Abstract
Background:
Short peripheral intravenous catheters are the most common invasive procedure used to deliver medications, blood products, and fluids to patients, and failure has the potential to impact the quality of care.
Methods:
This study compared IVs stabilized with current transparent film dressing to those using an engineered stabilization device. The first phase of the study evaluated documentation; data from patient records were found less than optimal but most complete for securement and removal, the two fields most critical to the study. The second phase measured IV dwell time, restart rates, and time between IV loss and restarts.
Results:
Although there were no statistically significant differences in restarts, the IV dwell time was longer when the engineered device was in place, helping to extend the life of the IV and prevent interruption of care. In the absence of data on the cost of infections and other complications, the use of an engineered device increased the cost of IV starts.
Conclusions:
Adding an engineered stabilization device increases the cost of peripheral IV starts, but contributes to reducing IV restarts and preventing IV complications due to destabilized IVs. Although there were no statistically significant differences in restarts, the IV dwell time was longer when the device was in place, helping to extend the life of the IV and prevent interruption of care. Complete and accurate documentation and improved quality depended upon the ability to abstract unit-level data, which is vital for capturing the appropriate healthcare indicators. Clinical nurses must be involved in the decision-making regarding health records and operability at the unit level.
Keywords
Introduction
Up to 90% of hospitalized patients require some form of intravenous (IV) therapy.1–3 Considered a routine nursing procedure to improve treatment outcomes, 4 keeping an IV in place without complications remains a challenge. 5 Unstable IVs put patients at risk, increase healthcare costs and decrease quality.6,7
In 2016, the Infusion Nurses Society (INS) introduced and subsequently continued the recommendation to use a non-bordered polyurethane dressing in combination with an engineered stabilization device, or ESD8,9 as transparent dressings or tape are not explicitly designed to prevent catheter dislodgment. They allow micro-movements within the blood vessels resulting in vein injuries like disconnection, infiltration, extravasation, infection, and phlebitis. These complications potentiate the need for unscheduled restarts that can result in treatment delays, additional cost, safety concerns, nursing interruption, and most importantly, patient discomfort and dissatisfaction.
Clinicians identified dislodgement as the most observed safety issue, 6 and 84 articles cited over 30% of IVs as having an infection or inflammation-related complications. 5 With a reported failure rate of 35% to 50%, this invasive procedure is not without potential patient harm. 4 At the time of the study, the transparent adhesive dressing with supportive tape was still the most commonly used procedure. 10
Stabilizing IVs was identified as one of the top 10 patient safety concerns for 2021 by the Emergency Care Research Institute, citing an analysis of 27,320 IV failures, including 6119 infections and 21,201 events of non-infection harm. 11 Risk reduction requires methods to improve IV securement and improved documentation of clinical interventions and processes.7,9,11 A study of IV complications in a pediatric sample found no differences between patients with ESDs compared to transparent dressings. 12 A systematic review of six trials comparing securement methods (including transparent dressings) found no statistical differences in documented failure, dislodgement, infiltration, or time to failure. 13 Another trial that compared four types of securement reported similar results, with no differences as evidenced by IV failure, occlusion, phlebitis, or dwell time; overall, 41% of patients had IV failure, again without any differences between groups. 3
The purpose of this study was to compare a commercially available stabilization device with current standard practice of transparent tape on peripheral IV dwell time, restart rates, and time between IV loss and restarts. A secondary aim was to assess the equipment costs between the two methods.
Methods
Design
The study was a descriptive comparative intervention study using a convenience sampling strategy and data from the electronic health record (EHR) after the study phases were completed. The STROBE checklist was used to guide reporting transparency. 14 There were two phases; documentation assessment for quality of IV documentation followed by implementation of the intervention. The study was approved by the medical center’s Institutional Review Board and met the criteria for waiver of documented written informed consent (IRB ID: R20180034).
Setting
The study was completed over 6 months on a 46-bed surgical unit in a Midwest nonteaching Magnet® recognized medical center.
Participants
The IVs to be assessed during the study period included all newly admitted patients with peripheral IVs that remain in place for 48 h or more. Newly admitted was defined as a patient admitted from any other unit or by direct admission with a peripheral IV in place. There were on average 29 patients with peripheral IVs on any given day. Of those, 86.2% were expected to meet the inclusion criteria, providing an estimated 350 IVs during each study period.
The study compared IVs using a traditional transparent film dressing to those secured with a commercially available ESD, Guard360 (PrimeGuard Medical, LLC Chicago IL). There wasno change in IV insertion practice except for the method of stabilization. Once the peripheral IV was inserted and the ESD in place, a Tegaderm transplant film dressing was placed over the catheter insertion site and catheter hub. The Tegaderm dressing did not cover the ESD or connection hub between catheter and IV tubing (see Figure 1).
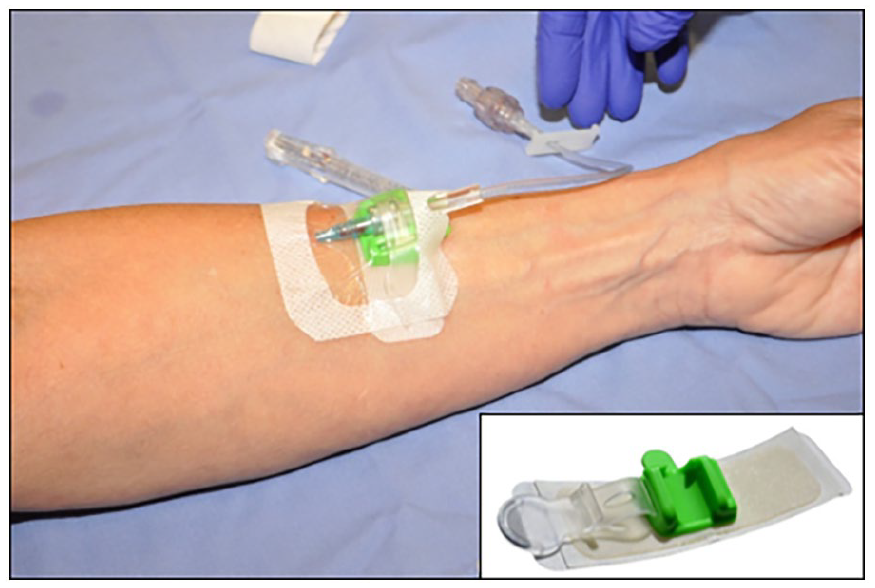
Commercially available engineered stabilization device used in the study. Medical Peripheral IV Securement Device Guard360 (PrimeGuard Medical, LLC Chicago IL).
Data analysis
Demographic characteristics were described, and differences were assessed by Student’s t-test and Chi-square. Outcomes were assessed by group assignment; the control group included IVs that were secured with the current practice of transparent film dressing. The intervention group included IVs secured with the ESD device. All data were analyzed using the statistic functions in Excel-XLStat. Probability values were all set at 0.05.
Outcome variables
Data were obtained from the Electronic Health Record (EHR) that included age, gender, body mass index, admission date, discharge date, IV start date, IV removal date, IV securement fields, patency/maintenance fields, site signs/symptom fields, and removal indications fields. From this information, percent of complete documentation, IV dwell time, number of restarts, and time to restart were calculated.
Documentation completeness
Number of completely documented IVs divided by the total number of IVs was used to calculate the completion rate. Frequency and percent were used to describe documentation completion rates.
IV dwell time
Dwell time was calculated in days and hours by subtracting IV start date from IV end date. Means and standard deviation were used to calculate group dwell time. Student’s t-test was used to calculate differences between groups.
Restart rate
Restart due to destabilization was defined from the EHR removal indication list. Of the 10 reasons in the list, four were deemed proxies for destabilization: Lumen/catheter not patent, removed inadvertently, removed per patient, and site symptomatic. The IV was categorized as either destabilized or normal loss. If other indications were checked and did not include 1 of the 4 destabilization criteria, the IV removal and restart were not considered to be a restart due to stabilization. Frequency and percent were used to describe restarts by group. Chi-square test for independent samples was used to compare restart the control and intervention groups.
Time between IV loss and IV restart
For IVs that were categorized as restart, time documented for removal was subtracted from the time the new IV was started. This was calculated in minutes. Differences between groups were analyzed by Student’s t-test.
Equipment cost
The cost-effectiveness ratio was used to determine the incremental costs associated with one additional restart where the costs for the ESD therapy minus costs for standard therapy divided by the difference of restarts from the ESD group minus restarts in the standard group. Material costs and RN time to start and document were included using the average hourly rate for an IV therapy nurse. Additional analyses used annualized costs associated with estimated annual unit IVs with estimated restarts based on the study outcomes. Cost differences were measured.
Documentation assessment phase
Phase 1 focused on improving the EHR documentation of IV therapy by providing unit and vascular team nurses in-service education and hands-on instruction with the ESD, not then in use the facility. In preparation, the health informatics team assessed the ability to extract data from the EHR to measure outcomes. Checkboxes in the Adult Assessment Intervention field included Access/Monitoring Devices; in turn, this provided an IV Device tab with expandable fields for insertion/securement and assessment information, signs and symptoms experienced by the patient or observed by the nurse, and removal indications. To assess documentation of IV loss, the informatics team extracted data on 100 patients admitted to the study unit. In this initial dataset, 22 patients had IV restarts; only 10 (45%) had complete documentation. To improve documentation and thus the sample size, documentation in-service training was completed by the clinical nurses involved in the study. In four pilot 2-week trials, documentation remained stable with only slight increases despite repeated reviews and appeals. Documentation was not optimal but was the most complete for the securement and removal fields, at 43% and 51.4%, respectively, the two most critical to the study.
Intervention phase
Phase 2 was a 2-week period where IVs that met inclusion criteria were secured with the ESD. Patients who had a patent IV stabilized with transparent film dressing (the current standard of care) in place upon arrival were assigned to the control group; no current patent IV was changed over to an ESD unless it failed and a restart was needed. If a new peripheral IV was inserted, the ESD was placed per the manufacturer’s recommended procedures, that is, the polyurethane transplant film dressing was placed over the catheter insertion site but not the catheter hub, which was stabilized with the device. After the intervention period, a de-identified list of all patients filtered by study unit and IV use code was abstracted by medical record number and admission/discharge dates
Results
During the intervention period, 338 IVs were documented. Of those, 256 were placed with current practice, with 174 (67.9%) with complete documentation. The intervention group had 43 IVs with 27 (62.7%) with complete documentation. The final control and intervention group of 174 and 27 were used for analysis (Figure 2). There were no statistically significant differences for age [59.8 (16.8) vs. 59.4 (21.7) years; p = 0.9] and body mass index [30.9 (8.9) vs 30.1 (9.0); p = 0.7] or female gender (45.9% vs 44.4%; P = 0.5) between groups (Table 1).
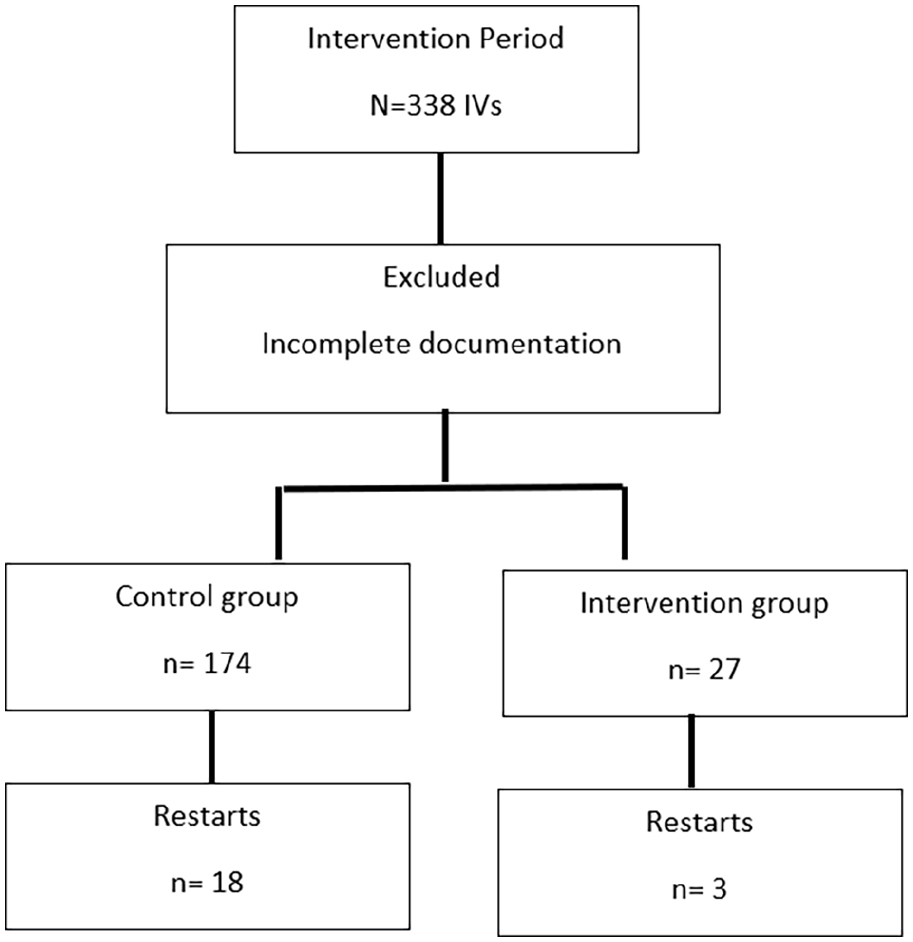
Flow diagram of study sample.
Summary of demographic characteristics and primary outcome variables for study groups.
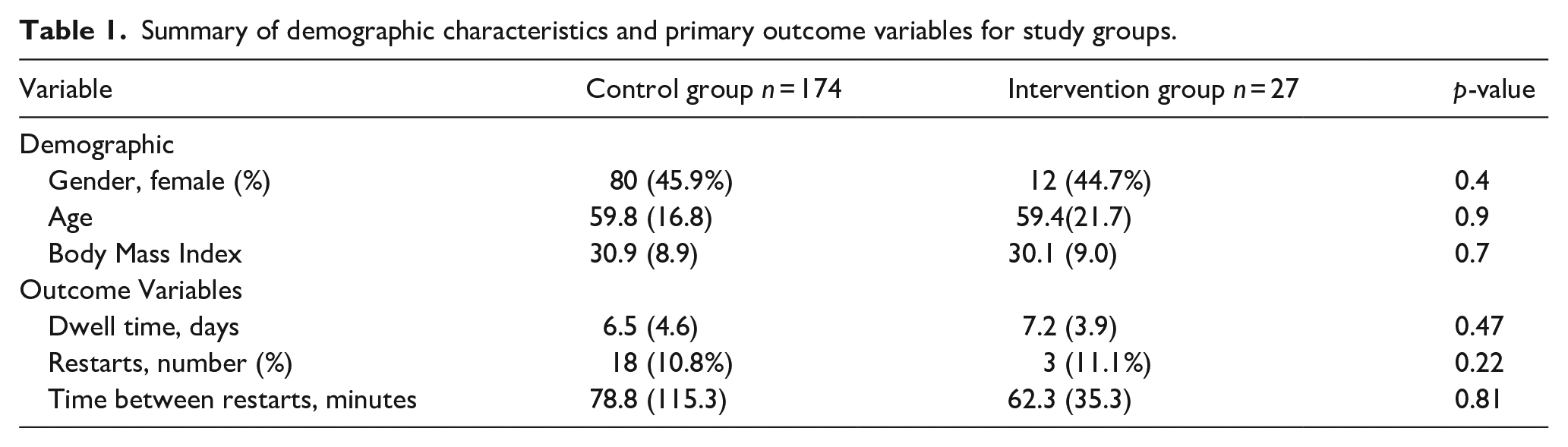
Analysis found no statistically significant differences between the control and intervention groups on the measures documented: IV dwell time [6.5 (4.6) vs 7.2 (3.9) days; p = 0.47]; restarts [18 in the control group (10.3%) vs three in the intervention group (11.1%); (Σ2 (1, 21) = .3.8, p = 0.22]; or mean time between the loss of one IV and the restart of another IV [78.8 (115.3) minutes for the control group vs 62.3 (35.3) minutes for the intervention group (t (19) = 2.1, p = 0.81].
Material costs for starting an IV in the standard group were $11.40 and $12.90 for the ESD group. The mean time for a nurse to start and document IV placement was 24 min; using the average hourly wage for nursing of $30.84, the cost for starting an IV with transparent dressing was $23.74; for using an ESD, $25.24. Based on the costs for starts and restarts, there was a $23.44 incremental cost associated with one additional IV restart (Table 2).
Itemized costs during the study period with incremental cost-effectiveness for each additional restart and annualized costs for study unit using estimated annual IVs and restarts.
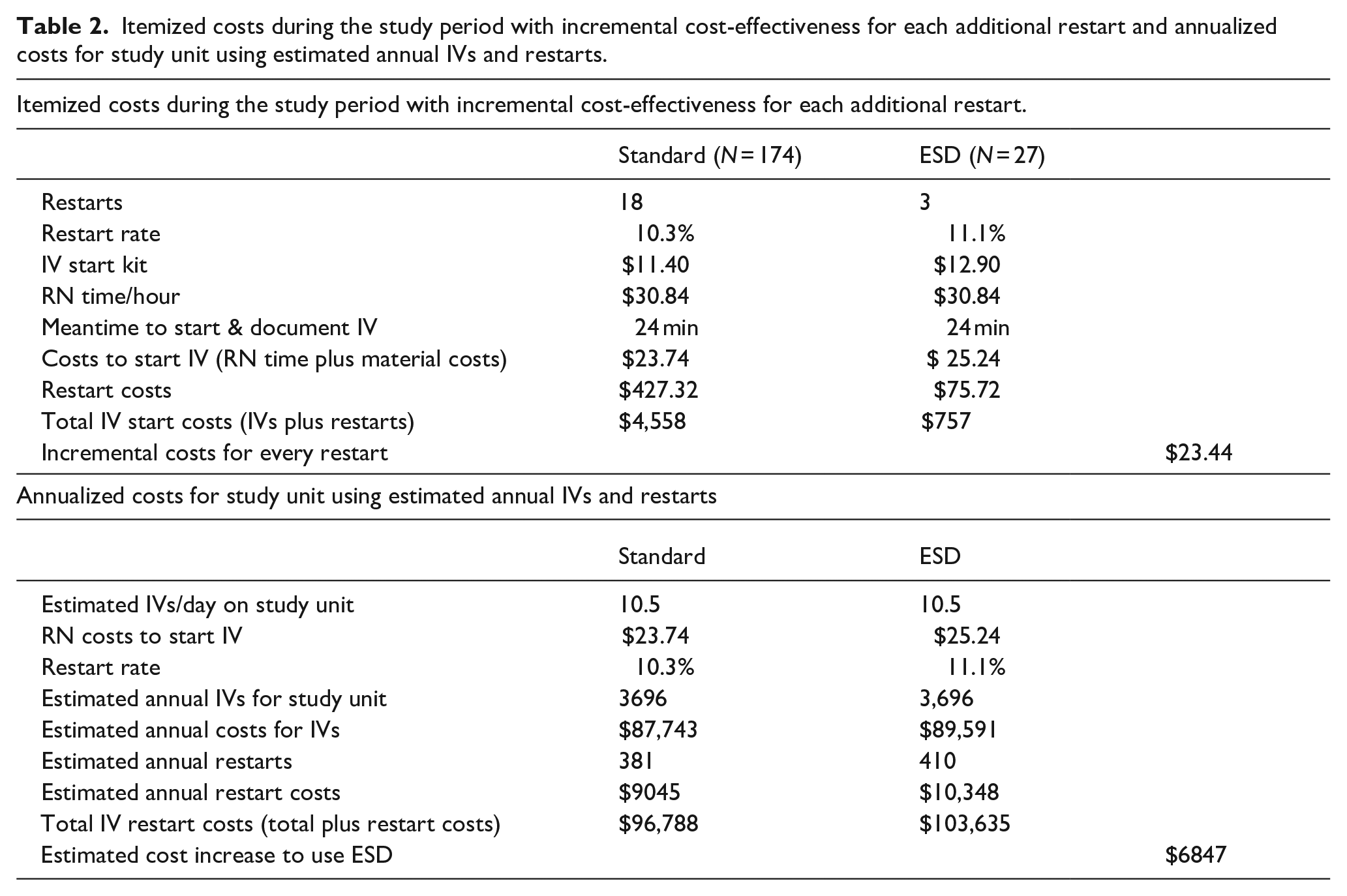
Using the new estimation of 10.5 IVs per day (compared to the estimation of 29 IVs/day during the pre-study evaluation), the unit would have approximately 3696 new IVs per year, creating a cost of $87,743 for IVs secured with transparent tape versus $93,287 for ESDs ($5544 per year). Assuming a 10.3% restart rate for IVs with transparent tape dressings and an 11.1% restart rate for ESDs, the unit would need an extra 381 IVs ($9045) for the former and 410 IVs ($10,348) for ESDs. Overall using the ESD would increase annual costs by $6847 (Table 2).
Discussion
There were no statistically significant differences in the IV restart rate, dwell time or time to restart. Restart rates were higher than reported in the literature for both the traditional transparent tape dressing (10.3% vs 6.9%) and the ESD (11.1% vs 3.1%) 17 ; given our reduced sample size in the ESD group, this may not reflect an accurate estimation of events and may limit generalizability.
Time to restart was shorter for the ESD group. A significant standard deviation in the control group may have impacted the statistical findings. In this case, using a comparison of medians may have been the more appropriate analysis. Restarting a displaced IV may not be controlled by the type of securement used but rather a reflection of the system in place on the unit to have an IV restarted. These data were difficult to assess as often the time stamp in the EHR reflected the time in charting rather than the actual time the IV was ended or restarted, making the assessment of this variable difficult.
Adding a single ESD during the intervention period increased costs by $23.44, extrapolated to an annualized cost of over $6800. Other studies using real-time data capture and long observation periods may be able to ascertain budget-neutral costs. Following peripheral IV complications and prevention of at least one acquired infection may be one way to justify the increased costs. A reduction in restarts in the ESD group by one restart would reduce the restart rate to 7.4% and would have decreased the annualized restart costs by $3445, falling below the annualized restart costs in the standard of care group.
Nurses need to be able to track the direct and indirect consequences and costs as the use of the ESD comes with additional costs. Unit-level data are essential to identifying stabilization strategies that will improve the length of dwell time for a short peripheral IV. Using a matched sample or randomizing patients would be ideal.
Limitations
The most immediate limitation was incomplete IV documentation, a problem recognized in practice and the literature.15,16 Despite multiple attempts at improvement in Phase 1, inconsistent documentation of IV removal indications and destabilization loss required that 201 IVs (41% of the IVs that met our inclusion criteria) be eliminated from the analysis. It is, therefore, likely that our findings do not fully describe destabilization or the role of securement methods in IV loss.
Another limitation was that documentation of peripheral IV catheter infections and other complications was not available for analysis. Including these adverse events could provide evidence of the impact of improved IV stabilization on costs, outcomes, and quality of care.
Implications
Stabilization is instrumental in reducing IV restarts as well as preventing IV complications due to destabilized IVs. By increasing the dwell time of an IV, there can be additional time for nurses to focus on other issues. Interruption of care with an IV restart hampers the efficient and effective plan of care. Thus, stabilization would reduce missed medications or delay care.
Unit level data is essential for clinical nurses to identify daily practice changes that improve outcomes and quality of care. While not specific to IV documentation, one implication can impact the nurses’ ability to track clinical practice outcomes. Clinical nurses must be involved in the decision-making regarding health records and operability at the unit level. If clinical nurses want to impact unit-level quality indicators, the ability to abstract unit-level data is essential.
Conclusion
Adding an engineered stabilization device increases the cost of peripheral IV starts but contributes to reducing IV restarts and preventing IV complications due to destabilized IVs. Although there were no statistically significant differences in restarts, the IV dwell time was longer when the device was in place, helping to extend the life of the IV and prevent interruption of care. Understanding the efficacy and effectiveness of the securement method requires full and complete documentation within the EHR. With more than 300 million IVs used to care for hospital patients in the United States a year, 18 the rewards of improving this nursing procedure will be significant for nurses, patients, and quality care.
Supplemental Material
sj-pdf-1-jva-10.1177_11297298221098331 – Supplemental material for Comparative intervention assessing a catheter stabilization device on peripheral intravenous line loss
Supplemental material, sj-pdf-1-jva-10.1177_11297298221098331 for Comparative intervention assessing a catheter stabilization device on peripheral intravenous line loss by Gabriela M Wilson, Rebecca P Winsett, Bhumika Modi, Ru Jia, Terilea Patton and Debra Silberberg in The Journal of Vascular Access
Footnotes
Acknowledgements
The authors thank Jaymee Cannon BSN RN CMSRN for coordinating the training activities involved in the study, and to the Health Informatics Department of St. Vincent Evansville, Indiana, for their assistance with abstracting data from the electronic health record
Declaration of conflicting interests
All authors declare no conflict of interest. Test usage of this device on the selected St. Vincent Evansville unit was approved by hospital leadership.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
Author biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
