Abstract
Purpose:
The purpose of this study is to evaluate the safety and efficacy of the mechanical thrombectomy with the Indigo System in the treatment of thrombosed arteriovenous fistulas and grafts.
Methods:
A retrospective search of endovascular procedures performed from November 2018 to June 2020 was conducted. Inclusion criteria were: acute arteriovenous fistula or graft thrombosis that underwent endovascular mechanical thrombectomy with Indigo System. The following information was collected from each case: sex, age, fistula modality, fistula location, treatment modality, and outcomes. Endpoints evaluated were: technical and clinical success rates; primary, assisted primary, and secondary patency rates; complication rates.
Results:
Twenty-six mechanical thrombectomy procedures for declotting of arteriovenous fistula thrombosis, using the Indigo System, were performed in 22 patients. Technical and clinical success was achieved in 23/26 cases (88%). Mean follow-up was 9 months (range 11–539 days). The 6-month primary, primary assisted, and secondary patency rates were 71%, 86%, 93% and the 12-month primary, primary assisted, and secondary patency rates were 71%, 72%, 80%, respectively. No technical or device-related complications were observed during thrombectomy, however two venous ruptures occurred on the angioplasty of the underlying stenosis.
Conclusion:
In conclusion, vacuum-assisted thrombectomy of acutely thrombosed arteriovenous fistulas and grafts with Indigo System is safe and effective, providing good short term patency rates.
Introduction
Vascular access thrombosis is a leading cause of morbidity in end stage renal disease (ESRD) patients that undergo haemodialysis, accounting for 65%–85% of access losses.1–3 In these cases of acute thrombosis, a timely intervention to attempt access salvage is recommended in order to achieve better patency rates. 1
Several techniques and devices have been described for the recanalization of acutely thrombosed arteriovenous fistulas and grafts (AVFGs), including surgery and mechanical or pharmacomechanical thrombectomy. 4
Among the devices used for mechanical thrombectomy is the Indigo System (Penumbra, Alameda, California), a vacuum-assisted device with continuous aspiration, originally developed for use in stroke and further developed for peripheral interventions, including venous thrombectomy. So far there are only a few studies describing the device use in thrombosed AVFGs, with a limited number of patients.5,6
The purpose of this study is to evaluate the safety and efficacy of the mechanical thrombectomy with the Indigo System in the treatment of thrombosed AVFGs.
Methods
Study design
A retrospective search of endovascular procedures performed from November 2018 to June 2020 was conducted. This study was reviewed and approved by the institutional review board. The requirement for patient informed consent was waived.
Inclusion criteria were: acute arteriovenous fistula or graft thrombosis that underwent endovascular mechanical thrombectomy with Indigo System (Penumbra, Alameda, California).
The following information was collected from each case: sex, age, fistula modality (i.e. graft or native vein fistula), fistula location, treatment modality, and outcomes.
Endpoints evaluated were: technical and clinical success rates; primary, assisted primary, and secondary patency rates; complication rates.
Definitions
Definitions for success and patency were based on the Quality Improvement Guidelines for Percutaneous Image-Guided Management of the Thrombosed or Dysfunctional Dialysis Circuit as follows 7 :
Anatomic success: restoration of flow combined with a <30% residual diameter stenosis for any significant underlying stenosis.
Clinical success: resumption of normal haemodialysis for a minimum of at least one session following percutaneous intervention.
Post intervention primary patency: the interval of uninterrupted patency after endovascular intervention within a dialysis circuit to thrombosis or repeated endovascular intervention.
Post intervention assisted primary patency: the interval of uninterrupted patency after an endovascular intervention until dialysis circuit thrombosis or a surgical intervention of the access circuit.
Post intervention secondary patency: the interval of patency after and endovascular intervention until the dialysis circuit is surgically declotted, revised, or abandoned.
Procedure
Arteriovenous fistula thrombosis was diagnosed by the attending dialysis nephrologist due to absence of thrill on the fistula or failed dialysis. Patients then underwent duplex ultrasound to confirm the presence of thrombosis or stenosis.
Intervention was performed in one of two different angiographic units (Siemens AG, Forchheim, Germany; Philips Medical Systems, Best, The Netherlands). Patients underwent general anesthesia or local anesthesia with 2% Lidocaine and sedation at the anesthesiologist discretion. Access site was the venous side of the fistula: when thrombosis occurred on a limited segment of the vein, a single ultrasound guided puncture of the vein in an area without thrombus was performed. When there was extensive thrombosis occurring in the entire venous segment, double puncture was done. After angiography, systemic heparinization with 5000 UI of unfractionated heparin was initiated and mechanical thrombectomy was performed with the Indigo System (Penumbra, Alameda, California). Following thrombectomy, angioplasty of the underlying stenosis was performed with conventional balloon, high-pressure balloon or covered stent at the physician discretion. After declotting, patients were admitted to the nephrology unit for dialysis within 24 h. Clinical follow-up was performed during dialysis sessions (Figure 1).

Patient with acute thrombosis of a brachio-cephalic fistula on the left arm. (a) Ultrasound guided retrograde puncture of cephalic vein. (b) Angiography depicting thrombosis of the cephalic vein. (c) Angiography after vacuum-assisted mechanical thrombectomy of the cephalic vein depicting partial recanalization. (d) Angiography after additional vacuum-assisted mechanical thrombectomy of the cephalic vein depicting recanalization of the cephalic vein with underlying perianastomotic stenosis. (e) Angiography after angioplasty of the perianastomotic stenosis with a high pressure balloon depicting venous patency without residual stenosis. (f) Angiography of the entire cephalic vein depicting venous patency without residual thrombosis or stenosis.
Results
From November 2018 to June 2020, 26 mechanical thrombectomy procedures for declotting of arteriovenous fistula thrombosis, using the Indigo System, were performed in 22 patients (Table 1). Of these patients, 14 were male (63%) and 8 were female (36%), with an average age of 68 years (range 31–81). All procedures were performed within 24 h of diagnosis. Mechanical thrombectomy was performed with Indigo catheter 8F in 25 cases and Indigo catheter 6F in 1 case.
Patient demographics.
Technical and clinical success were achieved in 23/26 cases (88%). Procedures were entirely endovascular in 25 cases and hybrid in one case of a patient with brachiocephalic fistula presenting with aneurysmal dilatation next to the arteriovenous anastomosis and an extensive amount of thrombus in cephalic vein. Declotting of the cephalic vein was performed and a new proximal anastomosis was made.
An underlying stenosis was observed in all patients except one (25/26 cases, 96%), and these patients underwent percutaneous transluminal angioplasty. Covered stents were used in five cases (19%), three of them due to residual stenosis and two of them due to venous rupture after angioplasty. In two of the residual stenosis cases, the covered stent was placed at the venous anastomosis of a PTFE graft, and in the third case it was placed at the cephalic arch of a native brachio-cephalic fistula. In five patients, a low dose (5–10 mg) of recombinant tissue plasminogen activator (rtPA) was used during the procedure. No technical or device-related complications were observed during thrombectomy, however two venous ruptures occurred on the angioplasty of the underlying stenosis.
In only one patient, the first thrombectomy procedure was unsuccessful, therefore mean follow-up of the remaining 21 patients was calculated in 9 months (range 11–539 days). Reinterventions were performed in six patients, (two angioplasties and four thrombectomies). At the end of follow-up, one patient had died, 18/21 fistulas were still patent and two had occluded after a mean time of 7.2 months (events at 83 and 350 days). The 6-month primary, primary assisted and secondary patency rates were 71%, 86%, 93% and the 12-month primary, primary assisted, and secondary patency rates were 71%, 72%, 80%, respectively (Figure 2).
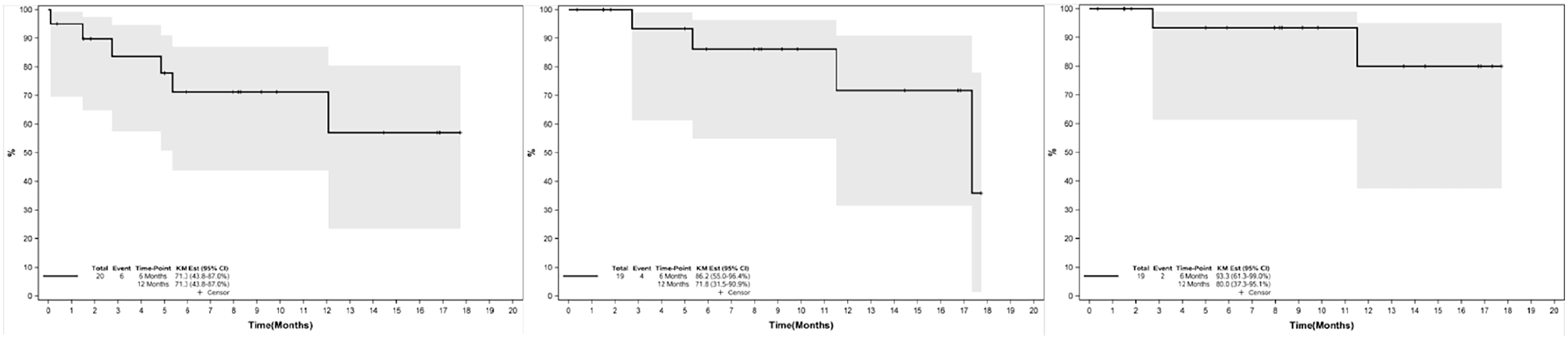
Primary, assisted primary and secondary patency rates for patients with acute thrombosis of an arteriovenous fistula or graft that underwent vacuum-assisted mechanical thrombectomy with Indigo System.
Discussion
This study is one of the largest series on the use of the Indigo System for the thrombectomy of arteriovenous fistulas and grafts. Anatomic and clinical success rates were 88%. In the largest study with the device, a multicenter European cooperation that included 35 patients, the authors observed technical and clinical success rates of 97% and 91%, respectively. 5 In a recently published Italian study describing vacuum-assisted thrombectomy on long segments of thrombosed arteriovenous grafts in 10 patients, the authors found a clinical success rate of 80%. 6
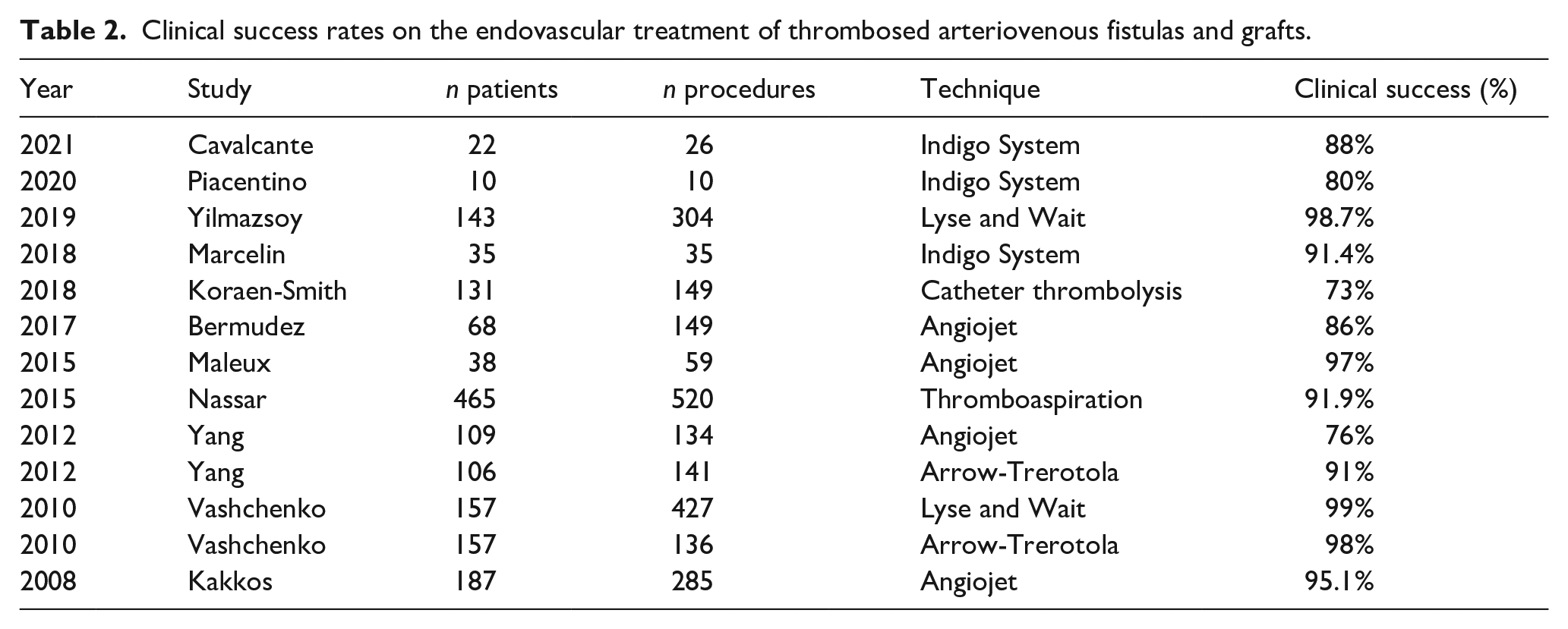
Several different techniques and devices have been used for thrombectomy of AVFGs. Recent studies have demonstrated excellent technical and clinical success rates, around or above 90% in most series (Table 2).5,6,8–15
Clinical success rates on the endovascular treatment of thrombosed arteriovenous fistulas and grafts.
In our study, 6- and 12-month primary patency rates were 71%. In literature, these rates range from 40% to 78% in 6 months and from 23 to 66% in 12 months.5,9–14 In the two previous series with the device, 6-month primary patency rates observed were 71% in the European multicenter study and a low rate of 37.5% in the Italian study, which may be explained by the fact that long thrombosis on grafts were treated (mean 30 cm).5,6
Primary assisted and secondary patency rates were even better in our series, emphasizing the benefit of intervening on a dysfunctional or thrombosed fistula. Our 6- and 12-month assisted primary rates were 86% and 72% and secondary rates were 93% and 80%, respectively. In other studies, 12-month primary assisted and secondary rates ranged from 30% to 70% and 57% to 87%, respectively.5,9–12,14,15 In the other studies with the device, 6-month primary assisted and secondary rates were 80% and 88.5% at the European multicenter study and 50 and 62.5% respectively.5,6
Among the other devices available for AVFGs thrombectomy, the largest series in literature describe the use of the Angiojet (Boston Scientific, USA) and the Arrow-Trerotola (Teleflex, USA). Other devices reported in this scenario are the Cleaner XT (Argon Medical Devices, USA), the Aspirex (Straub Medical, Switzerland), and the ClariVein (Merit Medical, USA).16–18
The Angiojet is a rheolytic thrombectomy device, in which high-speed jets of saline are injected in a retrograde direction through the catheter, causing a negative pressure (Venturi effect), responsible for thrombus fragmentation and aspiration.9,12 Recently Chan and Goh published a systematic review on the use of the Angiojet in thrombosed AVFGs, which included 836 patients (431 grafts and 234 autogenous fistulas). The average rates of technical and clinical success were 89.1% and 86.7%, while the average rates of primary patency at 6 and 12 months were 42.5% and 30.5%. At 6 and 12 months, assisted primary rates were 75.1% and 74.5% and secondary patency rates were 47.3% and 35.3%, respectively. 4
The Arrow-Trerotola, on the other hand, promotes thrombectomy through a rotational mechanism, causing maceration and fragmentation of the thrombus.8,9 In a study comparing the two devices, clinical success rates were higher with Arrow-Trerotola than Angiojet (91 vs 76%, p=0.002) and it was also associated with a shorter procedure duration (56 vs 88 min, p < 0.001). Post-intervention 6- and 12-month primary patency rates, however, were not different between the devices: 43% and 29% with Arrow-Trerotola and 45% and 12% with Angiojet, respectively. 9
Endovascular techniques for the treatment of thrombosed AVFGs are not exempt from complications, and hematoma, early rethrombosis, vascular rupture, arterial embolism, among others, may occur.5,9,11,12,14 In a recent systematic review of the literature and meta-analysis, including 3000 cases of endovascular treatment of fistula thrombosis, the authors described 4.6% of minor and 0.6% of major complications. 19 In our study, complications occurred in two patients, all of them vascular ruptures during angioplasty of the underlying stenosis and were treated immediately with covered stents. In the other series published with the device, Marcelin et al. 5 described one major complication, a vein perforation, which was treated surgically and Piacentino et al. 6 observed two complications, one hand ischemia due to distal arterial embolization, treated surgically and a bleeding at the access site, treated with suture.
The main limitation of this study is its retrospective nature, the small number of patients and its limited follow-up. Nevertheless, it is important to depict the results of a not much studied device and the value of mechanical thrombectomy in the salvage of AVFGs. Further studies with a larger number of patients and a longer follow up are necessary, as well as studies comparing different devices.
In conclusion, vacuum-assisted thrombectomy of acutely thrombosed AVFGs with the Indigo System is safe and effective to remove sufficient volume of acute thrombus to restore flow, usually without the need for additional pharmacological thrombolytics and provides good short term patency rates.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical standards
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
