Abstract
Introduction:
Although several guidelines recommend that prolonged administration of vancomycin should be preferably carried out by a central venous access, vancomycin is often given peripherally. Whether such risk may be affected by different modalities of administration in terms of dilution and time of infusion, it is a matter of controversy.
Methods:
This single-center prospective study enrolled all consecutive patients requiring prolonged intravenous infusion of vancomycin (1 g/day) using long peripheral cannula “mini-midline.” Patients were randomized in study group (4 mg/ml) and control group (20 mg/ml). All patients were systematically evaluated every 24 h by the Visual Exit-Site Score and a daily ultrasound scan of the veins of the arm.
Results:
The daily ultrasound evaluation showed venous thrombosis at the distal tip of the cannula in all patients, in both groups. After this finding in the first 14 patients, the study was interrupted. All thromboses were completely asymptomatic and occurred in absence of any sign of catheter malfunction. The onset of thrombosis was significantly earlier in the control group (ranging from 24 to 48 h) than in the study group (ranging from 48 to 96 h), with an average of 30 ± 11 versus 68 ± 16 h (p < 0.001).
Conclusion:
Continuous intravenous infusion of vancomycin should be preferably delivered by a central venous access, as largely recommended by current guidelines, since peripheral infusion is inevitably associated with venous thrombosis, independently from the type of peripheral venous access device adopted (short peripheral cannula vs long peripheral cannula) and from the extent of dilution.
Introduction
Vancomycin is a glycopeptide antibiotic widely used in clinical practice, for treatment of severe local or systemic infections due to multi-resistant Gram-positive bacteria such as MRSA (methicillin-resistant Staphylococcus Aureus) or for treatment of infections by Gram-positive bacteria in patients with known allergy to penicillin and cephalosporin.
When infused in a peripheral vein, vancomycin is known to be associated with a relevant risk of chemical phlebitis (i.e. thrombophlebitis), due to its irritant effect on the endothelium.1,2
Although several guidelines recommend that prolonged administration of vancomycin should be preferably carried out by a central venous access,2,3 vancomycin is often given peripherally. Whether such risk may be affected by different modalities of administration in terms of dilution (concentration) and time of infusion, it is a matter of controversy.
The toxicity of vancomycin at different concentration has been studied in vitro on human umbilical vein endothelial cells 1 : this experimental study showed a significant increase of cell death when vancomycin concentration was >2.5 mg/ml, and that local toxicity was also time-dependent (significantly higher cell death after 48–72 h of treatment than after 24 h). At 5 mg/ml, cell death was close to 100%, and there was no difference between 48 versus 72 h of contact. Also, vancomycin toxicity did not seem to be caused exclusively by its low pH.4,5 Furthermore, using a fixed daily dose of vancomycin (>1 g/day), continuous infusion induced more cell toxicity than intermittent infusion; vancomycin toxicity was less dose-dependent with intermittent infusion than with continuous infusion. 1
Administration by central venous access neutralizes the potential toxicity of vancomycin on the endothelium since the drug is immediately diluted in the blood. For this reason, current guidelines recommend administering vancomycin via a central venous catheter, in particular if the planned concentration is >5 mg/ml, and/or if given by continuous infusion,6–9 and/or if there is history of thrombosis, hypercoagulability, decreased venous flow, or end-stage renal disease 10 requiring preservation of the veins of the arm.8,11,12 In one clinical study, 13 continuous infusion of vancomycin had a higher safety profile and a significantly lower incidence of nephrotoxicity compared with patients receiving intermittent infusion, whilst the clinical efficacy and mortality were not significantly different.
Although some studies have shown occurrence of thrombophlebitis even after 24 h of peripheral infusion, 14 other studies state the feasibility of peripheral infusion, suggesting that intermittent infusion (one or twice a day over a period of at least 60 min), may reduce endothelial damage.1,15
In one clinical study, short-term (<6 days) intermittent administration of vancomycin via a long peripheral cannula in polyurethane was found to be equally safe and less expensive than via central venous catheter.16,17
In short, endothelial toxicity of vancomycin during peripheral administration may be theoretically modulated by its concentration, by the modality of infusion (continuous vs intermittent) and by the type of peripheral venous access device (short cannula vs long peripheral cannula).
This study was focused on the risk of local thrombosis/thrombophlebitis in critically ill patients receiving continuous intravenous infusion of vancomycin via a long peripheral cannula in polyurethane, comparing two different modalities of drug dilution.
Methods
The study was carried out in the Intensive Care Unit of a University Hospital during a period of 6 months, after approval by the facility’s Ethic’s Committee.
All patients requiring prolonged (more than 7 days) intravenous infusion of vancomycin were included in the protocol. In all patients, the antibiotic was administered via a 3 Fr (20 Ga.) “long peripheral cannula” (“mini-midline”), in polyurethane, 8 cm long (LeaderFlex™, Vygon, France), placed in a deep vein of the upper arm. All cannulas were inserted according to a standardized insertion bundle including: pre-procedural systematic ultrasound scan of the deep veins of the upper arm (so called RaPeVA—Rapid Peripheral Vein Assessment) 18 ; proper hand hygiene; skin antisepsis with 2% chlorhexidine in 70% isopropyl-alcohol; maximal barrier precautions; choice of an appropriate vein in terms of caliber (3 mm or more), position (in the middle third of upper arm: so called “green area” according to Zone Insertion Method (ZIM™), 19 and depth (less than 2.5 cm); proper visualization of median nerve and brachial artery before venipuncture; real-time ultrasound guided puncture and cannulation of the vein; sutureless securement (Grip-Lok®, TIDI Products LLC, Neenah, USA); exit site protection with transparent semipermeable membranes.
Patients were randomized in two groups: in the control group, vancomycin was given by continuous infusion at standard dilution (1 g in 50 ml saline: i.e. 20 mg/ml), while in the study group the drug was diluted five times (1 g of vancomycin in 250 ml saline: i.e. 4 mg/ml); in both groups, the planned therapeutic dosage was 1 g/day. No other drugs were infused into the same infusion route to avoid incompatibility with other drugs.20,21
All patients were systematically evaluated every 24 h during the period of treatment, searching any sign of inflammation of the exit site according to Visual Exit-Site Score, 22 and ruling out the occurrence of asymptomatic venous thrombosis (thrombophlebitis) by daily ultrasound scan of the veins of the arm. Bi-dimensional scan was coupled with Compression Ultra-Sonography (CUS). 23
For each patient, the following data were recorded: age and gender, size, length, and type of device used, vein chosen for insertion, vein diameter and depth, number of attempts for vein insertion, results of daily Visual Exit-Site Score and CUS, incidence of venous thrombosis.
Statistical analysis
All data were included in an Microsoft Excel 2016 (Version 16.0.4229.1024) 24 spreadsheet for record and analysis. Continuous data were expressed as mean and standard deviation (SD). Categorical data were reported as absolute numbers and percentages (%). Differences between groups were assessed with parametric tests for independent using the t test. A minimum of 15 patients for group was esteemed as required to achieve statistical significance. An analysis of the preliminary results halfway through the study was planned.
Differences between groups were regarded as statistically significant when p value was <0.05. MedCalc Statistical Software version 18.10 (MedCalc Software bvba, Ostend, Belgium) was used for statistical analysis.
Results
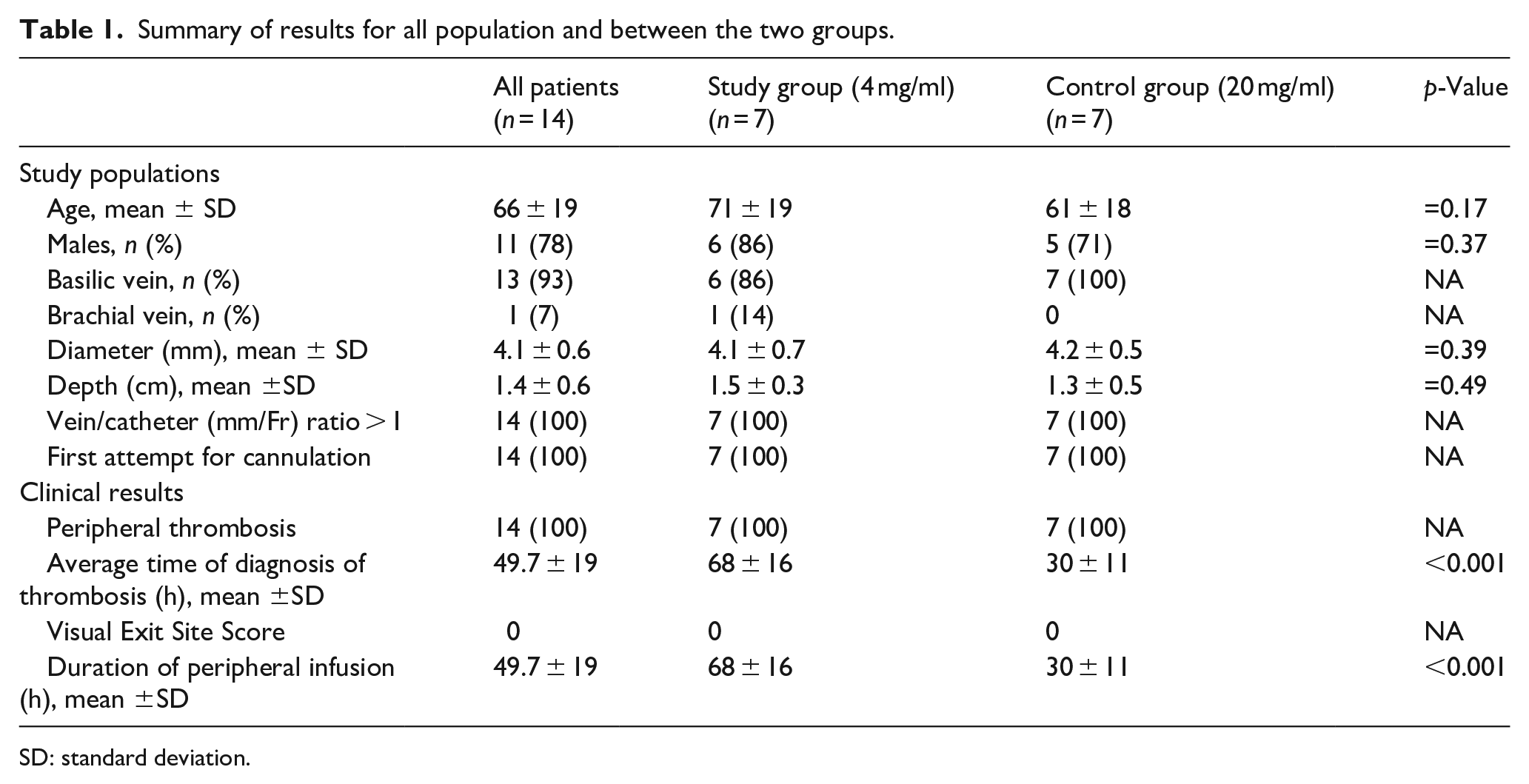
Only 14 patients were included in the study: seven in the study group and seven in the control group (20 mg/ml). Main characteristics between the two groups are shown in Table 1. Long peripheral cannulas were inserted in the basilic vein in most of patients (93%), except for one patient, where the brachial vein was cannulated.
Summary of results for all population and between the two groups.
SD: standard deviation.
There were no significant differences in gender (P = 0.37) and mean age (71 ± 19 vs 61 ± 18 years, p = 0.17) between two groups. There was no significant difference in mean vein diameter in study group versus control group (4.1 ± 0.7 mm vs 4.2 ± 0.5 mm respectively, p = 0.39). All cannulated veins were more than 3 mm in diameter. The mean depth of the veins was 1.5 ± 0.3 cm in the study group and 1.3 ± 0.5 cm in control group (no vein deeper than 2.5 cm), without any significant difference between two groups (p = 0.49). All veins were cannulated at first attempt in the green area according to ZIM™. 19
The daily evaluation by CUS showed venous thrombosis (Figure 1) at the distal tip of the cannula in 100% of patients, in both groups (Table 1). After this finding in the first 14 patients, the study was interrupted.
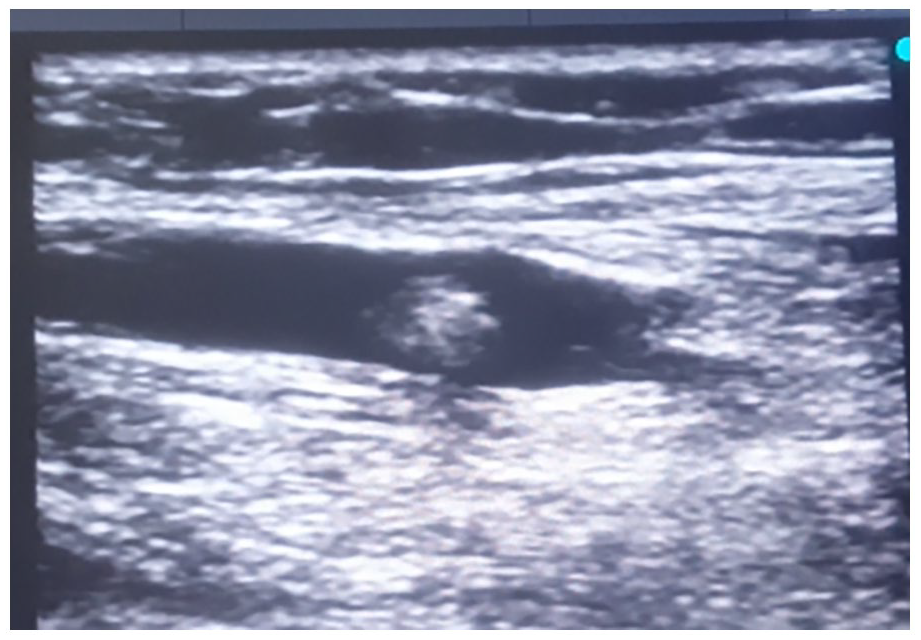
Ultrasound longitudinal scan of the basilic vein showed venous thrombosis at the distal tip of the long peripheral cannula.
All thromboses were completely asymptomatic and occurred in absence of any sign of catheter malfunction. The onset of thrombosis was significantly earlier in the control group (ranging from 24 to 48 h) than in the study group (ranging from 48 to 96 h), with an average of 30 ± 11 versus 68 ± 16 h (p < 0.001).
As the thrombosis was detected, the peripheral venous access was removed, and the vancomycin infusion was continued through a central line. Therefore, the duration of peripheral infusion of vancomycin corresponds to the average time of diagnosis of thrombosis.
The daily evaluation of the Visual Exit Site Score was negative (score zero) in all patients of both groups, for all the duration of the treatment.
Discussion
This study showed that the intravenous continuous infusion of vancomycin via a peripheral venous access—even using a long peripheral cannula in polyurethane—is inevitably associated with thrombophlebitis, regardless of its dilution.
The site of the thrombotic event (at the distal tip of the cannula) strongly suggests that the pathogenesis is exclusively related to the direct chemical injury of the drug on the vein wall. This contention is further supported by the fact that the vein/catheter ratio 25 was optimal in all patients and that the insertion-related trauma to the vein was minimal (all veins were cannulated at first attempt). Endothelial toxicity of vancomycin is known to be concentration-dependent. Concentrations greater than 2.5 mg/ml resulted in a significant increase in vascular endothelial cell death, with a median lethal dose of 5 mg/ml over 24 h-infusion. 1
Recent in vitro data also found that even flow variations of low concentrations of vancomycin infusion (between 1 and 4 mg/ml), simulating multi-infusion conditions, induced cellular stress independently by vancomycin concentration and that these variations did not significantly influence local toxicity compared to a fixed concentration. 21
Moreover, in an animal model, also when using midline catheters, the vancomycin infusion resulted in severe vascular injury and premature catheter failure. 26
As suggested by these findings, a possible explanation of these results is that the peripheral use of vancomycin could induce an excess death rate of endothelial cells surrounding the catheter outlet, where the endothelial cells are subjected to a slow and continuous flow of the drug, due to the weak blood flow in peripheral veins of patients in bed and sedated. Long peripheral cannulas are defined by current guidelines 27 as peripheral venous access devices longer than 6 cm (i.e.: longer than short cannulas) and shorter than 15 cm (i.e.: shorter than midline catheters). Two previous studies have suggested that vancomycin infusion via a long peripheral cannula in polyurethane—at a dilution of 4 mg/ml—may not associated with symptomatic thrombophlebitis or venous thrombosis, both during short-term (<6 days), 16 or long-term therapy (average of 7.5 days, range 1–25 days). 27 Both the device used in these two studies and the device of this study are long peripheral cannulas (8–10 cm) in polyurethane, a material notoriously less thrombogenic than polytetrafluoroethylene (often used for short peripheral cannulas). Therefore, these results are apparently in contrast with the previous studies of Caparas et al.16,28,29 Notably, the latter investigators did not consider the use of ultrasound for detection of asymptomatic thrombosis, while this study protocol included a routine daily venous scan by ultrasound. Lastly, it is possible that the different modality of infusion (continuous vs intermittent) may account for the difference.
This study reported equal risks of thrombosis with high (20 mg/ml) and low (4 mg/ml) concentration of vancomycin. There is some controversy about the actual importance of dilution, rather than pH, as a factor in the pathogenesis of thrombophlebitis.30,31 An interesting result of this study is the different time of onset of thrombosis, significantly shorter when the dilution was 20 mg/ml rather than 4 mg/ml. There were no differences in severity of vessel injury between different concentrations of vancomycin used, because all patients had venous thrombosis, but this damage appeared to be time dependent. Probably the damage mechanism was the same, but the onset time was earlier for higher concentrations.
Considering that the overall pH of the solution is only minimal affected by such dilution, and that the osmolarity was also very similar (as a matter of fact, slightly higher in the study group), it appears that the irritant effect of vancomycin on the endothelium must be secondary to some other mechanisms (non-related to pH or osmolarity), as the cellular stress, which are somehow affected by dilution.
Limitations
Main limitation of this study is the small sample size. However, since all enrolled patients presented an early asymptomatic venous thrombosis, the authors decided to interrupt the study and complete vancomycin treatment via a central venous access.
Also, it is not actually known how many of the asymptomatic thrombotic event may eventually become symptomatic and/or cause catheter failure. Most of patients were sedated and unable to complain of pain; nonetheless, it was not possible to detect any other clear sign of symptomatic thrombosis (redness, local swelling, ipsilateral edema of the arm, etc.) and no catheter malfunction (the volumetric infusion pumps used for the protocol did not record any alarm for distal occlusion). These results may not be generalized to larger patient samples.
Finally, the authors considered that continuous infusion of vancomycin cannot be automatically extended to intermittent infusion. Further investigation comparing continuous versus intermittent therapy would provide greater understanding of vancomycin administration in various administration methods and different patient cohorts.
Conclusion
To avoid vessel injury, continuous intravenous infusion of vancomycin should be preferably delivered by a central venous access, as largely recommended by current guidelines, since peripheral infusion is inevitably associated with venous thrombosis, independently from the type of peripheral venous access device adopted (short peripheral cannula vs long peripheral cannula) and from the extent of dilution. When vancomycin is administered at a standard dilution (20 mg/ml), thrombotic complications are expected to occur rapidly (after 24 h of infusion), even using a long peripheral cannula.
For a better definition of the margin of safety of intermittent infusion of vancomycin by the peripheral route, further clinical studies are needed. In future studies, we strongly recommend considering all non-drug related factors which may be associated with catheter related thrombosis (inadequate ratio of caliber between vein and catheter; excessive trauma to the vein at the time of cannulation; inappropriate securement of the line). Finally, further research is also needed to establish the safety of using long peripheral catheters or midline catheters for intermittent or continuous infusion of irritant or vesicant drugs.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
