Abstract
Background:
Peripheral intravenous catheter (PIVC) insertion often fails on the first attempt. Risk factors include small vein size and dehydration, causing vein deformation and displacement due to puncture resistance of the vessel. The authors developed a short, thin-tipped bevel needle and compared its puncture performance with needles of four available PIVCs using an ex vivo model.
Methods:
The PIVC with the thin-tipped short bevel needle was compared to four available PIVCs using an ex vivo model which simulated the cephalic vein of the human forearm. The ex vivo model consisted of a porcine shoulder and porcine internal jugular vein, and was used for evaluation of the rate of vein deformation and vessel displacement during needle insertion.
Results:
An ex vivo model was created with a vessel diameter of 2.7–3.7 mm and a depth of 2–5 mm. The thin-tipped short bevel PIVC needle was associated with a significantly lower compressive deformation rate and venous displacement compared to the needles of the other four PIVCs.
Conclusion:
The thin-tipped short bevel needle induced lower compressive deformation and displacement of the vein than the conventional needles. This needle has the potential to improve the first-attempt success rate of peripheral intravenous catheterization in patients with difficult venous access.
Keywords
Introduction
Peripheral venous puncture is one of the most widely used clinical procedures. 1 Up to 81% of hospitalized patients undergo peripheral intravenous catheter (PIVC) placement.2–4 However, this procedure can be difficult and requires technical skills from a healthcare professional. A recently published meta-analysis reported that the first attempt success rate is about 83% and 76% with and without ultrasound, respectively. 2 Multiple attempts due to difficult PIVC placement induce patient distress and pain, complications, and extra treatment costs.5–8
There have been multiple reports on risk factors for patients whose veins present difficulty in placing PIVCs. Those factors include small vein size, vascular fragility, dehydration, collapse of the vessel, and thinned subcutaneous tissue.3,9–15 Peripheral venous puncture in pediatric patients with small and fragile blood vessels is said to be more likely to fail because the veins tend to deform or displace due to the mechanical stress of the needle tip. 10 In addition, it is reported that the veins in elderly patients with decreased subcutaneous tissue strength tend to wiggle and roll under the skin due to the mechanical force generated by the needle. 12 Additionally, several reports have described posterior wall puncture, one of the causes of failure in placing PIVCs, due to compressive deformation during needle puncture of peripheral veins when using ultrasound-guided PIVC placement.16,17 Munshey et al. 16 explained that posterior wall puncture likely occurs in pediatric patients, whose veins are small in diameter and easily compressed. Gottlieb et al. 17 noted that vascular puncture in emergency patients who do not have sufficient venous distension can increases the risk of injury to the posterior vessel wall.
There are many methods aimed to increase the success rate of PIVC cannulation in patients with difficult venous access in clinical settings, such as mechanical stimulation, heating with a warmed towel, venous distension with a tourniquet or an electrical stimulation device, and visualization of the vessel using near-infrared (NIR) and ultrasound (US) devices.18–21 Mechanical stimulation and warming of the puncture site are common procedures, but their effectiveness is still unclear. Venous distension is an effective part of peripheral intravenous cannulation, but may take a long time in elderly and dehydrated patients.20,22 Since the veins are especially fragile in the elderly, there is also a risk of vein injury if the arm is tied with too tight a tourniquet.22,23 Visualization of blood vessels with NIR or US devices is commonly used for patients with difficult venous access, and the use of these devices may increase the puncture success rate.6,24–26 NIR devices can help visualize the potential presence of invisible blood vessels, but does not significantly improve the puncture success rate because they cannot measure the depth or size of blood vessels.24,27 US-guided puncture can improve the success rate of PIVC cannulation.2,25,28 The success rate of venous puncture with or without US-guided puncture in patients with difficult venous access and multiple previous failures is 75% and 49%, respectively. 2 However, US-guided puncture may be the most challenging for practitioners not familiar with ultrasonography and require proper training and persistent practice to attain competence.29,30 In addition, even with US-guidance, the success rate can be affected by vessel diameter.11,31,32 In one study, the puncture success rate in veins with a diameter <0.4 cm was lower than that in veins with a diameter ⩾0.4 cm (39% vs 63%). 11 Even if the vein is visualized, insertion of a needle into such a small target is typically difficult. Puncture of small and fragile blood vessels is considered more likely to fail because these vessels tend to deform or displace due to needle puncture. 10 Additionally, tenting of the vein wall, wherein the needle tip cannot penetrate the anterior vein wall, is often observed during needle insertion with US guidance, which can lead to double wall puncture (anterior and posterior vein walls) and failed cannulation. 33 Even well-trained healthcare professionals should be careful when puncturing blood vessels to avoid the risk of double wall puncture. 32 This phenomenon suggests that minimizing needle insertion force may reduce unsuccessful PIVC insertion.
In a previous study, a regular lancet needle with a very thin tip (bevel angle: 12°) was manufactured and its initial peak insertion force evaluated using porcine liver tissue. 34 The peak insertion force produced by the needle was 11% lower than that produced by a conventional needle. However, this needle had a regular bevel which was not short enough, preventing its use in PIVCs. Furthermore, the needle’s penetration force was not measured in the study. During the measurement of the insertion force, the bevel face did not completely penetrate the tissue, and only a part of the needle tip was inserted into the soft tissue. More importantly, the needle included a spiny projection on the bevel face, which can substantially increase the penetration force. Thus, needles for intravenous catheters need to have the shorter bevel length and the smooth bevel face, and the resistance forces must be measured until the bevel face of these needles including the catheters penetrates the object. Tanabe et al. 35 developed a short bevel needle with a very thin tip and evaluated it using a phantom. The thin-tipped short bevel needle was 24-gauge (outer diameter: 0.55 mm) without any spiny projections on the bevel face and achieved successful puncture at smaller angles than the needles of conventional PIVCs, even when the insertion site on the polyvinyl chloride tube phantom was off-center. However, since the mechanical properties polyvinyl chloride tubing are different from those of living tissue, they cannot be guaranteed to behave in the same way. Abolhassani et al. 36 reported that needle insertion into an artificial model such as a phantom could not mimic the interactive forces at play during needle insertion into inhomogeneous, viscoelastic tissue. Evaluating the deformation and displacement of a vessel during needle insertion requires use of an ex vivo model, which includes a vessel derived from a living body. However, no ex vivo model to evaluate the performance of peripheral needle puncture has been reported. If the thin-tipped short bevel needle was to achieve better puncture performance than conventional needles in an ex vivo model, it would be possible to identify indications for its use.
The aim of this study was to create an ex vivo model combining a porcine internal jugular vein (IJV) and a porcine shoulder to simulate the cephalic vein in the human forearm, and to compare the puncture performance of the thin-tipped short bevel needle with four types of conventional needle.
Methods
A PIVC with the thin-tipped short bevel needle and four others were tested in the ex vivo porcine model to assess vascular deformation rate and target vessel displacement. Five types of PIVC were compared: The PIVC with the thin-tipped short bevel needle (“needle A”), a Surshield Surflo II (Terumo Corporation, Tokyo, Japan) (“needle B”), a SuperCath 5 (Medikit Co., Ltd., Tokyo, Japan) (“needle C”), a BD Insyte Autoguard BC (Nippon Becton Dickinson Company, Ltd., Tokyo, Japan) (“needle D”), and an Introcan Safety 3 (B. Braun Aesculap Japan Co., Ltd., Tokyo, Japan) (“needle E”). A PIVC has a structure in which a needle having a smaller diameter than a catheter is combined in an inner lumen of the catheter. All PIVCs consisted of a 22-gauge polyurethane catheter (length: 25 mm) and a 24-gauge stainless needle with a short bevel. Moureau’s vessel health and preservation program recommends that an outer diameter of a catheter be 33% or less of a diameter of the vein. 37 The diameter of 22-gauge catheter corresponds to less than 33% of the vessel diameter in the ex vivo model. The study did not involve any living entity, and accordingly no ethical statement is required.
Optical microscopic and electron microscopic images of needles
The needle tips were imaged with 150× optical microscopy (OM) (VHX-6000, Keyence Corporation, Osaka, Japan) and 1000× scanning electron microscopy (SEM) (S-3400N, Hitachi High-Tech Corporation, Tokyo, Japan). The authors measured the thickness of the needle tips through SEM observation.
Ex vivo model
The ex vivo model, a modification of the model of Tsukune et al. 38 was used to measure vascular deformation and target vessel displacement of the five needles. Because Tsukune’s model envisioned the internal jugular vein of the central vein, the authors simulated the cephalic vein of the forearm. Since the diameter of the cephalic vein in the forearm is 2–4 mm and the depth of the cephalic vein in the forearm is 1–6 mm, the authors positioned a blood vessel with a diameter of 2.7–3.7 mm and placed it at a depth of 2–5 mm.39–41 Porcine shoulder (50 mm × 20 mm × 40 mm) and porcine IJV (outer diameter approximately 2 mm, length 80–100 mm) were purchased from Tokyo Shibaura Zouki Co., Ltd. (Tokyo, Japan). The IJV was inserted into a tunnel that was formed in the shoulder musculature using a stainless steel rod (outer diameter of 3 mm) and one end of the vein was ligated with a 3-0 suture. Since the peripheral venous pressure of the human forearm is approximately 1.7 ± 0.5 kPa, the embedded porcine vessel was filled with water produced by reverse osmosis to reach a hydrostatic pressure of 1.3 kPa, resulting in their diameters expanding from 2 mm to 2.7–3.7 mm. 42
The model was set on a jig to allow a puncture angle of 30° (Figure 1), and the PIVC was attached to the load cell of the compression testing machine (AGS-1kNX, Shimadzu Corporation, Kyoto, Japan). As in the previous study, the authors set the puncture speed at 50 mm/min. 35 Echo gel was applied to the surface of the model. The 40-mm long IJV embedded in the porcine shoulder was punctured in the midline, and deformation and displacement of the vessel during puncture was monitored by US (eZono-4000, Jena, Germany). In order to track the position of the tips of the needles, the probe was moved by a motor (43 mm/min) along with the movement of the needle. The test was started before the vessel was punctured and ended when the needle passed through the posterior wall of the vessel. US images obtained with this model were reviewed by highly experienced nurses who had more than ten years of work experience and had conducted US-guided PIVC puncture in clinical settings more than 50 times.
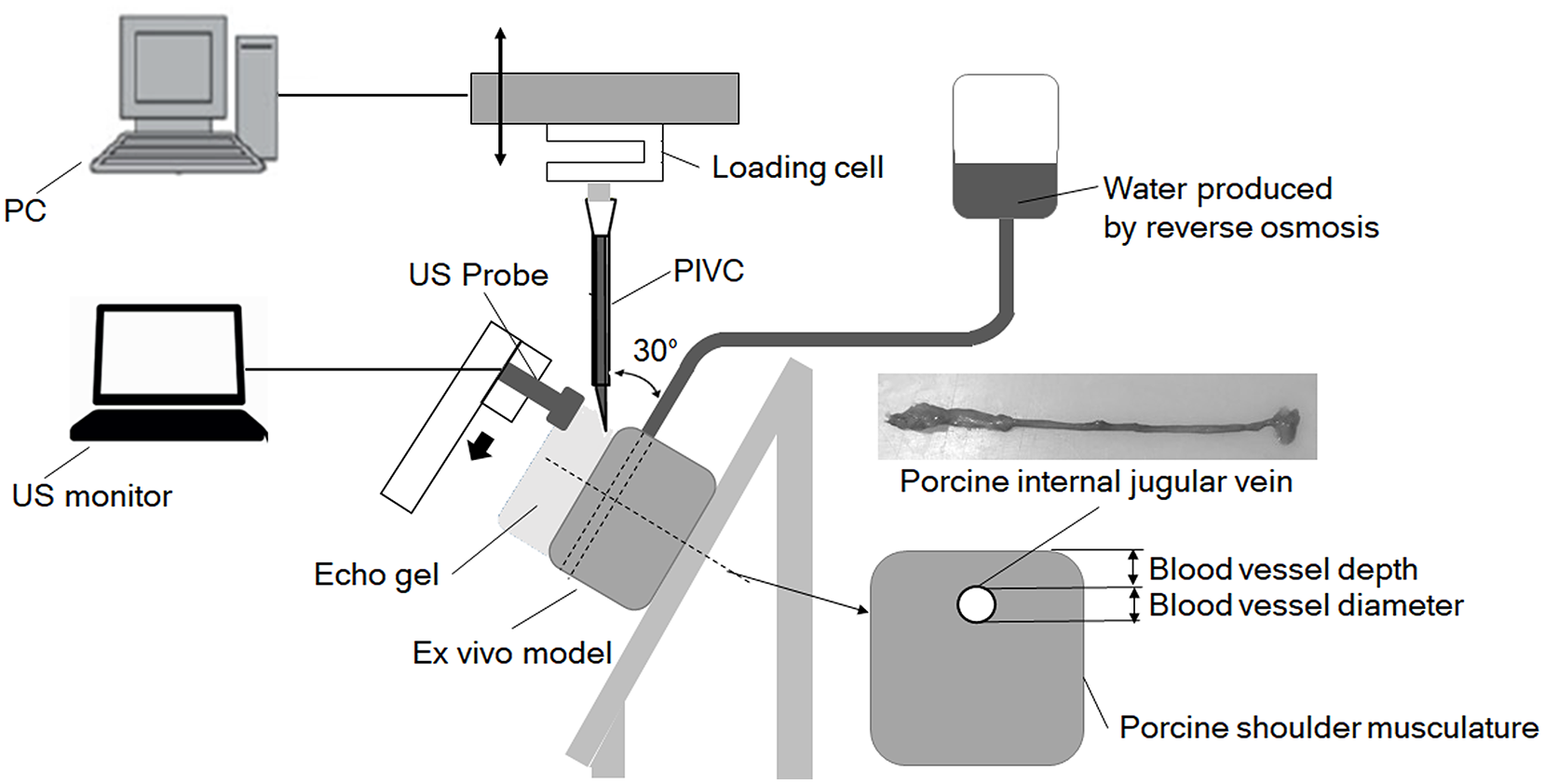
Experimental setup for measurement of the compressive deformation of the vein and its displacement.
Data analysis
Compressive deformation rate and displacement of vein
Compressive deformation of the vein was defined as the change in vertical diameter (Figure 2(a)). The degree of change in deformation of the vein was defined as the maximum compressive deformation divided by the initial vessel diameter. The maximum compressive deformation occurred just before the needle tip entered the anterior wall of the vessel. The timing of needle entry into the vessel was determined from the force-displacement curve of the compression testing machine. US images at baseline and the point of maximum deformation were used. Open-source image-processing software (ImageJ version 1.53a, National Institutes of Health, Bethesda Maryland, USA) was used to measure the dimensions. 43 Vessel displacement was defined as changes in the centroid of the vein before and after insertion (Figure 2(b)). The centroid of the vein was calculated by Image J by specifying the contour of the vessel in the US image. Statistical analyses were performed to determine differences in the rate of compressive deformation and vein displacement. The results were analyzed for statistical significance using Dunnett’s test. Values of p < 0.05 were considered statistically significant. All statistical analyses were performed using SPSS version 25 (IBM, New York, USA).

Schematic images of the outcome measurement method: (a) deformation of the vein and (b) displacement of the vein.
Results
Optical microscopic and electron microscopic imaging of the needle tips
OM and SEM images of the needle tips and their sizes are shown in Figure 3. The bevel lengths of the five needle tips ranged from 1.6 to 1.9 mm. The bevel angles of the five needle tips ranged from 22° to 43°; the bevel angle of the thin-tipped short bevel needle was the smallest. The thickness of the tips of needle A, needle B, and needle C were less than 1 μm, whereas those of needle D and needle E were 7 and 5 μm, respectively.

Comparisons of needle tip designs among five types of PIVC 1000× SEM images showing the bevel angles.
Ex vivo model
In the ex vivo model, the diameter of the vessel could be set between 2.7 and 3.7 mm and the depth of the vessel between 2 and 5 mm. There were no significant differences in the diameter and the depth among the five PIVC groups (p = 0.524 and p = 0.594, respectively). The nurses confirmed that the ex vivo model is capable of reproducing both the displacement and deformation of the vein occurring in patients in clinical situations.
Compressive deformation rate and displacement of the vein
The rate of compressive deformation and the vessel displacement of the five types of PIVCs are shown in Figures 4 and 5, respectively. The PIVC with the thin-tipped short bevel needle showed significantly lower rates of compressive deformation and vessel displacement compared to needles B through E (p < 0.05).
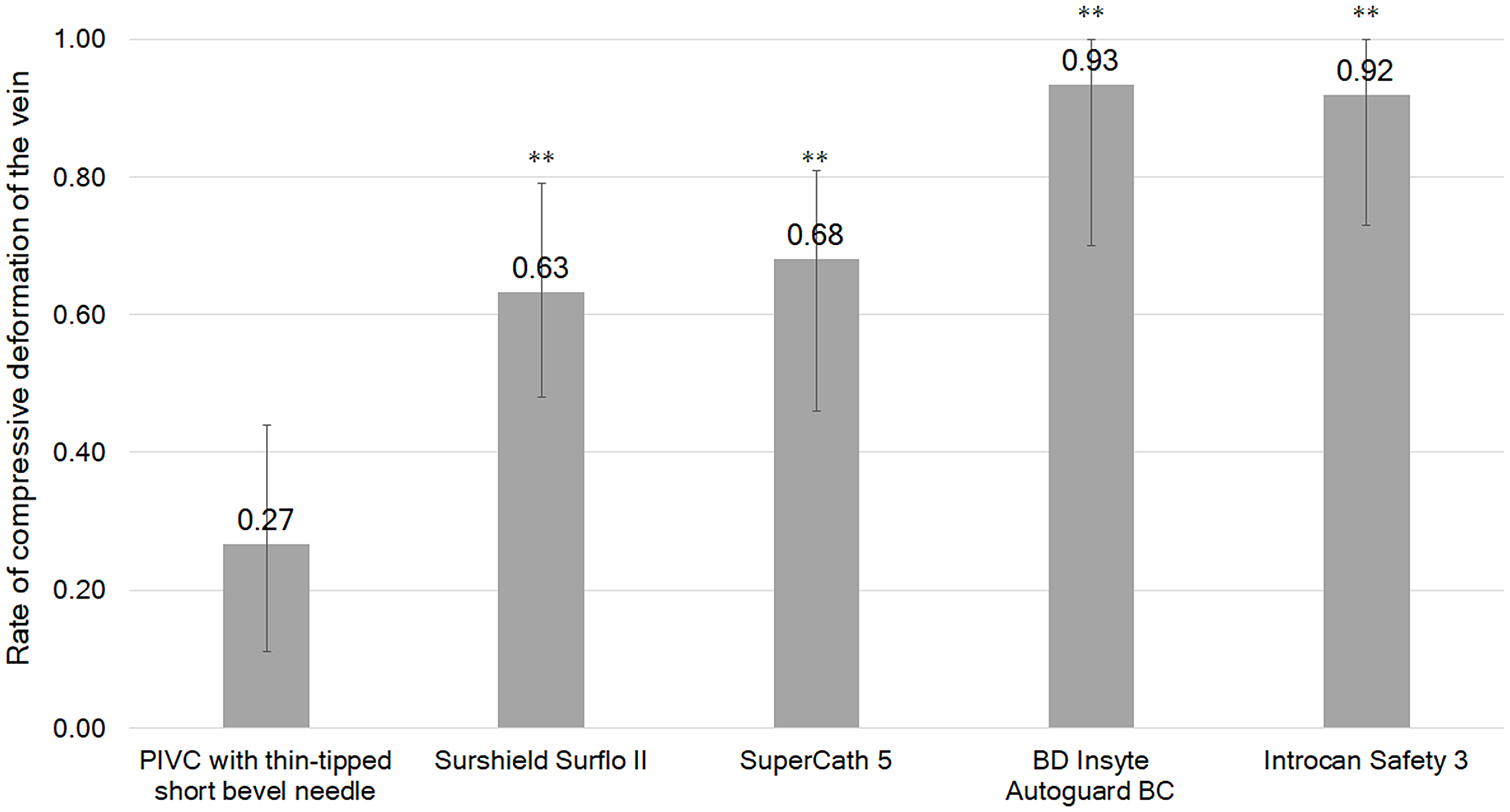
Measurements of the mean, maximum, and minimum rate of compressive deformation of the vein when punctured with the needle tip (N = 10; **p < 0.01, and statistically significant compared with the PIVC with the thin-tipped short bevel needle (needle A)).
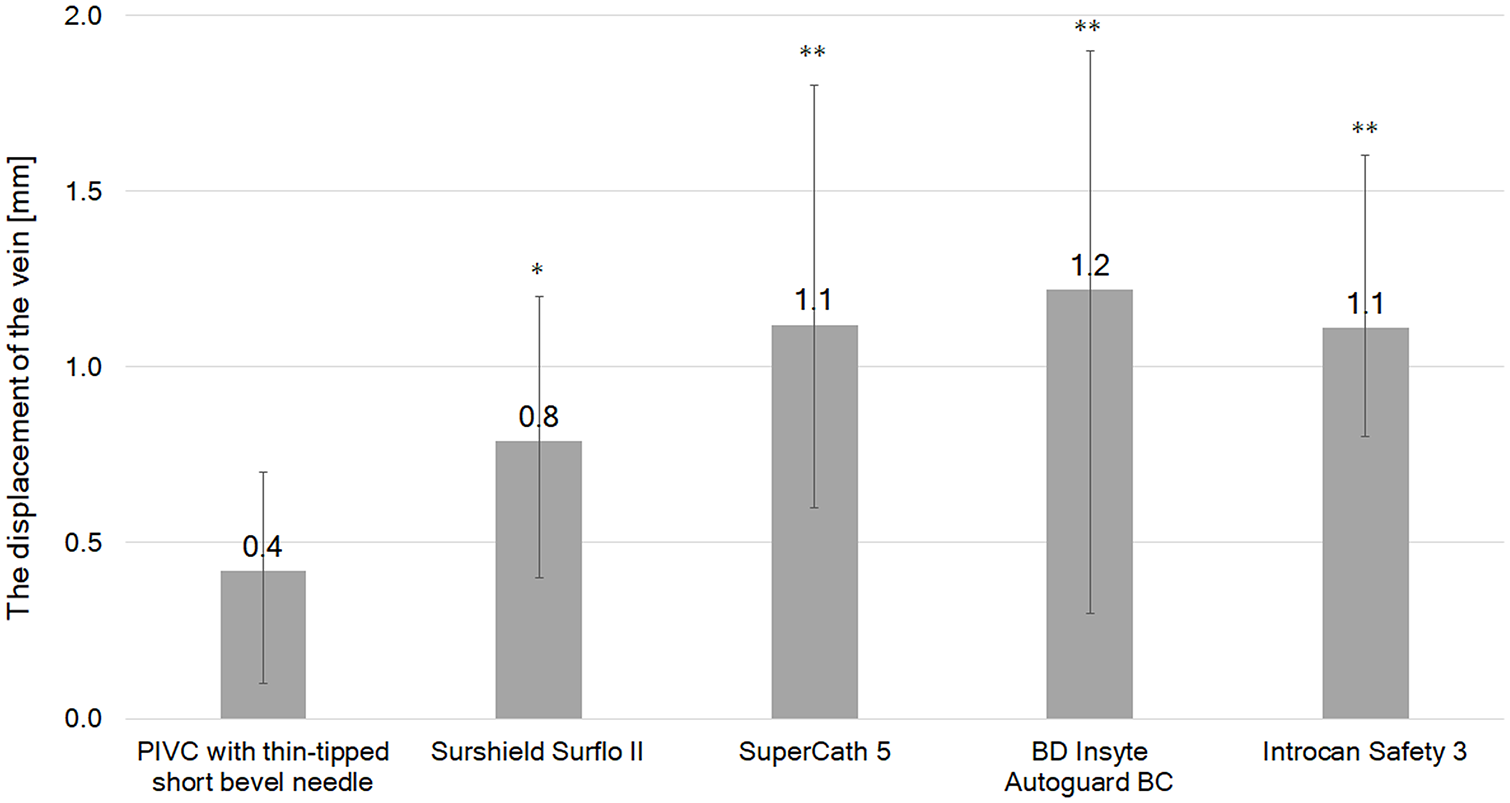
Measurements of the mean, maximum, and minimum displacement of the vein when punctured with the needle tip (N = 10; *p < 0.05, **p < 0.01, and statistically significant compared with the PIVC with the thin-tipped short bevel needle (needle A)).
Discussion
This is the first study to create an ex vivo model simulating the peripheral vein of the human forearm using animal vein, and to compare the behaviors of living tissue when punctured with a PIVC having a thin-tipped short bevel needle and four other needle types. The vascular compressive deformation rate of the PIVC equipped with the thin-tipped short bevel needle was significantly lower than that of the four other needles. In addition, vessel displacement was significantly smaller with the thin-tipped short bevel needle than with that of the four other needles.
To evaluate the puncture performance of the thin-tipped short bevel needle for PIVC using living tissue, an ex vivo model was created using the porcine internal jugular vein. In a previous study, Davidson et al. 44 combined a rubber tube and a turkey leg to make a simulator for training in venous puncture with US. Since this model used a turkey leg, which is a living tissue, it can provide near-clinical images and is useful for training in the acquisition and interpretation of US images. However, the model used a rubber tube as the vein, preventing evaluation of the behavior of the vein when punctured with a needle. The authors reproduced the deformation and displacement of blood vessels during needle puncture by using an ex vivo model consisting of porcine shoulder muscle tissue with an appropriately placed porcine IJV.
The 22° tip angle of the thin-tipped short bevel could have contributed to the smaller deformation and displacement of the vein. Needles used in clinical settings are mainly divided into two groups, and a regular bevel and a short bevel are used according to the purpose. The regular type of bevel needle commonly used for intramuscular and subcutaneous injections has a small bevel angle (approximately 20°) and low puncture resistance, but its longer length increases risk of inadvertent tissue trauma. 45 In contrast, the short bevel needle reduces risk of inadvertent tissue trauma, but its large bevel angle (typically over 30°) produces higher puncture resistance. Generally, short bevel needles are used for access to blood vessels because the whole bevel length can be easily inserted into the vein. 45 Therefore, there is a problem that the resistance force is relatively large compared to that generated by regular bevel needles, and the vein is deformed and displaced. 10 The thin-tipped short bevel needle the authors developed has a short bevel and a small bevel angle of 22°, and therefore has advantages of both regular and short bevel needles, including reduced possible risk of inadvertent tissue trauma and lower puncture resistance. Although it is technically possible to manufacture a bevel angle smaller than 22°, the angle was designed with that of a regular bevel as the lower limit (20°). If a bevel angle is too small, there is a risk of the needle-tip bending when puncturing patients with resistant skin. Thus, an angle of approximately 22° can be both the minimum and optimal angle for intravascular needles. The deformation rate and the displacement of the vein tended to increase as the tip angle increased, but when comparing needle C with needles D and E, the outcomes of needle C were smaller than those of the others even if the tip angle was large. SEM images of the tips of needles C, D, and E showed that they were less than 1, 7, and 5 μm, respectively. The quality of electropolishing as part of the needle manufacturing process can affect the sharpness of the needle tip and reduces puncture resistance. 46 Not only the angle of the needle tip but also the precision of sharpening the tip may be involved.
The PIVC equipped with needle A was associated with significantly smaller compressive deformation and displacement of the vein than the four conventional needles. Although it is recommended to use small-gauge PIVCs and US-guided puncture to assist venipuncture in patients with difficult venous access, smaller-gauge PIVCs that do not allow administration of drugs or other materials at the prescribed rate may limit treatment options. 6 The US-guided puncture technique is effective in patients with difficult venous access, but its effectiveness in patients with small blood vessels is limited. Therefore, a certain number of needle insertion experience is required to acquire a suitable technique.2,11,24,28,31,47 There is a need for devices that are not limited in treatment and do not require special training. Since our thin-tipped short bevel needle causes less deformation and displacement of the vessel during puncture than conventional needles, it has the potential to increase the success rate of venous puncture in patients with small blood vessels that are difficult to puncture at the blood vessel center and collapse easily. In addition, the thin-tipped short bevel needle differs from the conventional PIVCs only in the needle tip, meaning that no special training may be required, and use in clinical settings can be started immediately. On the other hand, peripheral venous cannulation is a procedure that strongly relies on the skill of the healthcare professional. Various techniques and devices to improve the success rate of venipuncture are only effective if they are performed properly. Used by properly trained healthcare professionals, this thin-tipped short bevel needle can increase the first-attempt success rate of puncture in patients with difficult venous access.
The puncture conditions used in this study, such as puncture angles, puncture speeds, venous pressure, were fixed, and did not cover actual clinical conditions. Further, the animal blood vessel used in this study did not completely match the deformation and the displacement of human blood vessels. However, it is considered that the behavior of blood vessels in living tissues can be reproduced. Furthermore, the deformation and the displacement of veins may change depending on conditions such as puncture speed, puncture angle, and venous pressure, but the order of the mean values in this study does not change.
Conclusions
To evaluate the puncture performance of PIVC needles, the authors created an ex vivo model which reproduces the deformation and displacement of the cephalic vein of the patient’s forearm during needle insertion. In this model, the thin-tipped short bevel needle caused less deformation and displacement of the vein than the other needles tested. Our results suggest that the thin-tipped short bevel needle may be effective in improving the first-attempt success rate of venipuncture in patients with difficult venous access by reducing the deformation and displacement of the vein, when used by properly trained healthcare professionals. Further studies are needed to prove the effectiveness of this needle in clinical situations.
Footnotes
Acknowledgements
The authors thank Libby Cone, MD, MA, from DMC Corp. (www.dmed.co.jp <![]() >) for editing drafts of this manuscript. The authors wish to acknowledge the contribution of Mr. Yuusuke Shibata for critical revision of the manuscript.
>) for editing drafts of this manuscript. The authors wish to acknowledge the contribution of Mr. Yuusuke Shibata for critical revision of the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Hidenori Tanabe, Kousuke Oosawa, Manabu Miura, Shinichi Mizuno, Takayuki Yokota, Takehiko Ueda, and Yasunobu Zushi are employees of Terumo Corporation. Ryoko Murayama and Mari Abe-Doi are researchers of a laboratory supported by Terumo Corporation.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was a joint research venture between Terumo Corporation and The University of Tokyo and was funded by Terumo Corporation.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
