Abstract
Background:
Umbilical vessels present after birth allow a unique central access for both venous and arterial catheterization, yet the catheterization complications can be misdiagnosed as the complications of prematurity per se.
Methods:
A prospective observational study of 41 used polyurethane umbilical catheters, both venous and arterial was conducted in a tertiary neonatal intensive care unit. The study consisted of bedside ultrasound imaging and post-removal microbiological and microstructural analysis to assess the in vivo catheters’ changes and their clinical significance.
Results:
The study has shown that catheters’ surface thrombosis and bacterial colonization happen more often within umbilical venous than within arterial catheters (31% vs 8% in both cases) and are inversely proportional to the patient’s gestational age (thrombosis: Me: 28 weeks vs no thrombosis: 32 weeks; p = 0.05, bacterial colonization: 27 weeks vs no colonization: 30 weeks; p = 0.013), respectively. The clots formed near the catheter’s tip are correlated with catheter’s bacterial colonization. Chemical analysis with energy dispersive spectroscopy showed a higher calcium composition in used catheters (19.89% vs 0%, p = 0.016) and structure analysis in the scanning electron microscopy proved that within hours catheters become covered with an external coating of a constant thickness, not affected by the catheterization time.
Conclusion:
The following observations give a better insight to the complex in vivo interactions and call for a more intense bedside-monitoring of the indwelling devices.
Introduction
Umbilical catheters (UCs) have changed the face of neonatology. With their introduction to clinical practice, continuous blood pressure monitoring, taking blood, and parenteral nutrition or drug administration have become easier and safer for the smallest patients like never before. The great advantages of their usage are no need for venipuncture or puncture of the artery, sparing the other central vessels, and relatively simple operating technique.
However, as with every medical procedure, umbilical vessels’ catheterization may generate serious complications. According to the latest systematic review, 13.4% of umbilical vein catheters (UVC’s) and 9% of umbilical artery catheters (UAC’s) are associated with an adverse effect. The most common complication is malposition of UVC. 1 Other adverse effects mentioned in the literature are central line-associated bloodstream infections (CLABSIs), thromboembolism, air embolism, detachment of the cannula, and many others.2,3
As some of the above-mentioned neonatal complications may be lethal, emphasis should be put on investigation on that topic. We hypothesize that such life-threatening complications as thrombosis or CLABSIs are possibly related to the changes in catheter’s microstructure. Many years have passed since the UCs were introduced in clinical practice and still there is no sufficient data on the microscopic structure of umbilical catheters and their predisposition to damage.
Nowadays there are new methods of chemical analysis and modern imaging available in the scientific world. That is why in our study we decided to take a closer look at catheter structure and analyze its potential role in generating the observed complications. The aim of our study was to deepen the understanding of correlation between thrombosis, bacterial colonization, and microstructural changes of the catheter.
Methods
Study design
A prospective observational study was conducted in a tertiary neonatal intensive care unit (NICU) in the Department of Pediatrics, Jagiellonian University. The participants were consecutively recruited NICU patients of any weight or gestational age who had an umbilical catheter (venous or arterial) inserted. Informed consent was obtained from the parents. The analyzed umbilical catheters were composed of polyurethane and impregnated with barium for radiography visualization (Vygon umbilical catheters, 2.5 and 3.5Fr).
UVC placement and ultrasound imaging
Inserting of the catheter was performed in a sterile manner: the periumbilical area was disinfected with a chlorhexidine solution. Afterward the cord clamp was held with sterile gauze and sterile drapes were applied. To minimize the blood loss an umbilical tape was tied around the base of the cord. The cord was cut with a sterile scalpel to expose the umbilical vessels and with sterile forceps the catheter was slowly inserted under the supervision of ultrasound. The site preparation, skin antisepsis, and insertion procedures were consistent with the 2021 INS Guidelines. 4 The tip navigation and location protocols were consistent with Neo-ECHOTIP protocol by Barone et al., 5 although the linear probe was used from a frontal thoracic view. The correct position for the tip was at the junction between the inferior vena cava and the right atrium. After confirming the proper position of a catheter, each catheter and securing suture were secured together with tape. After the procedure was complete, the antiseptics were removed by saline solution. Ultrasound imaging was performed directly after insertion, within the first 24 h and then every 1–2 days or more often if the patient was unstable. The catheters were in the next days observed in vivo by ultrasonography for thrombi formation or malposition. Bedside examinations were performed using a Philips HD11 ultrasound system with a linear probe either by a certified neonatologist or a trained pediatric resident; all examinations were recorded and verified by a second examiner. The types of fluids infused into the catheters were parenteral nutrition and neonatal drugs. No blood products were administered into the catheters and no antibiotic prophylaxis of CLABSI was used.
Post-catheterization analysis
Upon routine removal the umbilical catheters were collected, immersed in the distilled water for excess blood/drugs removal, dried out, and cut into three pieces (Figure 1(a)). All procedures were performed under sterile conditions. Distal 1 cm of the catheter was cut vertically (tip of the catheter, Figure 1(c)) and transverse (lumen of the catheter, Figure 1(b)) for microstructure analysis. The following 5 cm of the catheter was sent for microbiological analysis.
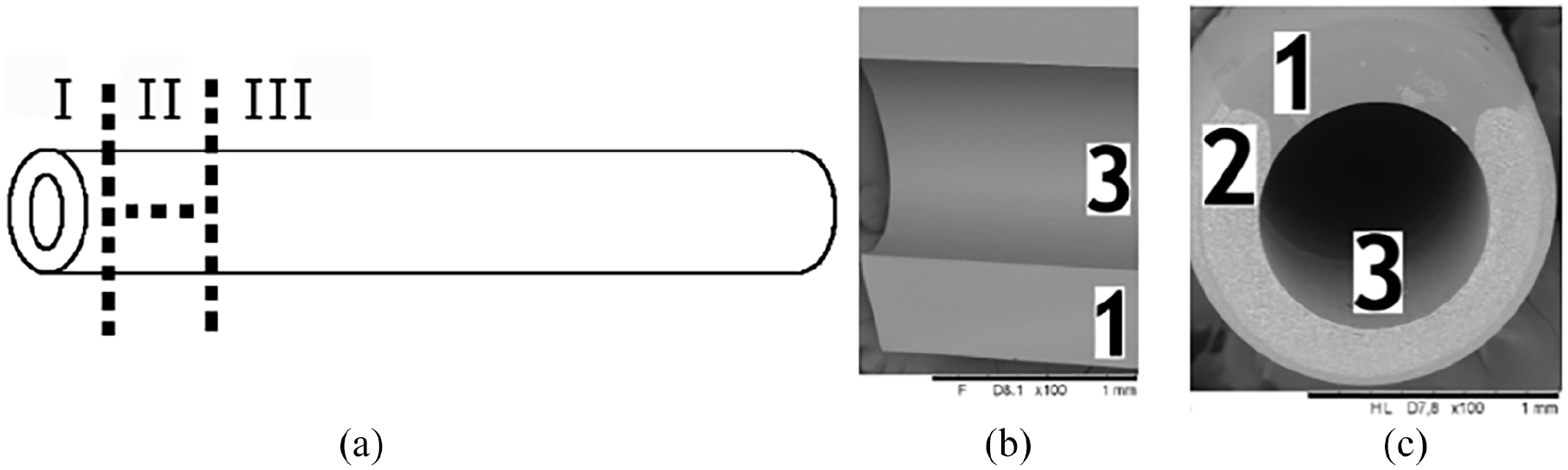
(a) Catheter’s preparation: I—catheter’s tip microstructure analysis, II—catheter’s lumen microstructure analysis, III—microbiological analysis; microstructure images: (b) longitudinal section (c) transverse look at the tip; 1—contrast-free part, 2—contrast part, 3—lumen.
Microstructure analysis
The catheters’ structure was characterized by non-destructive analysis using Scanning Electron Microscope (SEM, Hitachi TM3000) coupled with Energy Dispersive X-ray Spectroscope (EDS) for local chemical analysis. Used catheters were compared to the new ones, and the new ones had a default dual structure (3/4 of the catheter’s cross-section composed of the heterogenous barium-polymer, and ¼ of the catheter composed of the homogenous polyurethane (Figure 1(c)).
Microbiological analysis
Catheters’ bacterial colonization was analyzed using Brunn’s and Maki method, defined as growth of bacterial colonies on an agar plate cultivated in a sterile environment.
Statistical analysis
Statistical analysis was performed with the use of SPSS version 27 software (Armonk, NY, USA). Results were presented based on the parameters of descriptive statistics, including either mean values and standard deviations (SD), or median values with range for continuous variables and numbers with percentages for categorical variables. Qualitative values were compared by Fischer exact test. To assume the differences in continuous variables between studied groups Mann-Whitney U test were used. The correlation analysis was performed using the Spearman test. Differences were found as statistically important if the probability of type I error α was lower than 0.05
Outcome variables
The screening for thrombosis and bacterial colonization were qualitative. The structural analysis was both qualitative and quantitative. The presence of the following structural changes was detected (Figure 2):
The tips were coated to a various degree, so a quantitative assessment scale was used (0: lack of coating; 1: <25% of the surface coated; 2: 25%–50%; 3: 50%–75%; 4: >75%; 5: the whole surface coated). Additionally, the chemical composition of the coating on the catheter’s tip and within the inner surface was analyzed.

Structural changes observed with Scanning Electron Microscopy: (a) coating, (b) clot, (c) stricture, (d) flakes, (e) fringes, and (f) forehead roughness.
Results
Forty-one used polyurethane umbilical catheters (29 venous and 12 arterial) from a total of 33 patients were prospectively evaluated. The patients' gestational ages ranged from 23 to 41 weeks (median 29 weeks), and their birth weights ranged from 630 to 3620 g (median 1290 g). The main reason for umbilical catheterization was prematurity (73%, n = 24; including 11 extremely premature patients); other causes were therapeutic hypothermia monitoring (15%, n = 5), meconium aspiration syndrome (n = 3), and one congenital disorder. The catheterization time ranged from 2 to 34 days, with a median time of 8 days for UAC and 10 days for UVC. The exceptionally long 34-days catheterization time was observed in a patient born in extremely critical condition with birth weight <750 g. Due to circulatory insufficiency and extreme edema no other central catheter could be inserted. Maintaining the umbilical venous catheter despite the recommended catheterization time was a life-saving procedure for the child.
During catheterization in 31% (n = 9) of UVCs and 1 (8%) UAC a catheter external thrombosis was observed. Thrombotic catheters were characterized by longer median catheterization time (12 vs 8 days; p = 0.006), and younger gestational age (Me: 28 vs 32 weeks; p = 0.05).
All removed catheters were sent to the microbiological laboratory. Positive culture was observed in 10 cases—nine (31%) of UVCs turned out to be colonized with bacteria whereas the colonization occurred in only 1 (8%) UAC catheter. Among 10 positive cultures, coagulase-negative staphylococci were detected in nine cases and Gram-negative bacteria in one case. Out of eight colonized UVC, three resulted in CLABSI (37.5%); the only one colonized UAC caused general bacterial infection (100%).
Colonized catheters had a longer average catheterization time (15 vs 8 days; p = 0.001), younger gestational age (27 vs 30 weeks; p = 0.013), and lower birth weight (1000 vs 1430 g; p = 0.07). The patients in the thrombosis and colonization group varied—only three patients had a thrombus upon ultrasound together with a positive catheter culture.
Around 24/41 catheters (59%) with time were found in a different location than upon insertion. In 7/41 (17%) of cases the position was critical enough to remove the catheter based on the obtained ultrasound image.
Microstructure analysis
Upon microstructure analysis all the catheters had an external surface and tip coating in varying degrees (labeled 0–4). Median degree of UVC coating for the forehead was 3 and the median degree of UAC coating for the forehead was 2 (p = 0.05). Median degree of external surface coating was 2 for both UVC and UAC (p = 0.49). There was no correlation between degree of coating and catheterization time, however there were correlations between degree of external surface coating and gestational age (R = −0.295; p = 0.05) and birthweight (R = −0.385; p = 0.02). Twenty-two (54%) catheters had a forehead roughness observed—a feature related to the manufacturing process observed in new catheters as well. There was a negative relationship between forehead roughness and degree of forehead coating (p = 0.048).
Thirty-nine (95%) catheters presented with an inner surface coating. The coating was thick enough to narrow the catheter lumen in 17% cases. There was no difference in the prevalence of catheter narrowing between the UVC and UAC, colonized versus non-colonized catheters or catheters with thrombi confirmed by ultrasound versus no-thrombi catheters. Neither catheterization time, maturity of the newborn and birthweight influenced the risk of catheter’s lumen narrowing.
Apart from thrombi formed on the external part of the catheter observed with ultrasound screening, microscopic analysis revealed clots within the catheter’s lumen near the tip in the case of 10 catheters. The most important risk factor of clots formation was the positive result of microbiological culture (6/10 (60%) vs 4/31 (12,3%); p = 0.006). Moreover, irregular homogenic structures that can be responsible for the initiation of the clotting process (flakes) were observed in the case of 30 (73%) catheters.
Energy dispersive X-ray spectroscopy
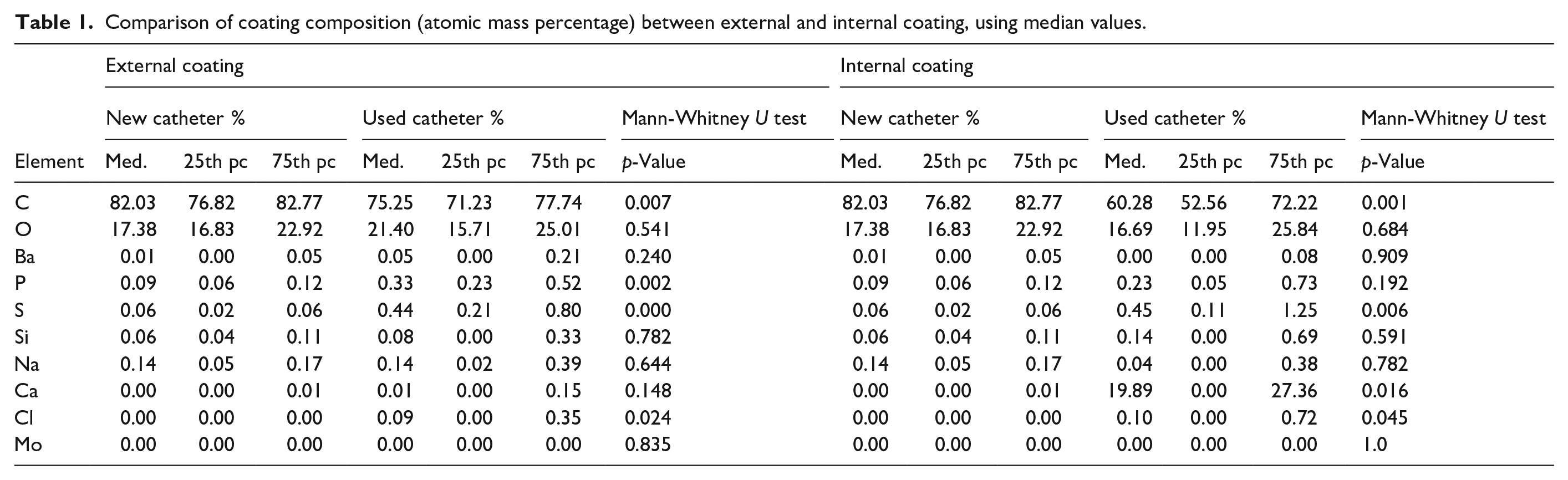
The elemental composition of used catheters coating was analyzed using Energy Dispersive X-ray Spectroscopy and compared to the surface of new catheters. There was a significant difference between new and used catheters. The external coating was characterized by statistically lower carbon composition and higher phosphorus, sulfur, and chloride composition. The results of spectrum analysis of the internal coating were presented in Table 1. The calcium content was significantly different between new and used catheters (Graph 1). Moreover, calcium exceeded 10% in the case of 21 (52%) internal coating analyses. Higher calcium content was correlated with longer catheterization time (R = 0.213; p = 0.1).
Comparison of coating composition (atomic mass percentage) between external and internal coating, using median values.
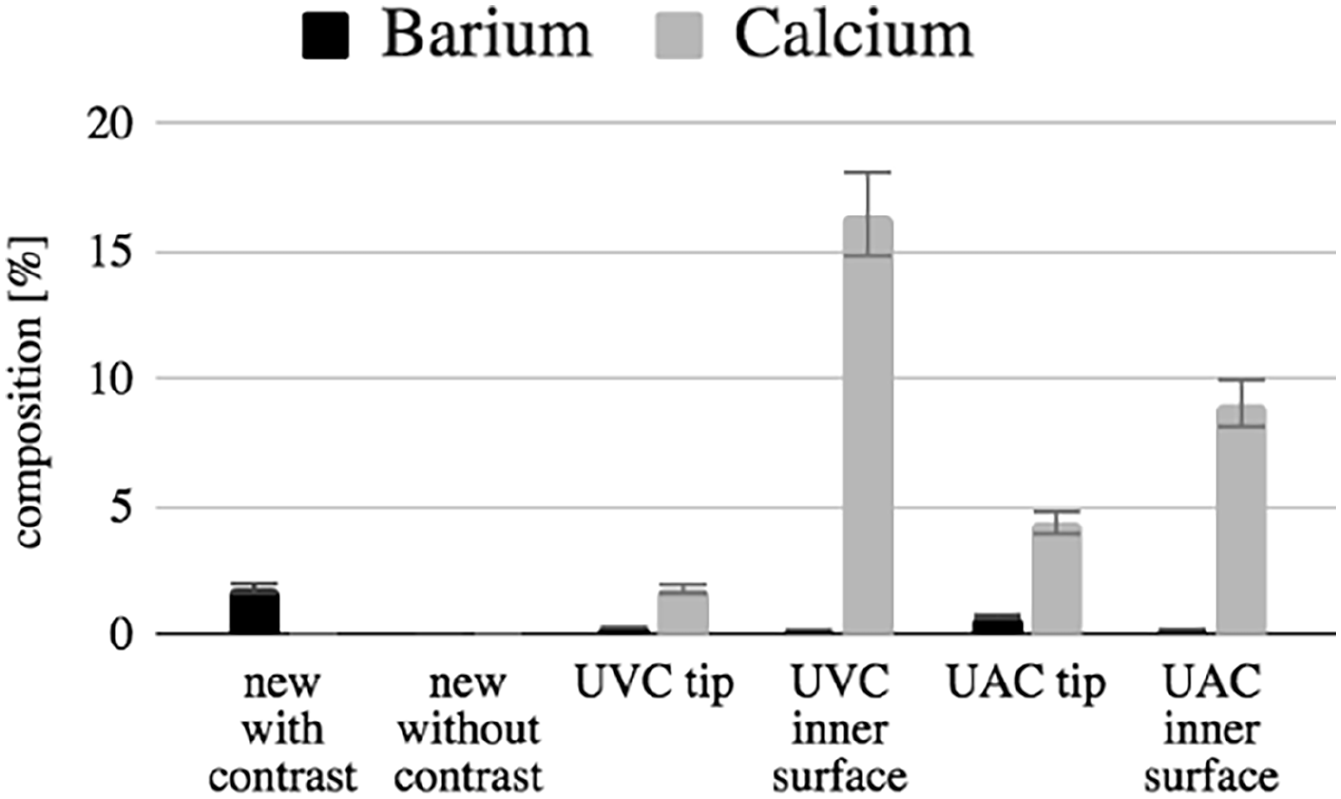
Comparison of barium (default contrast) and calcium (build-up with time) content within new and used catheters.
Discussion
This study provides a complex prospective analysis of the structural changes of umbilical catheters resulting from the complex in vivo interactions, regarding clinically significant thrombosis observed upon ultrasound imaging, microbiological smears with a sepsis-inducing potential and microstructural changes observed with electron microscopy. There is a lack of similar studies in the literature, and the subject seems worth investigating since it serves as an accurate universal model of vascular line’ colonization and thrombotic potential.
The strength of this study is its innovation and multidimensionality. The fact that it is a single center small descriptive study without sophisticated statistical methods is its major limitation. Nevertheless, it provides a new approach to the complexity of catheterization complications.
The study showed that a predisposition to external catheter thrombosis and bacterial colonization occurs mainly in the umbilical venous catheters and is directly proportional to catheterization time and inversely proportional to gestational age of the patient. This can be explained by deceleration of the blood flow around venous catheters compared to the arterial ones and additional flushing of the UAC’s since they are facing the flow upstream in the aorta. Additionally, the arterial catheters are usually fixed more tightly than the venous ones, what can also be a factor diminishing the risk of thrombosis. Prematurity is a known risk factor for thrombosis due to impaired fibrinolysis 6 and a cause of altered resident microbiota. 7 Colonization of catheters with mainly Coagulase-negative staphylococci that we have shown goes along with the well-known fact that many invasive infections of preterms are caused by skin-colonizing bacteria. 8 Moreover, administration of osmolar hyperalimentation fluid increases the risk of the mentioned events.
Our research revealed that all used catheters to some degree have an outer and inner catheter’s surface coating. Venous and arterial catheters differed in the intensity of forehead coating, which can be associated to the already mentioned mechanical flushing of UAC tip located upstream in the aorta. Surprisingly there was no correlation between degree of external surface coating and catheterization time, which may be coherent with the recent reports that all intravascular catheters are physiologically covered with circulating fibronectin forming a fibroelastic sleeve, that is not a thrombi.9,10 This process starts within 24 h of catheterization 11 and is a formation of connective tissue that does not uncontrollably build up like a thrombus. In our study the external coating was correlated with gestational age and birthweight once again suggesting an altered physiology of a preterm infant. The internal coating was not influenced by any of the studied factors, which indicates it may be associated with a chemical reaction with administered fluids or with a different agent that was not analyzed in our study.
Apart from the catheter's surface coating, heterogeneous spongy-structured structures near the catheter’s tip were observed and identified as thrombi. As described in the literature, colonization of indwelling devices without yet present evidence of bacteremia is often expressed by thrombi formation 12 and its presence should be a red flag for a potential sepsis occurrence. Moreover, the location of the potentially colonized thrombi—near the tip—can explain the pathway from a catheter’s colonization to a generalized infection (by the thrombi detachment and migration to the circulation).
Calcification of catheters with time (already after a couple days) suggests a polyurethane tendency to absorb calcium ions, leading to the loss of catheter’s elasticity and a higher risk of thrombotic events. Both of those features suggest a need for a regular ultrasound monitoring of the catheters, as they may cause vessel injury or thrombotic process.
As umbilical catheters were collected randomly, once again13,14 with our study we proved that around 3/4 of umbilically catheterized patients are premature children. Knowing the complexity of prematurity complications, we suggest that they could be partially avoided with implementing catheters’ monitoring.
Limitations of the study
The main limitation of this study is that the use of the catheters in the analyzed group does not reflect the recent standard of care suggested by the recent literature 4 that suggests considering limiting UVC dwell time to 4–7 days and UAC to 5 days, in a perfect clinical setting. Unfortunately the catheter removal is not always possible due to lack of other insertion sites (edematous patient, extremely low birth weight) thus sometimes clinical practice differs from the guidelines. Our research was conducted before the mentioned guidelines were published. The heated discussion of umbilical catheterization time was also present in our unit pushing us to create a catheters’ monitoring algorithm based on our clinical observations, where a catheter could be maintained for a longer time as long as it was repeatedly observed with a bedside ultrasound.
We have made an additional analysis limiting the dwell time according to current recommendations; however, we ended up with the same results of a smaller group, thus we decided not to include it in the following paper.
We believe that our research has a significant value since it confirms the standard of care suggested by the recent literature. It proves that the coating of catheters happens quickly within hours after insertion into the vessel and therefore regular monitoring is needed.
Conclusions
Polyurethane umbilical catheters undergo changes in vivo that can lead to thrombosis, sepsis, and tissue damage. After insertion into the vessel within days they bind calcium and lose their elasticity, and within hours they become covered with an external coating of a constant thickness, not affected by the catheterization time; it may be a fibroelastic sleeve observed in the vessel after umbilical arterial catheter removal. Additionally catheters’ surface thrombosis and bacterial colonization happen more often within umbilical venous catheters and are inversely proportional to the patient’s gestational age. The clots formed near the catheter’s tip are correlated with catheter’s bacterial colonization, potentially leading to sepsis when detached.
The following observations call for a more intense bedside-monitoring of the indwelling devices. We hope that our results may lead to the further development of new technologies in this field and at the same time reduce the adverse effects caused by umbilical catheterization in newborns.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was conducted with funding from the National Science Centre, Poland as a part of project no. 2016/21/N/NZ5/01442.
Ethical approval
The research was performed with approval from the Jagiellonian University Bioethics Committee (no. 122.6120.287.2015) and informed parental consent.
