Abstract
Background:
The superficial runoff veins anatomically run alongside the cutaneous nerves in the volar forearm. Consequently, the surgical repair of vascular access (VA)-associated venous aneurysms attached to the cutaneous nerves could cause intraoperative nerve injury. Therefore, we proposed a cutaneous nerve-conscious VA-related aneurysmal repair assisted by nerve ultrasonography. This ultrasonography aids in the preoperative examination of the courses of the nerves neighboring the aneurysm.
Methods:
Sixteen consecutive patients who underwent surgical revisions of VA-related aneurysms (14 venous aneurysms and two arterial pseudoaneurysms) were enrolled. The locations of aneurysms derived from preexisting arteriovenous fistulas included 11 radiocephalic arteriovenous fistulas in the wrist or distally, three radiocephalic arteriovenous fistulas at the antecubital fossa, and two brachiocephalic arteriovenous fistulas at the antecubital fossa or in the distal upper arm. A preoperative ultrasonographic scan of the cutaneous nerve trunks that ran peripherally toward and along the aneurysms was attempted to avoid nerve injuries during operations. Basically, the aneurysms were resected from the proximal to the distal ends after being separated from the preserved adjacent nerves.
Results:
The cutaneous nerve trunks that coursed toward and along the aneurysm could be identified by preoperative ultrasonography and could be preserved intraoperatively based on ultrasonographic findings in all patients. In four patients, the cutaneous nerve branch was unexpectedly or intentionally severed for a seamless surgical process because the nerve branch that divided from the cutaneous nerve trunk was strongly attached to the aneurysm. They subsequently suffered from hypoesthesia, but did not experience neuropathic pain.
Conclusion:
Preoperative ultrasonographic examination of the cutaneous nerve facilitated the intraoperative prevention of cutaneous nerve injury in VA-related aneurysmectomy. Nerve-sparing VA surgery assisted by preoperative nerve ultrasonography based on the understanding of topological anatomy might contribute to the reduction in postoperative neuropathy and enhance comfort in the daily life of hemodialysis patients.
Keywords
Introduction
In vascular access (VA) surgeries of the upper limb, meticulous handling is required in some operative fields in order to avoid peripheral nerve injuries.1–3 The following nerves are encountered during the dissection of inflow arteries beneath the fascia: (1) the median nerve, which runs alongside the brachial artery in the upper arm and antecubital fossa; (2) the ulnar nerve, which courses along the ulnar artery in the distal forearm; and (3) the superficial radial nerve (SRN), which passes close to the radial artery in the mid-forearm.1,3 Furthermore, while dissecting the outflow cutaneous veins, we can encounter the following nerves: (1) the SRN, which passes alongside the cephalic vein (CeV) in the wrist; (2) the lateral antebrachial cutaneous nerve (LACN), which lies parallel to the CeV at the antecubital fossa; and (3) the medial antebrachial cutaneous nerve (MACN), which runs beside the basilic vein (BaV) in the distal upper arm.1–5 However, there are very few reports of sensory cutaneous nerve lesions associated with VA surgery. Cutaneous nerve injury is probably underreported because it causes only mild neurological deficits and does not affect daily life. 3
Nevertheless, the cutaneous nerves providing sensory innervation that lie in contact with the superficial cutaneous veins, which function as the outflow routes of arteriovenous fistulas (AVF), may be unexpectedly injured during VA surgery. 3 VA-related venous aneurysms frequently compress and adhere to the adjacent cutaneous nerves. Thus, it seems plausible that unintentionally severing their trunks and arborizing branches during aneurysmal resection could lead to massive sensory deficits in the forearm and hand. To avoid such lesions, nerve ultrasonography (US), which is necessary for US-guided regional anesthesia consisting of peripheral nerve block for executing VA surgery, was employed to assess the cutaneous nerve paths during preoperative evaluation performed in our hospital.4,5
Thus, this study mainly aimed to retrospectively assess the cutaneous nerve-preserving repair of VA-related aneurysms assisted by preoperative US visualization of the cutaneous nerves adjacent to the aneurysm to avoid intraoperative nerve injury. This study also sought to discuss the clinical utility, feasibility, and pitfalls of this procedure for the prevention of surgery-induced neuropathy.
Methods
Patient profiles
We enrolled 16 consecutive patients who underwent surgical repairs of VA-related aneurysms, assisted by preoperative US evaluation of the cutaneous nerves in the vicinity of vessels, between August 2015 and May 2020 and retrospectively analyzed the results. The study was conducted in accordance with the principles laid down in the Declaration of Helsinki and approved by the institutional ethics review committee (no. 2020-002). All patients provided written informed consent for their treatments as well as for the present publication. The patients’ characteristics are summarized in Table 1.
Patient characteristics.
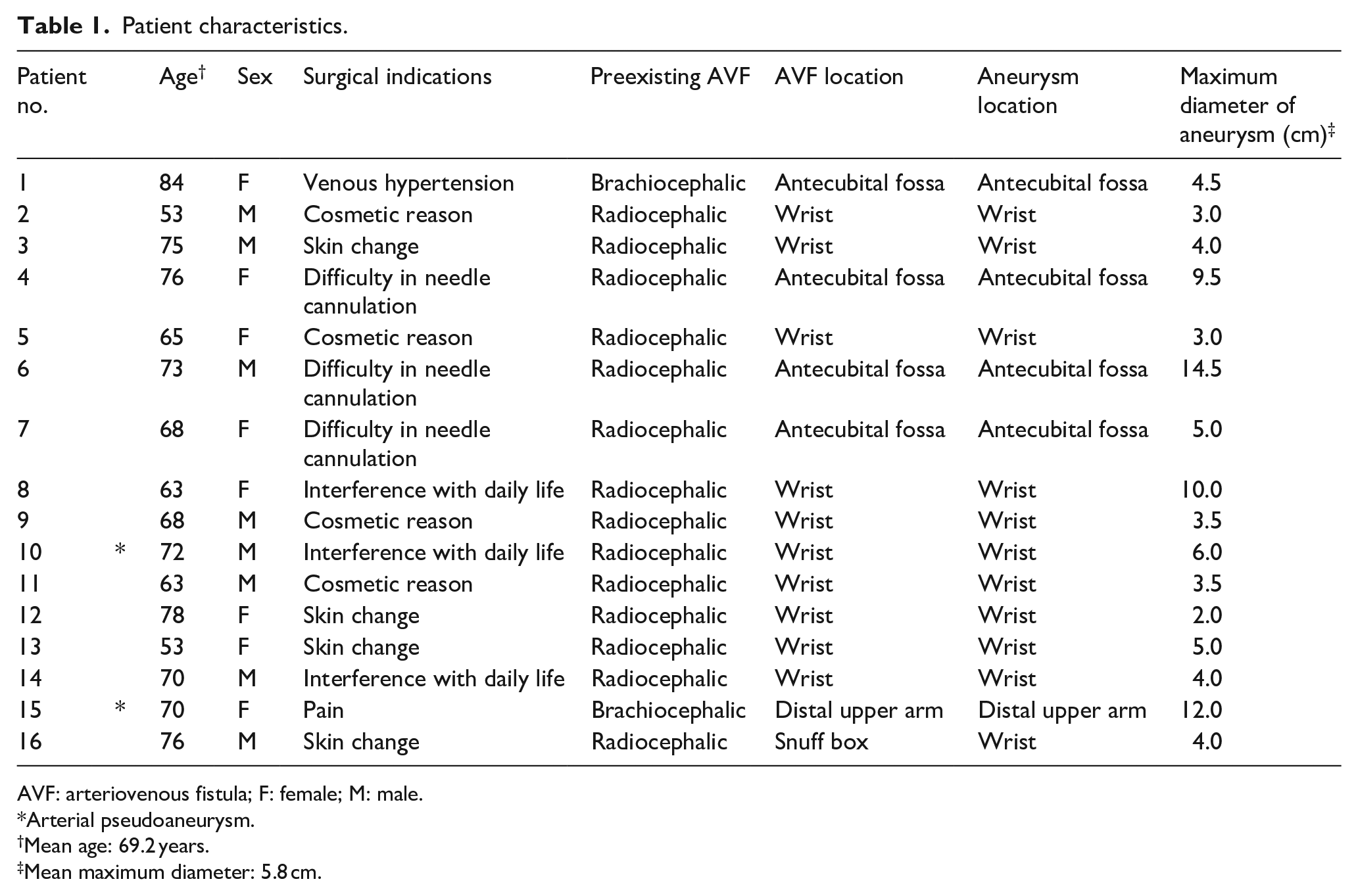
AVF: arteriovenous fistula; F: female; M: male.
Arterial pseudoaneurysm.
Mean age: 69.2 years.
Mean maximum diameter: 5.8 cm.
The mean patient age was 69.2 years. Two patients had brachiocephalic AVFs, whereas others had radiocephalic AVFs, from which the aneurysms emerged. The aneurysms were located at the wrist or distal to it in 11 patients, at the antecubital fossa in four patients, and in the distal upper arm in one patient, which were approximately consistent with the locations of preexisting AVFs. Two were pseudoaneurysms, one each at the radial artery in the wrist and at the brachial artery in the distal upper arm; the other aneurysms were outflow venous aneurysms in the vicinity of arteriovenous anastomoses. Their sizes ranged from 2.0 to 14.5 cm in the maximum diameter (mean, 5.8 cm). Ten patients were asymptomatic and indicated for the repair due to the lack of suitable veins for needle cannulation, interference with daily life, and cosmetic reasons.
Anatomical features of the cutaneous nerves in relation to the surrounding muscles and superficial veins in the upper limb
The cutaneous sensory nerves that closely accompanied the runoff veins of the AVFs in the upper limb are the SRN, LACN, and MACN. The SRN and LACN that run along the CeV provide sensory innervation to the dorsolateral area of the hand and the anterolateral area of the forearm, respectively. The MACN that courses along the BaV provides sensory innervation to the anteromedial skin of the forearm. The anatomical relationships of the SRN, LACN, and MACN with the surrounding muscles and cutaneous veins are illustrated in Figure 1.6–8
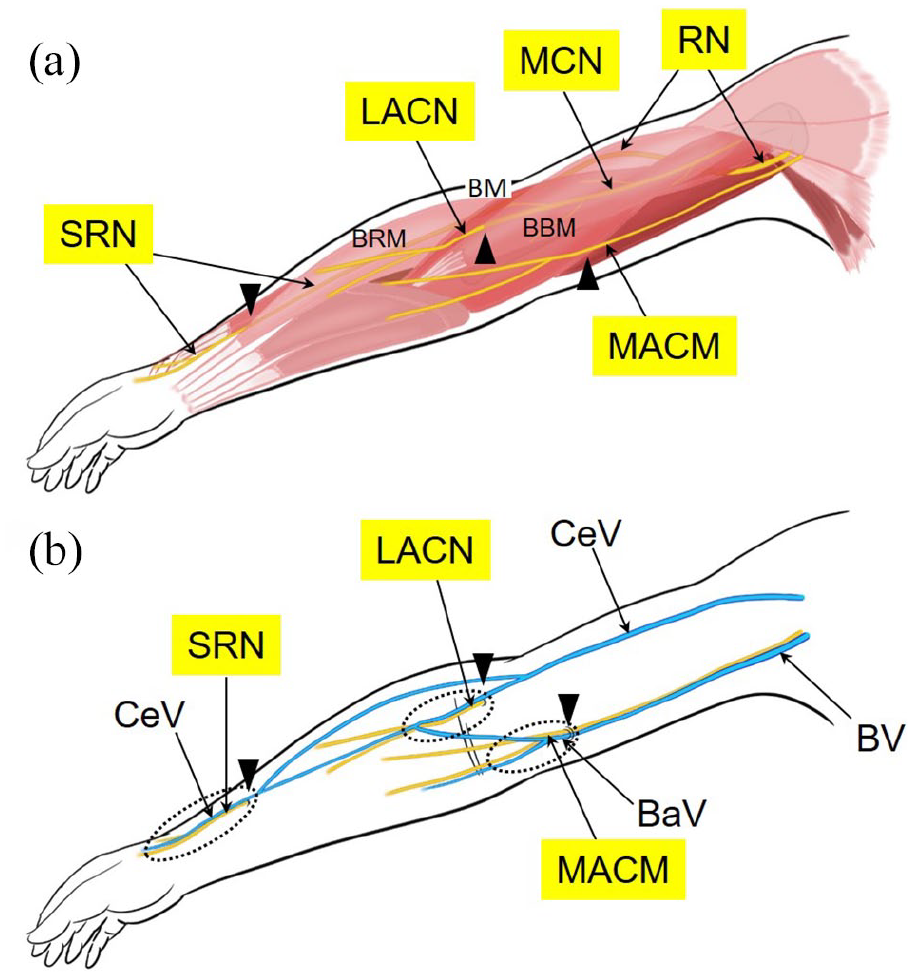
Anatomical relation of the cutaneous nerves to the adjacent muscles and superficial veins in the upper limb: (a) Topographical anatomy of the cutaneous nerves in relation to the muscles, (b) Topographical anatomy of the cutaneous nerves in relation to the superficial veins. The RN runs as the trunk of the SRN, after the posterior interosseous nerve branches off at the elbow, distally beneath the BRM and alongside the radial artery in the forearm and arises into the subcutaneous tissue in the distal forearm. It courses in the wrist beside the CeV and splits into arborized branches innervating the dorsum of the hand. The MCN passes between the BBM and the BM, providing motor innervation to the muscles. It pierces the fascia lateral to the biceps brachii tendon at the elbow, continues as the trunk of the LACN, just beneath the median CeV in the antecubital fossa, and branches over the anterolateral forearm. The trunk of the MACN at the axilla runs distally in the anterolateral side of and parallel to the BV and the BaV. It pierces the superficial fascia with the BaV in the distal upper arm and courses subcutaneously along it and arborizes over the anteromedial forearm. Dotted circles indicate the regions where each cutaneous nerve trunk runs adjacent to each superficial vein. Arrowheads indicate the sites where each cutaneous nerve arises to the subcutaneous space.
Preoperative US examination of the cutaneous nerves based on their anatomical landmarks
In addition to the functional and morphological examination of the VA in the upper limbs, the ipsilateral cutaneous nerves were continuously scanned in the short-axis view along the upper limbs using a linear 5- to 18-MHz transducer for real-time US guidance (HI VISION Avius; Hitachi Aloka Medical Ltd., Tokyo, Japan), as described in previous studies.4,5 In cases of wrist aneurysms, the SRN trunk was predominantly examined, and the branches of the LACN were assessed as backup. In cases of antecubital and distal upper arm aneurysms, the LACN trunk was mainly examined and the MACN trunk was additionally assessed. The SRN and/or LACN trunk were examined in all patients because the AVFs outflowed through the CeV alongside the SRN in the wrist and LACN at the antecubital fossa. The cutaneous nerve trunks adjacent to the aneurysms that would be resected were preoperatively visualized, considering their anatomical relations to their neighboring landmarks. The nerve trunks were traced from the regions proximal to the aneurysms because it was difficult to image the nerves—in particular, the branches arborizing from the trunk—at the region of the aneurysms as they were flattened and kept away from the skin surface. The proximal landmarks of the SRN trunk scan were (1) the brachioradial muscle and radial artery, beneath and parallel to which the SRN ran in the proximal forearm and mid-forearm and (2) the CeV, parallel to which it passed in the wrist. The landmarks of the LACN trunk scan were (1) the biceps brachii tendon, lateral to which the musculocutaneous nerve (MCN) passed and emerged as the LACN trunk subcutaneously above the elbow and (2) the CeV, parallel to which the LACN coursed at the antecubital fossa. The MACN scan landmark was the BaV, alongside which it ran in the upper arm. The supplementary video shows a representative landmark-based US tracing of the SRN trunk and LACN trunk in a healthy volunteer (Supplemental Videos 1 and 2).
Surgical procedures
The cutaneous nerve trunks, which had been preoperatively identified by US, were visually recognized at the regions proximal to the aneurysms using 3.5 × loupe magnification. After being separated from the preserved adjacent nerves, the aneurysms were resected from the proximal to the distal ends. We assessed the preoperative US images of the cutaneous nerve trunks and branches accompanying the aneurysm as well as their visibility and preservability during repair surgeries.
A single surgeon (H.M.) with more than 15 years of experience in VA surgery performed all except for two operations, in which he was involved as a supervisor. He also performed the preoperative US examination of the nerves and administered US-guided regional anesthesia.
Results
Clinical results are presented in Table 2.
Clinical results.
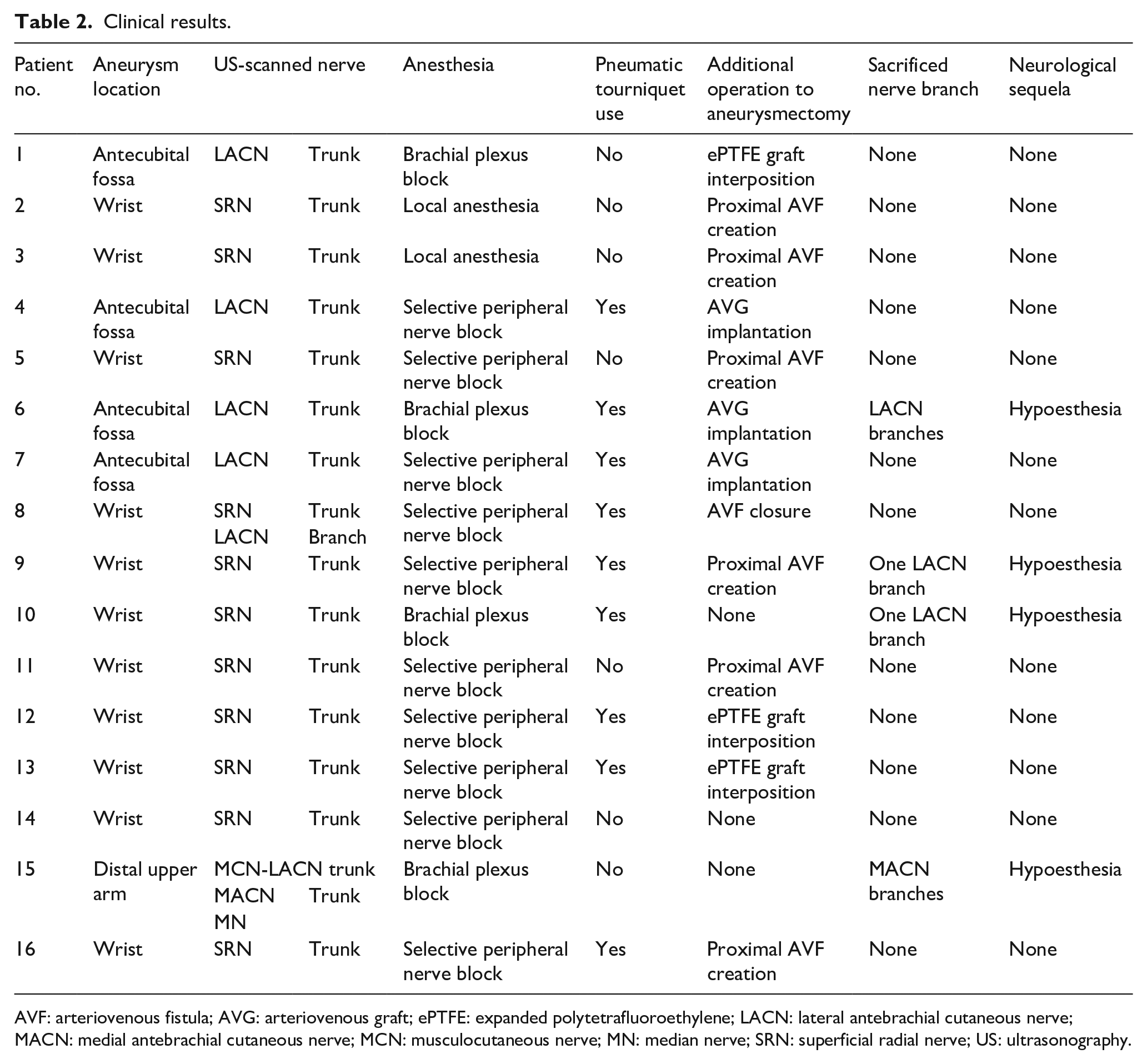
AVF: arteriovenous fistula; AVG: arteriovenous graft; ePTFE: expanded polytetrafluoroethylene; LACN: lateral antebrachial cutaneous nerve; MACN: medial antebrachial cutaneous nerve; MCN: musculocutaneous nerve; MN: median nerve; SRN: superficial radial nerve; US: ultrasonography.
Regional anesthesia, including the brachial plexus and peripheral nerve block that deprived sensory innervation to the operative fields, was used in all except for two patients in whom local anesthesia was administered. Pneumatic tourniquets were used in nine patients. Aneurysmectomy was performed in all patients, and VA reconstructions were added in 12 patients.
Aneurysm at the wrist
The SRN trunk was visualized by preoperative US and spared in all 11 patients with wrist aneurysms. In eight of them, the SRN was intraoperatively identified under a loupe magnification; in other patients, it was conserved without being exposed in the operative field. An instructive aneurysmectomy with additional proximal AVF creation is shown in Figure 2 (patient 16).
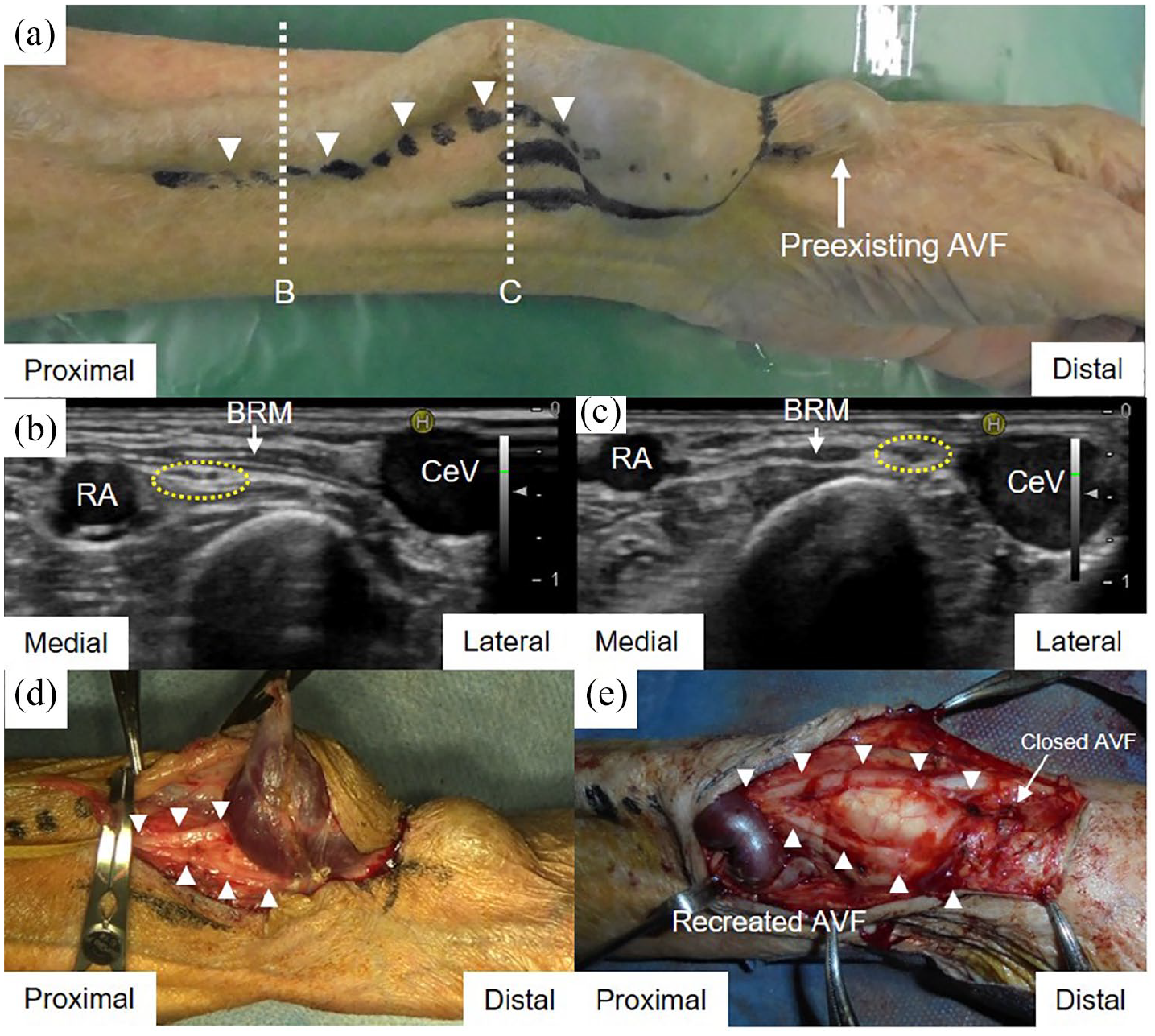
Surgical repair of the aneurysm at the left wrist: (a) A line was marked on the skin proximal to the aneurysm along the superficial radial nerve after the ultrasonographic scan (arrowheads). Dotted lines B and C indicate cross-sectional levels of the ultrasonographic scan that are shown in Figure 2(b) and (c), (b) At the level of the proximal one-third of the forearm, the superficial radial nerve (encircled by yellow dotted line) ran deeply beneath the BRM and parallel to the RA away from the CeV, (c) More distally, the superficial radial nerve (encircled by yellow dotted line) arose lateral to the BRM in the subcutaneous tissue and approached the CeV, (d) The aneurysm was dissected from the proximal to the distal end for primary recognition of the superficial radial nerve trunk (arrowheads) branching peripherally, and (e) After aneurysmectomy with the proximal AVF recreation, the superficial radial nerve (arrowheads) was spared without any injury.
In patient 8 with a wrist aneurysm extending to the dorsal mid-forearm, the LACN branches that had been scanned by US were preserved. They were isolated from the aneurysm under visualization, and the SRN was spared without being exposed. In patients 9 and 10, a small peripheral LACN branch that had not been preoperatively scanned was sacrificed after deliberation for a seamless aneurysmal excision. The patients experienced hypoesthesia in a small region at the volar lateral wrist innervated by the transected branch; however, neuropathic pain was not encountered.
Aneurysm at the antecubital fossa
The LACN trunk, lateral to the biceps brachii tendon, was scanned by preoperative US in all patients and was retained in three patients under visualization. A representative procedure is shown in Figure 3 (patient 4).
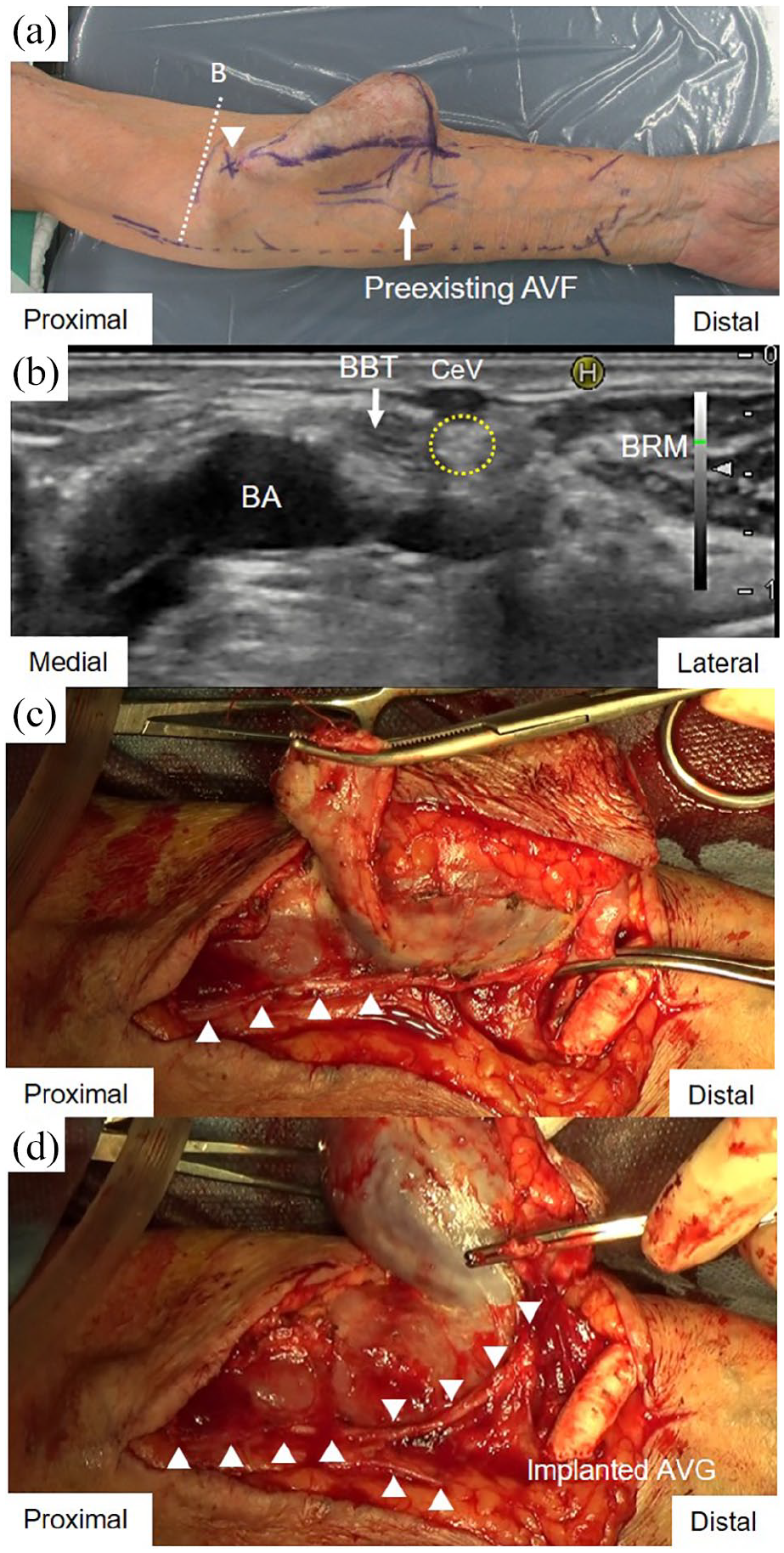
Surgical repair of the aneurysm at the left antecubital fossa: (a) The skin at the intersection between the lateral antebrachial cutaneous nerve and the outflow communicating branch of the deep vein was marked after ultrasonographic evaluation (arrowhead). Dotted lines B indicate cross-sectional levels of the ultrasonographic scan that are shown in Figure 3(b), (b) Above the elbow, the lateral antebrachial cutaneous nerve (encircled by yellow dotted line) arose subcutaneously, lateral to the BBT beneath the CeV, (c) After implanting the radiobasilic AVG in the forearm, the aneurysm was resected from the proximal to the distal portion, with the trunk of the lateral antebrachial cutaneous nerve (arrowheads) visually separated from it, and (d) The branches bifurcating from the trunk of the lateral antebrachial cutaneous nerve were preserved through the whole length (arrowheads).
On the contrary, the LACN branches distal to the LACN trunk in patient 6 were inadvertently transected, and the proximal nerve ends were translocated into the brachioradial muscle to avoid the formation of painful end-neuromas (Figure 4).
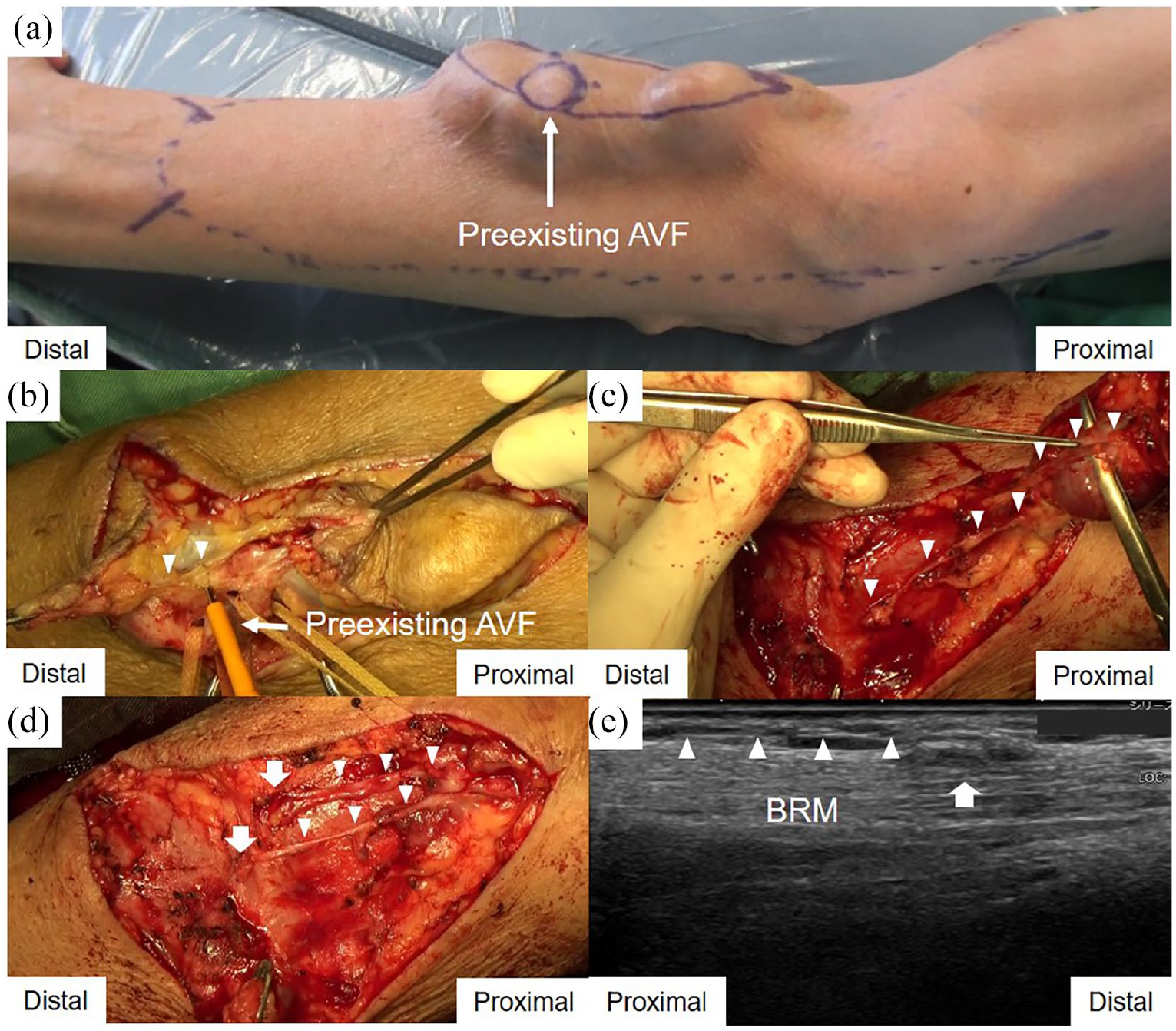
Surgical repair of a right antecubital venous aneurysm in the distal-to-proximal direction: (a) Aneurysm expanding distally and proximally from the radiocephalic AVF at the right antecubital fossa, (b) Aneurysmectomy from the distal to the proximal segment caused inadvertent transection of the lateral antebrachial cutaneous nerve branch (arrowheads). This is a still image just before its transection extracted from the intraoperative video, (c) The transected proximal branches (arrowheads) were separated from the aneurysm, which was being resected toward its proximal edge, (d) Nerve relocation into the muscles. The distal nerve ends were resected with the aneurysm, as severed nerve repair was not feasible. The proximal ends (arrows) of the transected nerve branches (arrowheads) were buried into the BRM, and the epineurium of each nerve branch was sutured to the epimysium of the muscle using three 7-0 polypropylene sutures to avoid the formation of painful neuromas at its ends, and (e) The proximal nerve end that was translocated into the muscle formed a small neuroma (arrow), which was imaged as a low echoic intramuscular nodule continuing from the proximal nerve branch (arrowheads) in a long-axis view at 3 years postoperatively.
At 3 years postoperatively, hypoesthesia in the ipsilateral anterolateral forearm persisted; however, neuropathic pain was absent even if end-neuromas were sonographically identified in the muscle.
Aneurysm in the distal upper arm
In patient 15, a pseudoaneurysm at the brachial artery in the distal upper arm was detected at the puncture site of an introducer sheath used during an unsuccessful endovascular treatment at 1 month prior to aneurysmal repair. It was observed to extend between the biceps brachii muscle and the brachial muscle and compressed the MCN, MACN, and median nerve. The aneurysmal entry site was sutured, and blood and clot inside the aneurysmal sac were removed with the MCN trunk covertly preserved outside the sac and with the distal MACN branches intentionally sacrificed for a smooth surgical process. The patient suffered from hypoesthesia in the proximal anteromedial forearm postoperatively and had no complaints of neuropathic pain.
Other postoperative complications did not occur. None of the patients experienced hypoesthesia in addition to that caused by the injured nerve. The preoperative issues of patients were resolved after surgeries.
Discussion
In the fields of orthopedic surgery and neurosurgery, injuries to the cutaneous nerves during operations of the hand and upper extremity are frequently reported. The focus is on the education of nerve anatomy to avoid injury and the management of painful neuromas caused by the injuries.7,9–11
In addition to very few reports on nerve injuries after VA surgery, severe nerve lesions are also considered rare despite their proximity to vessels in VA surgery of the upper limb.1,3 However, Brittinger reported that 5% to 20% of patients with radiocephalic AVF at the wrist suffered from the lesions of the SRN or LACN. The irritation of the superficial palmar branch of the ulnar nerve in patients with ulnar artery AVF in the distal forearm and the lesions of the MACN in patients with BaV AVF in the upper arm were recorded as benign lesions. 2 Therefore, the injuries involving the sensory cutaneous nerves, which usually run close to the cutaneous runoff vein, are underreported because sensory deficits caused by their injuries are considered trivial and underestimated in comparison to motor deficits caused by the lesions of the median, ulnar, and radial nerve (RN) trunks. 3
VA-related venous aneurysms develop subcutaneously in contact with the sensory cutaneous nerves and compress it more than a non-arterialized superficial vein. To our knowledge, there are a few case reports of repair surgery for preoperative neurological symptoms that affected the motor as well as the sensory perception of the median nerve and the RN.12–14 Among studies on the surgical management of VA-related aneurysms, Furukawa reported that five out of 26 patients who underwent surgical revisions of VA-related aneurysms had complications of minor peripheral neuropathy; however, other reports had not described postoperative neuropathy.15–17 Therefore, there is a possibility that surgeons were unlikely to be concerned about intraoperative cutaneous nerve preservation and postoperative cutaneous neuropathy because venous aneurysms rarely induced preoperative neurological symptoms. In contrast, VA-related aneurysms of the brachial artery have been reported to compress the median nerves, leading to compartment syndrome; thus, they were cautiously repaired.18,19
High-resolution nerve US has become an established technique for the study of peripheral neuropathies, which include entrapment, tumoral, posttraumatic, and postsurgical injuries, and is increasingly used for diagnosis and surgical planning. 20 Hence, to preserve the nerve, we instituted nerve US to examine the anatomical relationship of VA-related aneurysm with the neighboring cutaneous nerve before its repair. The evolution of nerve scanning techniques also influenced the use of US-guided selective peripheral nerve block as anesthesia for VA surgery.4,5
We demonstrated that, depending on the site of the aneurysm, the target cutaneous nerve trunk could be identified by preoperative US examination. The US identification of the peripheral nerves, for which an understanding of their topographical anatomy is prerequisite, could contribute to nerve-sparing surgery as a complementary procedure. Cartwright et al. examined reference values for the cross-sectional area of the peripheral nerves, which were measured using US. They reported that the cross-sectional area (disclosed as mean ± standard deviation) of the RN at the antecubital fossa was 9.3 ± 2.4 mm2, that of the MCN in the upper arm was 6.9 ± 2.5 mm2, and that of the vagal nerve was 5.0 ± 2.0 mm2. 21 Although a high-resolution US was capable of visualizing such small peripheral nerves, their anatomical landmarks in US scanning could further expedite their depictions as per Gruber et al. 22 In the present study, the cutaneous nerve trunks localized proximal to the aneurysms were identified according to their landmarks in all patients. In contrast, it was difficult to visualize the peripheral branches that split from the trunks with few exceptions, because the thinner branches peripherally coursed away from the landmark superficial outflow veins.
This study made us realize some key techniques for preserving the cutaneous nerves during aneurysmal resection. First, the important step was the direction of resection of the aneurysm. The aneurysm was resected from the proximal to the distal end after confirming the location of the cutaneous nerve trunk proximal to the aneurysm. Successful excision without nerve injury was performed. Its excision in the distal-to-proximal direction posed the risk of severing nerve branches in the middle. In a case of distal-to-proximal resection of a venous aneurysm in the antecubital fossa, two LACN branches in the mid-forearm were transected accidentally. Other techniques, including aneurysmal resection without exposing the nerve, dissecting the subcutaneous tissue deliberately and longitudinally so as to not cross the nerve path, and avoiding electrocautery that caused thermal nerve damage, may help reduce nerve lesions.1,9,11 Medical devices such as pneumatic tourniquet could help increase the visibility of the cutaneous nerve in bloodless operative fields, in addition to loupe magnification.4,11
The clinical significance of preventing cutaneous nerve injury is to avoid postoperative neuropathies, including sensory deficits and potential neuropathic pain caused by neuroma formation at an injured nerve. In our study, four patients had severed single or multiple cutaneous nerve branches and complained of postoperative hypoesthesia depending on the number and site of the impaired nerve branch. The more peripheral and fewer the severed nerve branches, the narrower the extent of hypoesthesia. No patients have complained of neuropathic pain thus far. One of them underwent a preventive relocation of the proximal nerve end into a local muscle to avert the formation of painful end-neuromas. 23 Neuropathic pain, defined as pain resulting from a lesion or disease in the central or peripheral nervous system by the International Association for the Study of Pain, is also caused by peripheral nerve injury, which could form neuromas.9,23 Persistent pain arising from an end-neuroma, which occurs at the proximal transected nerve end, occurs in 3% to 5% of patients with peripheral nerve injury.9,23 Therefore, we speculate that neuropathic pain due to surgical nerve injury following VA surgery may be latent in hemodialysis patients. One widespread method to deal with a transected proximal nerve that could not be repaired includes its transposition into an appropriate muscle. This was considered to prevent immature regeneration into the skin, minimize scar tissue formation, and reduce its susceptibility to mechanical stimulation.23,24 The most important step in their prevention is to avoid intraoperative nerve injury because even minor damage to the most peripheral nerves in the digits could lead to neuropathic pain.9,23,25 VA surgery should ensure the preservation of the SRN, LACN, and MACN trunks to avoid the risk of extensive sensory blunting and potential neuropathic pain. If the trunk were transected, we recommend nerve suture or proximal nerve-end translocation to a local muscle under loupe magnification based on recommendations from orthopedic and neurosurgical articles.9–11,23–26
Our study has some limitations. First, the number of enrolled patients was small. Second, the dearth of scholarly articles relevant to VA surgery-related neuropathies made us evaluate our clinical results with extensive orthopedic and neurosurgical experiences.7,9–11,20,23–26 Thus, we should promote nerve-conscious VA surgery by drawing attention to the neighboring cutaneous nerves. We expect that this would enhance uncomplicated and favorable outcomes of VA surgery in patients on hemodialysis.
Conclusion
Preoperative US examination of the neighboring cutaneous nerves proximal to the aneurysm, based on their anatomical landmarks, followed by the proximal-to-distal resection of the aneurysm could avoid intraoperative nerve injuries. These steps could help refine the resection of VA-related aneurysms to minimize the incidence of postoperative neuropathy.
Footnotes
Author contributions
All authors discussed the results and contributed to the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This report was approved by the patients, who provided written informed consent for publication.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
