Abstract
Objective
The purpose of this study was to compare the efficacy and safety of aflibercept (AFL) versus ranibizumab (RAN) for the treatment of diabetic macular edema (DME).
Methods
The PubMed, Embase, Cochrane Library, and CNKI databases were searched up to September 2022 to identify prospective randomized controlled trials (RCTs) comparing AFL with RAN for the treatment of DME. Review Manager 5.3 software was used for data analysis. We used the GRADE system to evaluate the quality of the evidence for each outcome.
Results
A total of 8 RCTs involving 1067 eyes (939 patients) were included; there were 526 eyes in the AFL group and 541 eyes in the RAN group. Meta-analysis revealed that there was no significant difference between RAN and AFL in the best-corrected visual acuity (BCVA) of DME patients at 6 months (WMD: −0.05, 95% CI = −0.12 to 0.01, moderate quality) and 12 months after injection (WMD: −0.02, 95% CI = −0.07 to 0.03, moderate quality). Additionally, there was no significant difference between RAN and AFL in the reduction of central macular thickness (CMT) at 6 months (WMD: −0.36, 95% CI = −24.99 to 24.26, very low quality) and 12 months after injection (WMD: −6.36, 95% CI = −16.30 to 3.59, low quality). Meta-analysis showed that the number of intravitreal injections (IVIs) for AFL was significantly lower than that for RAN (WMD: −0.47, 95% CI = −0.88 to −0.05, very low quality). There were fewer adverse reactions to AFL than to RAN, but the difference was not significant.
Conclusion
This study found that there was no difference in BCVA, CMT or adverse reactions between AFL and RAN at 6 and 12 months of follow-up, but AFL needed fewer IVIs than RAN.
Introduction
Diabetic macular edema (DME) is a common ocular complication in diabetic patients, 1 and it is the first cause of blindness in adults in Europe and United States. 2 The incidence of macular oedema in patients with type 2 diabetes and patients with type 1 diabetes is 14% and 29%, respectively.3,4 Based on the high incidence of DME and the risk of blindness, the management of DME is particularly important. Currently, the treatment is based on oral medication, laser photocoagulation, intravitreal injections (IVIs) and vitrectomy,5,6 and the clinical efficacy of anti-vascular endothelial growth factor (VEGF) agents and dexamethasone implants has attracted considerable attention.7,8 Hu et al. 9 found that vitrectomy can improve the vision of DME patients and reduce macular oedema, and vitrectomy combined with internal limiting membrane peeling can significantly reduce macular oedema without increasing the incidence of complications. Anti-VEGF drugs are believed to improve the permeability of retinal and choroidal microvessels, reduce intraretinal and subretinal fluid accumulation, and improve patients’ vision, gradually becoming the first-line treatment for DME. 10 Corticosteroid sustained-release agents are also a hot topic in the treatment of DME due to their long duration of maintaining effective drug concentration, simple operation, and reliable efficacy. 11 Boyer et al. found that intravitreal injection of dexamethasone, which is a sustained-release agent, can reduce macular oedema and improve patients’ vision. 12 Malcles et al. 13 found that implantation of Ozurdex (dexamethasone implant) can improve macular oedema, reduce macular fovea thickness and have good safety in DME patients. Although some patients may experience an increase in intraocular pressure after receiving Ozurdex treatment, 13 most patients can effectively control intraocular pressure with intraocular pressure-lowering drugs.
VEGF is considered to be closely related to the occurrence of DME.14,15 Intravitreous anti-VEGF therapy is considered an effective intervention. 16 However, anti-VEGF therapy requires repeated injections, which increase medical expenditure and the risk of infection.17,18 Based on the guidelines of the Food and Drug Administration (FDA) for the treatment of DME and the level of evidence that has been disclosed, we chose to compare the efficacy of ranibizumab (RAN) and aflibercept (AFL) for the treatment of DME. RAN is a special humanized fragment antigen-binding region of an antibody designed for eye use that has high affinity and can inhibit all VEGF-A subtypes.19,20 AFL is a type of VEGF recombinant fusion protein that can inhibit the protein subtypes of VEGF-A and VEGF-B. 21 In addition, AFL can prolong the injection interval, effectively maintaining vision and promoting the recovery of retinal anatomy.22,23 There are still no evidence-based medical data to compare the efficacy of AFL and RAN for the treatment of DME, which makes it difficult for clinicians to choose between the two drug prescriptions. This meta-analysis evaluates the efficacy and safety of AFL versus RAN in the treatment of DME, providing an evidence-based recommendation for clinical practice and health economics applications.
Materials and methods
Database and literature search strategy
We searched the PubMed, Embase, Cochrane Library, and China National Knowledge Infrastructure (CNKI) databases up to September 2022 to identify randomized controlled trials (RCTs) comparing AFL and RAN for the treatment of DME. The search terms included ranibizumab, aflibercept, diabetic macular edema, macular edema, and diabetes. The search strategy is shown in supplementary material 1.
Inclusion and exclusion criteria
The inclusion criteria were as follows: 1) the study design was a prospective RCT; 2) the intervention consisted of at least two arms, one arm of which one was AFL (2.0 mg) and the other was RAN (0.5 mg); 3) the included cases were diagnosed with DME and had no other complications (e.g., retinal detachment) or other factors that cause tractional oedema (e.g., epiretinal membranes); 4) continuous outcome variables were reported as the mean ± standard deviation (SD); and 5) the outcome indicators reported at least one endpoint of concern for this study.
The exclusion criteria were as follows: 1) duplicate publications; 2) studies from which data could not be extracted or combined; and 3) different studies published for the same case group.
Data extraction and quality evaluation
Two evaluators (XHS and WZ) screened and cross-checked the literature independently in accordance with the inclusion and exclusion criteria. Any disagreements between the two evaluators (XHS and WZ) were resolved by discussion with the third evaluator (HYC). The following data were extracted from the included studies: author, publication year, sample size, age, follow-up time, glycosylated haemoglobin level, diabetes course, outcome indicators and corresponding measurement data, and literature quality evaluation elements.
The Cochrane risk of bias tool was used to assess the quality of the included literature. 24 The evaluation dimension includes 7 items, which can be determined as low risk, high risk or unclear risk.
Outcome parameters
A total of 4 outcome indicators were evaluated in this study: best-corrected visual acuity (BCVA), central macular thickness (CMT), number of intravitreal injections (IVI) and adverse reactions.
Data analysis
Review Manager 5.3 software was used for data analysis in this study. Continuous variables are represented by weighted mean deviation (WMD) and 95% confidence interval (CI). We determined the level of heterogeneity of each outcome using the I2 statistic. I2 ≤ 50% indicated a low level of heterogeneity, and in such cases, the fixed effects model is used for pooled data analysis. Otherwise, the random effects model is used for pooled data analysis. If the number of studies reporting an outcome was greater than or equal to 10, a funnel chart was constructed to evaluate publication bias.
Quality of evidence
We used the grading method recommended by the Grading of Recommendations, Assessment, Development and Evaluations (GRADE) system 25 to grade the outcome indicators of this study. According to the GRADE grading system, we evaluated each outcome indicator from five aspects: bias risk, inconsistency, indirectness, imprecision and publication bias. The evidence quality of outcome indicators can be categorized as high, moderate, low and very low.
Results
Search results
We retrieved 945 articles from four databases, including 314 from PubMed, 104 from the Cochrane Library, 496 from Embase and 31 from CNKI. After removing duplicate publications and screening the titles and abstracts, 52 articles remained for full-text screening. After reading the remaining 52 research papers, we ultimately included 8 RCTs.26–33 The titles of the 44 studies and the reasons for their exclusion are shown in supplementary material 2. The document retrieval process is shown in Figure 1.
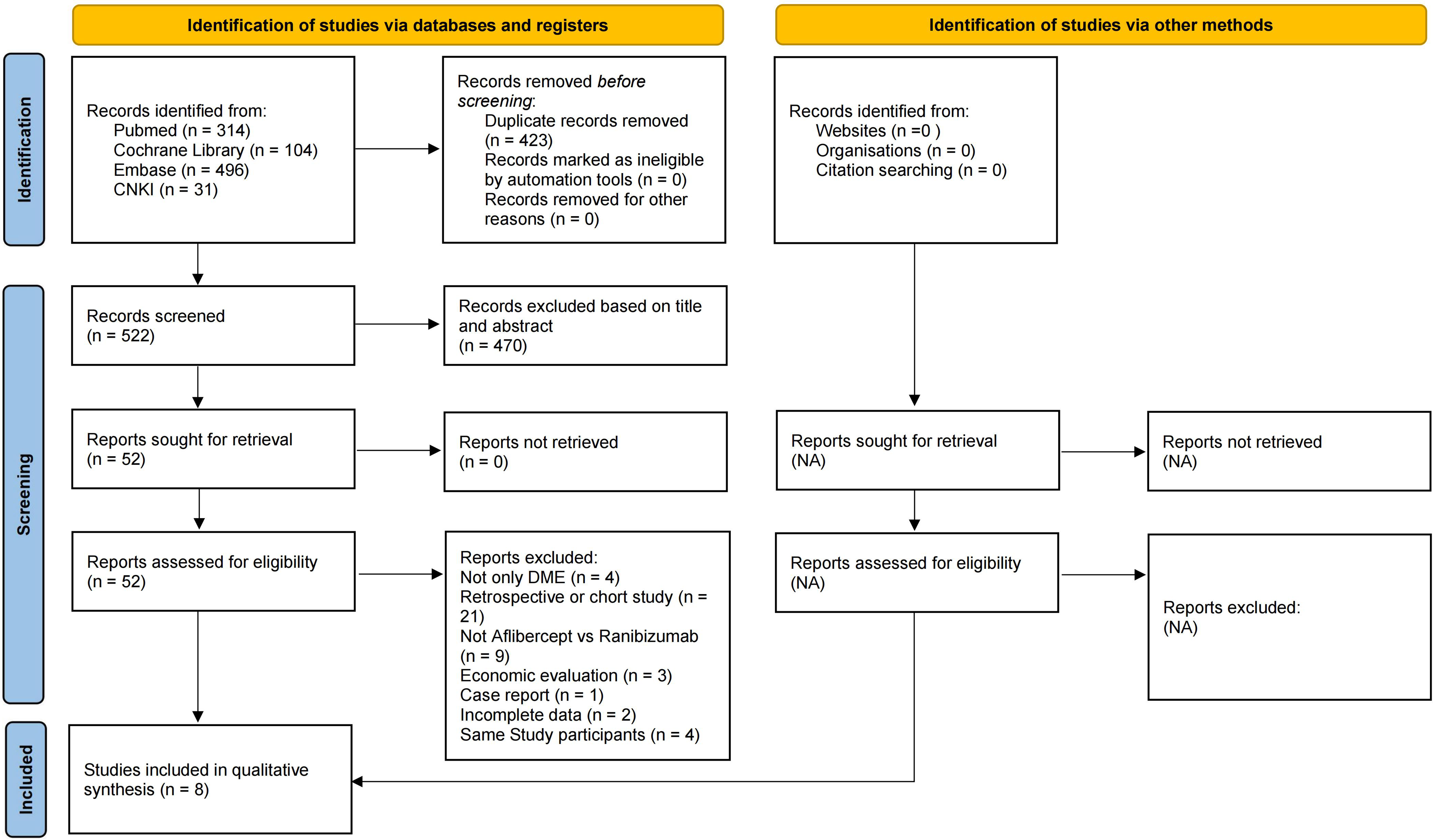
Preferred reporting items for systematic reviews and meta-analyses (PRISMA) flow diagram.
Characteristics of the included studies
Eight RCTs26–33 involving 1067 eyes (939 patients) were included in this meta-analysis; there were 526 eyes in the AFL group and 541 eyes in the RAN group. The average follow-up period was 11.5 months. There was no statistically significant difference in age or baseline data between the two intervention groups. The basic characteristics of the included studies are shown in Table 1.
Characteristics of the included studies.
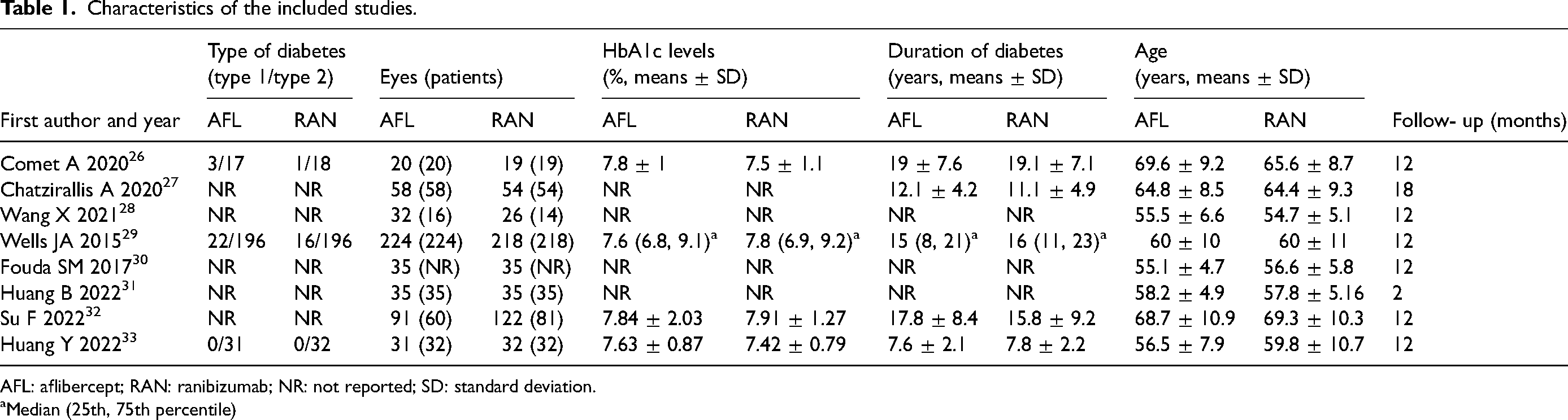
AFL: aflibercept; RAN: ranibizumab; NR: not reported; SD: standard deviation.
Median (25th, 75th percentile)
Risk of bias of the included studies
Three studies26,28,32 did not adopt the random allocation method, and thus, these studies were judged as having a high risk of bias. Two studies30,31 only mentioned randomization but did not specify specific random allocation methods, so they were judged as having an unclear risk of bias. The other three studies27,29,33 explicitly adopted random methods, such as the random number table method. Regarding the blinding method, only one study 29 had a low risk of bias, and the other seven studies26–28,30–33 all had an unclear risk of bias. All included studies26–33 had a low risk of bias with respect to incomplete data, selective reporting and other risks (Figure 2).
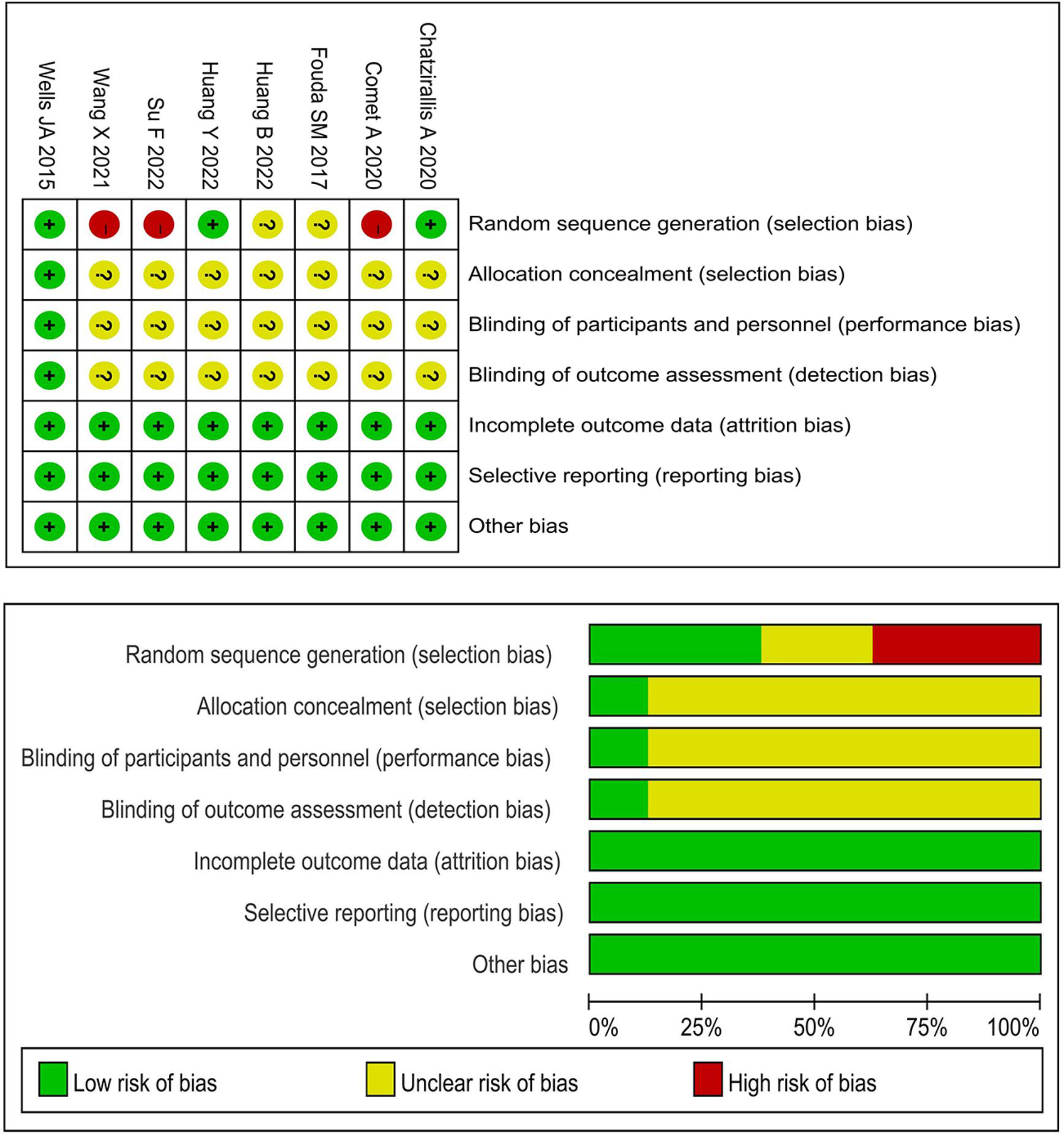
Risk of bias.
Meta-analysis
BCVA
A total of 6 RCTs26,28–30,32,33 reported BCVA, of which 4 studies28,30,32,33 converted BCVA into logarithm of the minimum angle of resolution (logMAR) vision. There was a low level of heterogeneity among the BCVA comparisons at 6 months (I2 = 0%, P = 0.47) and 12 months after injection (I2 = 0%, P = 0.44); therefore, a fixed effects model was adopted. There was no significant difference between RAN and AFL in improving the vision of DME patients at 6 months (WMD: −0.05, 95% CI = −0.12 to 0.01, P = 0.12) and 12 months after injection (WMD: −0.02, 95% CI = −0.07 to 0.03, P = 0.46) (Figure 3). Two studies26,29 adopted Early Treatment Diabetic Retinopathy Study (ETDRS) letter scores. Meta-analysis showed that there was no significant difference between AFL and RAN in BCVA (ETDRS) at 12 months after injection (WMD: 1.82, 95% CI = −0.16 to 3.79, P = 0.07) (Supplementary material 3).
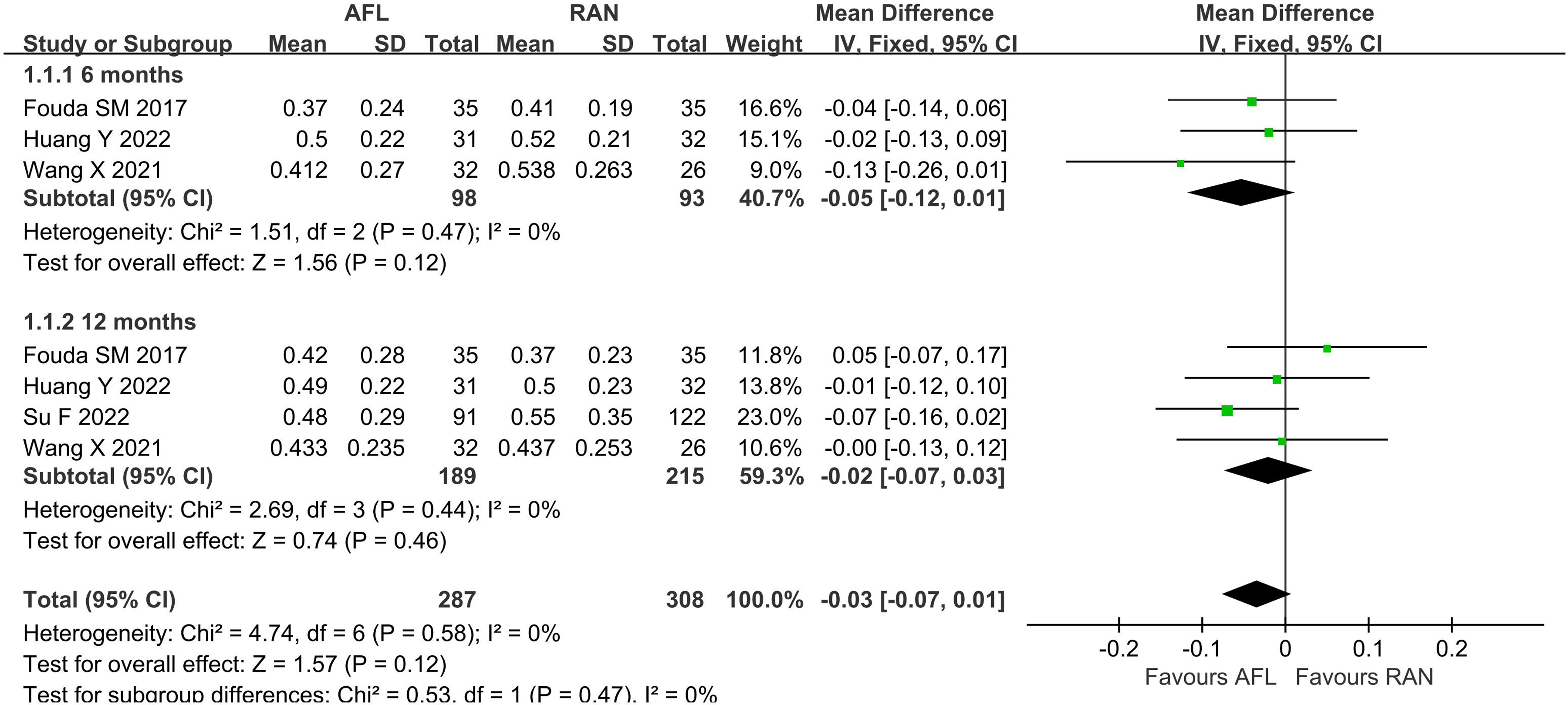
BCVA (logMAR) in AFL versus RAN (6- and 12- month follow-ups).
CMT
A total of 4 studies26,28,30,33 reported comparative data of CMT at 6 months after treatment. There was a high level of heterogeneity among the studies (I2 = 53%, P = 0.10); therefore, a random effects model was used. Meta-analysis revealed that there was no statistically significant difference between AFL and RAN in CMT changes at 6 months after treatment (WMD: −0.36, 95% CI = −24.99 to 24.26, P = 0.98) (Figure 4).
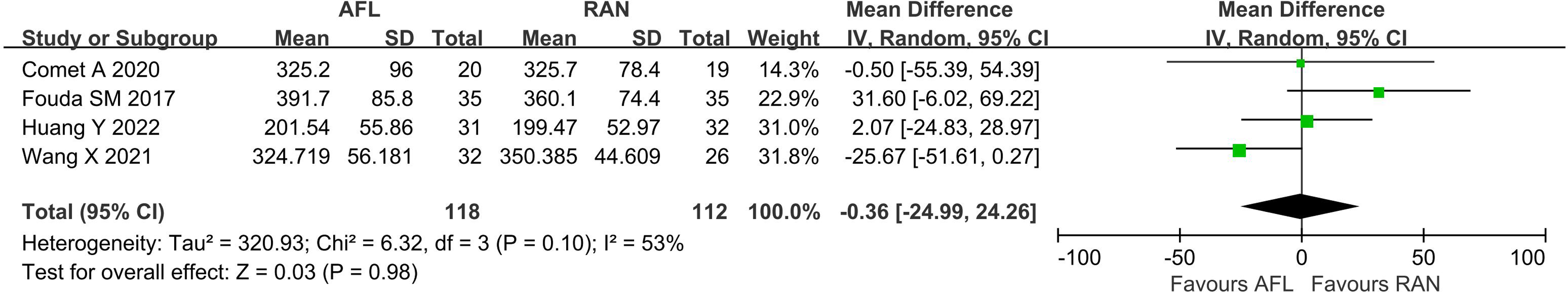
CMT in AFL versus RAN (6 months follow-up).
Six studies26,28–30,32,33 reported changes in CMT at 12 months of treatment. There was a low level of heterogeneity among the studies (I2 = 11%, P = 0.34); therefore, the fixed effects model was used. Meta-analysis revealed that there was no significant difference between AFL and RAN in CMT changes at 12 months after treatment (WMD: −6.36, 95% CI = −16.30 to 3.59, P = 0.21) (Figure 5).
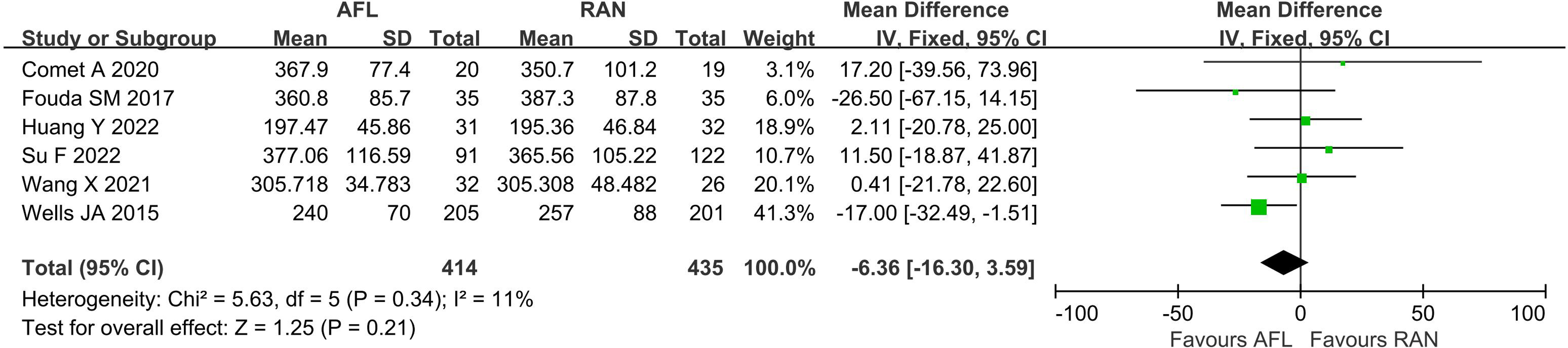
CMT in AFL versus RAN (12 months follow-up).
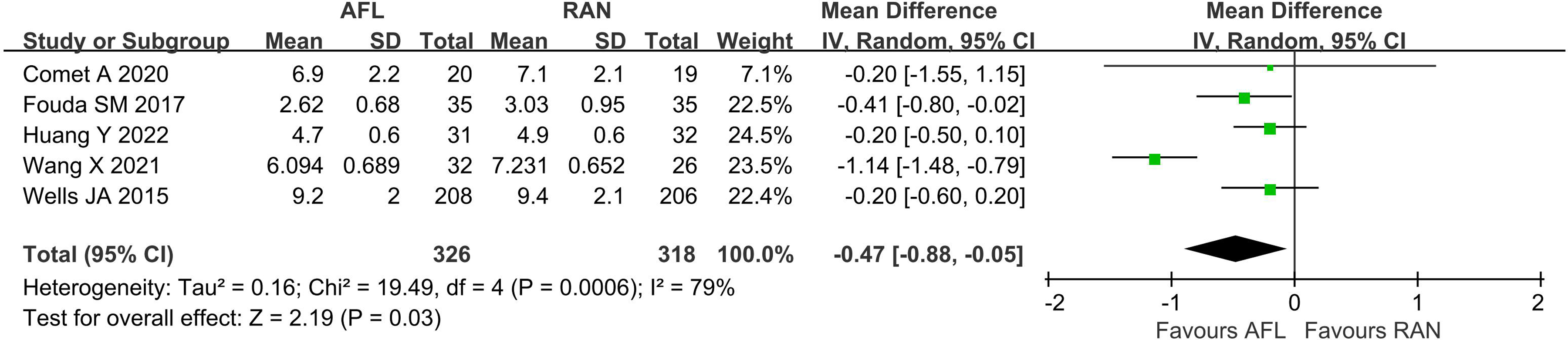
Number of IVIs in AFL versus RAN (12 months follow-up).
Number of IVIs
Five studies26,28–30,33 compared the number of IVIs within 12 months of follow-up. There was a high level of heterogeneity among the studies (I2 = 79%, P = 0.0006); therefore, a random effects model was used. Meta-analysis showed that the mean number of IVIs for AFL was minor than that for RAN (WMD: −0.47, 95% CI = −0.88 to −0.05; P = 0.03) (Figure 6).
Adverse reactions
All included studies26–33 examined the occurrence of adverse reactions. Six studies26–28,30–32 reported no adverse reactions in the AFL group and RAN group. Two studies29,33 reported adverse reactions in both groups, but there was no statistically significant difference between the two groups.
Quality of evidence
We evaluated the GRADE evidence level of the five outcome indicators examined in this study. The quality of evidence was moderate for BCVA (6- and 12-month follow-up). The quality of evidence was low for CMT (12-month follow-up). The quality of evidence was very low for CMT (6-month follow-up) and the number of IVIs. The factors that decreased the quality of evidence mainly included inconsistency, accuracy and limitations (Table 2).
Evaluation table of evidence level.
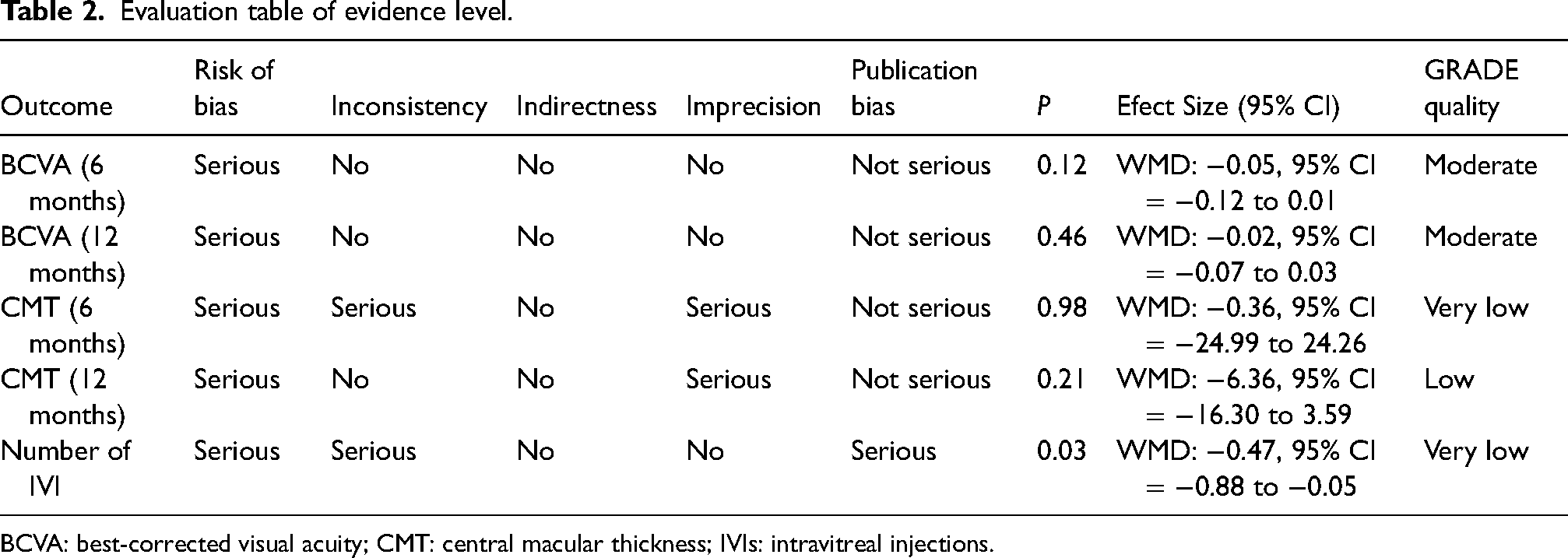
BCVA: best-corrected visual acuity; CMT: central macular thickness; IVIs: intravitreal injections.
Publication bias
Since fewer than 10 studies were included in this meta-analysis, we did not construct a funnel chart to evaluate publication bias.
Discussion
DME is related to the excessive permeability of retinal vessels, which leads to an increase in fluid accumulation in the retina and an increase in macular thickness.34,35 The binding of VEGF expression promotes the growth of vascular endothelial cells and vascular proliferation to face diabetic-based damage to the blood‒retinal barrier, but on the other hand, it leads to an increase in capillary permeability, with the formation of persistent intraretinal oedema.34,35 Previous studies36,37 about VEGF inhibition have proven that AFL and RAN have good efficacy in improving BCVA and CMT in the treatment of DME, but the difference between the two drugs remains unclear. Bressler SB et al. found that AFL, RAN and bevacizumab (BEV) can improve retinopathy in DME patients. 38 Compared with AFL or RAN, the treatment effect of BEV is worse, but no significant difference was observed between AFL and RAN (P = 0.51). 38 Two similar randomized phase III clinical trials39,40 found that AFL can improve the vision of patients with DME and reduce the central retinal thickness to a greater extent than lasers, indicating that AFL has potential clinical promotion value in the treatment of DME. The results of three systematic reviews and network meta-analyses41–43 published between 2014 and 2017 showed that there was still no clear and significant difference between AFL and RAN in the improvement of BCVA. Therefore, it is still necessary to obtain high-quality evidence regarding the comparative efficacy of AFL and RAN in DME patients. The results of this meta-analysis showed that there was no statistically significant difference between AFL and RAN in BCVA and CMT at 6 and 12 months after treatment. In terms of the number of IVIs, the number of injections of AFL was minor than that of RAN in the 12-month follow-up period, which indicates that AFL may have better health economic benefits and improve patient satisfaction. In addition, the treatments revealed safe, and there was no statistically significant difference in the incidence of adverse reactions between the two groups. Our study confirms the results shown by Wells et al., 44 who previously described that the number of AFL injections needed is minor than using RAN.
This study has some limitations. First, the blinding method and random allocation method of the included RCTs have defects, thus affecting the reliability of the conclusions of this study. Second, most of the samples included in the study were small, which may have led to the small-sample effect in the pooled analyses. Third, the follow-up time was 12 months, which is not conducive to our understanding of the long-term efficacy of drugs and long-term health economics effects.
Conclusion
In this study, we found that there are no differences between AFL and RAN in improving BCVA and CMT among DME patients, as they are both safe therapies, but the number of IVIs of AFL in the 12-month follow-up period was lower. Considering the limitations of the included studies and this meta-analysis, the conclusions of this study still need to be supplemented and verified by a large-sample, multicentre, high-quality RCT.
Supplemental Material
sj-docx-1-ejo-10.1177_11206721231178658 - Supplemental material for Aflibercept versus ranibizumab for diabetic macular edema: A meta-analysis
Supplemental material, sj-docx-1-ejo-10.1177_11206721231178658 for Aflibercept versus ranibizumab for diabetic macular edema: A meta-analysis by Haiyan Chen, Xuehui Shi, Wang Zhang and Qianqian Han in European Journal of Ophthalmology
Supplemental Material
sj-docx-2-ejo-10.1177_11206721231178658 - Supplemental material for Aflibercept versus ranibizumab for diabetic macular edema: A meta-analysis
Supplemental material, sj-docx-2-ejo-10.1177_11206721231178658 for Aflibercept versus ranibizumab for diabetic macular edema: A meta-analysis by Haiyan Chen, Xuehui Shi, Wang Zhang and Qianqian Han in European Journal of Ophthalmology
Supplemental Material
sj-docx-3-ejo-10.1177_11206721231178658 - Supplemental material for Aflibercept versus ranibizumab for diabetic macular edema: A meta-analysis
Supplemental material, sj-docx-3-ejo-10.1177_11206721231178658 for Aflibercept versus ranibizumab for diabetic macular edema: A meta-analysis by Haiyan Chen, Xuehui Shi, Wang Zhang and Qianqian Han in European Journal of Ophthalmology
Footnotes
Author contributions
HYC conceptualized and design the study. XHS and WZ performed data curation and investigation. HYC reviewed the manuscript. XHS and QQH performed the statistical analysis and reviewed the manuscript. All authors approve the final manuscript.
Consent for publication
All authors approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval and consent to participate
Systematic review did not require ethical clearance or informed consent.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
