Abstract
Tunneled femorally inserted central catheters (FICCs) are frequently required for central access in children when upper extremity vessels cannot or should not be cannulated. A recently published decision tool for tunneled FICCs identifies the medial thigh as the preferred exit site. In pediatric patients, this medial exit site may remain at risk of contamination from stool due to anatomic size, and there are no tools developed for FICC exit site decisions specific to children. We present our approach for the placement of the exit site in the far lateral region of the thigh and review previous FICC literature relevant to the pediatric population. In select patients, a lateral approach has the potential to decrease the risk of exit site contamination to prolong catheter viability and reduce patient harm.
Introduction
Protocols and guidelines support the use of tunneled femorally inserted central catheters (FICCs) in children with superior vena cava (SVC) obstruction, pre-existing SVC catheters, or certain forms of congenital cardiac disease.1–3 Traditional tunneling methods for FICCs identify the medial mid-thigh as the ideal exit site 2 (and/or entry site as in single-stick tunneling in small children and infants). However, a medial exit site placed high on the thigh may place the FICC at risk of contamination and subsequent risk of a central line-associated bloodstream infections (CLABSI).4,5 We present our approach to a lateral exit site for tunneled FICCs and review relevant literature in our pediatric population.
Case example
A 4-year-old (15 kg) male with a history of congenital diaphragmatic hernia and pulmonary hypoplasia presented with progressive pulmonary hypertension requiring critical care management. He had an SVC occlusion following extracorporeal membrane oxygenation as an infant and had a persistent left SVC draining to the coronary sinus. He had a prior history of a left lower extremity tunneled FICC with a medial exit site. Due to his acuity, he required a double lumen catheter so a tunneled FICC was planned. Prior to placement, the left femoral vein was found to be appropriately sized to accommodate a 4Fr catheter. The left femoral vein was also chosen to keep the right groin open for an access site for potential cardiac catheterization. The patient was adequately sedated for the procedure. For preprocedural planning the access site was marked. Next, a far lateral exit site away from the knee was selected and marked 10 cm distal to the access site. The planned tunnel track was assessed with ultrasound to verify the absence of anomalous vessels or lymph nodes. The left common femoral vein was accessed with a 21-g 7 cm needle using ultrasound-guidance. A 0.018-inch nitinol wire was inserted through the needle into the vessel. With ultrasound the wire was inserted so the tip was visualized at the inferior vena cava/right atrial junction. The excess wire was laid over the proposed exit site. Subtracting the difference of the wire length we were able to assess the total length of FICC required. A trimmed 4Fr double lumen polyurethane power-injectable catheter was attached to a tunneling device, inserted in the marked exit site, and advanced to the nitinol wire. A 4.5Fr peel away introducer was placed over the wire and the catheter was inserted through the introducer and into the vessel. The FICC was secured to the leg with a skin adhesive suture less device (Statlock®; BD) and the access site was then closed with cyanoacrylate glue (SecurePortIV®; Adhezion) and covered with reinforced adhesion skin closures (Steri-Strips®; 3M). (Figures 1 and 2). The catheter was utilized for 40 intensive care days and was without associated infection, infiltration, or thrombosis during its clinical lifespan.
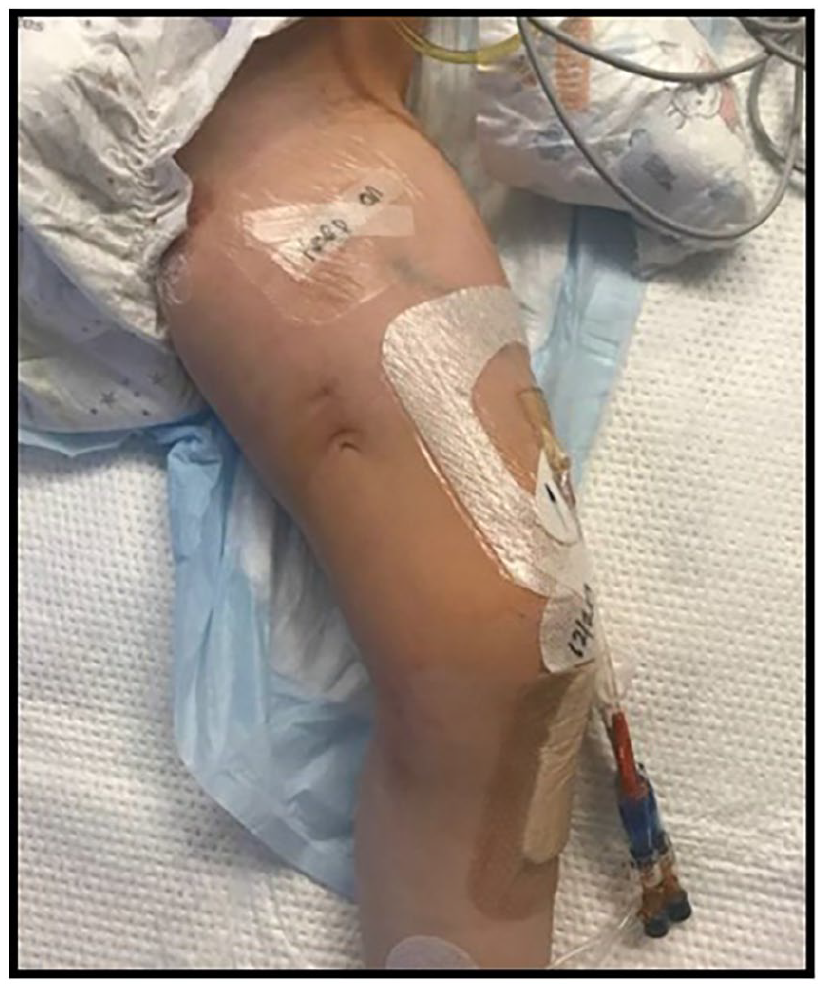
Leg in neutral position with view of access and exit site. Scar from prior FICC noted on medial thigh.
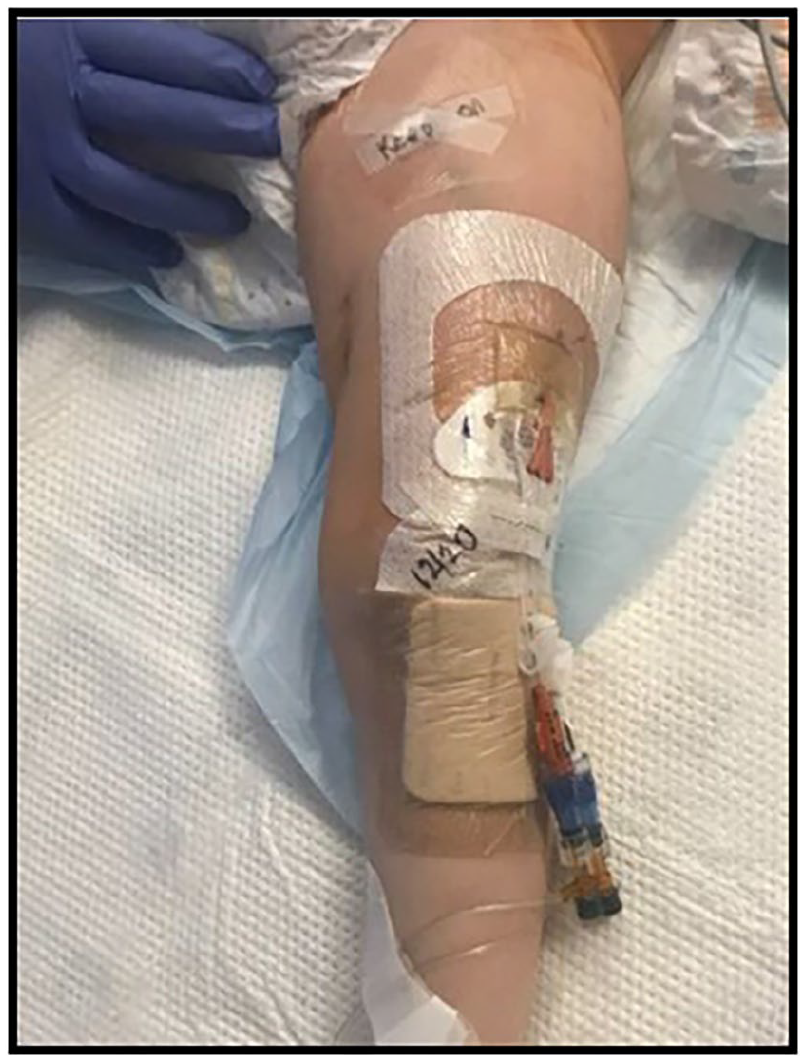
Leg rotated medially with view of exit site on lateral thigh.
Discussion
This case describes our use of a lateral tunnel exit site for FICC placement in a critically ill child. The current literature regarding FICC placement does not address potential unique needs within the pediatric population. Our approach to a lateral FICC exit site may reduce the incidence of line contamination from stool in the pediatric population.
In small children (<10 kg) and infants a lateral single stick approach may be employed to create the subcutaneous tunnel. 6 Larger children, such as in our presented case, will require a tunneling device to create the subcutaneous tunnel (Figures 3 and 4). To accomplish a lateral exit site, the anticipated access site to the vessel is visualized with ultrasound and marked. When using a tunneling device, the exit site is selected in a distal and lateral location that places the exit site in the middle or distal third of the thigh. The anticipated course of the tunneling, whether with needle or device, is interrogated prior to the procedure with ultrasound to identify blood vessels or lymph nodes to avoid. Generally, sedation and appropriate use of local anesthetics are required for these procedures.
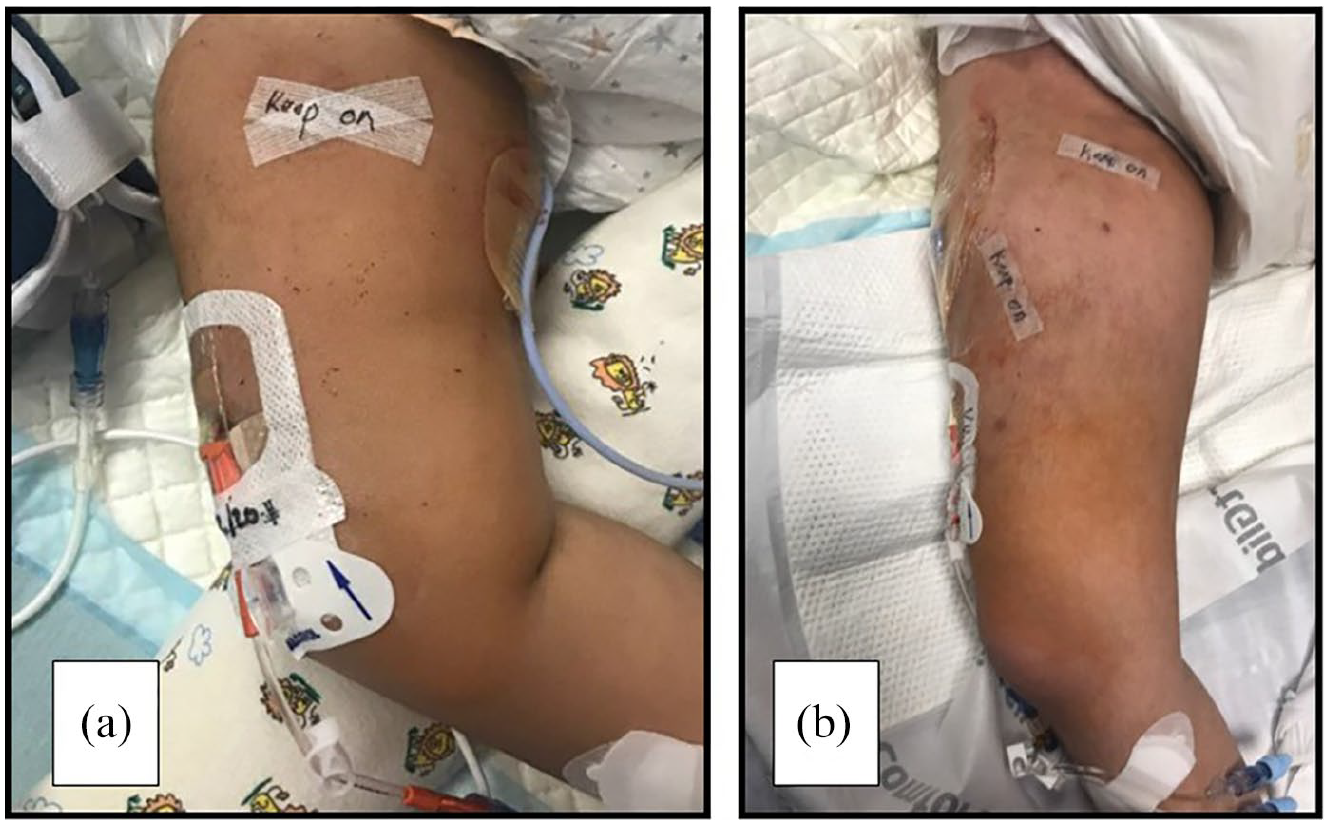
Other examples of FICCs with lateral exit sites: (a) an 18-month-old patient and (b) a 19-year-old patient.
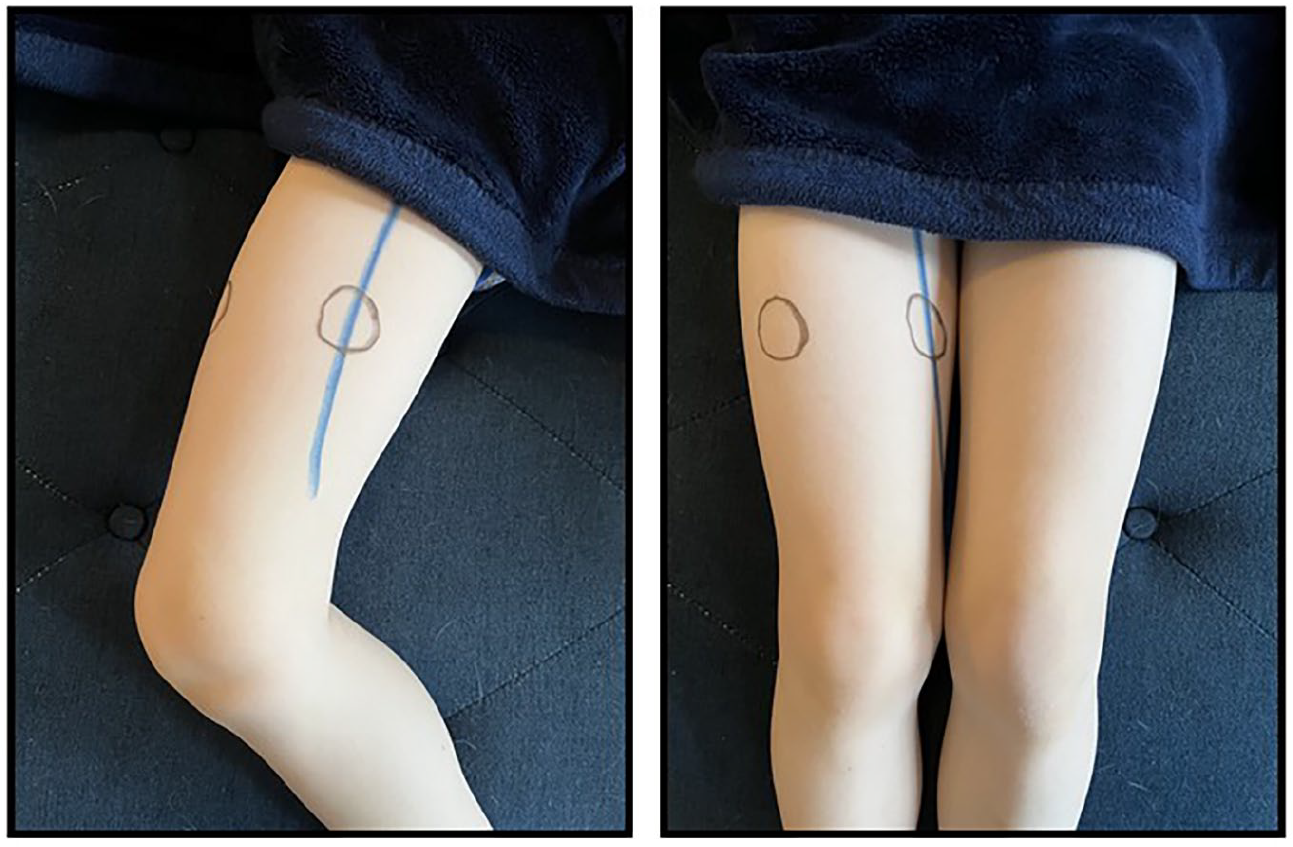
Blue line representing the superficial femoral vein with traditional medial exit site overlying the vein and lateral exit site each marked with circles. Figure on left with leg abducted and figure on right with leg adducted.
FICCs are infrequently used in pediatric clinical care but are a valuable option to provide life sustaining therapies in children with clinical indications for their use. 7 Previous descriptions of FICC placement suggest a tunnel site that lies within the medial plane of the mid-thigh.1,2 When placing a tunneled FICC an appropriate access site to the vessel is selected first. The use of Rapid Femoral Vein Assessment protocol can assist in the selection of an appropriate vessel. 8 Following the selection of the access site the exit site is then selected. Dawson’s Zone Insertion Method (ZIM) 9 of site selection for peripherally inserted central catheters can be applied to the femoral exit site, also referred to as the Femoral ZIM. 2
Tunneling is not always required in adult FICCs as directly accessing the superficial femoral vein in the mid-thigh is an appropriate approach assuming the vessel is of a sufficient diameter.3,10 With both tunneled and non-tunneled FICCs, a medial exit site may have become the standard due to its convenient procedural presentation in a patient whose leg is in a neutral position, as well as it being in plane with the vessel leading to a linear tunnel track. While a medial exit site may be convenient for the proceduralist, if placed too high it may render the FICC prone to stool contamination and possibly contribute to an increased risk of CLABSI. 11 The CLABSI rate in FICCs has been shown to increase at day 30 with enteric organisms causing most infections. 12 This suggests that stool contamination may be a significant contributor to the FICC CLABSI etiology. Simple diapering may not be adequate to contain a large stool burden.
Lateral exit sites in children, whether for single-stick or tunneled FICC placement, may be a method to mitigate the risk of catheter contamination. We suggest exit sites should be specifically documented for quality improvement processes targeted toward reducing patient harm. Comparing exit site data may elucidate both mechanisms of patient harm as well as the clinical impact of FICC technical performance in improving patient outcomes. Such data may result in evidence-based procedural best practice modifications specific to the pediatric population.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr. Himebauch receives support from the National Heart, Lung, and Blood Institute of the National Institutes of Health under Award Number K23HL153759. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Ethical approval
Local Institutional Review Board/Ethics Committee was not required for this manuscript as it contains less than five patients.
Informed consent
Written informed consent was obtained for placement of PICCs. No written consent was required by the author’s institution for a case presentation including deidentified patient data.
