Abstract
We report a new technique called “reimplantation of an artery with a hairpin turn (RAHT)” to reduce excessive vascular access flow. A 73-year-old woman on dialysis consulted us for vascular surgery because of an increased cardiac preload. Chest radiography and echocardiography revealed an excessive shunt flow in the brachial artery (flow rate, 2336 mL/min). Vascular echo-Doppler of the left upper limb showed that the radial artery made a hairpin turn at the arteriovenous fistula (diameter, 9 mm). Diameters of the radial artery proximal and distal to the arteriovenous fistula were 5.4 and 3.7 mm, respectively. We ligated and divided the juxta-anastomosis proximal radial artery and subsequently created an end-to-side anastomosis between the proximal radial artery and the distal radial artery. The anastomosis ostium in the distal radial artery (the recipient) was formed with a 4-mm longitudinal and gently curved incision. We performed RAHT so that the small anastomosis between both arteries and the small diameter of the distal radial artery juxta-anastomosis segment could reduce the vascular access flow. The flow rates in the brachial artery were 500 mL/min just after surgery and 560 mL/min at 2 months after surgery. Postoperative chest radiography and echocardiography confirmed a decrease in cardiac preload. We believe that this RAHT technique could be useful as one of the options to reduce the flow in patients who have excessive vascular access flow with a radial artery that makes a hairpin turn.
Keywords
Introduction
Because the radiocephalic arteriovenous (AV) fistula has a long-term patency, there are cases in which the long-term development of vascular access results in high flow. There is a risk of high-output heart failure when the vascular access flow rate significantly increases to over 1500–2000 mL/min or the flow volume/cardiac output ratio increases to over 30%–35%. 1 In the present case report, the dialysis patient had undergone surgery for an AV fistula in her left wrist 13 years before. As a result of continued patency of the vascular access for a long time, the vascular access blood flow continued to increase gradually and became excessive. In the process, the radial artery around the anastomosis continued to bend and developed a hairpin turn. To prevent heart failure progression, blood flow had to be reduced. To reduce the vascular access flow, we reimplanted the radial artery that made a hairpin turn in the following way: we ligated and divided the juxta-anastomosis proximal radial artery and subsequently created an end-to-side anastomosis between the proximal radial artery and the distal radial artery. We describe “reimplantation of an artery with a hairpin turn (RAHT)” as a new technique to reduce excessive vascular access flow.
Case description
A 73-year-old woman, who started dialysis because of chronic renal failure secondary to chronic glomerulonephritis 13 years previously, consulted us for vascular surgery to correct a continuously worsening cardiothoracic ratio and echocardiographic preload that were detected by her dialysis physicians. A radial artery–cephalic vein shunt was constructed in the left forearm 13 years before. On preoperative examination, chest radiography revealed heart enlargement with a cardiothoracic ratio of 57.2%, and echocardiography showed mild tricuspid regurgitation (TR), mitral regurgitation (MR), and a TR pressure gradient of 25 mm Hg. Vascular echo-Doppler indicated that the preoperative flow rate in the brachial artery was 2336 mL/min. The absence of ischemic findings in her fingers suggested that steal syndrome was not present.
On visual inspection of the left upper limb, the radial artery made a hairpin turn at the AV fistula. The AV fistula was 9 mm in diameter, and no stenosis was observed. The diameters of the radial artery proximal and distal to the AV fistula were 5.4 and 3.7 mm, respectively (Figure 1). For initial restoration treatment, she requested the utilization of the current vascular access route without intentional occlusion and wished to avoid surgery using vascular prostheses. Following a thorough discussion with the vascular surgeon and colleagues, the patient provided consent for treatment with the RAHT technique.
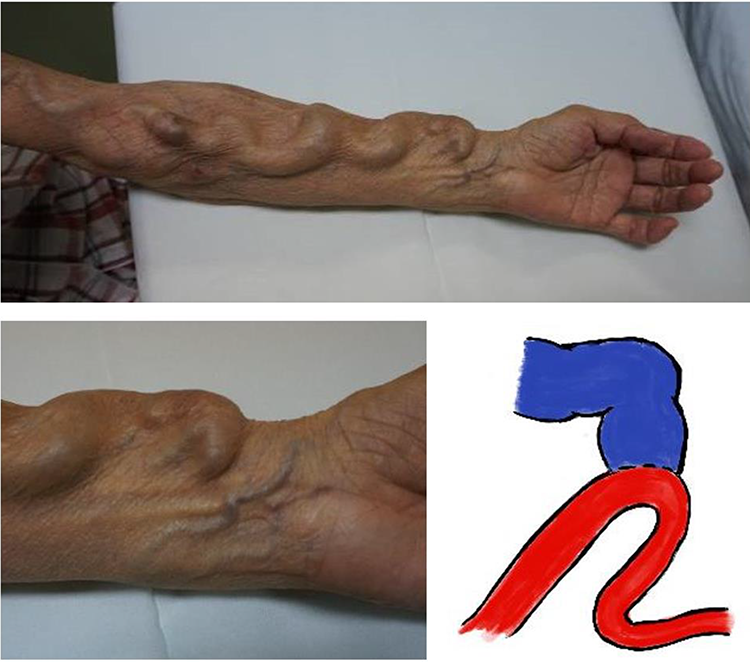
The findings of the left upper limb show that the radial artery makes a hairpin turn at the arteriovenous (AV) fistula.
The operation was performed with the patient under local anesthesia using a total volume of 110 mg of 1% lidocaine with a 2.5-cm longitudinal skin incision just above the radial artery that made a hairpin turn near the AV fistula. We controlled the radial artery at the proximal and distal ends of the AV fistula. Following intravenous administration of heparin (1500 units), the radial artery was dissected just above the AV fistula. We ligated and divided the juxta-anastomosis proximal radial artery and subsequently created an end-to-side anastomosis between the proximal and the distal radial artery using a single 6-0 polypropylene suture. The anastomosis ostium in the distal radial artery (the recipient) was formed with a 4-mm longitudinal gently curved incision. The radial artery between the former AV fistula and the new arterial end-to-side anastomosis was 15.1 mm in length (Figure 2). Intraoperative vascular echo-Doppler showed that the flow rate in the brachial artery after revision was 500 mL/min. The operation lasted 93 min, and the volume of blood lost was 10 mL.
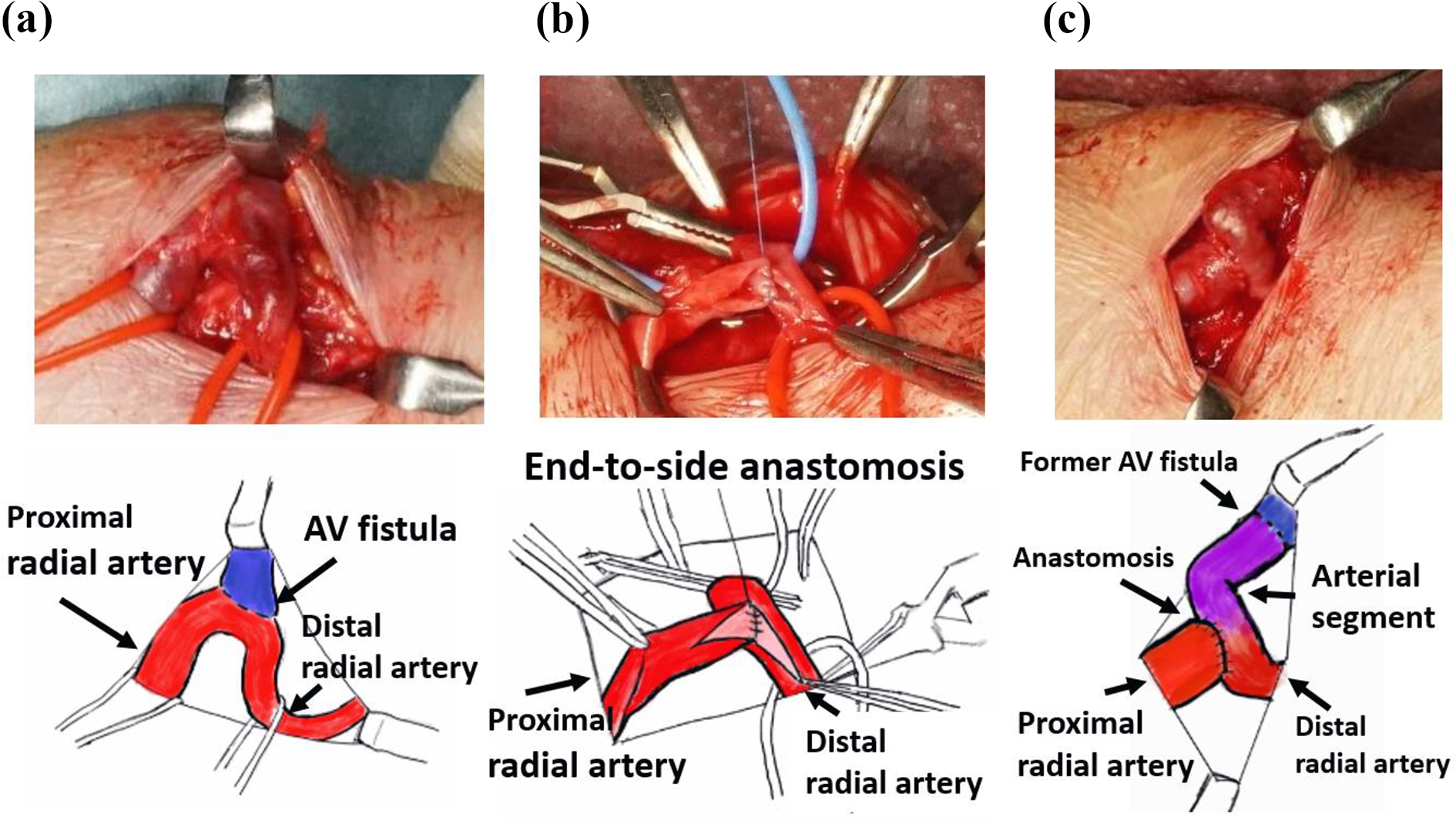
Surgical finding of reimplantation of an artery with a hairpin turn (RAHT). (a) A radial artery made a hairpin turn at the arteriovenous (AV) fistula. The AV fistula is 9 mm in diameter, and the radial artery diameters proximal and distal to the AV fistula are 5.4 and 3.7 mm, respectively, (b) end-to-side anastomosis between the proximal radial artery and the distal radial artery after dividing the juxta-fistula proximal radial artery, and (c) the final schema of the RAHT technique. The radial artery (indicated by purple in the figure on the right) between the former AV fistula and the new arterial anastomosis is used as the arterial segment with a distance of 15.1 mm.
Two months after the operation, vascular echo-Doppler showed that the flow rate in the brachial artery was 560 mL/min and the diameter of the radial artery between previous AV fistula and new anastomosis was 4.0 mm (Figure 3). The cardiothoracic ratio decreased to 52%, and echocardiography showed an improvement in TR and MR and a decrease in TR pressure gradient to 18 mm Hg, suggesting a decrease in the cardiac preload.
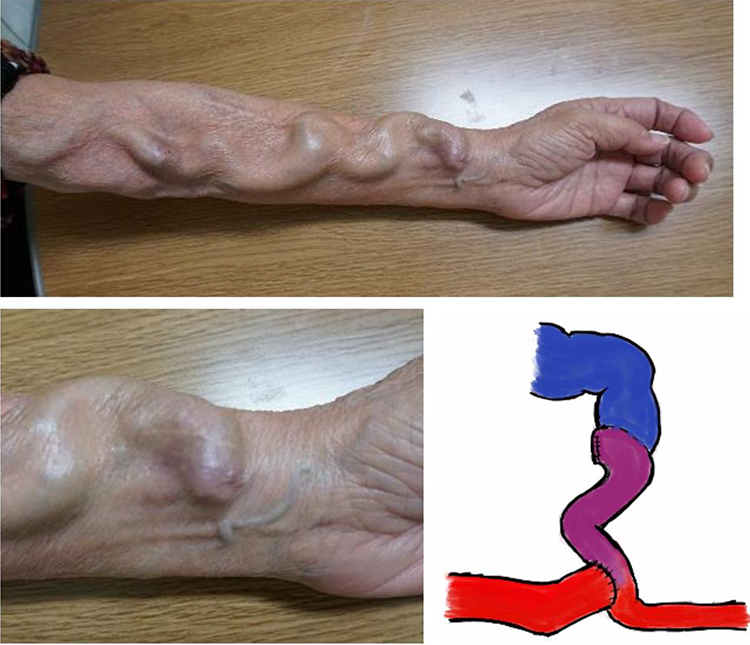
Findings of the left upper limb at 2 months after the operation. The arterial segment in purple color (lower and right) was at the beginning part in the new vascular access route. The diameter of the arterial segment was 4.0 mm.
Discussion
In this case, the AV fistula with excessive vascular access flow was located at the apex of the radial artery that made a hairpin turn near the wrist. We dissected the radial artery at the proximal end of the AV fistula and revised the cut edge of the dissected radial artery to the side of the non-dilated radial artery distal to the AV fistula. The concept of the RAHT technique is that the non-dilated arterial segment of the distal radial artery between the previous AV fistula and the new anastomosis between arteries reduce the vascular access flow. The surgical procedure needs one anastomosis, and the caliber difference of the radial artery proximal and distal to the AV fistula was not a major problem owing to the end-to-side anastomosis. Postoperative chest radiography and echocardiography confirmed a reduction in cardiac preload at least 2 months after surgery.
Several techniques have been developed to reduce excessive vascular access flow in the radiocephalic AV fistula region, such as venous banding, arterial banding/ligation, artificial blood vessel replacement surgery, re-anastomosis, among others. 2 The proximal radial artery ligation (PRAL) method, a simple procedure for reducing the excessive vascular access flow, is reported to have excellent blood flow reduction and patency rates. 3 However, in this case, the ligation of a healthy radial artery was a psychological burden for the surgeons and the patient. Therefore, we devised and performed the RAHT technique as the first stage of the surgery. Because reduction surgery for the excess vascular access flow would often cause the flow to increase eventually in the case of no obstruction, the PRAL method could be selected as a second or subsequent step when the blood flow increased again after RAHT. When considering multi-step surgery, it seems reasonable that complex procedures should precede simpler procedures because the adhesions and tissue scarring that occur in every surgery are expected to make subsequent surgery more difficult. PRAL is contra-indicated in case of poor retrograde blood flow from the ulnar artery and palmar arch, as demonstrated by positive Allen test and echo-Doppler; in such conditions, the RAHT technique would probably be applicable.
The advantage of the arterial segment in a vascular access was that it is less prone to expand chronically than a vein and could be expected to keep reducing the vascular access flow for a long time. Compared to prostheses, arterial segments are prone to dilation. However, in this case, the anastomosis between arteries was continuously sutured with a single polypropylene thread. Since the polypropylene thread does not stretch significantly, the anastomosis does not expand excessively. Therefore, the anastomosis ostium was expected to reduce the blood flow for a long time. In addition, in procedures requiring small-diameter conduits and anastomosis as represented by coronary artery bypass surgery, autologous arteries and veins have better patency rates than artificial grafts. 4 In the RAHT technique, a similar phenomenon is expected.
Concerning the mechanism by which the RAHT technique reduced the vascular access flow, we inferred that the end-to-side anastomosis ostium and subsequent arterial segment played an important role. Nojima et al. 5 reported that the mean length of 4-mm-diameter artificial grafts implanted at the anastomoses of high-flow AV fistulas was 41 mm, leading to a mean flow reduction rate of 39%. In our case, the length of the arterial segment was 15.1 mm, which reduced the blood flow of the brachial artery from 2336 to 500 mL/min (21%). This difference seemed to be attributed to the cross-sectional area of the anastomosis. Compared to autologous arteries and veins conduits, artificial grafts cannot form smaller diameters or smaller anastomoses because of graft failure caused by thrombus and intimal thickening. 6 In this case, the anastomosis ostium as the recipient was formed by a 4-mm longitudinal and unilateral gently curved incision on the side of the distal radial artery, which is smaller than the 4-mm round anastomosis formed by Nojima et al.’s artificial graft technique. The RAHT technique could reduce blood flow through a small anastomosis that is difficult to create with artificial grafts. With regard to the blood flow reduction effect of the conduit, Minion et al. 7 pointed that the diameter and length of the vessel providing resistance followed Poiseuille’s law. In this case, the diameter of the radial artery of the distal part of AV fistula, which we used as an arterial graft, was 3.7 mm and 0.68 times that of the artery of the proximal part of the AV fistula. As for the length of the segment to reduce the flow, it was defined that banding treatment could sufficiently adjust the vascular access flow even in a short section of approximately 5 mm. 8 Determining the optimal length of an arterial segment in RAHT technique requires a large number of cases, which is a task for the future.
This RAHT technique has limitations, which makes it difficult to be adopted in cases without a meandering radial artery. However, there are occasional cases in which the radial artery is exposed to prolonged high flow and meanders. Furthermore, in previous operations, the incision of the fascia just above the AV fistula and the retraction of the artery at the AV fistula by the superficial vascular access vein were supposed to promote radial arterial flexion, eventually leading to a hairpin turn at the AV fistula. In such cases, the RAHT technique is expected to be a useful treatment option.
Conclusion
The RAHT technique has four characteristic points: first, the autologous arterial segment located in the beginning part of the vascular access; second, one end-to-side anastomosis between the arteries to enable small anastomosis; third, the availability for both Allen test positive and negative patients; and finally, preservation of the run-off vein length required for needle punctures during hemodialysis. We conclude that this RAHT technique could be useful as one of the options to reduce the flow for patients who have excessive vascular access flow with a radial artery that makes a hairpin turn.
Footnotes
Acknowledgements
The authors are grateful to Dr Norihide Sugano (Tokyo Metropolitan Health and Hospitals Corporation Ohkubo Hospital) for his kind support and insightful advice in this article.
Author contribution
S.K. conceived and designed the study, performed the analysis, and prepared the manuscript. All authors discussed the results and contributed to the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This report was approved by the patient, who provided written informed consent for publication.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
