Abstract
Background
Pediatric Thyroid Eye Disease (TED) is a rare autoimmune condition primarily associated with Graves’ disease. Although usually milder than adult TED, it can still cause functional and psychological morbidity. Pediatric management remains unclear due to the absence of specific guidelines and concerns about treatment-related risks such as growth suppression.
Methods
We conducted a systematic review and meta-analysis following PRISMA guidelines, searching MEDLINE, Embase, and Emcare from inception to March 2024. Studies included interventional and observational reports involving patients ≤18 years with TED. Data were synthesized narratively and quantitatively. Meta-analyses were conducted using random-effects models, with heterogeneity assessed via I2 statistics and meta-regression. Primary outcomes included visual acuity (VA), proptosis, Clinical Activity Score (CAS), and adverse events.
Results
Thirty-two studies comprising 810 pediatric patients (mean age 11.7 years, 64.8% female) were included. The most common symptoms were exophthalmos (99.5%), eyelid retraction (73.1%), and dry eye (66.3%). Treatments ranged from antithyroid drugs and corticosteroids, to orbital decompression and biologics. Meta-analysis showed mean exophthalmos reductions of 4.69 mm for decompression, 4.25 mm for steroids, and 1.75 mm for biologics. Substantial heterogeneity and low certainty of evidence limited interpretability. Interventions were performed earlier than recommended, with no significant adverse effects reported.
Conclusions
Despite most pediatric TED cases being mild, a subset of patients requires more intensive management. This review, comprising predominantly of case reports and case series with very low certainty evidence, reveals gaps between practice and recommendations, highlighting the need for pediatric-specific guidelines informed by systematic evidence.
Introduction
Pediatric Thyroid Eye Disease (TED) is a rare manifestation of autoimmune thyroid disorders in children, most commonly associated with Graves’ disease (GD). The incidence of TED in children is 0.79 cases per 100,000 children yearly. 1 Prevalence among children with Graves’ disease is 0.25%, peaking between 10 and 15 years of age.2,3 Similar to adults, there is a strong female predominance. 2
Pediatric TED typically presents with milder ocular signs compared to adults, including eyelid retraction (23–91%) and proptosis (4–92%). 4 Radiological images show extraocular muscle enlargement and orbital fat proliferation in children with TED. 5 Unlike adults, children rarely have restrictive myopathy or optic neuropathy. Despite its milder course, TED in children can have a profound psychological impact due to cosmetic disfigurement and social challenges. 6 Furthermore, children tend to present with behavioral issues of hyperactivity, impaired school performance and nocturnal enuresis. 7
Management strategies for TED differ between adult and pediatric populations due to differences in presentation, severity and treatment effects. In adult populations, the 2021 European Group on Graves Orbitopathy (EUGOGO) consensus statement categorizes disease severity and encompasses lifestyle changes and localized and systemic treatments, like radiotherapy, surgical intervention, steroid therapy, biologics, and early risk factor modifications. 8 However, pediatric-specific severity classifications have not been formally validated. In clinical practice, adult frameworks are often extrapolated. Mild pediatric TED typically presents with eyelid retraction, soft tissue signs, and mild proptosis without functional impairment. Moderate-to-severe disease may include progressive proptosis, inflammatory lid edema, diplopia, or visual field defects. Sight-threatening disease, although uncommon, includes compressive optic neuropathy (CON) or severe corneal exposure. The absence of pediatric-specific severity criteria contributes to variability in management approaches.
The current pediatric guidelines (such as the 2022 European Thyroid Association (ETA) guideline for the management of pediatric Graves’ disease) are lacking large-scale trials and are rather focused on restoring euthyroidism alone, given the milder course of disease in pediatric patients. 4 Compared to adult TED, pediatric disease is generally less inflammatory, with fewer cases of restrictive myopathy or CON. 9 Conservative management is the recommendation for pediatric TED treatment, given the potential effects of systemic treatment in youth, such as the association between systemic corticosteroids with growth suppression and systemic side effects, 4 orbital radiotherapy with risks of potential secondary malignancies, and surgical outcomes being affected by ongoing development in children. 10 In short, there is a striking discrepancy between the structured guidelines for mild, moderate, and severe diseases in adult populations and the conservative approaches in pediatric populations. Establishing evidence-based guidelines for pediatric TED is crucial to ensure consistent care and safe escalation of treatment through proper evaluation of existing studies, especially given the current lack of guidance for managing cases that require more than conservative intervention. A systematic review synthesizing the currently fragmented evidence base for pediatric populations is thus the initial step in highlighting areas where pediatric presentations diverge from adults. This review aims to summarize the existing management strategies for pediatric TED to provide a better understanding of the presentations, management and outcomes for pediatric TED.
Methods
A systematic review was conducted following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statements. 11
Inclusion and exclusion criteria
This systematic review included interventional studies, such as randomized controlled trials, and observational studies, including cohort studies, case-control studies, case series and case studies. We included peer-reviewed primary studies that investigated treatments delivered to pediatric patients (18 years old and younger) with a primary diagnosis of TED. The studies described treatments addressing exophthalmos, such as steroids or other therapies. There were no restrictions on the country or publication language of the studies.
Studies were excluded if they involved non-human subjects, in vitro research, more than 50% of their population were above 18 years old, their data could not be reliably extracted, they contained duplicate or overlapping data, or they were literature reviews, systematic reviews, abstract-only papers, conference proceedings, editorials, theses, letters to the editor, or books.
Search strategy and sources
A comprehensive search strategy was developed in collaboration with a librarian at McMaster Health Sciences Library (J.R.). The searches included all the Medical Subject Headings (MeSH) terms that refer to TED and pediatrics, and was conducted in Medline, Embase, and Emcare on March 14, 2024 (Appendix A). The electronic search was supplemented by hand-searching the reference lists from included studies. Captured citations were exported to Covidence (Veritas Health Innovation, Melbourne, Australia) for further screening. Figure 1 displays the detailed PRISMA flowchart.
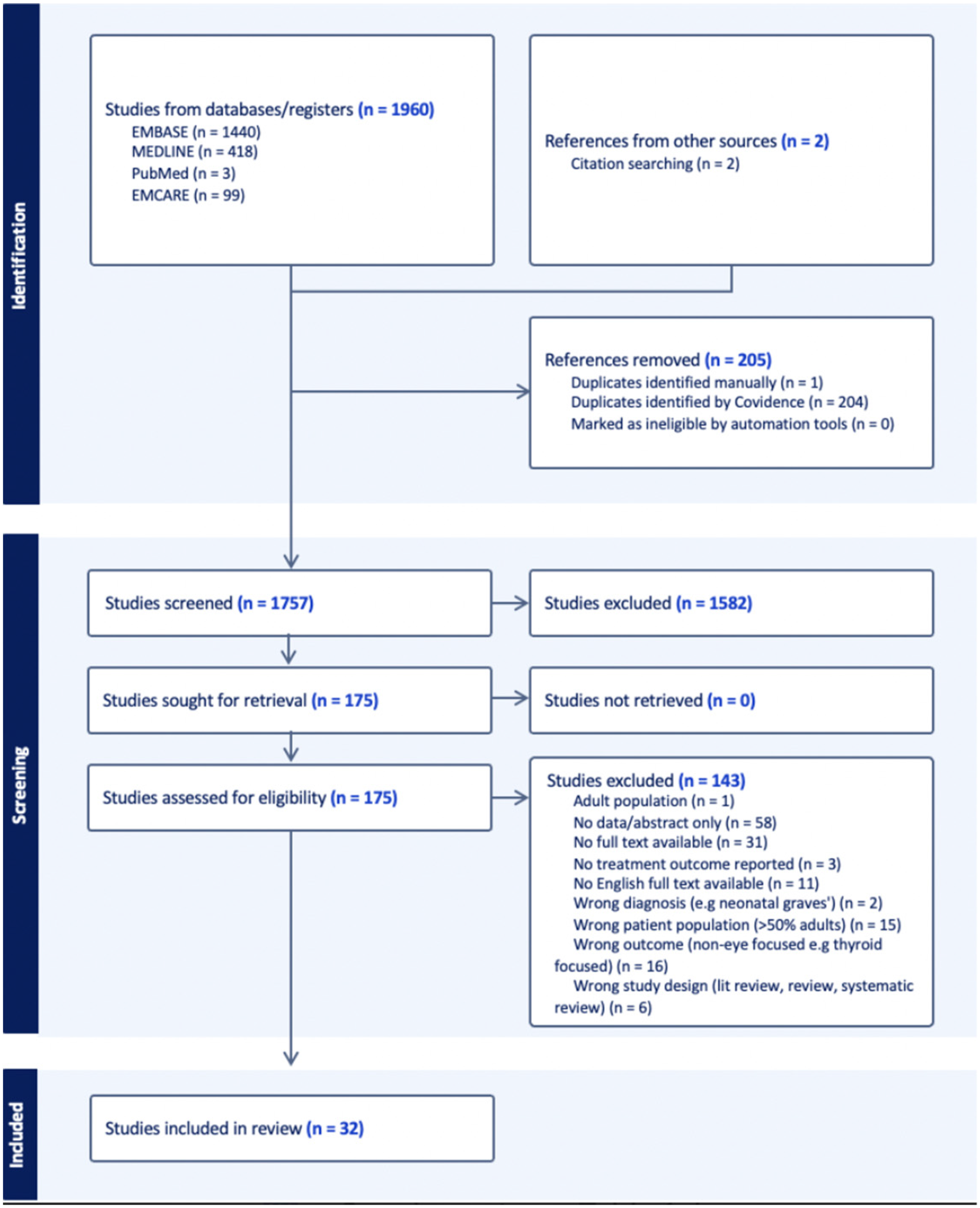
PRISMA flowchart for systematic review of pediatric thyroid eye disease studies.
Screening and study selection
The results were imported into Covidence systematic review software (Covidence, Inc). Duplicates were removed, and systematic screening was done by two independent reviewers (A.L., A.H.). Any disagreements during the screening process were resolved through discussion, and a third reviewer made a final decision (M.M.). Full-text articles were retrieved and assessed independently by two reviewers (M. M., A.G.). Inter-rater agreement was computed at each level of screening (κ = 0.28 (95% CI 0.23–0.33)) for title/abstract screening, κ = 0.79 (95% CI 0.66–0.89) for full-text review) and disagreements were resolved by consensus.
Data extraction
Two independent reviewers extracted data from all included studies (A.L., A.G.). The extracted variables included the types of treatments described in the studies. Medical treatments included steroids (such as oral prednisolone and IV methylprednisolone), radiotherapy, selenium supplementation, IV immunoglobulin, cyclosporin, and teprotumumab. Surgical treatments included orbital decompression, strabismus surgery, and eyelid surgery. In addition to treatments, data on study characteristics, including study design, setting, population demographics, and baseline disease characteristics, were also collected. Any discrepancies during the data extraction process were resolved through discussion. If data were unclear or missing, attempts were made to contact the study authors to obtain clarification or additional information.
Outcomes of interest
The primary outcomes for this review were visual acuity (VA), disease activity as measured by the Clinical Activity Scale (CAS), changes in proptosis measured in millimeters and thyrotropin Receptor Antibody (TRAb) results. Secondary outcomes included presenting ocular and systemic symptoms, and comorbid conditions. While treatments addressing TED were of primary interest, other treatments primarily for hyperthyroidism rather than TED, such as radioiodine therapy and procedures targeting cosmetic outcomes, were also included.
Risk of bias assessment
The risk of bias in the included studies was assessed independently by two reviewers using the JBI Checklist for Case Reports and the JBI Checklist for Cohort Studies with disagreements resolved through discussion and consensus (A.H., M.M.). The JBI checklists examine potential biases in group selection, diagnostic clarity, measurement of exposure and outcomes, follow-up duration, confounding variables, intervention details, adverse events and more. The use of the JBI tools allowed for a consistent and comprehensive assessment across both observational and case report study designs.
Data analysis
Data were synthesized both narratively and quantitatively. Quantitative analysis was conducted using one-way ANOVA and pooled mean differences with 95% confidence intervals were calculated. Heterogeneity between studies was assessed using Cochran's Q test and I2 statistics, with random-effects models applied when I2 > 50%. Pre-specified sources of heterogeneity, such as treatment group and patient age, were evaluated using meta-regression analyses. To further explore treatment effects, subgroup analyses were performed based on pre-specified hypotheses to assess the impact of treatment type and age on the degree of exophthalmos reduction. Sensitivity analyses using bootstrapped confidence intervals were also conducted to examine the robustness of the findings. For outcomes amenable to meta-analysis, statistical models were employed based on the level of heterogeneity. Random-effects models were applied when heterogeneity was significant, as indicated by an I2 statistic greater than 50%. The certainty of evidence for key outcomes was assessed using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) framework. Sensitivity analyses were conducted to evaluate the robustness of the synthesized results. R Studio software was used for statistical and meta-analysis.
Results
A total of 810 patients were included (64.8% female, mean age 11.73). Baseline characteristics are summarized in Table 1 and Supplementary Table 3. There were 8 retrospective cohort studies,7,10,12–17 17 case reports,3,18–32 5 case-series,6,33–36 1 longitudinal study 5 and 1 experimental study. 37 Exophthalmos was the most common presenting concern (89.9%). 99.7% presented with ocular signs and symptoms including exophthalmos (99.5%), eyelid retraction (73.1%), and dry eye (66.3%) most frequently. Systemic symptoms were noted in 77.8%, especially thyroid goiter (54.9%), weight loss (38.8%), and autoimmune conditions (37.8%) (Supplementary Table 3). Most patients were hyperthyroid (71.9%, n = 497/691); none were hypothyroid. The mean pre-treatment CAS was 0.79, with a mean symptom duration of 32.2 months. TRAb values were reported before in 2/33 studies, ranging from 14.6 to 36300 IU/mL.17,37 Two studies15,17 supported the clinical utility of TRAb in predicting remission or disease course in pediatric Graves’ disease while two studies12,37 found limited or no independent prognostic value.
Summary of patient demographics.
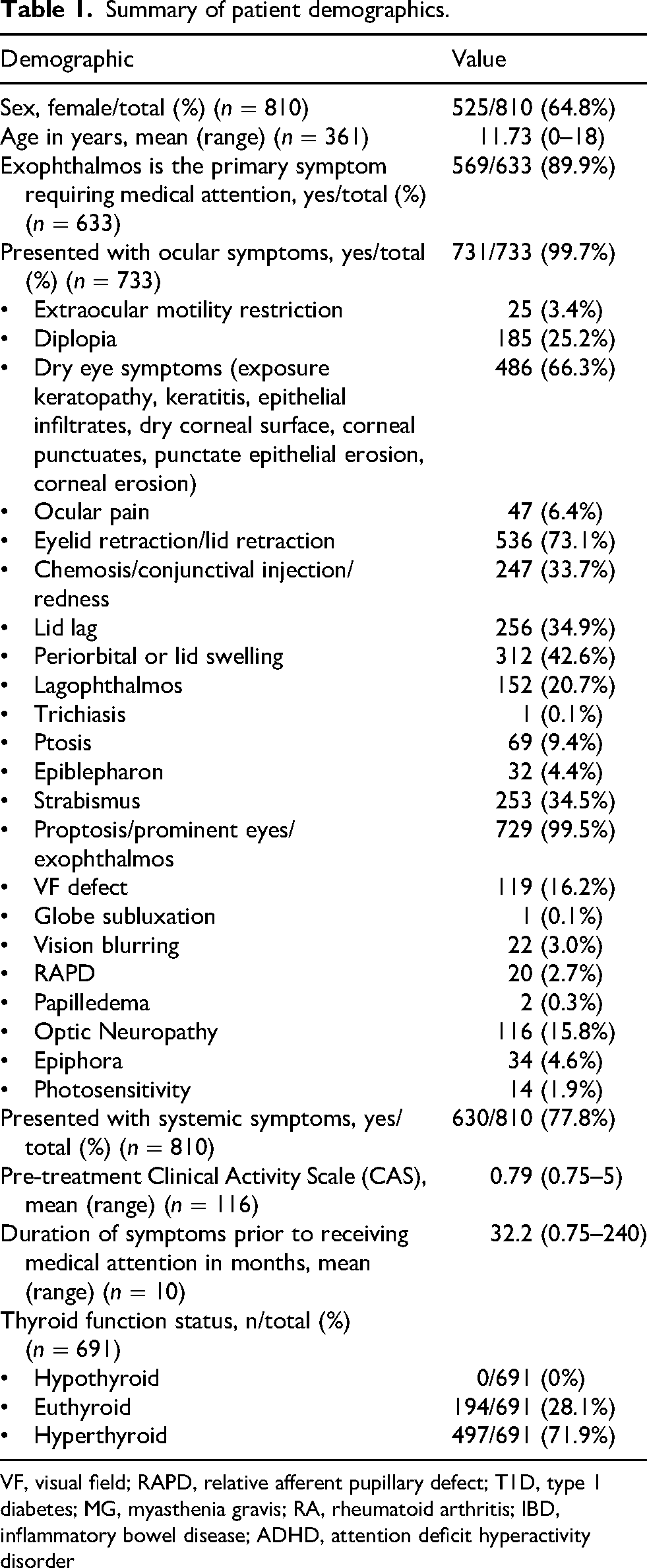
VF, visual field; RAPD, relative afferent pupillary defect; T1D, type 1 diabetes; MG, myasthenia gravis; RA, rheumatoid arthritis; IBD, inflammatory bowel disease; ADHD, attention deficit hyperactivity disorder
The treatment classes include antithyroid drugs (e.g., carbimazole, methimazole, propylthiouracil) (n=680), decompression surgery (n=205), strabismus surgery (n=278), antibody therapy (teprotumumab, rituximab, tocilizumab, n=31), steroid therapy (n=119), radioactive iodine therapy (n=267), thyroidectomy (n=220), and conservative management (n=18).
Indications for treatment
Treatment indications were split into a total of six categories: vision/extraocular movement problems, lids/edema/surface problems, exophthalmos, cosmetic, systemic/associated issues, and treatment resistance/failure (Tables 2 and 3). Decompression surgery, steroid therapy, and antibody therapy were each used for nearly all indication categories at least once. The only exception was antibody therapy, which was not used for vision or extraocular movement problems, but it was the only treatment used for cosmetic concerns.
Patient indications for thyroid eye disease treatments – by treatment groups.

Indications: 1 - Vision/Extraocular Movement Problems (Includes Strabismus); 2 - Lid/Edema/Surface Problems; 3 - Exophthalmos; 4 - Cosmetic; 5 - Systemic/Associated Issues; 6 - Treatment Resistance/Failure
N = 784, because the remaining excluded patients belonged to papers that did not provide extractable information on indications for treatment.
*These numbers represent the total count of patients associated with each indication. They may be larger than their subcategories because many papers did not specify their indication beyond the high-level concept (e.g., “Antithyroid,” rather than the drug type).
†The number here is larger than the total patient number because so many patients had multiple indications under the “Other” category. This category is shared by Tables 2 & 3.
Patient indications for hyperthyroidism treatments – by treatment groups.
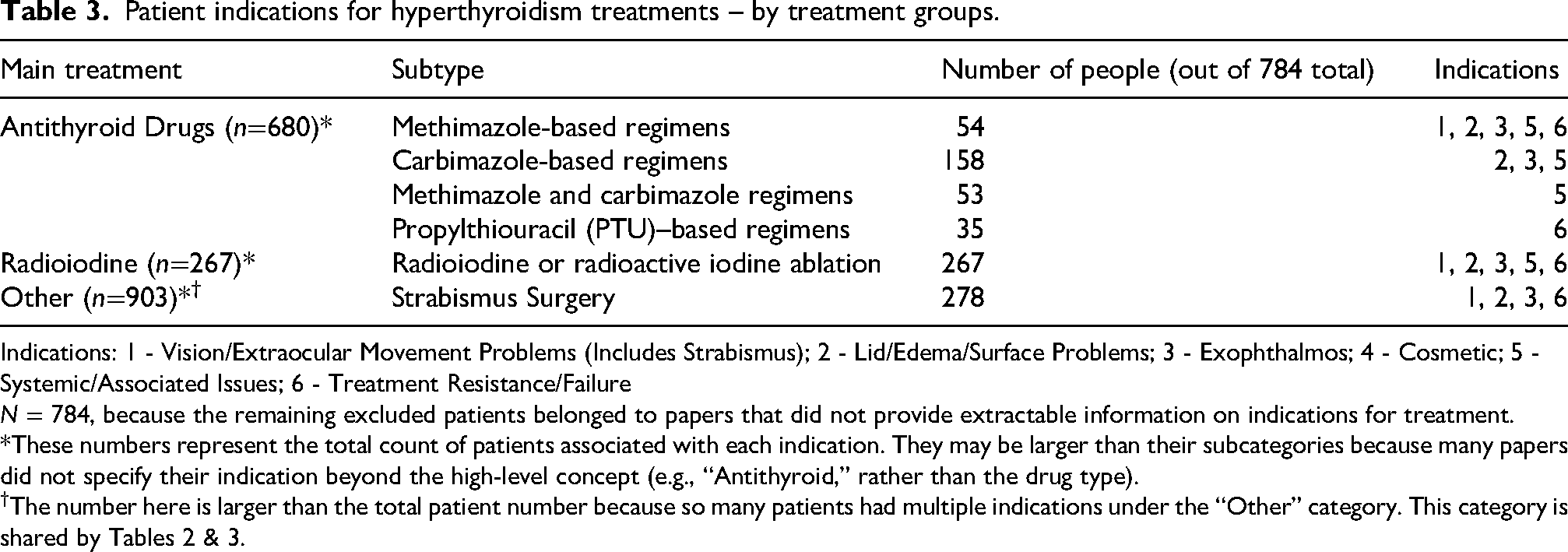
Indications: 1 - Vision/Extraocular Movement Problems (Includes Strabismus); 2 - Lid/Edema/Surface Problems; 3 - Exophthalmos; 4 - Cosmetic; 5 - Systemic/Associated Issues; 6 - Treatment Resistance/Failure
N = 784, because the remaining excluded patients belonged to papers that did not provide extractable information on indications for treatment.
*These numbers represent the total count of patients associated with each indication. They may be larger than their subcategories because many papers did not specify their indication beyond the high-level concept (e.g., “Antithyroid,” rather than the drug type).
†The number here is larger than the total patient number because so many patients had multiple indications under the “Other” category. This category is shared by Tables 2 & 3.
Visual acuity (VA) changes
Patients experienced a mean LogMAR change in best corrected VA of −0.067 (range 0 to −0.2 change), with a final follow-up LogMAR VA of 0 (Snellen ratio: 20/20). 58% remained the same or did not improve in VA, while 42% improved. No patients regressed in VA, though most studies did not report VA (n=772). Follow-ups ranged from 3 to 120 months (mean: 27.26 months).
Meta-Analysis on exophthalmos reduction
Exophthalmos reduction across treatment modalities demonstrated a mean reduction of 4.69 mm (95% CI: 4.58–4.87) following decompression surgery, 4.25 mm (95% CI: 2.6–6) with steroid therapy, and 1.75 mm (95% CI: 0–3.5) with biologic agents (Figure 2, Supplementary Table 1). Heterogeneity was substantial in studies involving decompression surgery, as indicated by an I2 value of 99.67%. Sensitivity analyses using bootstrapped confidence intervals further supported the observed variability in treatment effect estimates, with the greatest fluctuations noted in studies with small sample sizes. Analysis of variance (ANOVA) revealed no significant difference across groups (F = 2.43, p = 0.149). Standardized mean differences (SMDs) between treatment pairs were as follows: antibody vs. steroid, −0.58; antibody vs. decompression, −1.60; and steroid vs. decompression, −0.55. Meta-regression analyses found no significant influence of treatment type or patient age on the degree of exophthalmos reduction.
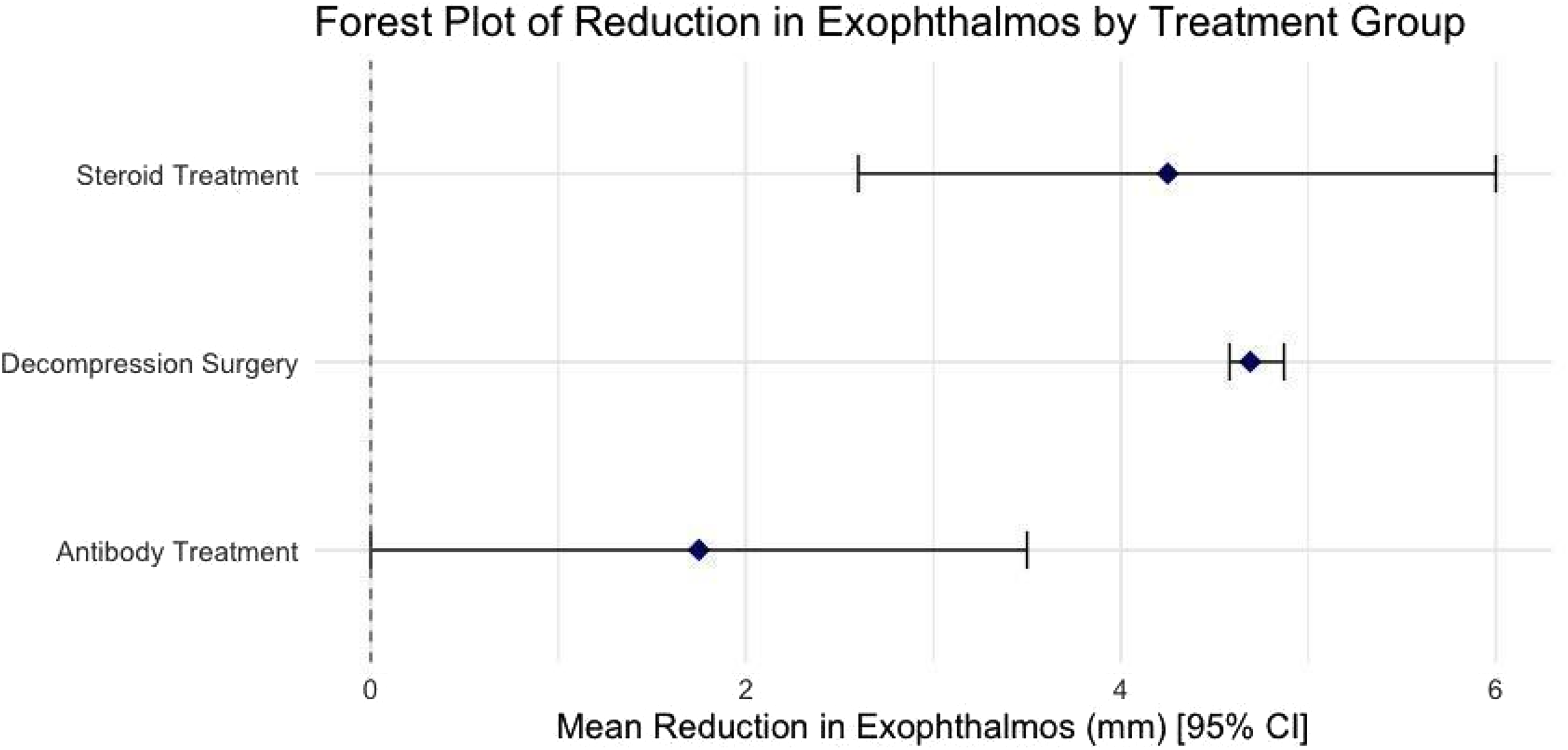
Forest plot of mean exophthalmos reductions for each treatment group.
Certainty of evidence
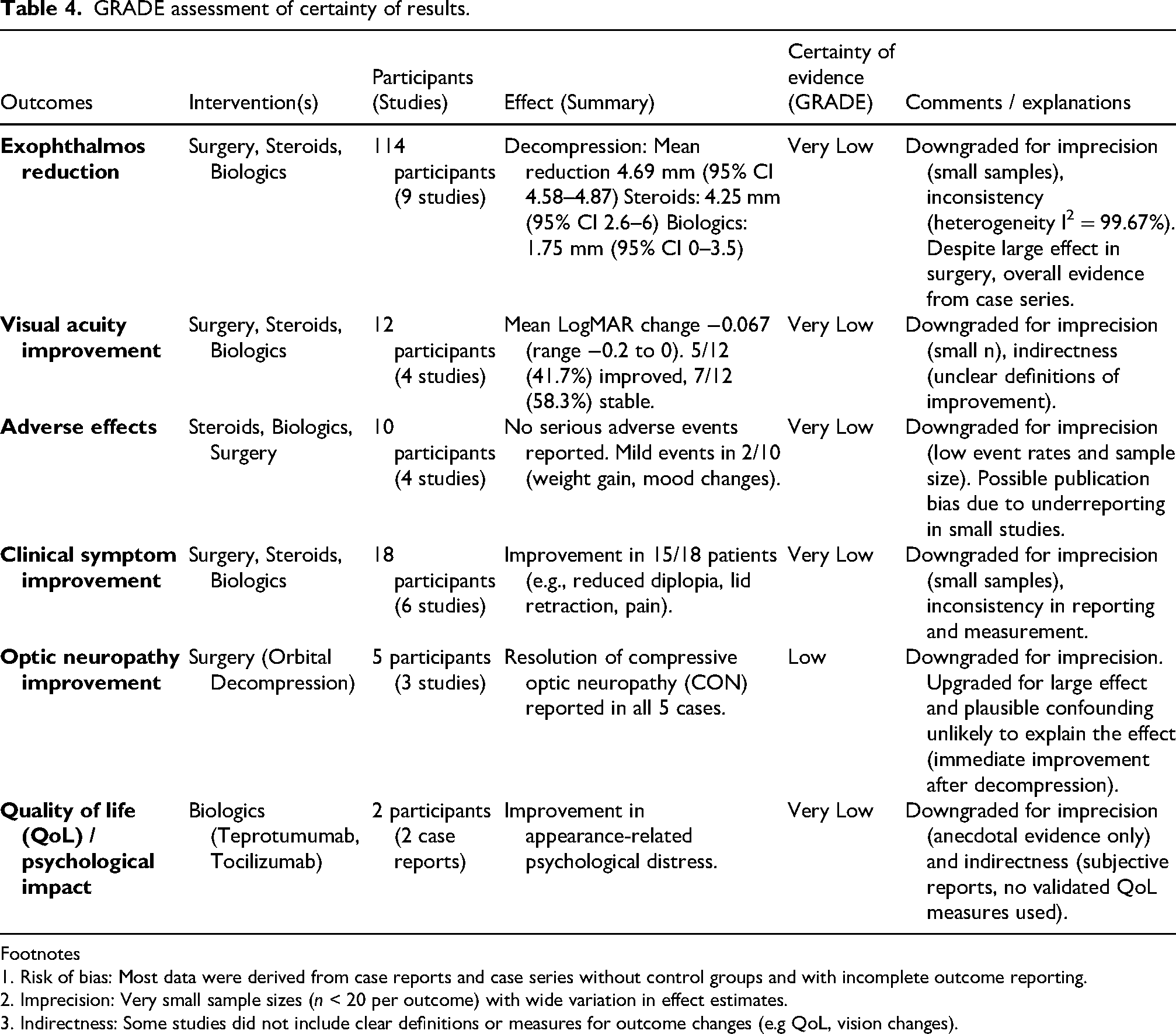
Overall certainty was very low due to small sample sizes, imprecision, and reliance on case reports (Table 4). Optic neuropathy improvement reached low certainty due to consistently large effects after decompression.
GRADE assessment of certainty of results.
Footnotes
1. Risk of bias: Most data were derived from case reports and case series without control groups and with incomplete outcome reporting.
2. Imprecision: Very small sample sizes (n < 20 per outcome) with wide variation in effect estimates.
3. Indirectness: Some studies did not include clear definitions or measures for outcome changes (e.g QoL, vision changes).
The most frequent deficiency on the JBI checklist was Q7 (reporting of adverse events), which led to GRADE downgrades for risk of bias and publication bias (supplementary Table 4). For observational reports, confounding and lack of adjustment further led to increased uncertainty. Small sample sizes resulted in imprecision downgrades. Consequently, certainty was very low for most outcomes, with optic neuropathy improvement rated low (large, immediate post-decompression effect despite RoB/imprecision). Due to the predominance of case reports and series, formal assessment of publication bias was not feasible; however, there remains a concern for selective reporting of positive outcomes and underreporting of adverse events with case reports.
Discussion
TED is driven by orbital fibroblast activation via TSH receptor (TSHR) and IGF-1 receptor (IGF-1R) signaling pathways 38 causing inflammation and fibrosis. Interleukin-6 (IL-6) has also been identified as a key mediator in TED pathogenesis, becoming a possible target for immunomodulatory therapy. 38 Though usually milder than adult cases, these mechanisms cause psychosocial distress and functional impairments in pediatrics, including increased rates of anxiety, inattention, and depressive symptoms. 37
While TED is extensively studied in adults, the impact on pediatric patients remains under-characterized. Our review synthesizes clinical presentation, management and outcomes in pediatric TED to help inform evidence-based management in a vulnerable and understudied group.
Of the 810 patients included in our review, 733 had ocular symptoms, commonly presenting with exophthalmos (99.5%) and eyelid retraction (73.1%), while the initial cause for presentation varied from orbital apex syndrome to poor weight gain and irritability. 38 Though the mean CAS was low (0.79), supporting a milder inflammatory phenotype overall, the extended pre-diagnosis course (mean 32.2 months) suggests prolonged disease activity before care, possibly allowing fibrotic progression. Optic neuropathy (15.8%) and diplopia (25.2%) were notable functional complications. These findings suggest that while pediatric TED may appear quiescent, it can progress insidiously and warrant timely intervention. Chua et al. similarly noted that many pediatric TED patients undergo surgical rehabilitation rather than acute anti-inflammatory therapy, highlighting how delayed recognition affects treatment course. 10 The findings for TRAb monitoring were mixed, with an equal number of studies suggesting the utility of TRAb monitoring for prognosis and clinical decisions, while others warn against relying on this measurement alone for management decisions.12,15,17,37
Management strategies aligned with disease severity, which correlates with symptom profile (Table 5). Most patients with mild/inactive disease were managed with antithyroid drugs (680/810 patients) or with observation and lubrication (18/810).3,18,21–23 Moderate-to-severe active disease, characterized by lid edema and exophthalmos, were managed with intravenous corticosteroids as first-line therapy for reducing inflammation and stabilizing vision. Dose varied at 15 mg/kg/day for 3 days then again a week later, or a weekly 250 mg infusion for 6 weeks, followed by 125 mg for another 6 weeks. 19 Studies recommended a 12-week IVMP course for active or progressive proptosis of 250–500 mg weekly for 6 weeks, followed by 125–250 mg weekly for another 6 weeks.19,34 For dysthyroid optic neuropathy with visual field defects, a high-dose protocol was advised of 15 mg/kg/day or 1000 mg/day for 3 days, repeated one week later.19,34 Notably, no steroid-related side effects were reported, possibly due to prednisolone use over more growth-suppressing alternatives or underreporting (Supplementary Table 2). 39 Among patients presenting with lid retraction, management consisted of medical therapy (prednisolone and antithyroid drugs) and rarely, surgical interventions (epiblepharon correction, strabismus surgery, orbital decompression, or mullerectomy and lateral canthoplasty) following failure of conservative management. Formal upper eyelid retraction repair, as commonly performed in adult TED, was not reported in pediatric cohorts.
Pediatric GED treatment algorithm.
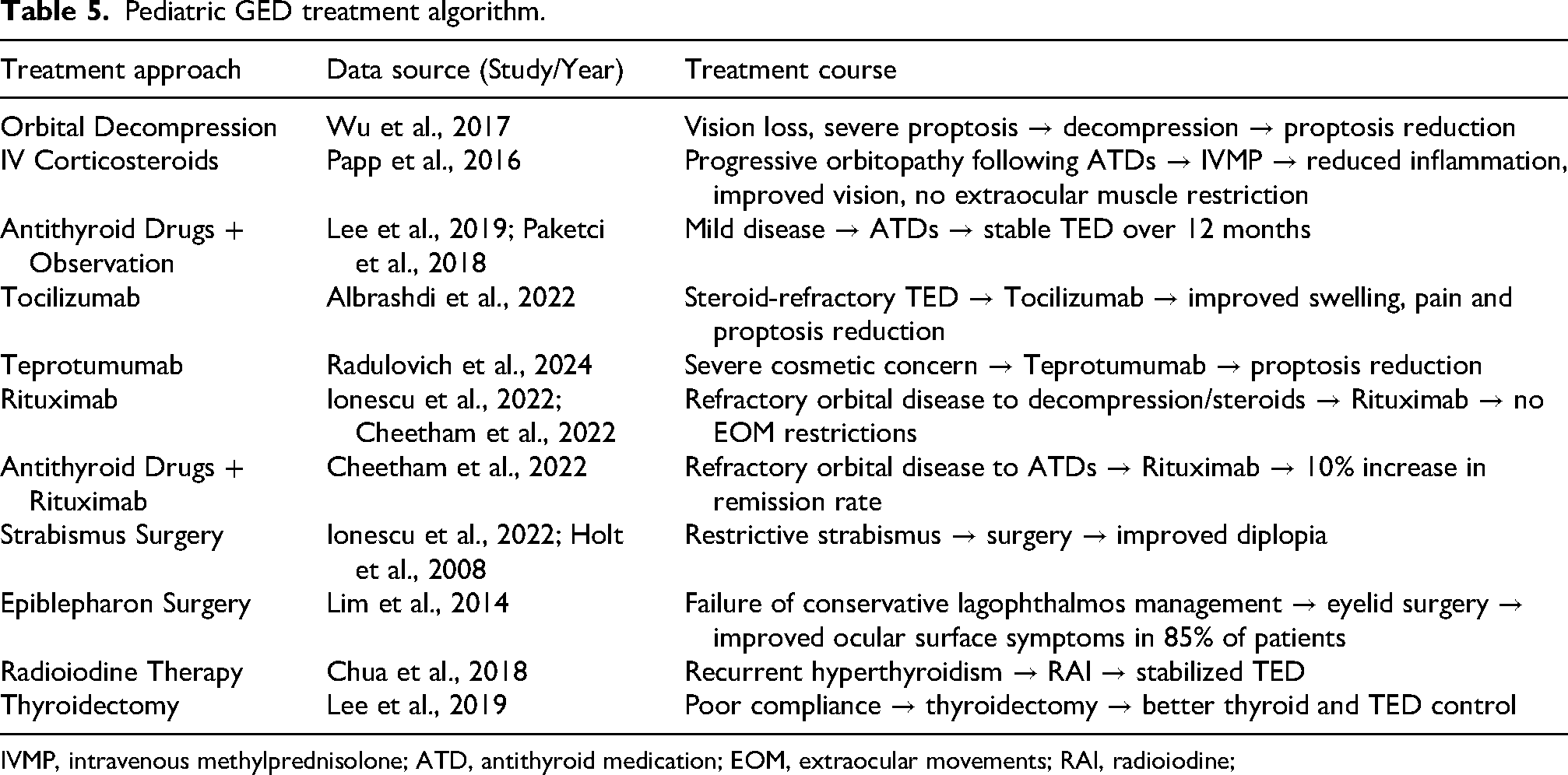
IVMP, intravenous methylprednisolone; ATD, antithyroid medication; EOM, extraocular movements; RAI, radioiodine;
Biologics were used sparingly, generally in refractory cases. Teprotumumab, 20 and rituximab 34 have been employed in limited pediatric patient studies, for refractory or persistent disease. Tocilizumab (4–8 mg/kg monthly×4 doses) improved proptosis, pain, and lid signs.26,32 Teprotumumab (510 mg then 1020 mg every three weeks×8 infusions) was reported to improve CAS scores by 4 points, resolved diplopia, improved proptosis by 3.5 mm and provided cosmetic benefit by 24 week follow-up. 20 Nevertheless, this evidence remains limited, and previous literature has noted detrimental effects of teprotumumab, particularly in younger animals, concerning reduced growth, smaller thymus/spleen, and abnormal bone growth. As such, there are recommendations against its empirical use in pediatric populations. 39 Rituximab, used with anti-thyroid drugs, improved remission rates by 10% in a multicentre retrospective observational case series of 115 pediatric patients.34,40–42 Side effects included fatigue and amenorrhea (Supplementary Table 2). While promising for treatment-resistant cases or where contraindications for steroids exist, more pediatric-specific data are needed to confirm long-term safety and efficacy.
Surgical interventions were pursued in severe cases such as CON and severe proptosis. Orbital decompression surgery (26%), often followed steroid therapy and showed consistent proptosis reduction and vision stabilization, though the GRADE assessment of certainty of these results remains low.10,33,35,43 Though recommended after age 15, the median age in our review was 14, with some patients as young as 9. This discrepancy may reflect clinician judgment in severe cases outweighing growth concerns. Similarly, strabismus and epiblepharon surgeries for comfort and function were typically delayed ≥6 months post-inflammation in keeping with protocols, and provided high functional and cosmetic success.10,34
Definitive thyroid therapies were reserved for systemic disease control, including radioiodine (33%) in older adolescents and thyroidectomy (28%) in younger children or those with poor medication adherence.
Compared to adults, pediatric TED management remains more conservative due to growth and development concerns. Yet in practice, systemic therapies and surgery are sometimes used earlier, indicating a gap between theory and real-world management. Clinicians may escalate treatment earlier based on disease burden or patient preference. Overall, treatment in children tends to be delayed compared to adults, despite evidence that earlier intervention may be beneficial. These discrepancies highlight the need for pediatric-specific guidelines grounded in both evidence and clinical practice.
Additionally, TED exerts a psychosocial burden on those afflicted. In adult populations, many validated instruments exist that have consistently shown QoL detriments in social functioning, mental health, and emotional distress.40–42 In pediatric populations, changes in appearance may lead to more significant and unique psychosocial challenges. Further development of validated pediatric QoL tools may help to expand on these effects in one's formative years.
To our knowledge, this review is the most comprehensive synthesis to date of pediatric TED, drawing from studies across all ages and regions. Despite a rigorous methodology, the interpretation of these findings is limited by the predominance of case reports, which may be subject to heterogeneity in populations and overall lack of generalizability. Furthermore, outcome reporting was not standardized, with a lack of validated QoL measures, restricting the scope of meta-analysis. This review should therefore be used with caution rather than definitive guidance.
Conclusion
These findings highlight the need for earlier recognition and individualized management of pediatric TED to prevent long-term complications. Clinicians should tailor treatment based on age, disease severity, and progression risk. Future research may prioritize prospective, multicenter studies with standardized outcomes and pediatric-specific QoL measures. Moving forward, establishing dedicated registries to advance evidence-based guidelines that reflect the unique needs of children, along with conducting further multicenter prospective studies, should be prioritized.
Supplemental Material
sj-docx-1-ejo-10.1177_11206721261440985 - Supplemental material for Management and presentation of pediatric thyroid eye disease: A systematic review and meta-analysis
Supplemental material, sj-docx-1-ejo-10.1177_11206721261440985 for Management and presentation of pediatric thyroid eye disease: A systematic review and meta-analysis by Abbie C Lai, Caberry W Yu, Amir-Ali Golrokhian-Sani, Maya Morcos, Angelica Hanna and Santa Heede in European Journal of Ophthalmology
Footnotes
Acknowledgements
We would like to thank Abdullah El-Sayes for his valuable assistance with the screening process and Jo-Anne Roberts (J.R.) (Health Sciences Librarian, McMaster University) for her expertise and support in developing the search strategy for this review.
Ethics approval and informed consent statements
Not required at Hamilton Integrated Research Ethics Board for systematic reviews.
Author contributions
A.L.: Methodology, Data Curation, Investigation, Visualization, Formal Analysis, Investigation, Writing – Original Draft; C.W.Y.: Methodology, Project Administration, Supervision, Writing – Review & Editing; A.G.: Data Curation, Investigation, Visualization, Formal Analysis; M.M.: Data Curation, Investigation, Visualization; A.H.: Data Curation, Investigation; S.H.: Conceptualization, Methodology, Supervision.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Data available within the article or its supplementary materials
Supplemental material
Supplemental material for this article is available online.
Appendix A. Search Strategy
1. graves ophthalmopathy/
2. (endocrine ophthalmopath* or congestive ophthalmopath* or dysthyroid ophthalmopath* or edematous ophthalmopath* or graves* eye disease or graves* ophthalmopath* or graves orbitopath* or infiltrative ophthalmopath* or infiltrative ophthalmopath* or myopathic ophthalmopath* or (thyroid adj2 ophthalmopath*) or thyroid eye disease*).ti,ab,kw,kf.
3. GRAVES DISEASE/
4. limit 3 to yr="2004–2005"
5. 1 or 2 or 4
6. pediatrics/ or neonatology/
7. adolescent/ or child/ or child, preschool/ or infant/ or infant, newborn/ or infant, large for gestational age/ or infant, low birth weight/ or infant, small for gestational age/ or infant, very low birth weight/ or infant, extremely low birth weight/ or infant, postmature/ or infant, premature/ or infant, extremely premature/
8. (adolescen* or teen* or youth or youths or juvenil* or pubescen* or child* or kid or kids or school-age* or schoolage* or schoolchild* or preschool* or pre-school* or toddler* or boy or boys or girl or girls or minor or minors or infan* or newborn* or new-born* or baby or babies or neonat* juvenil* or p?ediatric*).mp.
9. (("1" or "2" or "3" or "4" or "5" or "6" or "7" or "8" or "9" or "10" or "11" or "12" or "13" or "14" or "16" or "17" or "18") adj2 age).ti,ab,kw,kf.
10. (("1" or "2" or "3" or "4" or "5" or "6" or "7" or "8" or "9" or "10" or "11" or "12" or "13" or "14" or "16" or "17" or "18") adj2 (year* adj2 old)).ti,ab,kw,kf.
11. or/6–10
12. 5 and 11
13. exp cohort studies/
14. cohort$.tw.
15. controlled clinical trial.pt.
16. epidemiologic methods/
17. limit 16 to yr="1966–1989"
18. exp case-control studies/
19. (case$ and control$).tw.
20. (case$ and series).tw.
21. case reports.pt.
22. (case$ adj2 report$).tw.
23. (case$ adj2 stud$).tw.
24. or/13–15,17–23
25. 5 and 11 and 24
1. endocrine ophthalmopathy/ or graves disease/
2. pediatrics/ or neonatology/
3. adolescent/ or juvenile/
4. infant/ or baby/ or newborn/
5. child/ or infant/ or preschool child/ or school child/ or toddler/
6. "minor (person)"/
7. (("1" or "2" or "3" or "4" or "5" or "6" or "7" or "8" or "9" or "10" or "11" or "12" or "13" or "14" or "16" or "17" or "18") adj2 age).mp. [mp=title, abstract, heading word, drug trade name, original title, device manufacturer, drug manufacturer, device trade name, keyword heading word, floating subheading word, candidate term word]
8. (("1" or "2" or "3" or "4" or "5" or "6" or "7" or "8" or "9" or "10" or "11" or "12" or "13" or "14" or "16" or "17" or "18") adj2 (year* adj2 old)).mp. [mp=title, abstract, heading word, drug trade name, original title, device manufacturer, drug manufacturer, device trade name, keyword heading word, floating subheading word, candidate term word]
9. 2 or 3 or 4 or 5 or 6 or 7 or 8
10. exp cohort analysis/
11. exp longitudinal study/
12. exp prospective study/
13. exp follow up/
14. cohort$.tw.
15. exp case control study/
16. (case$ and control$).tw.
17. exp case study/
18. (case$ and series).tw.
19. 10 or 11 or 12 or 13 or 14 or 15 or 16 or 17 or 18
20. 1 and 9 and 19
Same search terms as Embase.
Total: 1957
w/out duplicates: 1754
[
