Abstract
Insertion of Peripherally Inserted Central Catheters (PICCs) is potentially associated with the risk of immediate/early adverse events, some of them minimal (repeated punctures) and some relevant (accidental arterial puncture or nerve-related injury). Several strategies adopted during the insertion process may minimize the risk of such events, including late complication risks such as infection, venous thrombosis, or catheter dislodgment and/or malposition. This paper describes an update version of the SIP protocol (Safe Insertion of PICCs), an insertion bundle which includes eight effective strategies that aims to minimize immediate, early, or late insertion-associated complications. These strategies include: preprocedural ultrasound assessment utilizing the RaPeVA (Rapid Peripheral Venous Assessment) protocol; appropriate skin antiseptic technique; choice of appropriate vein, adoption of the Zone Insertion Method™; clear identification of the median nerve and brachial artery; ultrasound-guided puncture; ultrasound-guided tip navigation; intra-procedural assessment of tip location; correct securement of the catheter, and appropriate protection of the exit site. This updated version of the SIP protocol includes several novelties based on the most recent evidence-based scientific literature on PICC insertion, such as the clinical relevance of the tunneling technique, the use of ultrasound for intra-procedural tip navigation and tip location, and the new technologies for the protection of the exit site (cyanoacrylate glue) and for the securement of the catheter (subcutaneous anchorage).
Keywords
Introduction
Peripherally Inserted Central Catheters (PICCs) are widely used for administration of chemotherapy, antibiotics, parenteral nutrition, for short-, medium-, and long-term venous access and for frequent blood sampling amongst both hospitalized and non-hospitalized patient populations. The insertion of PICCs is a frequently performed procedure in clinical practice, currently associated with a low risk of complications.1–11 In the last two decades, many factors have improved the safety of this procedure, the most important being the increasingly widespread use of ultrasound (US) in different phases of PICC insertion. Ultrasound is used for preliminary venous assessment, real-time venipuncture, and immediate detection of puncture-related complications (tissue hematomas, intramural hematomas of the vein, etc.) 5 ; US is also appropriate for “tip navigation” (i.e. to verify the correct direction of the guidewire and/or catheter while the device is advanced into the vascular system), for “tip location” (i.e. to assess the central position of the tip), and for the diagnosis of most late non-infective complications (fibroblastic sleeve, catheter-related venous thrombosis, tip migration, etc.).1–18
Beyond US, other strategies and technologies, such as the preference for open-ended, non-valved, power injectable, polyurethane catheters, the appropriate choice of the exit site, skin antisepsis with 2% chlorhexidine in 70% isopropyl alcohol, use of maximal sterile barrier precautions, intracavitary ECG for tip positioning, sutureless securement, and cyanoacrylate glue all have increased the safety profile and the cost-effectiveness of the procedure.1–16
An insertion bundle consists of several recommendations, based on scientific evidence, capable of acting synergistically to provide maximal safety, positive clinical outcome, and cost-effectiveness of a given procedure. When placing a PICC, the purpose of an insertion bundle is to minimize any complication directly or indirectly related to the procedure, such as accidental arterial injury, incorrect tip location, arrhythmias, catheter-related venous thrombosis, and catheter-related infections.
An insertion bundle was proposed for PICCs several years ago, 16 and more recently, for Centrally Inserted Central Catheters (CICCs) 19 and Femorally Inserted Central Catheter (FICCs). 20 This paper describes a new updated version of the SIP protocol (Safe Insertion of PICCs), including eight different recommendations designed to guarantee a safer, successful, and more cost-effective procedure (Table 1).
The eight steps of the SIP Protocol.

Preprocedural ultrasound evaluation: The Rapid Peripheral Vein Assessment (RaPeVA)
Pre-procedural evaluation begins with an adequate anamnestic evaluation (e.g. history of previous vascular devices or of difficult venipunctures).
The choice of the vein is of the utmost importance and should be obtained after a rational and objective systematic evaluation of the anatomical characteristics of the vascular system of each patient, using ultrasound.19–22
The Rapid Peripheral Vein Assessment (RaPeVA) protocol is a systematic approach to US evaluation of the veins of the arms and of the cervico-thoracic region before PICC insertion.16,19 RaPeVA consists in several steps that can be performed in a short time and bilaterally. The superficial and deep veins of the forearm and arm are visualized by linear US transducer with a frequency range of 7–12 MHz, suitable for the study of superficial tissues (no deeper than 2–3 cm). The transducer is placed transverse to the axis of the limb and perpendicular to the skin, to obtain an optimal panoramic view of the veins in their relationship to other structures, importantly arteries and nerves. The most appropriate veins for PICCs are often in the following order of preference: the basilic vein (in the bicipital-humeral groove), the brachial veins (inside the neurovascular bundle of the arm), and the axillary vein (in the yellow zone according to the Zone Insertion Method™ (ZIM™). 23 In obese patients, the cephalic vein at the lateral, mid-arm region may also become a practical option since the other veins may be too deep. During RaPeVA, the operator must (a) rule out venous abnormalities, such as thrombosis, stenosis, external compression, anatomical variations of size and shape of the veins, (b) choose a vein with an appropriate diameter to reduce the risk of catheter-related thrombosis, and (c) obtain a full anatomic evaluation for optimal selection of the puncture site and the exit site.11,12,20–22
The seven steps of RaPeVA are performed in the following systematic approach (Table 2):
- Step 1: visualization of the cephalic vein at the antecubital fossa (Figure 1)
- Step 2: sliding of the probe from the radial side to the ulnar side until the artery and brachial veins are identified and the confluence between the antecubital vein and basilic vein is seen (Figure 2)
- Step 3: identification of the basilic vein along the bicipital-humeral groove, sliding the probe upwards (Figure 3)
- Step 4: examination of the nerve-vascular bundle of the arm (Figure 4)
- Step 5: moving laterally over the biceps muscle, visualization of the cephalic vein (Figure 5)
- Step 6: rapid examination of the axillary vein in the infraclavicular area (Figure 6)
- Step 7: visualization of the internal jugular, subclavian, and brachiocephalic vein in the supraclavicular area (Figure 7).
The seven steps of the Rapid Peripheral Vein Assessment (RaPeVA).

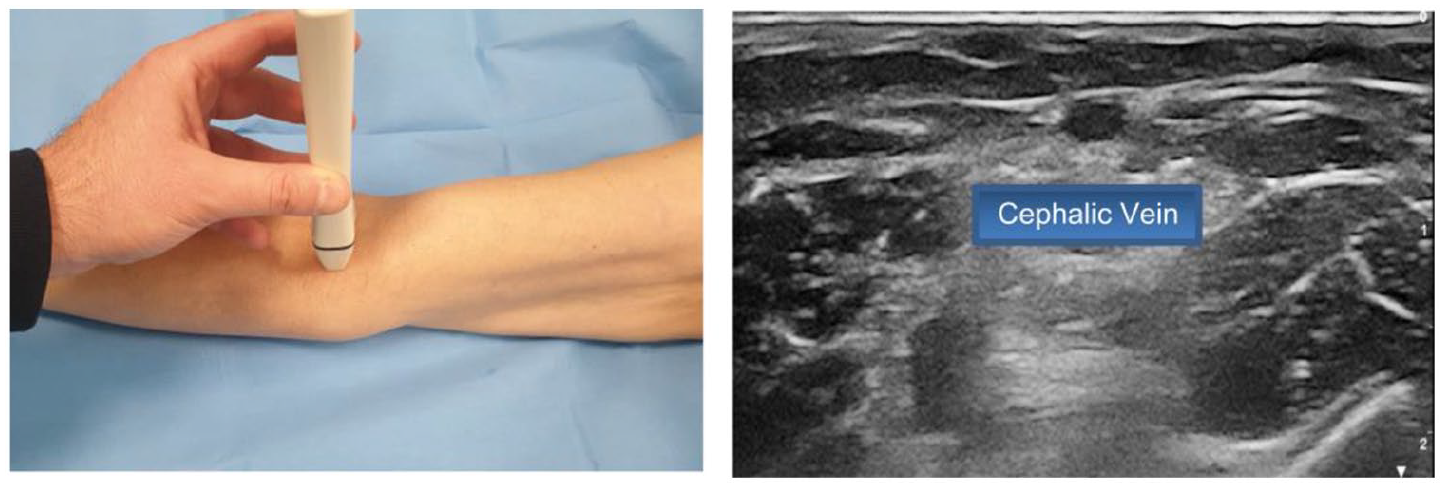
Step 1: Visualization of the cephalic vein at the antecubital fossa.
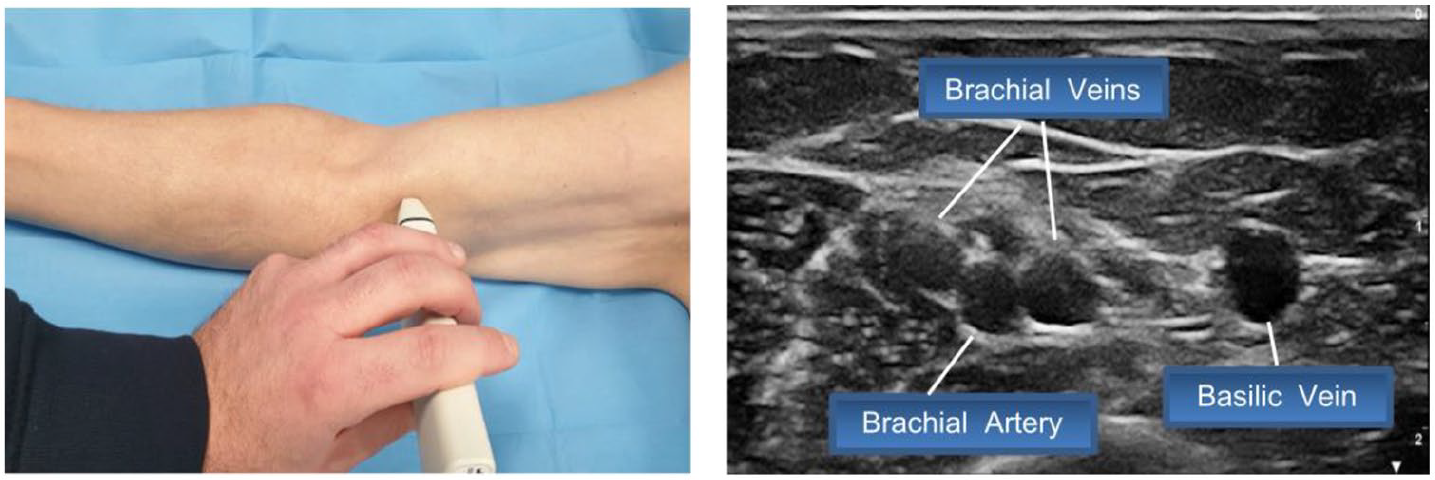
Step 2: Identification of the artery and brachial veins and visualization of the confluence between the antecubital vein and basilic vein.
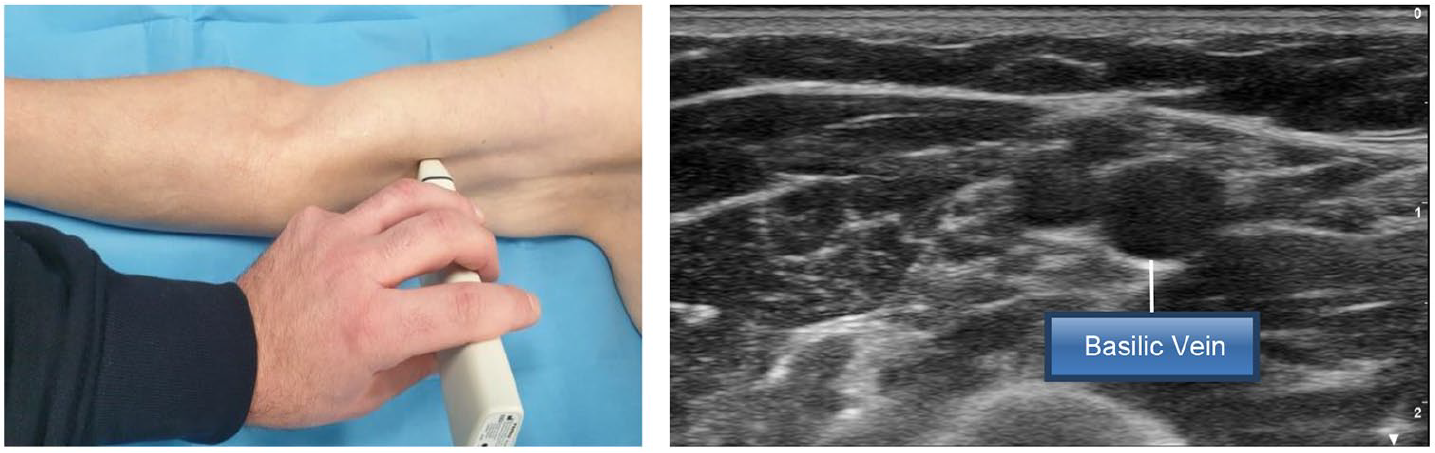
Step 3: Identification of the basilic vein along the bicipital-humeral groove, sliding the probe upwards.
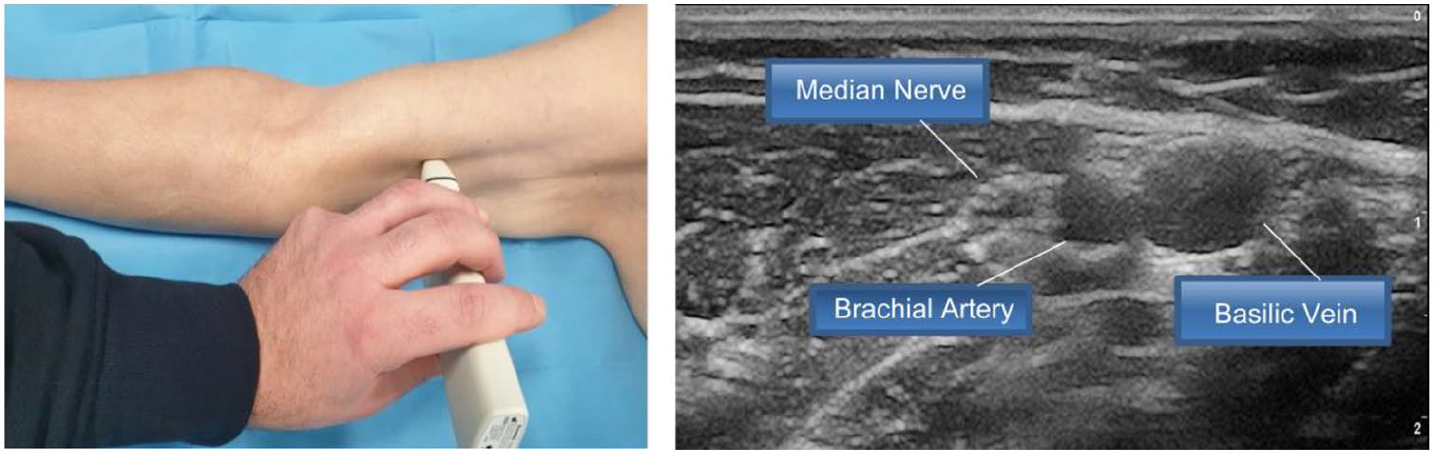
Step 4: Examination of the nerve-vascular bundle of the arm.
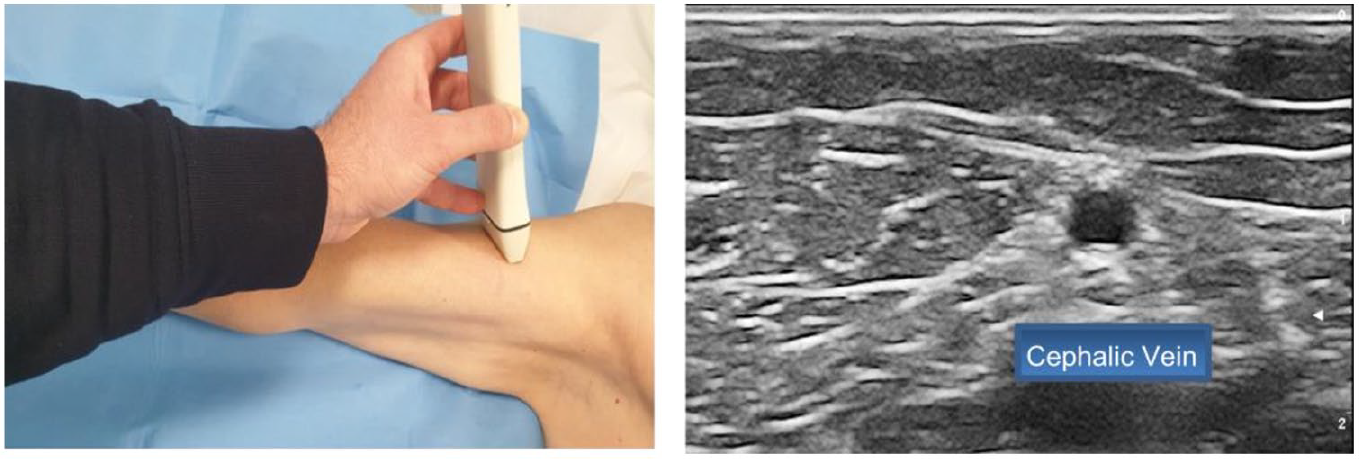
Step 5: Visualization of the cephalic vein, moving laterally over the biceps muscle.
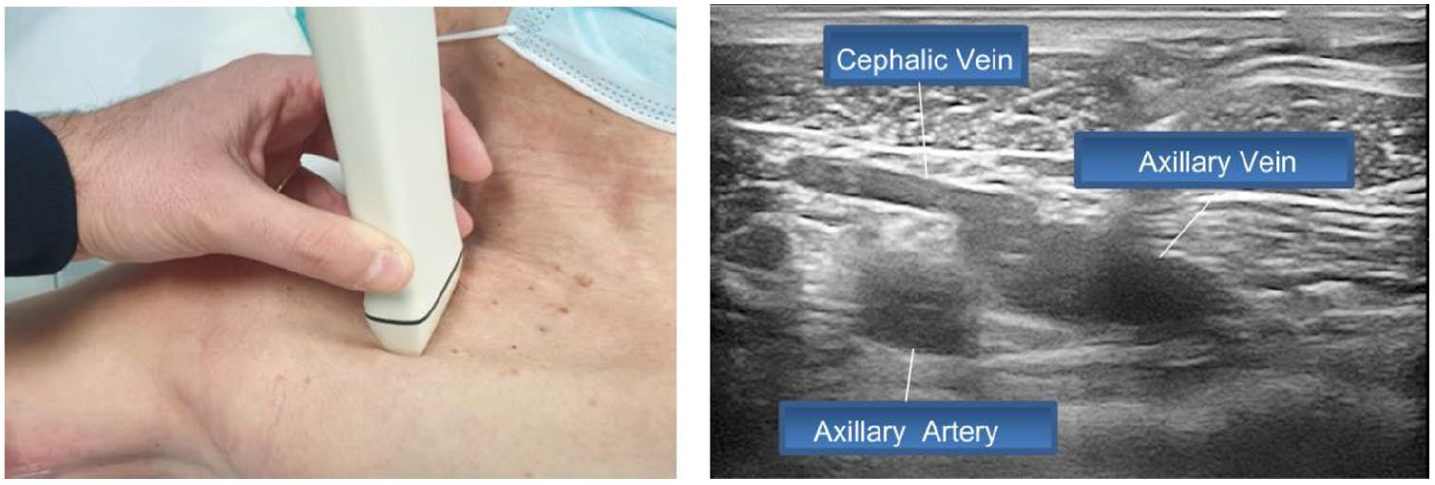
Step 6: Rapid examination of the axillary vein in the infraclavicular area.
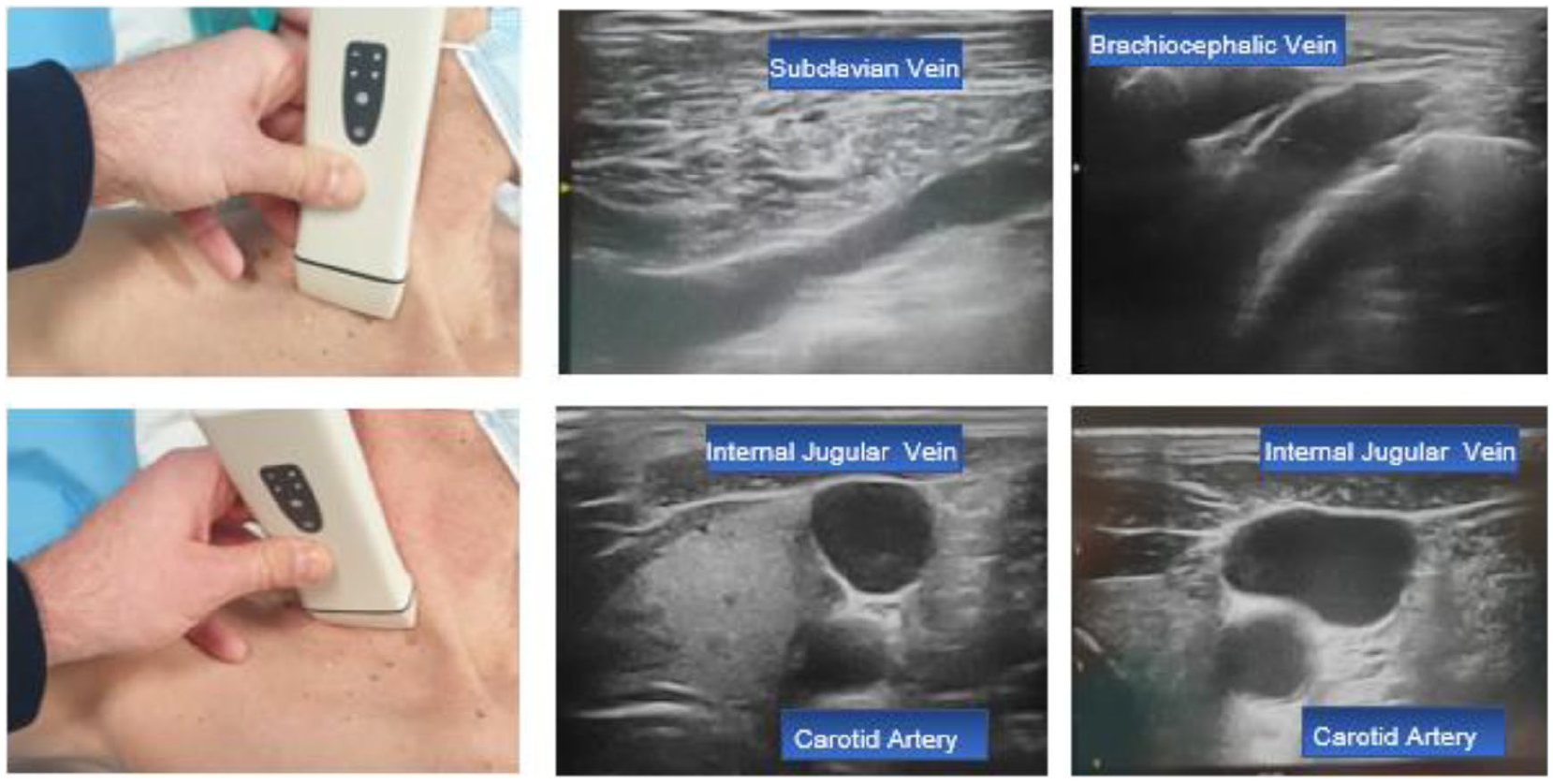
Step 7: Visualization of the subclavian, brachiocephalic, and internal jugular vein in the supraclavicular area.
The RaPeVA protocol ensures that the clinician systematically considers all possible venous options, choosing the best vessel and puncture site, based upon assessment findings. The most appropriate puncture site is frequently assessed in the ZIM™ green zone 23 ; however, if the most appropriate site may be in the ZIM™ yellow zone, tunneling of the catheter should be considered, according to the RAVESTO protocol, establishing an exit site in the green zone. 24
Appropriate aseptic technique
The second step concerns the correct antiseptic technique used during the placement of a PICC. Hand hygiene is preferably performed with hydroalcoholic gel. In special cases, or when the hands are visibly dirty, the hydroalcoholic gel must be preceded by washing with soap and water, according to current international guidelines on infection prevention. For skin antisepsis prior to device insertion, 2% chlorhexidine in 70% isopropyl alcohol should be used. Povidone iodine, in either aqueous or alcohol solution, has a role primarily if there is a known allergy to chlorhexidine. Regarding the application technique of the antiseptic, no clinical difference in microorganism reduction between the concentric circle versus the back-and-forth techniques has been demonstrated when both techniques are used equally on clean and healthy skin. 25
As recommended by all current guidelines, the risk of bacterial contamination must be reduced by adopting maximal barrier precautions, that is cap, facemask, sterile gown and gloves, full-size sterile drape over the patient, plus sterile cover for the US probe (long enough to cover both the probe and the cable when on the sterile field).11,12,25,26
Choice of the appropriate vein and the Zone Insertion Method™ (ZIM™)
The choice of the optimal vein to cannulate is pivotal. An important parameter to consider is the diameter of the vein, evaluated without a tourniquet, and this should be at least three times the diameter of the catheter. The intent is to maintain an ideal catheter-vein ratio (1:3 or less) as to reduce the risk of catheter-related thrombosis. Considering that 1 French (Fr) catheter size corresponds to 0.33 mm, a 3Fr catheter will require a vein diameter of at least 3 mm, a 4Fr catheter will require a diameter of at least 4 mm, etc.9,27,28
The risk of infection or dislodgment of a PICC is often influenced by the choice of the exit site. This SIP protocol suggests utilizing the ZIM™ consistently. 19 This method involves the calculation of the length of the arm (distance between the acromion and the olecranon) and the subsequent division of the arm into three bands, a yellow zone (proximal third), a green zone (middle third), and a red zone (distal third). The red zone is an area with a high risk of catheter dislodgment because of movements of the elbow. For this reason, the red zone is to be avoided as both a venipuncture and catheter exit site. The yellow zone corresponds to the axillary region and is may often be an area with higher bacterial contamination of the skin, due to the proximity of the axilla. 19 To achieve an ideal exit site, venipuncture should always be performed in the green zone; however, in clinical practice, this may not always possible due to smaller caliber veins or unfavorable anatomical relationships (proximity to the brachial artery and/or median nerve). In these cases, it is possible to overcome the problem through tunneling or pseudo-tunneling, according to the RAVESTO protocol. 24 For the tunneling of the catheter, blunt tunnelers should be preferred, since they are associated with minimal risk of localized bleeding, even in patients with coagulation disorders or reduced platelet counts. 29
In short, RaPeVA provides a standardized approach to choosing a peripherally located puncture site, while the ZIM™ optimizes the exit site.
Clear identification of median nerve and brachial artery
Clear ultrasound identification of the median nerve and brachial artery is an important process for clinicians during the preliminary assessment, and even more so during venipuncture. The median nerve is usually located close to the brachial artery (often immediately above it), and appears as a hyper-echogenic, non-compressible structure with an internal “multicore cable” looking structure. Accidental arterial puncture may be associated with local hematomas of varying degrees, but are reversible, while accidental injury to the median nerve may be associated with more serious, even permanent sequelae.11,27 The ultrasound identification of these structures requires adequate ultrasound instrumentation (especially for correct identification of the nerve) and appropriate training.16,19–22
Ultrasound-guided venipuncture
Ultrasound-guided venipuncture is now considered the “gold standard” for performing central venous catheterization. 12 For ultrasound-guided insertion of PICCs, a short axis view of the vein and an out-of-plane venipuncture is considered the preferred choice, due to a panoramic view of the surrounding structures. 30 The procedure should always be performed using the modified Seldinger technique (“catheter through peel-away introducer”). The use of a micro-introducer kit, consisting of 21 Gauge echogenic needle, 0.018″ nitinol-tipped guidewire, and micro-introducer/dilator of correct size and length is strongly recommended, allowing for a minimally invasive and less traumatic approach during the vessel puncture and tissue dilation processes.
Ultrasound tip navigation
After the US-guided venipuncture and insertion of the micro-introducer has been performed, ultrasound may also be used for assessing the correct direction of the catheter toward the ipsilateral brachiocephalic vein and exclude any primary malposition (US-based “tip navigation”), by scanning the veins of the supraclavicular area.16,21,31 This process can be performed with the same linear US transducer used for the venipuncture and has been described in the ECHOTIP protocol.17,18 Tip navigation with ultrasound has proven to be safer, easier, more widely applicable, and less expensive than electromagnetic tip navigation. 32
Intra-procedural assessment of tip location
This important step of the updated SIP bundle incorporates the intraprocedural assessment of the central tip location position. Post-procedural control and manipulation of catheter tip is overtly discouraged by current guidelines11,12 and is considered a waste of time and resource allocation, including any potential harm caused to the patient. One of the most cost-effective and accurate intra-procedural methods for tip location is intracavitary electrocardiogram (IC-ECG). 33 Fluoroscopy, if available, is also an acceptable intra-procedural method, however may be inaccurate, is expensive, is often logistically difficult, and unnecessarily exposes patients to additional levels of ionizing radiation.11,12
The applicability of the IC-ECG method has been extended more recently to patients with atrial fibrillation, 34 however cannot be applied to non-atrial fibrillation patients with an absent P wave (patients with a pacemaker and/or implantable cardioverter-defibrillator (ICD) with underlying rhythm abnormalities). In these cases, another effective, inexpensive, and non-invasive intraprocedural method for tip location is an ultrasound scan using the “bubble test” (a rapid infusion of a few milliliters of “agitated” saline solution that allows for a better visualization of the catheter tip with US).12,35,36 This procedure requires a convex or phased-array US transducer and is performed using either a subcostal or apical view.17,18 However, US-based tip location is not expected to routinely replace IC-ECG, as it is less accurate than IC-ECG in adult patients, it requires specific clinician training, and its applicability/feasibility is not yet 100%.12,17,18,35
Correct securement of the catheter, proper protection of the exit site
Securement by sutures is now discouraged by many current guidelines.11,12,25,26 Suture-based securement of venous access devices has been frequently associated with higher risks of exit site infection, tissue injury and catheter dislodgment, as well as increased risk of accidental needlestick injury. Current preferred options for securement are (a) adhesive-based securement devices, (b) transparent dressings with an integrated securement device, and (c) subcutaneous tissue anchorage. Subcutaneously anchored securement is safer and more effective than skin-adhesive devices, and has been associated with lesser risk of infection, as it allows more complete skin antisepsis around the exit site during care and maintenance.37–40 In patient populations at high risk for catheter dislodgment (non-collaborative patients, diaphoresis, etc.) subcutaneous anchorage should always be preferred.37–39
The decision to optimize the exit site location constitutes the first corrective action that clinicians can provide to improve site protection. In this regard, tunneling is considered a fundamental insertion technique that allows clinicians to obtain an appropriate exit site, even when the most suitable venipuncture site is in the yellow zone. 12 At the time of PICC insertion, the best strategy to avoid local bleeding and extraluminal bacterial contamination is the sealing of the exit site with a cyanoacrylate glue. Glue may also have the additional advantages of reducing micro-movements or pistoning of the catheter at the exit site, often associated with local damage to the endothelium and increasing the risk of intravenous thrombus formation. 41 N-Butyl-2 cyanoacrylate (NBCA) is documented to be faster to solidify and with higher tensile strength than 2-octyl cyanoacrylate (OCA). The use of glue is currently recommended at the time of insertion and first dressing change, as the antibacterial protection of the exit site will also be safeguarded by using a chlorhexidine-impregnated sponge dressing. 42 Repeated weekly application (during dressing changes) of glue on the exit site may be theoretically associated with skin damage. 43 In tunneled PICCs, glue is used not only for sealing the exit site, but also for closing the skin at the venipuncture site. Glue has also proven useful in several patient populations, from neonates to adults,44,45 also offering a potential hemostatic and antimicrobial effect.46–50
A role in reducing exit site bleeding had been attributed to the reverse tapering design of some catheters, clinicians should be aware that reverse tapering does not represent an improvement in the safety and efficacy of these devices; the adoption of inverse conical PICCs should be weighed against their possible disadvantages. 51 The exit site should always be covered with a semi-permeable, transparent membrane dressing, preferably with a Moisture Vapor Transfer Rate (MVTR) of 1500 g/m2/day or more 52 —to ensure protection and breathability of the exit site, additional stabilization of the catheter, and reduction in the risk of skin-related injury. Dressing membranes with MVTR <1500 g/m2/day may be associated with accumulation of fluid and associated skin maceration, particularly in diaphoretic patients.
Appropriate catheter securement and protection of the skin and exit site are key factors for reducing the incidence of dislodgment, infection, venous thrombosis, and associated skin injuries.11,12,19,20
What’s new in this 2022 update of the SIP protocol
Compared to the SIP protocol proposed several years ago, 16 several recommendations were added, with the purpose of further reducing the risks and complications associated with PICC insertion:.
- A more accurate application of the Rapid Peripheral Vein Assessment (RaPeVA) protocol, which should not be limited to a systematic evaluation of the veins of the arms, but extended also to the deep veins of the cervico-thoracic region;
- A closer focus on the appropriate technique of skin antisepsis, to be obtained by 2% chlorhexidine in 70% isopropyl alcohol, emphasizing that no clinical difference in microorganism reduction between the concentric circle versus the back-and-forth techniques has been demonstrated when both techniques are used equally on clean and healthy skin. 25
- The adoption of the RAVESTO protocol, 24 which recommends to tunnel the PICC if the ideal puncture site is in the yellow zone according to the Zone Insertion Method™ (ZIM™) 23
- The preference for the use of blunt tunnelers, since they are associated with minimal risk of localized bleeding, even in patients with coagulation disorders or reduced platelet counts. 29
- The use of ultrasound for tip navigation16,21,31 and for tip location, preferably using the “bubble test,”17,18 considering this so-called ECHOTIP protocol may be very useful in association or as an alternative to the IC-ECG method.
- A wider use of subcutaneous anchorage for catheter securement, since this strategy is safer and more effective than stabilization with skin-adhesive sutureless devices,37–40 and that it should be preferred in patient populations at high risk for catheter dislodgment (non-collaborative patients, diaphoresis, etc.).37–39
- The consistent use of cyanoacrylate for sealing the exit site, so to avoid local bleeding and extraluminal bacterial contamination.
- The consistent use of transparent semipermeable dressings—preferring the membranes with high MVTR—so to ensure optimal protection and breathability of the exit site, additional stabilization of the catheter, and reduction in the risk of skin-related injury.
Conclusions
Early and late complications are most often caused by inadequate clinical decision-making at the time of PICC insertion, with examples such as the avoidance of US-guided venipuncture which may increase the risk of accidental arterial puncture, nerve injury, or patient injury; failure to verify correct location of the tip may increase the risk of catheter-related thrombosis; the choice of a suboptimal exit site may also expose the patient and device to bacterial contamination, increasing the associated infectious risks.1–18,25,26
When placing PICCs, these evidence-based strategies will facilitate the clinician in protecting the patient from insertion-related complications, whether immediate (puncture-related failures, arterial injury, hematoma, nerve injury) or early (arrhythmias, tip malposition, dislodgment) or late (infection, venous thrombosis). These safe, effective, and clinically beneficial strategies are all elements of the updated SIP insertion bundle described in this paper.
The use of a standardized, systematic insertion bundle is clinician friendly, saves time and resources, safeguards patient safety-related issues, and ensures procedural cost effectiveness. A consistent systematic adoption of all eight recommendations of the SIP protocol may improve clinician performance while also providing a useful and evidence-based educational tool when teaching the fundamentals of PICC insertion.
Footnotes
Author’s note
The authors are members of the GAVeCeLT—WoCoVA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
