practice emphasize that catheter insertion and management practices positively impact catheter removal and length of hospital stay. 10 In addition to complications becoming life-threatening, PIVCs can increase healthcare professionals’ workloads by extending hospital stays and the need for care and treatment and decreasing comfort.9,11–15
Nurses have significant responsibilities in the safe initiation, management, and termination of intravenous PIVC therapy.12,16–18 Although risk factors affecting PIVC dwell time have been identified in previous studies, the number of studies with tangible results that directly show factors affecting PIVC dwell time and removal is quite limited.12,13 This study was conducted to identify associations between PIVCs and clinical characteristics in the development of phlebitis in adult patients.
Methods
Study design
This was a prospective, observational study.
Population and sample of the study
The study population consisted of patients who had undergone PIVC insertion in the surgical and medical intensive care units and inpatient clinics of a 698-bed university hospital. Based on phlebitis rates from previous studies,12,13 the sample size was calculated (n = 168) using the G*Power 3.1.9.2 software package. The inclusion criteria were as follows: patients aged ⩾18 years who had been hospitalized and received treatment in the last 72 h, were open to communication and collaboration, and could provide informed consent were neither underweight nor obese according to Body mass index (BMI) classification, had undergone catheter insertion at the selected site for the first time, and in whom the catheter had been inserted in only one attempt.
Data collection tools
The patient information form consisted of questions about patients’ education and income, hospitalization and discharge dates, medical diagnoses, clinics, and the presence of chronic diseases.
The PIVC and treatment information form consisted of questions about PIVC size; whether three triple lumen adaptors were connected to the PIVC via a connector, intravenous flow regulator or an infusion pump; the anatomical site of insertion; the number of insertion attempts at the site; the type, dose, frequency, and route of administration of requested drug and fluid treatments; antibiotic administration and its frequency and quantity; total amount of fluids taken by the patient; PIVC dwell time; phlebitis development; grade of phlebitis; and reasons for PIVC removal.
The Visual Infusion Phlebitis (VIP) scale is a validated scale developed by Gallant and Schultz. 19 The Turkish adaptation study using the VIP scale was conducted by Pasalioglu and Kaya 20 and published previously. It included the grade of phlebitis based on observations of catheter-related complications that developed during the treatment via PIVC and the signs and symptoms that manifested according to the grade of phlebitis. The PIVC nurse observation form was used to record data from the VIP scale every 8 h.1,3,4
Data collection
The institution where the study was conducted followed the PIVC placement and maintenance procedures of the Centers for Disease Control and Prevention 2017. 21 PIVCs made of the same material (thermoplastic polyurethane) were used for all patients. Only catheter size varied due to individual characteristics. Transparent catheter dressings were used by the same clinical nurse to place and observe the PIVCs.
The patient information form and the PIVC and treatment information form were distributed among the patients. The researchers applied the VIP scale by observing the PIVCs every 8 h. Apart from this routine observation, the researcher conducted additional observations to diagnose phlebitis and evaluate catheters and catheter sites when patients complained. The data were recorded in the PIVC nurse observation form. The PIVC remained inserted as long as no phlebitis symptoms were observed; however, in patients with Grade 2 phlebitis symptoms, the PIVC dwell time was recorded, and the catheter was removed.
Ethical considerations
Ethics committee approval (date: 18.11.2020, number: 83156) and permission from the institution (date: 08.09.2020, number: 54230385) were obtained. Written consent was obtained from the nurses who volunteered to participate in the study. The principles of the Declaration of Helsinki were followed in this study.
Data analysis
The IBM SPSS Statistics 24 software package was used for the analysis. A normality test determined that the variables had a normal distribution. A parametric independent samples t-test was used to compare the two independent groups. Data with a non-normal distribution were analyzed using non-parametric methods. The Mann–Whitney U test was used to compare the two independent groups. The Kruskal–Wallis H test was used to compare three or more independent groups. Bonferroni correction was used for the paired comparisons of variables with significant differences in three or more groups. A p-value of <0.05 was considered statistically significant.
Results
Patient characteristics and demographics are presented in Table 1. Phlebitis developed in 29.2% (n = 49) of the patients, half of which were grade 2 (51%, n = 25), which developed within 48 h in 38.8% (n = 19). Of the patients (n = 49) who developed phlebitis had redness, 98% (n = 48) had tenderness, 89.8% (n = 44) had pain, and 55.1% (n = 27) had a palpable cord (Table 2). There was a statistically significant correlation between the status of phlebitis development and the use of an infusion pump (p = 0.026) and the anatomical site of PIVC insertion (p = 0.000). Phlebitis developed more often in patients in whom an infusion pump had been used and who had a PIVC inserted into the cubital region. There was a statistically significant difference in the development of phlebitis between age groups (p = 0.000), length of hospital stay (p = 0.000), presence of chronic diseases (p = 0.000), and the clinics where the patients were admitted (p = 0.011). Phlebitis development occurred more in patients aged ⩾65, who had been hospitalized for 5–6 days on average, had chronic diseases, and had been admitted to the intensive care unit (Table 3). Catheter dwell time was longer in patients aged <50 years (p = 0.000), had a PIVC inserted into the back of the hand (p = 0.004), did not develop phlebitis (p = 0.000), did not use antibiotics (p = 0.000), and were given less than 1000 ml of fluid (p = 0.000) (Table 4).
Patients’ demographic and clinical data (N = 168).
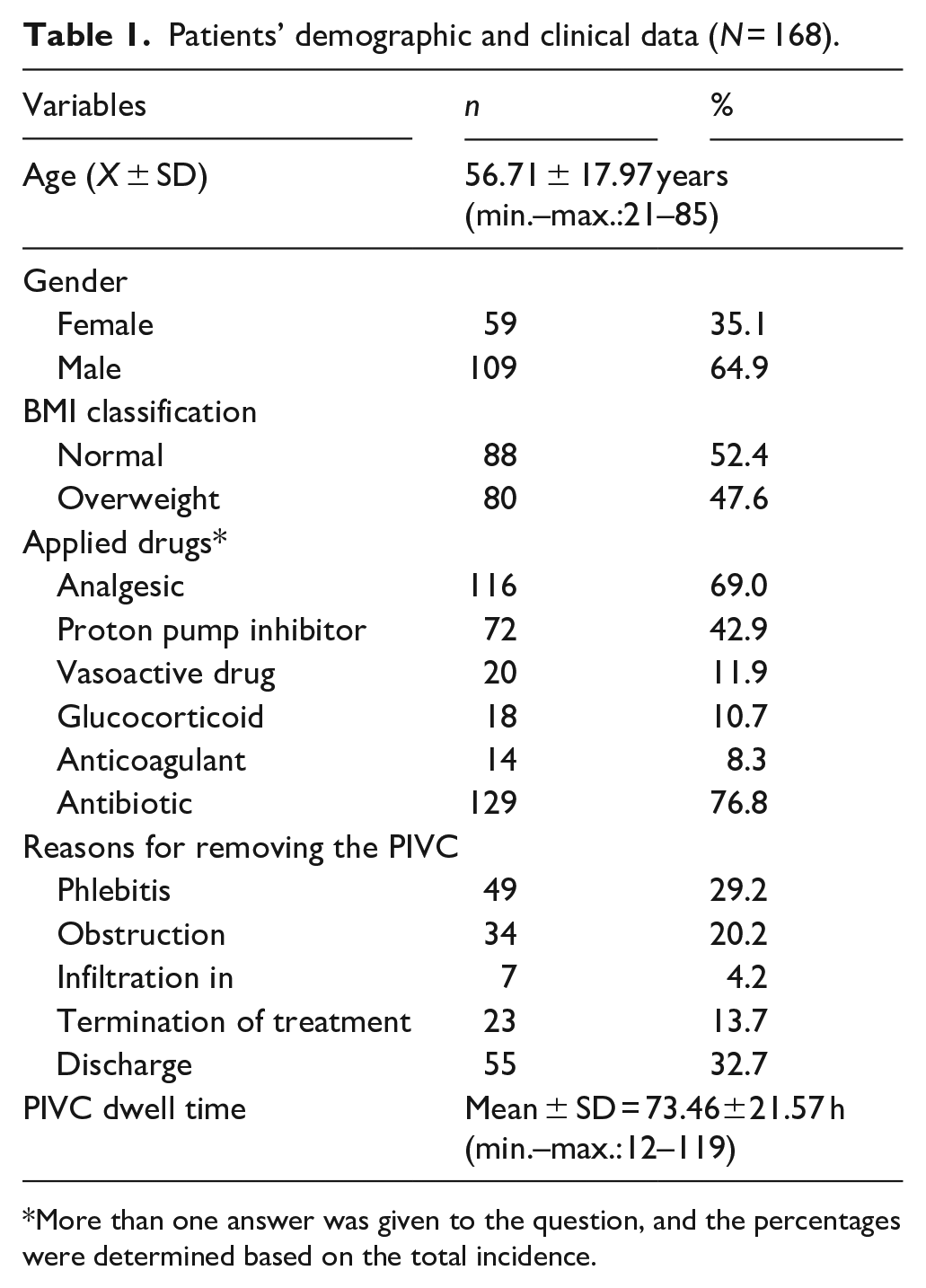
More than one answer was given to the question, and the percentages were determined based on the total incidence.
PIVC application and development of phlebitis in patients (N = 168).
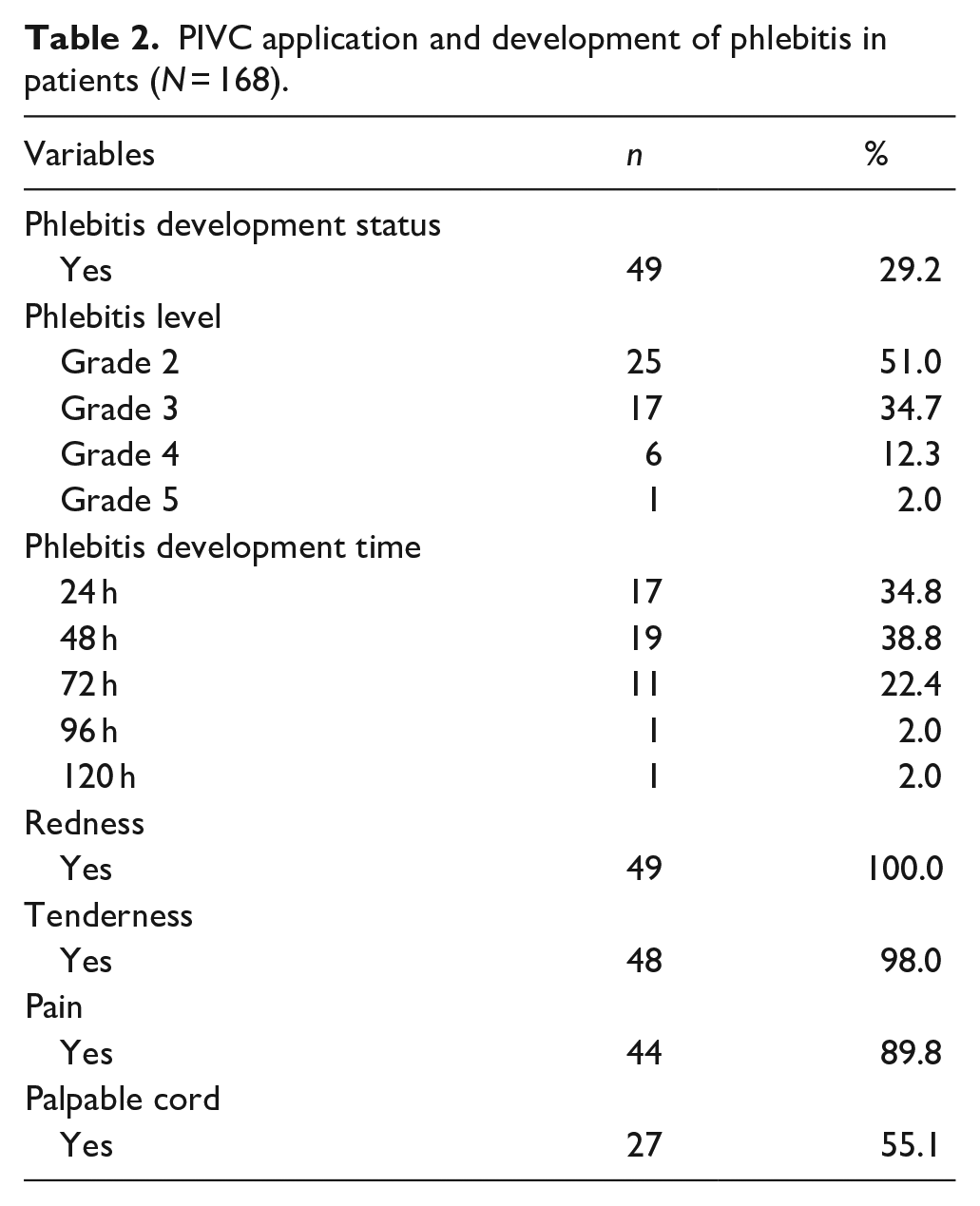
Phlebitis development status in patients according to PIVC characteristics (N = 168).
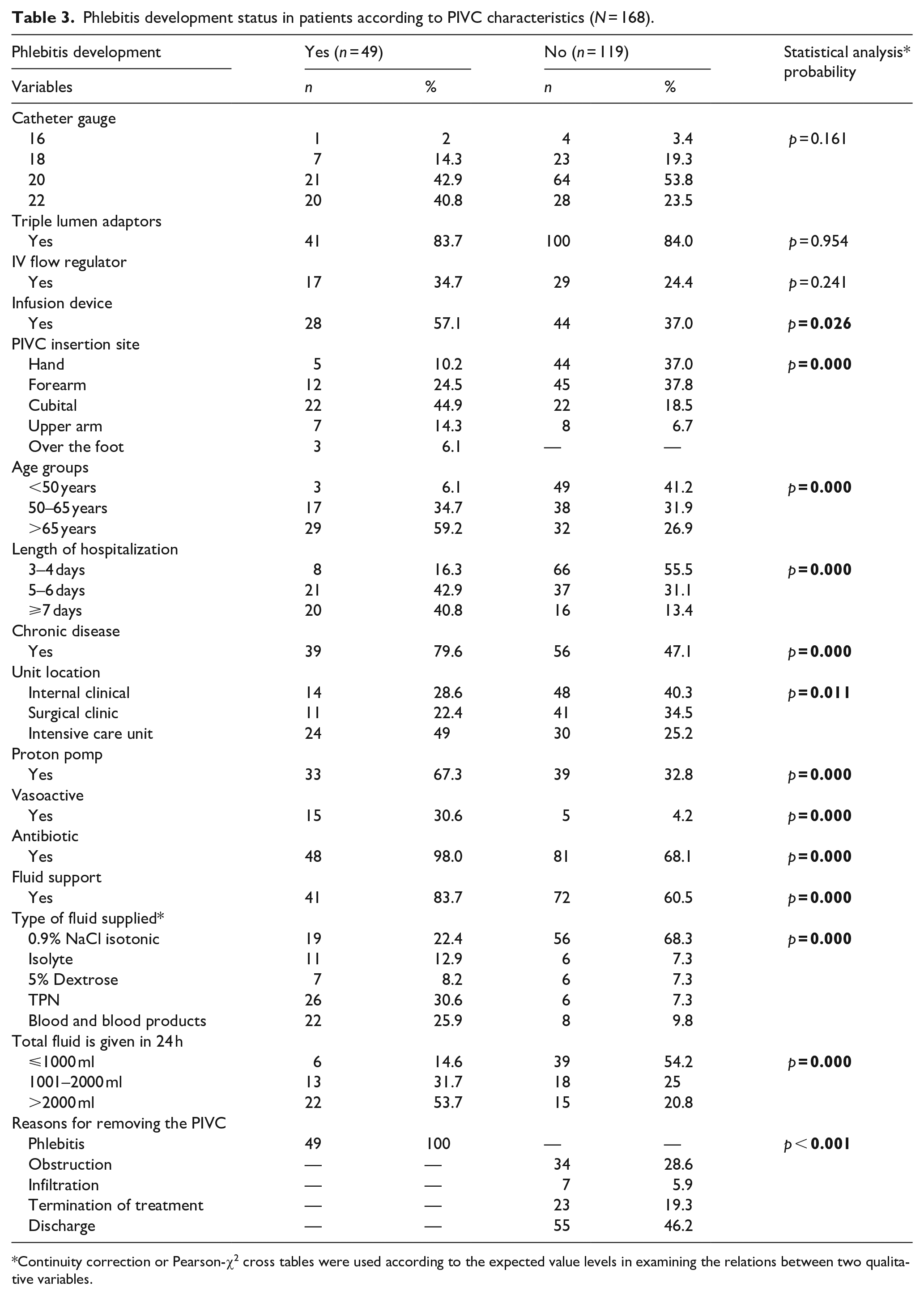
Continuity correction or Pearson-χ2 cross tables were used according to the expected value levels in examining the relations between two qualitative variables.
Comparison of patient’s individual, treatment, catheter characteristics, phlebitis development, and PIVC dwelling time (N = 168).
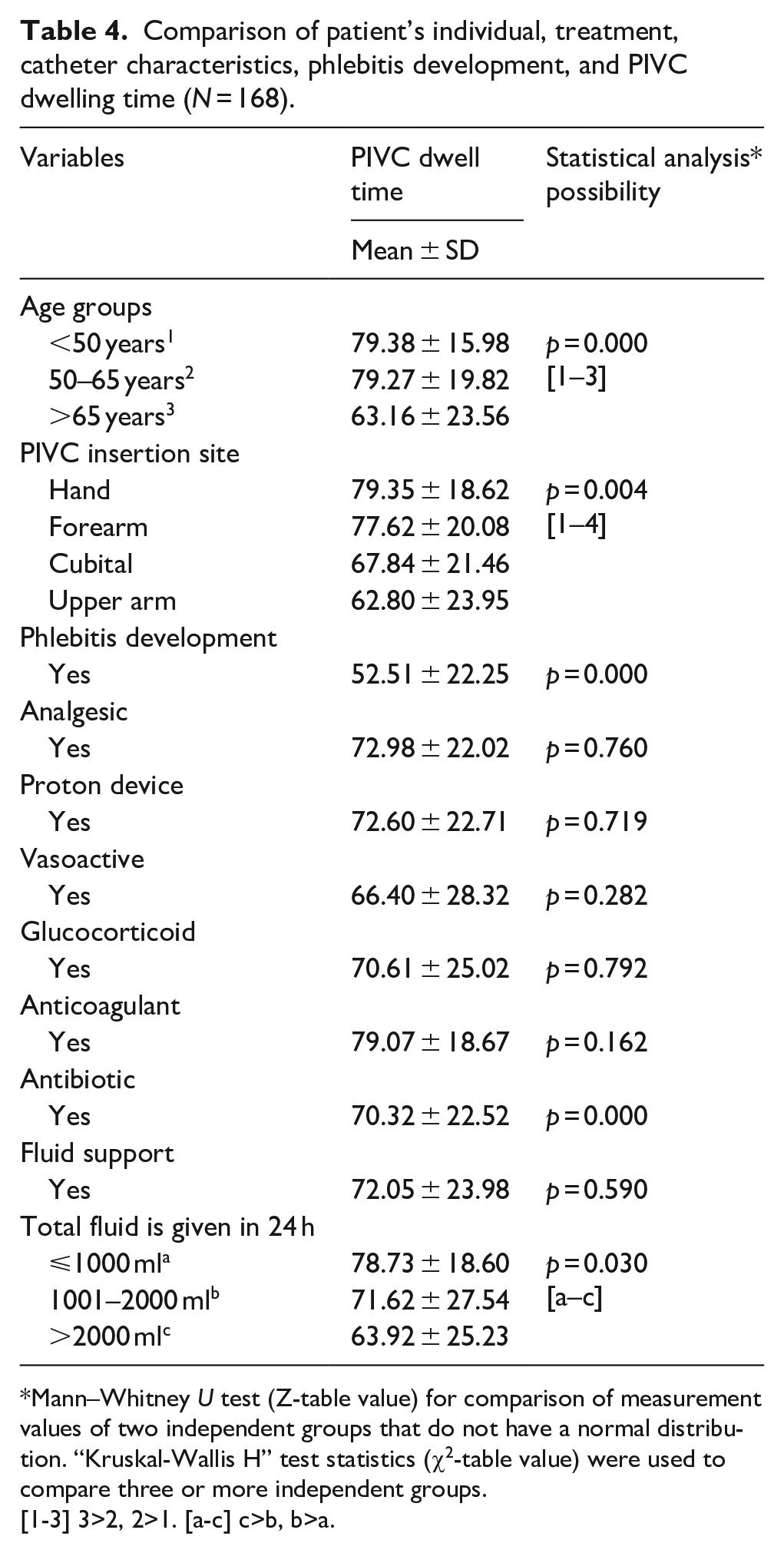
Mann–Whitney U test (Z-table value) for comparison of measurement values of two independent groups that do not have a normal distribution. “Kruskal-Wallis H” test statistics (χ2-table value) were used to compare three or more independent groups.
[1-3] 3>2, 2>1. [a-c] c>b, b>a.
Discussion
The findings of this study indicate that the presence of chronic disease, hospital length of stay, reason for hospitalization, amount of medication and fluid administration, catheter size, and related types of equipment influenced PIVC dwell time. It was determined that a 20-gauge catheter had been used in 50.6% of the patients. In the literature, the use of the smallest catheters and rapid introduction of drugs/fluids into the circulation are recommended to prevent the formation of venous insufficiency and vein damage. 22 In this study, all catheters used were shorter than 6 cm—that is, short peripheral catheters (SPCs). However, in the last decade, SPCs have frequently been preferred because they are inexpensive and easy to insert. But have found to lead to catheter problems and early removal due to complications.23,24 In this study, the use of SPCs had no negative effects. PIVCs were inserted into patients’ forearms more than other sites, which is recommended in order to reduce pain during dwell time, to prevent the dislodgement of the PIVC, and to prevent the blockage of fluid flow.25,26 The mean PIVC dwell time was 73.46 ± 21.57 h. Researchers have suggested that PIVC dwell time should be extended to 72–96 h if there is no risk of infection or phlebitis.10,21 Wei et al. 13 reported that the mean PIVC dwell time was 43–76 h. It was found that PIVCs had primarily been removed due to discharge, followed by phlebitis, then blockage. Similarly, in Atay and Yilmaz Kurt’s study, 27 phlebitis, blockage, and expiration date were among the reasons for PIVC removal. The most common reason reported for PIVC removal has been phlebitis.22,28,29
Phlebitis, which is characterized by redness, pain, and swelling—as well as purulent discharge in the case of bacterial phlebitis—was the most common among PIVC complications. 23 Regarding phlebitis grades, 28.2% of the patients in Berse et al.’s study 30 and 43.2% in Braga et al.’s study 31 developed Grade 1 phlebitis. It is clear that the incidence rate of phlebitis is higher than the rate accepted by the Infusion Nurses Society. 10 Phlebitis developed more frequently in patients who used an infusion pump. This indicates the importance of monitoring phlebitis development in adult patients using an infusion pump.30,32 Although the literature recommends inserting the PIVC into the forearm, the majority of patients who developed phlebitis had a PIVC in the elbow. 33 This divergence is due to the forearm being a more immobile site that is easier to support. 34
Patients who developed phlebitis were predominantly 65 years of age and older and were hospitalized for an average of 5–6 days. In a study by Atay and Yilmaz Kurt, 27 it was found that the duration of hospitalization increased the likelihood development of phlebitis; therefore, adult patients with long hospital stays should be followed up frequently for its development. Peripherally inserted central catheters (PICC) and midline are commonly used for medium- and long-term venous access. Midline catheters are rarely used despite being recommended for intravenous therapies lasting more than 6 days and should likely be considered in such cases. 12 Phlebitis developed more frequently in individuals with chronic diseases, and the majority of patients who developed phlebitis were hospitalized in the intensive care unit. Phlebitis occurred primarily in patients taking proton pump inhibitors, which are associated with disruption of vascular endothelium; therefore, phlebitis also occurred primarily in patients taking vasoactive drugs. Excessive use of such drugs could increase blood vessel inflammation and the risk of phlebitis.13,35 Nearly all patients who developed phlebitis received high doses of antibiotics. Therefore, patients being administered intravenous antibiotics23,30,35 should have regular surveillance for phlebitis. Approximately 84% of the patients who developed phlebitis had undergone fluid therapy, this was due to the pragmatic characteristics of the sample. Some patients who developed phlebitis were given Total Parenteral Nutrition (TPN), the concentration of this solution can damage the venous endothelium and induce the development of phlebitis. 34 Patients who developed phlebitis had been given more than 2000 ml of fluid. As the amount and frequency of fluids administered to patients increase, the possibility of developing phlebitis increases due to irritation and trauma that may occur in the venous system. In almost all the patients, the PIVC was removed due to phlebitis.36, 37 The literature has emphasized that PIVC dwell time directly affects patients who are at higher risk of developing phlebitis and thrombophlebitis.13,30,35 This finding is significant, as it shows how effective the PIVC dwell time is in the development of phlebitis.
Patients under 50 years of age and those in the 50–65 year group had longer PIVC dwell times. According to the Infusion Nurses Society (INS), 10 being over 65 is one of the primary factors in the development of phlebitis due to the delayed response of the immune system to trauma that comes with advanced age, changes in subcutaneous tissues and veins, insufficient hydration, and slowed cell renewal and wound healing.
PIVCs inserted into the back of the hand had longer dwell times. Some studies have shown less phlebitis development and longer PIVC dwell times in the forearm, antecubital site, and back of the hand insertions.2,20 PIVC dwell times were longer in patients who did not develop phlebitis. Recently, it has been recommended that instead of routinely replacing the PIVC, it should be replaced when removal is necessary, as this would improve patients’ quality of life, comfort, and satisfaction and reduce financial losses.9,11 Patients who were not given antibiotics had longer PIVC dwell times. This is due to the structure of antibiotics and their negative effects on the venous system. 37 PIVC dwell times were longer in patients who were given a total of ⩽1000 ml fluid in 24 h. It is thought that the risk of irritating the venous structure and developing complications increases as the amount, frequency, and speed of drugs/fluids administered at the PIVC site increase.
Limitations
This study has some limitations. Since it was conducted at only one institution, the results cannot be further generalized. No internal or external validation of the data collection tools developed by the authors was conducted. Infection, mechanical, and chemical phlebitis signs, and symptoms may be the same. In addition, it is not easy to distinguish between phlebitis and extravasation. This study’s results are reported without this distinction, which is one of the important limitations of the study. Another limitation is the lack of sufficient information about drug/antibiotic types. In this study, catheters made of the same material (thermoplastic polyurethane) were used for all patients. Since phlebitis development in patients is thought to be related to the material used, this was another limitation of the study.
Conclusion
Phlebitis and related complications play an important role in PIVC dwell time and are important reasons for their removal. This study found a correlation between PIVC application and phlebitis development. When the PIVC is inserted, the signs and symptoms of phlebitis should be monitored in terms of the type of phlebitis and the factors affecting it. Future studies should determine which infusions cause phlebitis. It is recommended that nurses use valid and reliable measurement tools to prevent complications that may develop due to PIVCs. PIVCs-related educational programs should be developed, and care and treatment interventions should follow current evidence-based guidelines.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
