Abstract
Background:
Tunneled central venous catheters (tCVC) exchange may be difficult in some situations. We retrospectively report our preliminary experience of a novel extra-catheter guide wire technique for exchange of dysfunctional tCVC.
Materials and methods:
Retrospectively analyze the demographics, treatment details and outcomes data of 39 patients received tCVC exchange from January 2018 to January 2019. According to whether guide wire could pass through the catheter lumen, patients were divided into in-catheter group and extra-catheter group. The technical successful rate, peri-operative complications, 1-month and 6-month catheter flow rate was recorded and compared between the 2 groups.
Results:
The final study population consists of 39 patients, including 16 in-catheter group and 23 extra-catheter group. The technical successful rate was 100% in both groups. All patients achieved restoration of line patency and completed at least 1 hemodialysis section. The 1-month (267.69 ± 20.12 vs. 274.13 ± 17.69, p = 0.604) and 6-month (255.81 ± 12.93 vs. 256.97 ± 11.20, p = 0.403) catheter flow rate was comparable between the 2 groups.
Conclusion:
The novel extra-catheter guide wire technique was helpful for in situ exchange of dysfunctional tunneled central venous hemodialysis catheters, especially when the guide wire could not pass through the catheter lumen.
Introduction
There were over 70,000 cases of end-stage renal disease (ESRD) in the United States in 2015 1 and the overall prevalence of chronic kidney disease in China had reached 10.8% in 2012. 2 Though arteriovenous access (arteriovenous fistula or arteriovenous graft) was recommended as the first choice for hemodialysis, tunnelled central venous catheters (tCVC) were preferred in some circumstances for long-term or indefinite duration. 3 It is reported that the proportion of tCVC used as long-term hemodialysis access was still high. 4
Patients with tCVC may suffer from catheter dysfunction. Fibrin sheath formation was the most common reason for long-term catheter dysfunction.5,6 The recommended treatment strategy for dysfunctional tCVC obstructed by fibrin sheath was catheter exchange with sheath disruption. 3 Normally, guide wire was used to pass through the catheter lumen, then the subsequent fibrin sheath disruption and catheter exchange could be carried out. However, it is not always possible for a guidewire to be passed through the catheter lumen when the lumen is occluded by thrombus.
We have tried a novel extra-catheter guide wire technique for in situ exchange of dysfunctional tCVC since 2018. The aim of this study is to retrospectively report our preliminary experience of the novel technique.
Materials and methods
Study population
From January 2018 to January 2019, 87 consecutive patients with dysfunctional tCVC were admitted to the Cardiovascular Surgery Department of China–Japan Friendship Hospital. Among these patients, 39 underwent in situ tCVC exchange. According to whether the guide wire could pass through the catheter lumen, patients were divided into 2 groups: in-catheter group and extra-catheter group. We collected and analyzed the demographics, treatment details and outcomes data of the 2 groups. The protocol was approved by the ethics committee of China–Japan Friendship Hospital and written informed consent was signed by all patients. All data was retrospectively collected.
Inclusion and exclusion criteria
Inclusion criteria were the following: (1) patients with ESRD who rely on tCVC placed in the internal jugular vein for hemodialysis; (2) catheter dysfunction was defined as inadequate to complete a hemodialysis section and unresponsive to patient repositioning, saline flushes, or lumen reversal; (3) patients who were planned to receive in situ catheter exchange; (4) patients with angiographically-demonstrated fibrin sheath formation.
Exclusion criteria included: (1) patients with primary catheter dysfunction, defined as dysfunction within 1 week after catheter insertion; (2) patients with peri-catheter infection not suitable for in situ catheter exchange; (3) in situ catheter exchange failed because of central vein occlusion or other reasons.
Catheter exchange process
The tCVC exchange process was performed under local anesthesia with patients in the supine position. We made a small incision around the jugular vein puncture site to isolate the catheter. Then pulled the catheter out from the jugular vein partially. We firstly punctured into the catheter lumen with a puncture needle (Cook, USA), then advanced the 0.035 inch guide wire (Radifocus, Terumo, Japan) through the needle into the catheter lumen. If the guide wire could pass through the catheter lumen smoothly, we used the guide wire for the next fibrin sheath disruption and catheter exchange process (these patients belong to in-catheter group). If the guide wire could not pass through the catheter lumen (these patients belong to extra-catheter group), we punctured through the catheter wall and punctured back. Next we introduced the guide wire through the needle, thus the guide wire was present as ‘U’ shaped through the catheter wall. Then we pushed the catheter back to the jugular vein and pulled the guide wire back slowly under the guidance of fluoroscopy. The guide wire tip then ‘jumped’ toward the front direction once back from the second puncture tunnel. We advanced the guide wire to the inferior vena cava to establish the working pathway and removed the dysfunctional catheter. A 10 French sheath (Avanti, Cordis, USA) was introduced through the guide wire and angiography was performed to illustrate the fibrin sheath. Then we advanced a noncompliant balloon (Advance, Cook, USA) to disrupt the fibrin sheath. At last, we exchanged the new tCVC (Palindrome, Covidien, USA), with location confirmed and flow rate checked.
Endpoints and follow-up
The primary endpoint was technical successful rate, restoration of line patency defined as flow rate ⩾300 mL/min, or adequate to complete a hemodialysis section. Peri-operative complications, 1-month and 6-month catheter flow rate was recorded and compared.
Statistical analysis
Mean ± SD was used for presentation of continuous variables and percentages for discrete variables. A two-sided independent sample t test was used for comparison of continuous variables. For discrete variables comparison, chi-square test or Fisher’s exact test was performed. The demographics, peri-operative variables and endpoints were compared between the 2 groups. SPSS version 23 (SPSS Inc., Chicago, IL, USA) was used for data analysis. A p value of <0.05 was considered statistically significant.
Results
Demographics and clinical features
The final study population consisted of 39 patients (14 men, mean age 67.05 ± 9.88 years old, Figure 1), of which 16 received in-catheter guide wire exchange, 23 received extra-catheter guide wire technique for in situ catheter exchange. The demographics and clinical features of the patients were comparable between the 2 groups (Table 1).
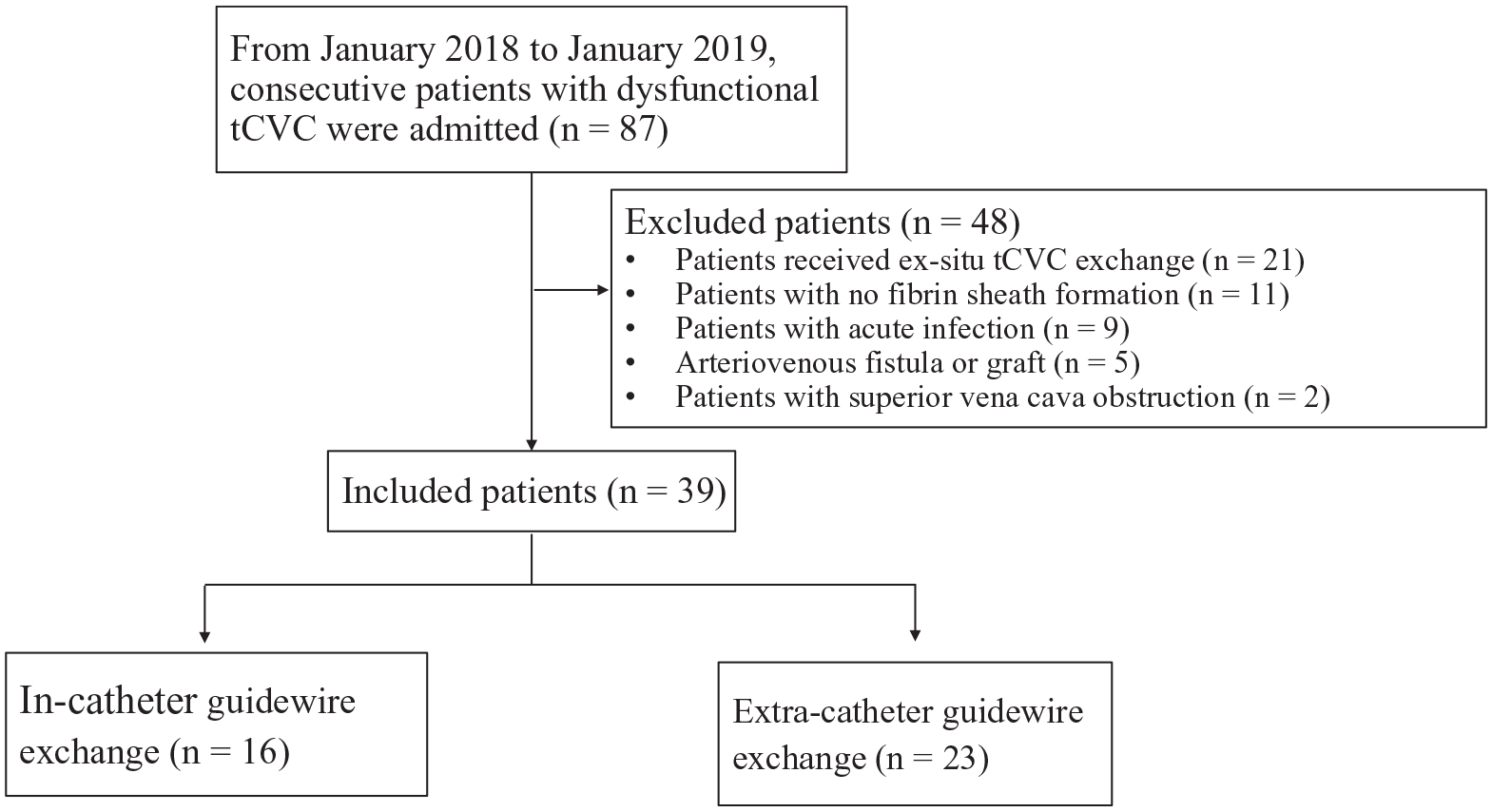
Flow chart for patient selection process.
Demographics and clinical features.
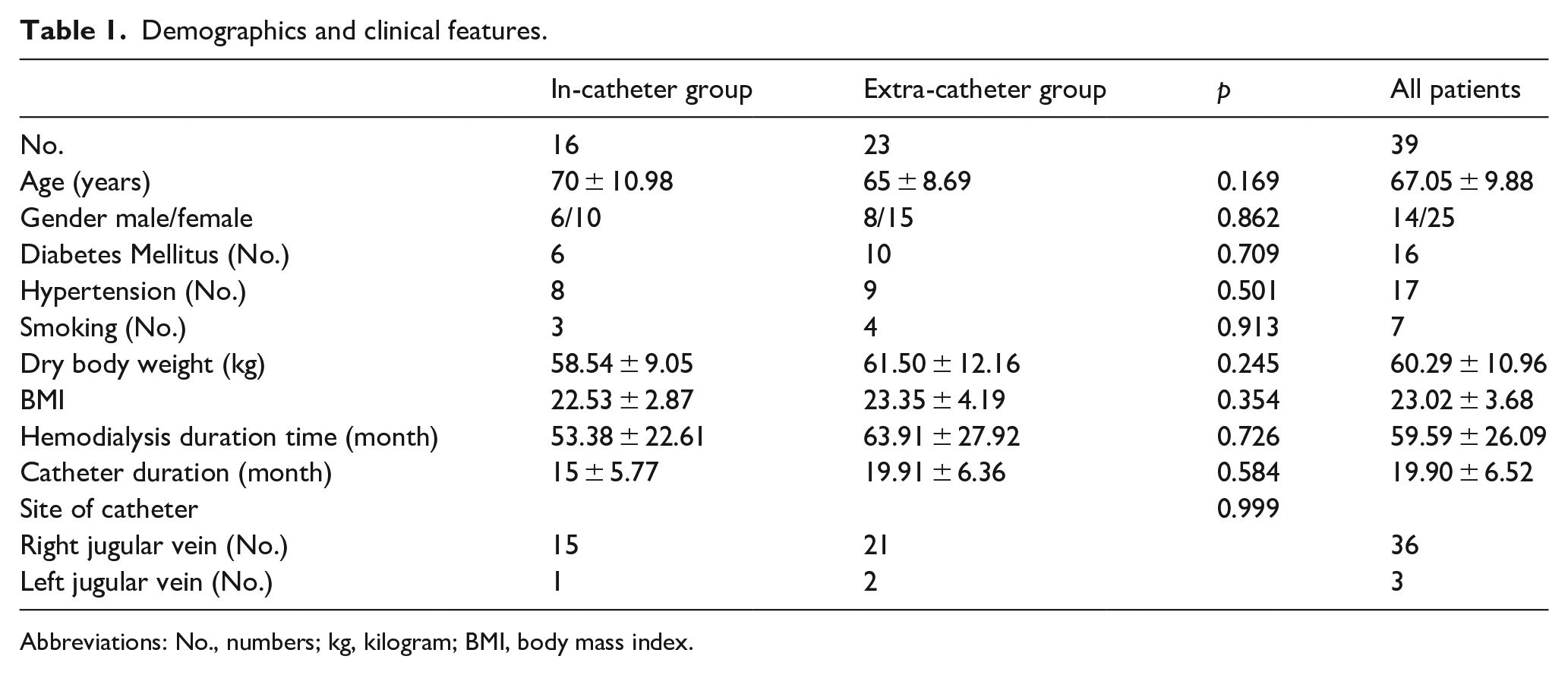
Abbreviations: No., numbers; kg, kilogram; BMI, body mass index.
Intra-operative variable
The typical process of extra-catheter guide wire technique for in situ catheter exchange was shown in Figure 2. Most of the tCVCs were located at the right jugular vein, except for one in in-catheter group and two in extra-catheter group were located at the left jugular vein. In general, we use 36 cm tCVCs for right jugular vein and 40 cm for left jugular vein. The diameter of balloon used to disrupt fibrin sheaths was 14 mm. The mean blood loss was 12.53 ± 2.32 mL in in-catheter group and 14.24 ± 3.21 mL in extra-catheter group. Most of the hemorrhage was from the previous tunnel between the jugular vein puncture site and the subclavian incision.
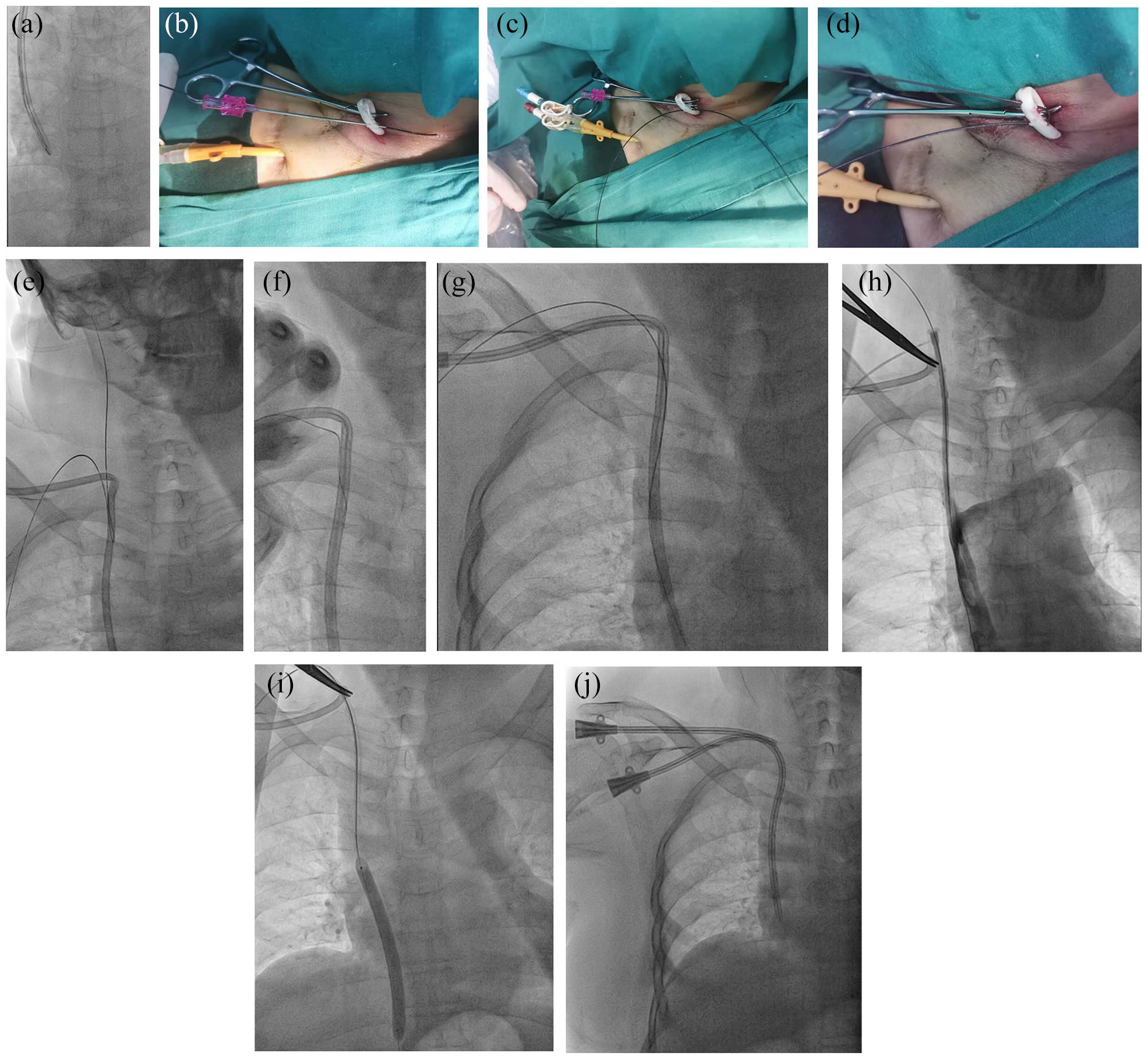
A typical case of extra-catheter guidewire for in situ exchange dysfunctional tCVC.
Primary endpoint
The technical success rate was 100% in both groups. For one patient in the extra-catheter group, a 40 cm catheter was too short for left jugular vein access, we exchanged another 43 cm catheter (Figure 3). All patients achieved restoration of line patency and completed at least one hemodialysis session.
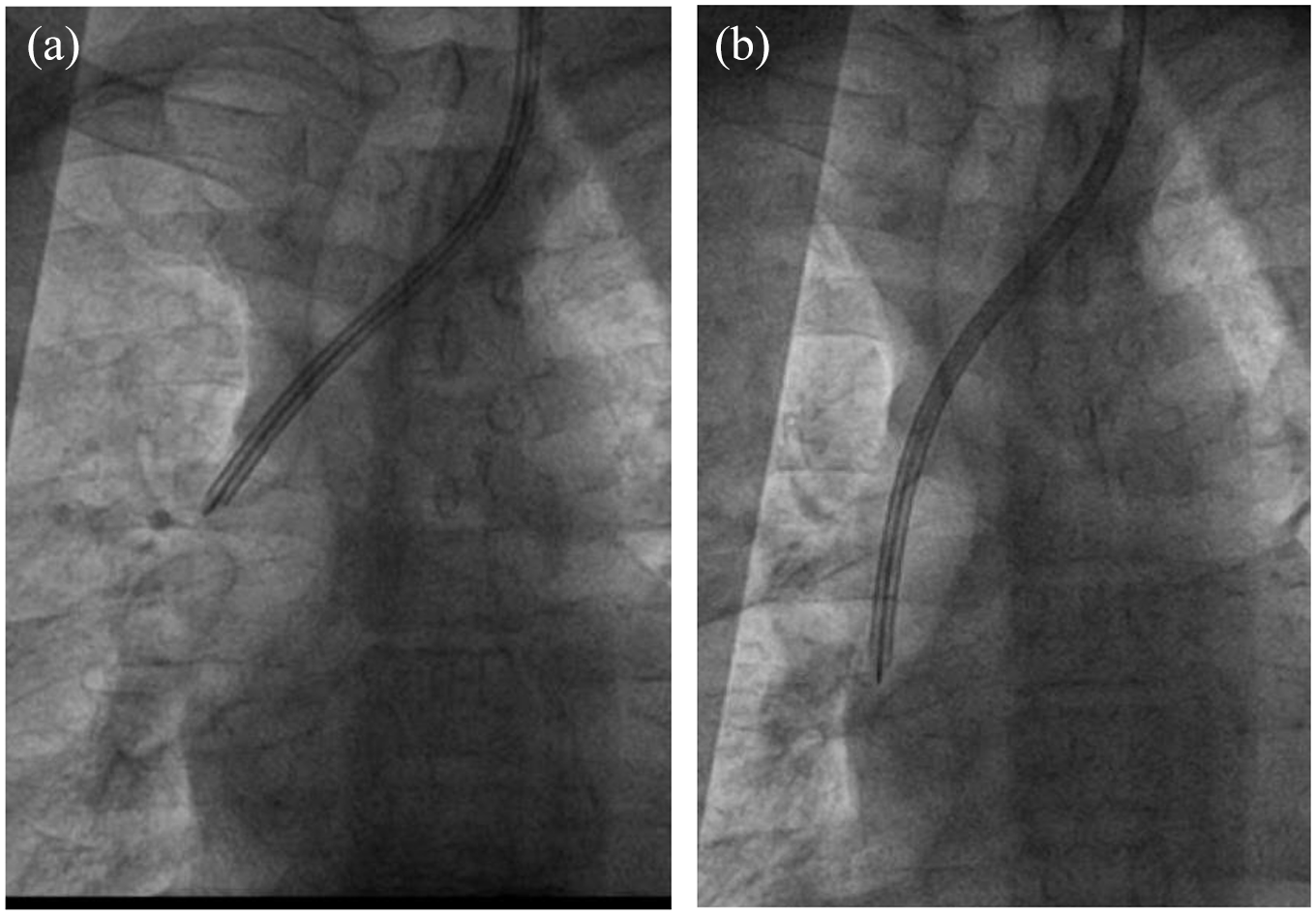
For 1 patient in extra-catheter group, a 40 cm catheter was too short for left jugular vein, we changed to another 43 cm catheter. (a) the 40 cm catheter was too short. (b) the 43 cm catheter was suitable.
Peri-operative complications
One patient in the in-catheter group and two in the extra-catheter group suffered from hematoma in the previous tunnel, all alleviated after incisional drainage. There were two documented catheter related infection and one catheter malfunction in the in-catheter group within one month. In the extra-catheter group, one catheter related infection and one catheter malfunction was documented within one month. All catheter related infection were alleviated with antibiotic treatment. For catheter malfunction, catheter function was restored with one mg/mL alteplase intraluminal dwell.
Follow-up results
The one-month and six-month catheter flow rate was comparable between the two groups (Table 2). Two patients in the in-catheter group and one in the extra-catheter group received catheter exchange because of thrombolytics-resisted catheter malfunction within 6 months.
1-month and 6-month catheter blood flow rate in the 2 groups.

Abbreviations: mL, millilitre; min, minute.
Discussion
Multiple interventions for treating central venous hemodialysis catheter malfunction were reported, 7 including thrombolytic therapy, fibrin sheath stripping, catheter exchange with or without sheath disruption. If catheter exchange in necessary, in situ exchange is preferred because the central vein resource is limited. When all the interventions failed to restore the catheter patency, de novo catheter placement is necessary.
After tCVCs insertion, local hemodynamic change and vessel wall shear stress fluctuation may stimulate fibrin sheath formation and intimal thickening. 8 The presence of fibrin sheath increases the risk of catheter malfunction and infection. 6 Different manoeuvres including stripping, internal snare and over-the-wire catheter exchange with or without balloon sheath disruption could be carried out to deal with fibrin sheaths. 9 In situ catheter exchange with sheath disruption was recommended by KQOQI. 10 It could achieve durable catheter patency, 11 without increasing bacteremia rates. 12 Li et al. 13 reported that catheter exchange through fibrin sheath crevice is an effective alternative strategy for in situ catheter exchange. This technique could achieve better 6-month maximum blood flow. All these catheter exchange methods were based on ‘guide wire first’ concept. However, sometimes a guide wire could not pass through the catheter lumen or side hole. In these situations, de novo catheter placement presents as the last choice. Our novel extra-catheter guide wire technique offers another option for these patients, which could increase technical successful rate for in situ catheter exchange and preserve more central venous resource. For patients that guide wire could pass through smoothly, this technique can also be used to increase the likelihood of passing the guide wire as the longer distance increases the chance that the wire will cross the fibrin sheath and this may result in better patency.
There are some key points that need attention. Firstly, we prefer that the first puncture tunnel parallel to the long axis of the catheter and the length in the catheter should be short. This makes it easy to advance the guide wire. Secondly, the second puncture tunnel should be a little further proximally to the first one to prevent complete withdrawal of guide wire. Thirdly, before pull the catheter back to the jugular vein, we should stretch the guide wire tight to make the ‘U’ shape more smoothly to prevent injuring surrounding tissues.
‘Stuck catheter’ refers to the condition in which a catheter is not removable from a central vein using standard methods. 14 For those patients, our technique may be useful to establish a guide wire pathway. A preliminary guide wire pathway is helpful in case of central vein rupture and stent graft implantation when removing the stuck catheter.
Catheter related right atrial thrombosis (CRAT) is an under-recognized complication of tCVC, which also could lead to catheter malfunction 15 and was reported to be associated with low tCVC tip. Higher incidence of pulmonary embolism and infections occurred in patients with CART. Our technique maybe not suitable for these patients. More studies should be carried out to explore the proper treatment strategy for patients with CART.
There are certain several limitations in our study. Firstly, this was an exploratory study with a relatively small sample size. Secondly, we use needle to penetrate the dysfunctional tCVC for 2 or more times. This potentially could increase the possibility of catheter fracture.
Conclusion
In conclusion, the novel extra-catheter guide wire technique was helpful for in situ exchange of dysfunctional tunnelled central venous hemodialysis catheters, especially in situations where the guide wire could not pass through the catheter lumen.
Footnotes
Acknowledgements
None.
Author Contributions
Zhang JB, Nie QQ, and Xu XJ contributed equally to this work; Zhang JB, Nie QQ, Fan XQ, and Ye ZD designed the research study; Zhang JB, Nie QQ, Xu XJ and Fan XQ performed the research; Zhang JB, He B, Wang XM, Wang F and Liu P analyzed the data and wrote the manuscript. All authors have read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Elite Medical Professionals project of China-Japan Friendship Hospital (NO. ZRJY2021-QM13).
