Abstract
Purpose
To describe a new method for delivering DMEK grafts into the recipient's eye with endothelium inward configuration using a no-forceps injection technique
Methods
We retrospectively review 11 patients that underwent DMEK surgery at our institution using a no-forceps injection technique. The graft was preloaded into an intraocular lens (IOL) cartridge and connected to an anterior chamber maintainer (ACM). A 5 ml non luer lock syringe was inserted into the other end of the ACM to create a one-flow system. The cartridge was inserted into the posterior end of an injector, and the graft was successfully delivered into the recipient's eye.
Result
Twelve eyes of 11 patients were included. Mean follow-up was 9.16 ± 1.3 months. At baseline, mean best corrected visual acuity (BCVA) was 0.76 ± 0.13 logMAr and mean endothelial cell density (ECD) was 2619.00 ± 115.89 cells/mm2. At follow-up, BCVA significantly improved to 0.22 ± 0.05 logMAR (p = 0.003). Although we observed a significant reduction in ECD at follow-up (1688 ± 182.20, p = 0.002), our patients lost only 35.69 ± 6.36% of endothelial cells.
Conclusion
Our technique can help surgeons safely deliver an endothelium-in graft into the recipient's eye. The method doesn’t require the use of a forceps, minimizing the risk of endothelial cell loss or graft damage.
Introduction
In the last decade, Descemet Membrane Endothelium Keratoplasty (DMEK) has gained popularity among surgeons to treat primary endothelial diseases because it is associated with a faster recovery after surgery, a better visual outcome, and a lower rejection rate compared to Descemet Stripping Automated Endothelial Keratoplasty (DSAEK).1,2 However, its adoption as a standard technique is still encountering difficulties, especially with less experienced surgeons, because of the steep learning curve in the preparation, injection, and unfolding of the graft.3–5 To overcome some of these issues, many eye banks have started preparing and shipping preloaded tissue.1,6,7 The graft is stripped by eye bank trained staff under controlled conditions, allowing an improvement in theatre efficiency, a reduction of the risk of tissue loss, and better quality control of the donor endothelium. 8
When the DMEK graft is stripped out from the cornea, it tends to scroll with the endothelium outside following its natural anatomical configuration given by the more rigid Descemet's membrane. 7 Preloaded tissues with endo-in configuration have the advantage to be easily inserted into the injector, but their unfolding inside the eye can be challenging, especially in complex cases.2,9,10 Further, since the cells of the tissue are in direct contact with the injector and, once delivered in the anterior chamber, with the iris and the cornea, there is a demonstrated higher risk of endothelial cell loss (ECL) and reduced graft survival after a successful surgery.2,11,12
In 2013 Muraine et al. 13 have published a new method to deliver the preloaded graft in an endothelium-in way. This configuration reduces the mechanical stress on endothelial cells and promotes a longer long-term survival of the graft.2,10,14 Further, the tissue spontaneously unfolds when injected into the AC because it tends to acquire its natural configuration with endothelium out. 10 Busin et al. have improved the delivery procedure by adopting for DMEK surgeries the bimanual pull-through technique.7,10 This method helps overcome certain issues of graft unfolding, but it requires the use of forceps to pull the graft into the recipient's anterior chamber, increasing the manipulation of the tissue and the risk of ECL.
In this scenario, there is an unmet need for new methods to deliver the DMEK graft minimizing the tissue manipulation and improving the surgeon's control over the procedure. Herein, we described a new technique for DMEK injection into the AC in a controlled way without manipulating the graft with the forceps.
Subjects and methods
Surgical technique
The tissue was harvested, preloaded with the endo-in configuration, and shipped to our cornea department at Liverpool University (UK) by the Veneto Eye Bank Foundation (Venice, Italy) as previously described. 9 After receiving the graft and checking the expiry date, the cartridge (Figure 1a) was mounted on the injector (Viscoject single-use BIO injector, Medicel, Switzerland). An anterior chamber maintainer (90 × 4 mm, 20 gauge, 45° bevelled tip, Beaver-Visitec International Ltd., Warwickshire, UK) was connected to the posterior end of the injector and pushed into the rubber plug at the rear exit (Figure 1b). The other end of the ACM was connected to a 5 ml non-luer-lock syringe (Figure 1c) (Video 1, Supplementary Files). We gently washed the tissue with balanced salt solution (BSS) from the front opening to ensure the removal of preservation media. The injector was then introduced into the anterior chamber through a single 2.7 mm 2 steps incision. The graft was advanced into the anterior chamber by applying gentle taps on the plunger of the 5 ml syringe which offered a slow and controlled flow of balance salt solution through the rubber plug of the injector (Video 2, Supplementary Files).
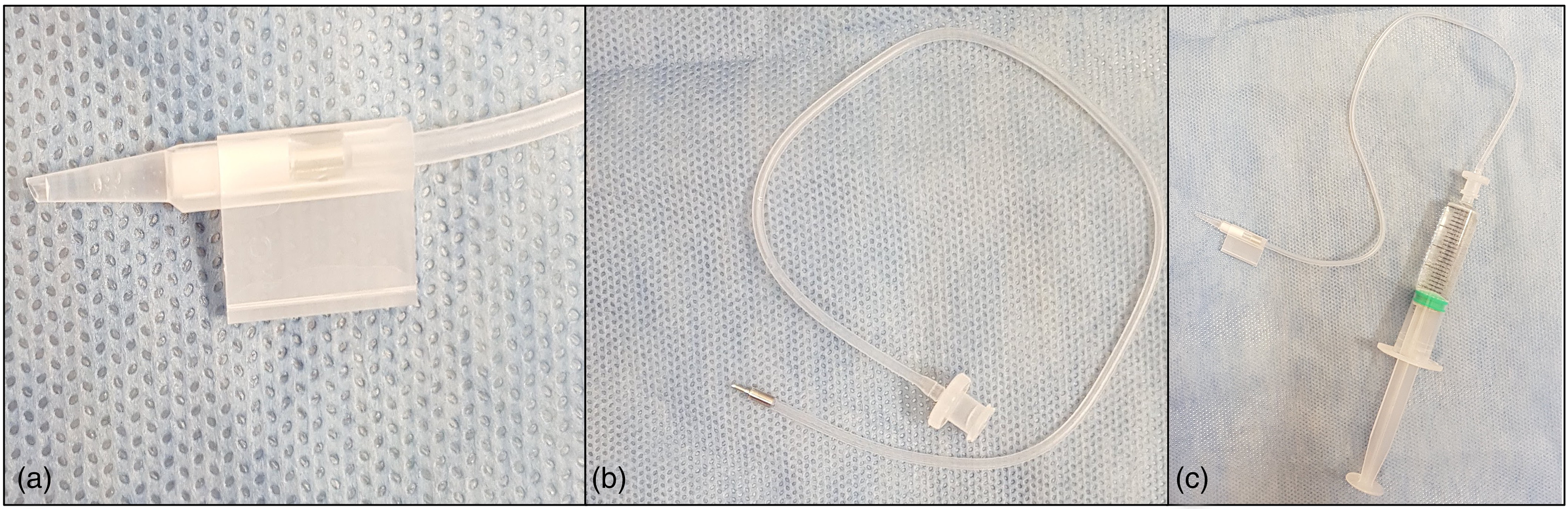
Photo of the cartridge used to inject the graft into the anterior chamber. The cartridge is mounted on an injector (not shown in the figure) to deliver the donor tissue into the recipient's eye (a). The anterior chamber maintainer is connected to the rubber plug at the rear exit of the cartridge (b). The other end of the anterior chamber maintainer is connected to a 5 ml non-luer-lock syringe to create a one-flow system (c).
Participants
This was a retrospective single-center study. All patients were enrolled at the Cornea Department of Liverpool University Hospitals, Liverpool, UK. Inclusion criteria were subjects undergone DMEK surgery with our new injection technique and a diagnosis of ECD dystrophy. Exclusion criteria where a history of previous corneal transplant or posterior segment surgery, glaucoma, and incomplete clinical charts. Twelve eyes of 11 patients were retrospectively reviewed. Slit lamp examination, retinal fundus examination, intraocular pressure (IOP), best corrected visual acuity (BCVA), and ECD were assessed at baseline and follow-up. ECD was evaluated using a non-contact specular microscope (CellCheck20, Konan, Konan Medical USA, Irvine, California, USA). Informed consent was obtained from all patients to use data for research purpose. Non-parametric tests were used to compare baseline and follow-up data. P was considered significant if < 0.05. Statistical Package for the Social Sciences V.24.0 (SPSS Inc., Chicago, Illinois, USA) was used for statistical analysis.
Results
We included 5 males and 6 females with a mean age of 72.16 ± 5.71 years (Table 1). Mean follow-up duration was 9.16 ± 1.3 months. DMEK surgery was performed in 6 right eyes and 6 left eyes, 7 subjects (63.6%) had a diagnosis of Fuchs endothelial corneal dystrophy (FECD), 3 (27.3%) had pseudophakic bullous keratopathy (PBK), and one (9.1%) had posterior polymorphous corneal dystrophy (PPCD). Five eyes (42.17%) underwent combined cataract and DMEK surgery, whereas 7 patients (58.3%) were already pseudophakic and underwent DMEK only. BCVA significantly improved from 0.76 ± 0.13 logMAR to 0.22 ± 0.05 logMAR when comparing baseline and follow-up assessment (p = 0.003) (Table 2). At follow-up, the ECD significantly decreased from 2619.00 ± 115.89 cells/mm2 to 1688.50 ± 182.20 cells/mm2 as compared to baseline (p = 0.002) with a mean endothelial cell loss of 35.69 ± 6.36% (Table 2). No correlation was observed between the endothelial cell loss and the follow-up duration (p = 0.36). Interestingly, there was no correlation between pre- and post-transplant BCVA (p = 0.2), but the graft ECD at follow-up significantly correlated with the number of endothelial cells assessed at baseline (p = 0.04, Spearman's correlation coefficient 0.60). No significant differences were observed when the final BCVA was adjusted for the baseline graft ECD (p > 0.05). Two eyes (18.2%) required rebubbling 1 week after the DMEK surgery for graft detachment, but this intervention had no effect on the final ECD compared to patients that did not require rebubbling (p = 0.883). Finally, we did not observe any significant difference between baseline and follow-up ECD between patients that underwent DMEK or combined phaco-DMEK surgery (p = 0.58).
Demographic and clinical characteristics.

Values reported as Mean ± SEM.
FECD: Fuchs Endothelial Corneal Dystrophy.
PBK: Pseudophakic Bullous Keratopahty.
DMEK: Descemet's Membrane Endothelial Keratoplasty.
Clinical outcomes.
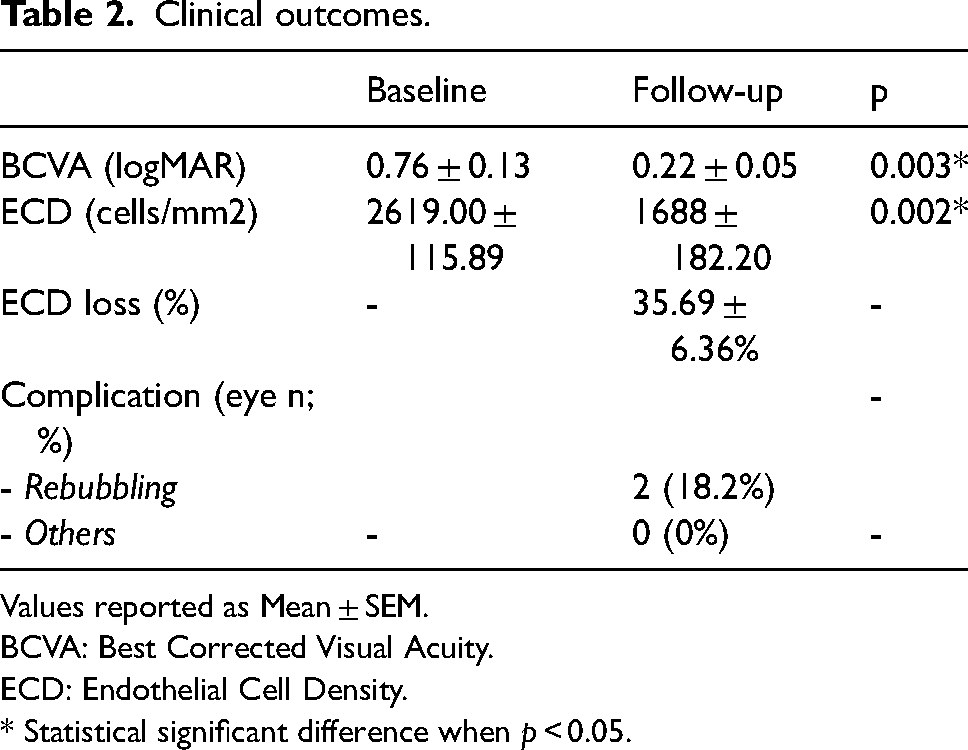
Values reported as Mean ± SEM.
BCVA: Best Corrected Visual Acuity.
ECD: Endothelial Cell Density.
* Statistical significant difference when p < 0.05.
This technique allowed us to successfully advance the DMEK tissue into the anterior chamber whilst maintaining it in the endothelium in configuration. As expected, most of the endothelium-in tissues usually unfold without any further manipulation once inserted into the eye. In our experience, the cases that do not open require few trans-corneal taps to completely unscroll. Informed consent was obtained from patients to use data for research purposes.
Discussion
Herein, we demonstrated a new technique to inject the DMEK graft into the recipient's eye in a controlled way. Our results show that this technique is safe and allow to significantly improve the BCVA reducing the need of tissue manipulation with forceps.
The introduction of preloaded DMEK grafts has brought many advantages like reduced tissue wastage, better quality control of grafts, and reduced surgical time. 15 Although many surgeons prefer to use endothelium-out tissues, this configuration of the graft has some issues. First, since the DMEK membrane spontaneously rolls with cells outward, it tends to maintain a tight scroll configuration inside the eye that can be difficult to unfold. 10 Second, the endothelial cells can be damaged by contact with the cartridge and, once the membrane is injected inside the recipient's eye, with the ocular anatomical structures like the cornea and the iris. 16
The endothelium-in configuration described by Muraine et al. 13 in 2013 tried to address some of the endo-outward problems. Indeed, the graft is prepared by forcibly folding the tissue in an endo-in shape. This technique has the advantage of minimizing endothelial cell loss (ECL) during transportation and delivery steps, reducing the manipulation time to unscroll the graft, and improving the visual acuity outcome. 10 Chong et al. showed an increased cell survival rate using a trifold endo-in technique as compared to endo-out. 14 Further, it has been suggested that DMEK surgery using preloaded endothelium-in grafts could be easier in challenging situations like AC IOL, large iris defects, and vitrectomized eyes. 17 In 2016 Busin et al. 7 presented their pull-through technique to facilitate the delivery of the graft into the eye recipient. This method has the advantage to improve the surgeon's control over the DMEK membrane during the injection step, and to allow the delivery of the graft maintaining its endo-in configuration. 2 However, the technique has the limitation of requiring a forceps to insert the donor tissue inside the anterior chamber.
With our technique, the graft is advanced inside the recipient's eye in a very slow and controlled method. Additionally, the transparent injector can be rotated within the anterior chamber during the advancement of the graft to maintain an orientation similar to the previously described methods. 18
Although different variables contribute to the success of a DMEK surgery, the endothelial cell count represents the main factor for long-term graft survival. 10 Thus, the use of forceps in the pull-through technique can increase the risk of damaging the EC and creating graft tears. 7 Busin et al. claim that their method requires only 3 or 4 forceps bites, with an estimated EC loss of 75 cells per forceps bite,2,7 but less experienced surgeons could have to manipulate more the graft, increasing the risk of cell death. 5 Further, since no difference was found in terms of ECL between endo-in and endo-out preloaded tissue preparation, 6 and between the endo-in pull-through method and the endo-out technique to inject a DMEK graft inside the anterior chamber,7,19 this could make novice surgeons desist from opting for endothelium-inward preloaded tissue. Additionally, the need for an ACM and the bimanual manipulation of the graft may prolong the learning curve and the surgical time. 17
Our technique has the main advantage to avoid the use of a forceps to deliver the DMEK tissue reducing the risk of ECL. At the same time, it guarantees a very good control of the graft orientation during the injection step, allowing the insertion of the tissue with the correct endo-in configuration and thus facilitating its natural unscrolling. Although we found a significant difference between baseline and follow-up ECD, however our patients showed a good mean cell density after the endothelial keratoplasty with a percentage of endothelial cell loss lower or comparable with what is described in literature.11,15,20–24 As expected, the number of cells at baseline significantly correlated with the ECD at follow-up. Considering that a DMEK graft loses up to 50% of cells during the first year of follow-up, clinicians should pay attention to donors’ cell count when they performed the surgery in young patients.
In conclusion, in this study we presented a new DMEK endothelium-inward delivery procedure. This technique offers optimal control over the graft during the injection step, allowing the positioning of the donor tissue inside the anterior chamber in the correct configuration. Further, it avoids the need for forceps use, reducing graft manipulation and the risk of ECL. In our case series, BCVA significantly improved after the DMEK surgery, and patients showed a good ECD at follow-up confirming the safety of our technique. The reduced cell loss and the easy and controlled delivery of the graft with our technique may increase the favourable benefits of the endo-in DMEK surgery. Further studies are needed to validate our technique.
Supplemental Material
Supplemental Material
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article: This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
