Abstract
A 14-week infant, with respiratory distress since birth, was referred to our institution. Chest radiography and ultrasonographic examination confirmed right-sided diaphragmatic eventration. Owing to difficulty in securing a peripheral venous access, a double-lumen 4-Fr central venous catheter (CVC) was inserted into the right internal jugular vein, under ultrasonographic guidance. Aspiration of blood from both ports confirmed intravascular placement. A frontal radiograph done after the procedure showed the catheter tip in the right atrium, hence it was withdrawn to a level just below the carina. Surgical plication of the right dome of the diaphragm was performed, following which an intercostal tube was placed. After 3 days, there was increased drainage of clear fluid. Biochemical analysis ruled out exudative effusion, hence displacement of the CVC into the pleural cavity was suspected. A frontal chest radiograph was done to confirm this, but it did not suggest CVC tip displacement. Bedside ultrasonography was done but the CVC tip could not be visualized. The patient was too unstable to perform a chest CT scan or echocardiography. Therefore, a bedside chest radiograph was taken while injecting 1 ml of iohexol (diluted with 4 ml of normal saline) into the CVC. This showed the contrast leaking out of the CVC, flowing into the mediastinal pleural space, and ultimately into the ICD tube, confirming displacement of the CVC tip. The catheter was immediately removed, and an alternate venous access was established.
Introduction
Pediatric (or neonatal) central venous catheterization is sometimes indispensable in critically ill patients, for fluid and drug administration, hemodynamic monitoring, or blood sampling. Current guidelines recommend that central venous catheters (CVC) should be inserted using real time ultrasound (RT-US) guidance. Even after successful placement, it is often challenging to safely maintain the CVC in place, especially in neonates and infants. A displaced CVC can have disastrous consequences, hence should be detected and managed promptly. We describe an infant with a displaced CVC that was not evident on bedside x-ray and ultrasonography, but another simple bedside investigation confirmed it.
Case description
A 14-week male infant with persistent respiratory distress since birth, was referred to our institution. The antenatal course had been uneventful. The infant had been delivered at term gestation via a lower segment cesarean section due to premature rupture of the membranes. The birth weight was 2700 g. The liquor was stained with meconium, and the infant required resuscitation following delivery. This included ventilation via bag and mask for 30 s, followed by intubation and invasive mechanical ventilation for 5 days. The infant was gradually weaned off the ventilator, but continued to have tachypnea with chest indrawing. There was no history of fever, cough, lethargy, stridor, forehead sweating or suck-rest-suck cycles. He was discharged after 15 days despite having tachypnea. At the age of 10 weeks, he was again hospitalized with increased respiratory distress, and required mechanical ventilation for respiratory failure. Multiple attempts at extubation were unsuccessful. Therefore, he was referred to our institution for further evaluation and management.
At admission to our institution, the infant was on the following ventilator settings: peak inspiratory pressure (PIP) of 16 cmH2O, positive end-expiratory pressure (PEEP) of 6 cmH2O, and fractional inspired oxygen (FiO2) of 40%. On these settings, there was no hypoxemia or carbon dioxide retention.
Weight at admission was 3.5 kg, corresponding to −4.98 z-score, length was at −1.30 z-score and head circumference at −0.91 z-score for age. There was no pallor, cyanosis, abnormal facies, or external congenital anomalies. On mechanical ventilation, there was equal air entry in both the lungs, and no crackles or wheeze. The infant was evaluated for causes of respiratory failure.
The initial leukocyte count was normal (5140 cells/mm3) and blood culture was sterile. The first chest X-ray in our institution showed a collapsed right upper lobe and elevated right dome of the diaphragm (Figure 1). Therefore, radiographs from the previous hospital admissions were obtained on request, and all showed elevation of the right dome of the diaphragm. Unilateral diaphragmatic palsy or eventration was considered. Bedside point-of-care ultrasonographic evaluation confirmed right-sided diaphragmatic eventration, and surgical plication was planned.
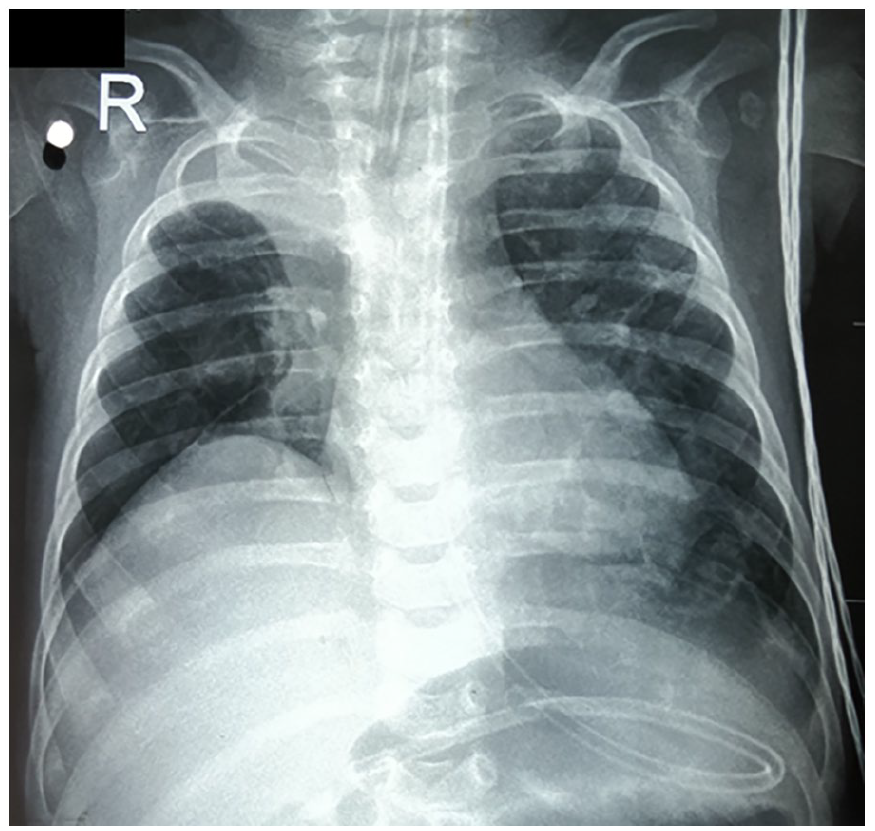
Frontal chest radiograph at admission to our institution, showing right lung upper lobe collapse and elevated right dome of diaphragm.
While preparing for surgery, it was difficult to obtain a second peripheral venous access as most of the superficial veins were not patent. Therefore, a polyurethane, double-lumen, 4-Fr central venous catheter (CVC) of 8 cm length (Certofix® Duo Paed S 408, manufactured by B. Braun, Germany) was inserted into the right internal jugular vein under ultrasonographic guidance, using the Seldinger technique. Aspiration of blood from both ports confirmed intravascular placement. A frontal radiograph immediately after the procedure showed the catheter tip to be in the right atrium (Figure 2), hence it was withdrawn so that the tip was just below the level of the tracheal carina. The CVC was then fixed in place using sutures and an adhesive securement device supplied with it. The insertion site was covered by a transparent adhesive dressing. The CVC was successfully used to administer intravenous medications and draw blood for investigations.
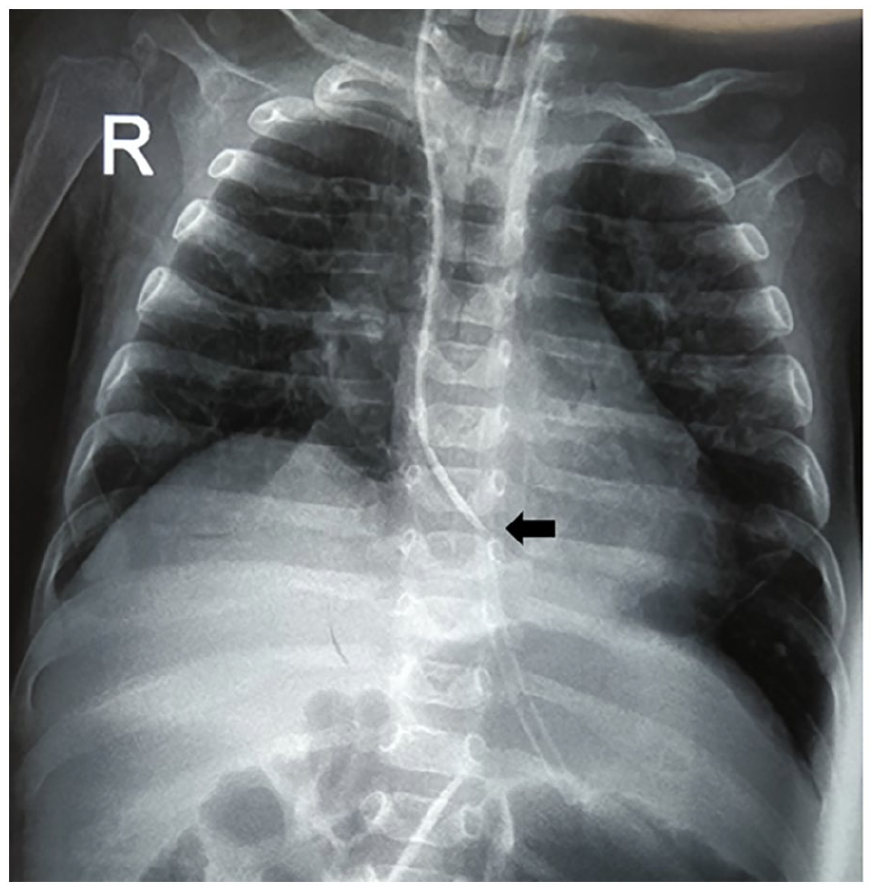
Frontal chest radiograph showing the central venous catheter tip in the right atrium immediately after its insertion (arrow).
The infant underwent thoracoscopic plication of the right dome of the diaphragm, under general anesthesia. During the procedure, only the lateral part of the right hemidiaphragm could be plicated. Hence, the infant continued to require ventilation support despite the procedure. An intercostal tube (ICT) placed in the pleural cavity, drained 100, 80, and 20 ml hemorrhagic fluid over three consecutive post-op days. However, on the fourth day, there was a dramatic increase in the drain output to 500 ml. The fluid was transparent, not bloody and had no flakes. As there was no fever, this seemed unlikely to be due to infection. There were no clinical signs of hemodynamic instability, although the infant appeared mildly dehydrated. Analysis of the fluid showed 118 cells/mcL with 41% neutrophils, 13% lymphocytes and 46% mononuclear cells, very low protein level of 0.1 g/dL and glucose level of 738 mg/dL. Bacterial culture of the fluid was sterile. Clinical judgment and these laboratory parameters raised the suspicion that the CVC tip had migrated into the pleural cavity.
A bedside frontal chest radiograph was done but the CVC did not appear to be displaced (Figure 3(a)). Bedside ultrasonography was performed, but the CVC tip could not be visualized. It was logistically difficult to shift the infant for a CT scan or echocardiography. Therefore, a bedside frontal chest radiograph was repeated, while injecting 1 ml of iohexol diluted with 4 ml of normal saline, into the CVC. It clearly showed the contrast leaking out of the CVC, tracking along the chest wall and pleura, and ultimately draining out through the intercostal tube (Figure 3(b)). This confirmed displacement of the CVC tip. The displaced catheter was immediately removed and venous access was established in the right femoral vein. The infant recovered uneventfully.
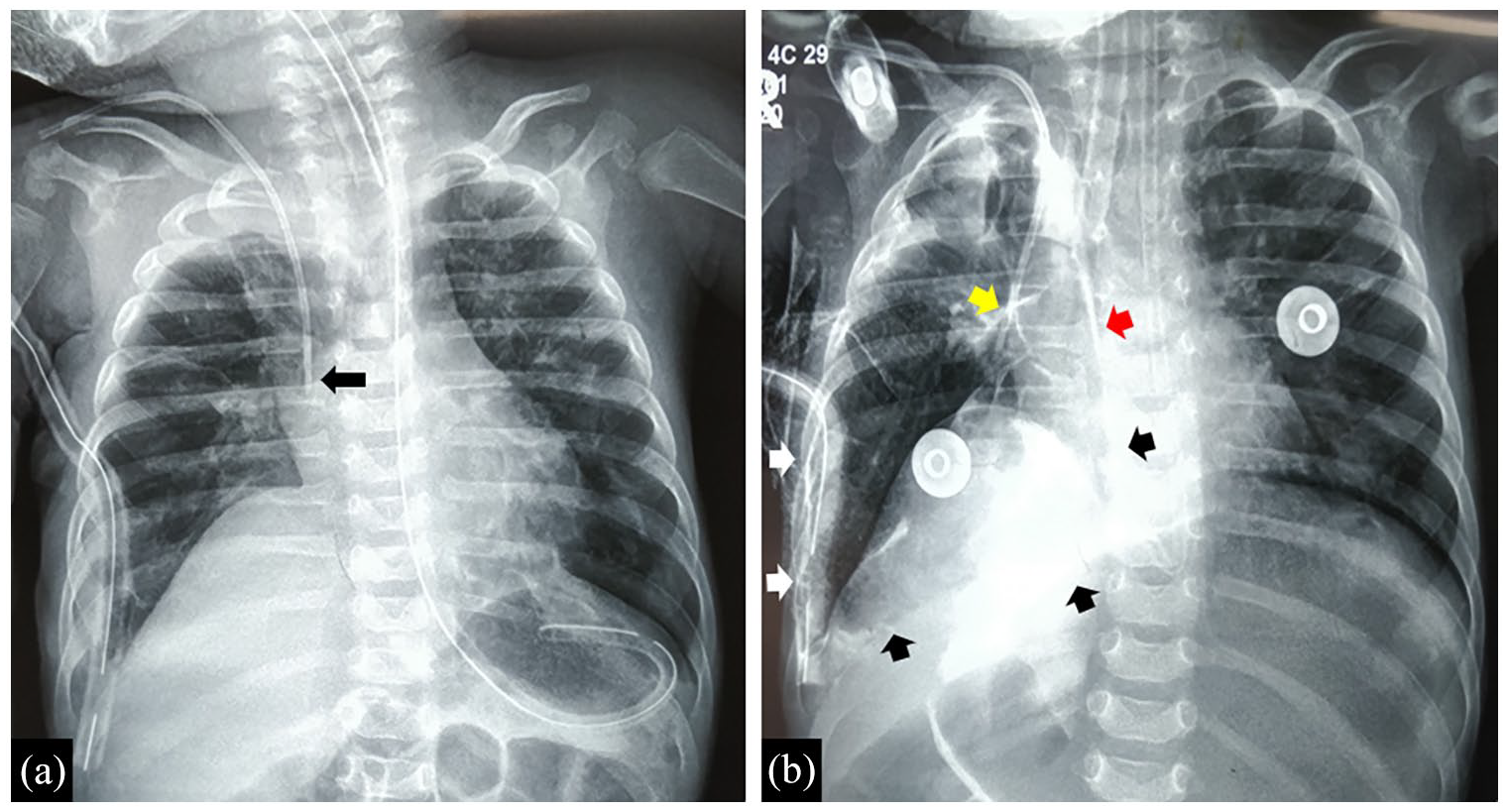
(a) Frontal chest radiograph done after diaphragmatic plication, showing intercostal tube in situ, and the central venous catheter tip (arrow) appearing to be in the correct position. (b). Frontal chest radiograph done while injecting intravenous contrast into the CVC, showing contrast emerging from the central channel (yellow arrow), side-channel (red arrow), tracking along the mediastinal and diaphragmatic pleura (black arrows), and draining into the intercostal tube (white arrows), thereby confirming catheter displacement into the pleural cavity.
Discussion
This case exemplifies one of the several complications associated with central venous catheterization in infants and young children. These complications may be classified as early or late complications. 1 Early complications include arterial puncture, arrhythmia, bleeding, nerve injury, catheter malposition or breakage, air embolism, or pneumothorax. Late complications include infections, thrombosis, and catheter dysfunction. In a prospective study conducted in a pediatric intensive care unit, the rate of catheter related complications was 23/155 (14.8%) and the rate of spontaneous catheter removal was 4/155 (2.6%). 1 As in the index case, there are a few reports of displaced CVC giving rise to pleural effusion, without the exact position of the catheter tip being identified. 2
There are several measures to make CVC insertion safer. Current guidelines published by the European Society of Anesthesiology (ESA), as well as the American Society of Anesthesiologists (ASA) recommend that CVC tip location should be performed using intracavitary electrocardiography (IC-ECG).3,4 This is considered the current gold standard for catheter tip control, and is also helpful to identify secondary malpositions. When intracardiac ECG is not applicable, the ESA 2020 guidelines recommend to use real-time ultrasound as a safe and effective tip location strategy. 3 The recently published ECHOTIP-Ped and Neo-ECHOTIP protocols fully describe how real time ultrasound can be used for such purpose.5,6
However, in our healthcare setting (as in most developing country settings), these facilities are often not available at the point of care. In fact, we do not even have an ultrasonography machine within the ward. Bedside ultrasonography has to be requested whenever required, and there may be delays due to this. The availability of equipment and trained personnel was further compromised during the COVID-19 pandemic, due to diversion of these limited resources to dedicated COVID wards. As a result, the position of the tip was not reconfirmed after it was withdrawn.
In hindsight, there are several measures that could have been used for safer maintenance of the CVC in the index case. For example, the fact that the tip location was not based on a real time technique (IC-ECG or RT-US), resulted in the CVC tip being too high when it was retracted, thereby creating the risk for damage to the vein wall. Ideally, the CVC tip should be placed at the junction between the superior vena cava and the right atrium. In settings where facilities are not readily available for the initial placement, these should be sought when CVCs have to be re-positioned for any reason, and also whenever any complication is suspected. In situations where the catheter tip is not identifiable on bedside ultrasonography, flushing with a small bolus of saline can help to visualize the tip. Thus, in the index case, real-time ultrasonography should ideally have been used, not only to locate the CVC tip during insertion, but also to confirm its position when it was pulled back. Flushing the CVC with saline would have helped to visualize the tip, and possibly observe its drainage into the pleural cavity, providing proof of dislodgement.
In the index case, it is not clear when the actual dislodgement of the catheter tip occurred, although it is reasonable to suspect that it happened on the fourth post-op day. Thankfully, the leakage occurred into the pleural cavity, and thence the intercostal tube. This protected the infant from potentially serious respiratory compromise. It is also fortunate that there was no major vascular injury or hemodynamic instability.
A correctly placed CVC can be secured more safely by tunneling the catheter in the infraclavicular area (especially where long-term use is considered). We did not do this as the CVC was intended for a relatively short duration of time. Alternatively, stabilization could be done using cyanoacrylate glue at the puncture site and a sutureless device rather than stiches.7,8 However, we fixed the CVC using only sutures and the securement device supplied with it.
Frontal chest X-rays, which used to be the most common modality to confirm correct placement (and by extension, suspected displacement) of a CVC, are now considered to be inaccurate for the purpose. 9 Fluoroscopy is another option, although it is not available at the bedside. 9 The increasing availability of bedside point-of-care ultrasonography has reduced the dependence on X-rays to confirm the position reliably. 10
Our experience in the index case should not be taken as an endorsement of using contrast injection as a method to identify CVC dislodgement. We emphasize that when intracavitary ECG or echocardiography are unavailable at the point-of-care, real-time ultrasonography should be used during CVC insertion, CVC maintenance, and when complications occur.
Footnotes
Consent
A written informed consent was taken from the parents for publication of this case report and the images.
Contributorship
KK and JLM were involved in the clinical care of the patient, KSS reviewed the radiology images, KK wrote the initial draft of the manuscript, JLM and KSS reviewed and finalized the manuscript. JLM will act as the guarantor.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
