Abstract
Introduction
Prechoroidal cleft has been described as a negative prognostic biomarker in patients affected with neovascular age related macular degeneration (nAMD). This peculiar finding consists of a lenticular hyporeflective space located between an outward bowing of Bruch's membrane and the base of a fibrovascular retinal pigment epithelium detachment (PED). Previous studies have reported the partial or complete regression of prechoroidal clefts after treatment with anti-vascular endothelial growth factor (VEGF) injections.
Case report
To report a case of complete anatomical regression of an unresponsive prechoroidal cleft after switching to intravitreal Brolucizumab. The patient maintained cleft regression over time and no adverse events (i.e., RPE tears, intraocular inflammation) were observed during follow-up.
Conclusions and importance
To our knowledge, this case report is the first to analyze the clinical efficacy of brolucizumab targeting prechoroidal clefts. Clinical implication and pathogenesis of prechoroidal clefts are yet to be fully elucidated.
Keywords
Introduction
Prechoroidal cleft is an optical coherence tomography (OCT) biomarker firstly described by Mukai et al. in 2014. 1 It is defined as an optically empty pocket located between an outward bowing of Bruch's Membrane (BM) and the base of a fibrovascular retinal pigment epithelium (RPE) detachment, visible in patients affected by neovascular Age-related Macular Degeneration (nAMD) and Polypoid Choroidal Vasculopathy (PCV). This biomarker has been described as an independent risk factor for the formation of RPE rips, subretinal hemorrhages and fibrotic scars.2–5
Although the complete pathogenic mechanism leading to the onset of this peculiar lesion is still controversial, overall, this AMD phenotype should be considered a risk factor for poorer long-term visual prognosis. To date, no histopathologic study has been conducted with a specific focus on this topic.
We report the case of a patient with Type 1 Macular Neovascularization (MNV) presenting with subretinal fluid (SRF) unresponsive to conventional intravitreal (IV) anti-VEGF treatment (three loading doses of Bevacizumab and Aflibercept respectively) who later showed complete prechoroidal cleft regression after switch to IV Brolucizumab. The aim of this report is to propose a rationale for the clinical efficacy of brolucizumab in resolving prechoroidal cleft in comparison with traditional anti-VEGF drugs, potentially serving as a first-line therapy for future intervention. To date, only a limited number of studies have investigated the treatment options and outcomes of this peculiar finding.1,5,7,8,13
Case report
A 63-year-old female patient presented with unilateral metamorphopsia to the University Eye Clinic of Genoa (Italy). Baseline BCVA was 20/32 in the right eye and 20/20 in the left eye. She underwent full ophthalmologic examination including fundus photography (FP), fluorescein angiography (FA), indocyanine green angiography (ICGA), and optical coherence tomography (OCT) and OCT-Angiography (OCTA).
FP imaging of the right eye (Figure 1-A) demonstrated the presence of a foveal dome-shaped PED in the setting of AMD with soft drusen. Early Phase FA (00:34 s, Figure 1-B) revealed the presence of subretinal fluid (SRF) and dye pooling in the late phases underneath a vascularized PED, confirmed by ICGA (00:37 s, Figure 1-C). Baseline OCT (Figure 2-A) demonstrated the presence of Type 1 MNV secondary to neovascular AMD with a vascular PED, SRF and intraretinal hyperreflective foci (HRF). The fellow eye displayed the presence of intermediate AMD and was unremarkable for neovascular lesions.
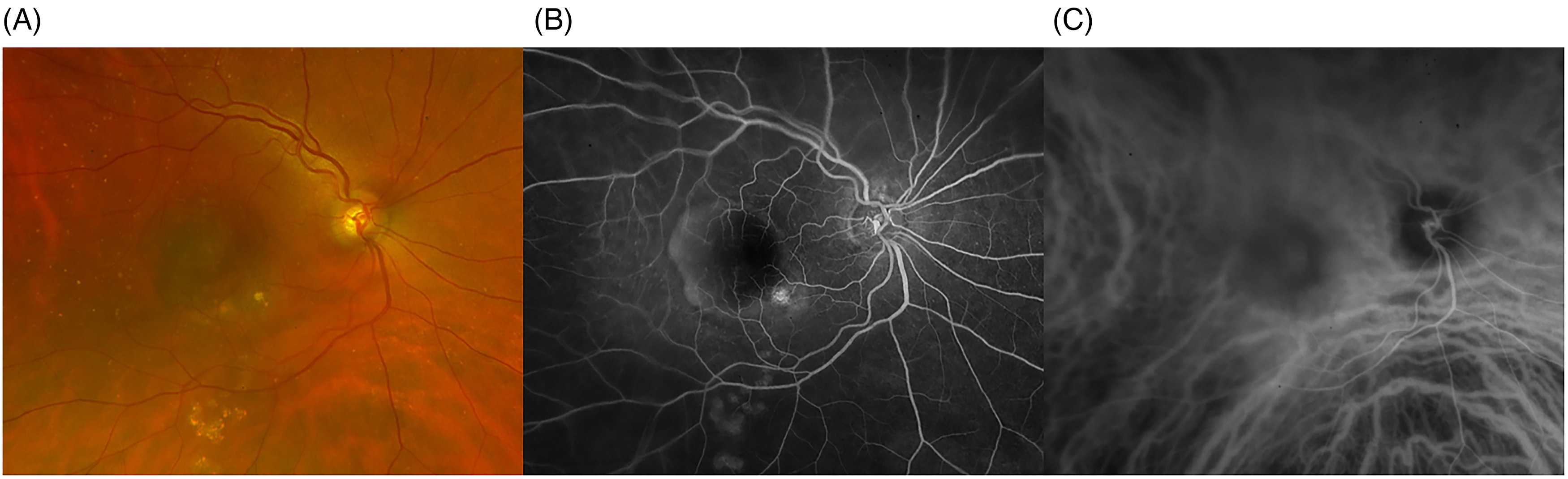
(A) Baseline Fundus photography (FP) of the study eye demonstrated a foveal dome-shaped PED in the setting of AMD with soft drusen. (B) Early Phase FA (00:34 s) revealed the presence of subretinal fluid (SRF) and dye pooling in the late phases underneath a vascularized PED. (C) ICGA (00:37 s) confirms the presence of occult Type 1 MNV secondary to neovascular AMD with a vascular PED and SRF.
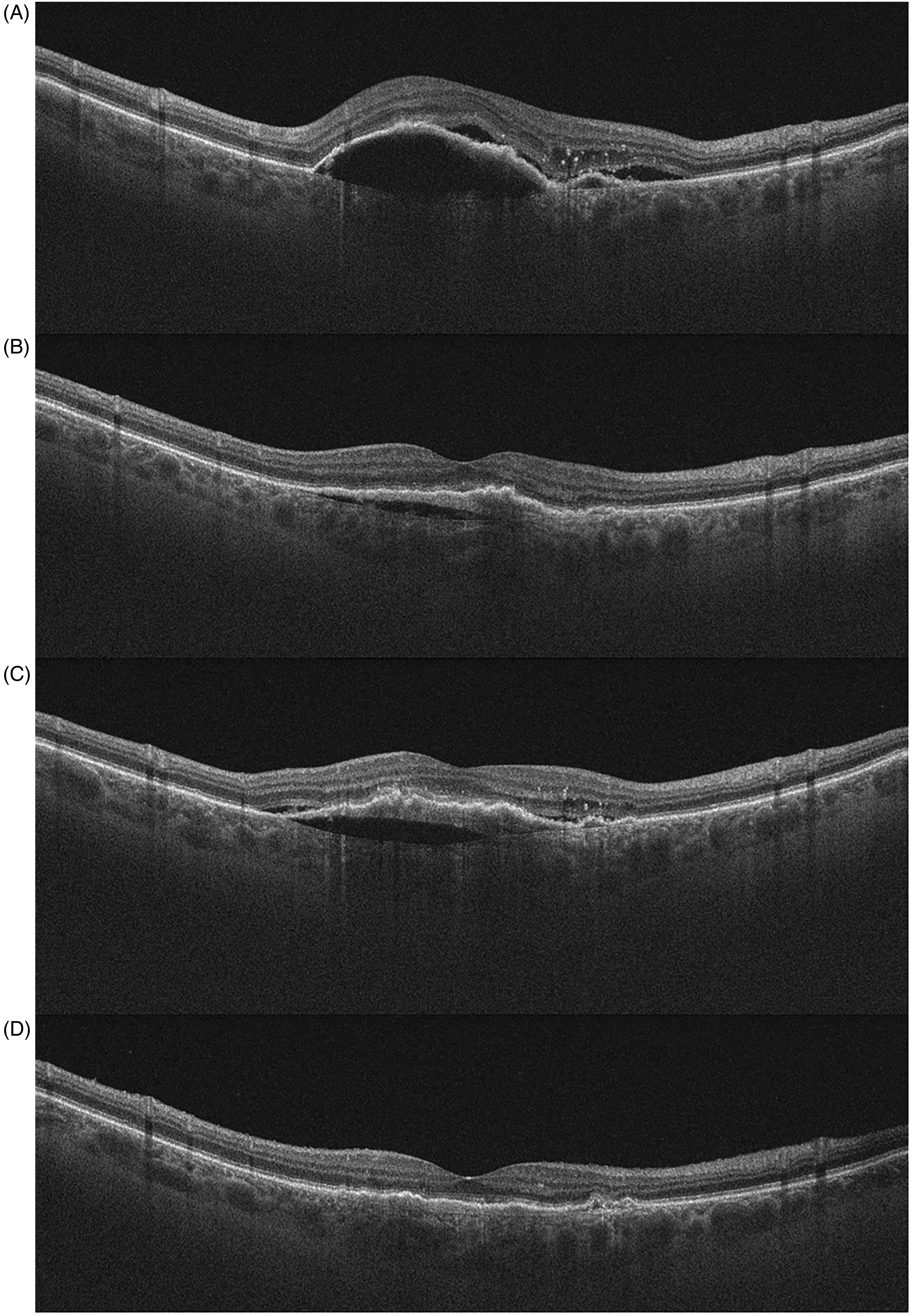
(A) Baseline OCT of the study eye demonstrated the presence of Type 1 MNV secondary to neovascular AMD with a vascular pigment epithelial detachment (PED), subretinal fluid (SRF) and intraretinal hyperreflective foci (HRF). (B) 4-month follow-up after 3 IV Bevacizumab displayed the development of sub-lesional prechoroidal cleft, despite reabsorption of SRF. (C) 9-month follow-up after three-doses of IV Aflibercept displayed an incomplete resolution of the lesion. (D) Final follow-up after switching to 3 monthly loading doses of IV Brolucizumab shows complete regression the of prechoroidal cleft.
Following the initial diagnosis, the patient was treated with three-monthly injections of Bevacizumab. OCT performed at 4-months follow-up (Figure 2-B) displayed the development of sub-lesional hyporeflective space between BM and the choroidal neovascularization after anti-VEGF therapy, consistent with prechoroidal cleft, despite reabsorption of SRF and decreased mean foveal thickness from 320 µm to 270 µm. Prechoroidal cleft was targeted with a switch to three-doses of IV Aflibercept, without clinical improvement at the 9-months follow-up (Figure 2-C).
Considering the risk profile of this phenotype we proposed the patient be switched to 3 monthly loading doses of IV Brolucizumab.
At the 13-months follow-up after completion of brolucizumab loading-doses (Figure 2-D) there was a consistent improvement on OCT imaging with complete regression of the prechoroidal cleft and SRF. Foveal thickness decreased to 250 µm and final BCVA was restored to 20/25. The patient underwent regular follow up visits and 2 IV extended injections (every 3 months): cleft regression was maintained over time (18-months follow-up), visual acuity was 20/25 and no adverse events (i.e., RPE tear, intraocular inflammation, retinal vasculopathy) were observed during follow-up.
Discussion and conclusion
Prechoroidal cleft has been reported to occur in 8,1 to 22,3% of nAMD patients3,4 (15% in a single report for Caucasian population). 5
This peculiar finding was first defined in a retrospective series of Asian patients diagnosed with nAMD (Type 1,2 and 3 MNV). The authors proposed that this anatomic space might be the result of fluid accumulation arising from active MNV components within the materials in PEDs. Of the 22 eyes included in the study, 54% showed a cleft reduction and 23% developed an RPE tear after treatment with photodynamic therapy (PDT; full dose full fluence) with a single injection of Ranibizumab administered 2 days prior. 1
Previously Neigel et al. 2 demonstrated the presence of a hyporeflective space, initially defined as a “cleft”, between the outward bowing of Bruch's membrane and the MNV complex in 6 patients prior to the formation of RPE tears. A similar finding was described by Kahn et al in relation to PCV. 6
More recently Kim et al 7 described a series of 29 patients pointing out a higher prevalence of Type 3 MNV with an insufficient response to anti-VEGF therapy among this subset. The same group later presented a larger series of 61 patients with a regression rate of 27,9% after anti-VEGF injections. The authors postulated that regression of prechoroidal cleft may occur with stabilization of neovascular lesions and progression to RPE atrophy, interpreting the cleft as a chronic structural alteration.
Cozzi et al. studied 27 Caucasian patients, proposing that the cleft represents a dynamic pocket of fluid, indirectly indicating MNV exudation and activity of the disease. 8
Recently, the introduction of the anti-VEGF agent Brolucizumab into the clinical practice (phase 3 HAWK and HARRIER trials) has raised concerns about balancing improved anatomical outcomes with potential risk of visually threatening adverse events, specifically vasculopathy. 9
Based on this review of the literature, we believe that the presence of a prechoroidal cleft represents a sign of inadequate control of subretinal exudation due to a poor fluid absorption in the vicinity of the choriocapillaris. Thus, based on a benefit-risk assessment, these lesions might benefit from a more effective treatment, even with the increased risk of possible adverse events.
Brolucizumab is composed of a humanized, scFv structure composed of 255 amino-acids, which inhibits all the isoforms of VEGF-A. 10 Brolucizumab molecular composition (26 kDa) provides better tissue penetration as compared with full—sized immunoglobulins G. 12 We propose that this molecule may penetrate into the sub-epithelium tissue plane in case of outward exudation from the MNV complex.
Interestingly, late phases of ICGA angiogram at the baseline visit in our patient did not show significant pooling of the dye into the prechoroidal lenticular space. This angiographic sign may indicate a minor choroidal contribution to the lesion formation, even in the case of a focal disruption of BM due to Type 1 MNV.
At this stage of the disease, Brolucizumab may have the potential to serve as a first-line therapy, with the aim of restoring the integrity of the BM-RPE complex (anchorage between RPE basal integrin receptors and BM matrix) 11 with a decreased number of injections.
Further studies will be needed to establish an optimal treatment strategy for this subset of patients and the long-term implications of Brolucizumab treatment in targeting prechoroidal clefts.
Footnotes
Authorship
All authors attest that they meet the current ICMJE criteria for authorship.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Patient consent
Oral informed consent was obtained from the patient for publication of this case report. This report does not contain any personal identifying information.
