Abstract
Background
Anterior Chamber bleeding without vitreous hemorrhage had been described after the removal of 23G vitrectomy cannulas. We report the case of an anterior chamber bleeding after an intravitreal Dexamethasone implant.
Case Report
One patient with macular edema due to central retinal vein occlusion in a vitrectomized eye underwent an intravitreal Dexamethasone implant. After the injection the patient suffered from anterior chamber bleeding without signs of vitreous hemorrhage. The complication resolved with a conservative treatment.
Conclusion
Anterior Chamber bleeding is a possible complication of dexamethasone implant, that can be treated in a conservative way.
Keywords
Introduction
The sustained-release 0.7 mg intravitreal Dexamethasone (Dex) implant (Ozurdex®, Allergan Pharmaceuticals, Irvine, CA, USA) is an efficient therapeutic option for treating Macular Edema (ME). The United States (US) Food and Drug Administration (FDA) approved the Ozurdex® for the treatment of ME secondary to: diabetic retinopathy, Retinal Vein Occlusion (RVO) and non-infectious posterior uveitis. 1
Regarding RVO, Euretina guidelines recommend Ozurdex® on a second-choice level for non-responders to anti-VEGF, or as first line in patient with a recent history of a major cardiovascular event or unwilling to come for monthly injections. 2 Nevertheless, none of the studies on which the guidelines are based had considered previously vitrectomized eyes. Few articles on this topic are present in literature. It is known that the pharmacokinetic of anti-VEGFs is different in a vitrectomized eye, with a reduction of the half-life. 3 However, for dexamethasone implant the pharmacokinetic profile is similar for vitrectomized and not vitrectomized eyes 4 and some authors have reached an improvement with Ozurdex® in a vitrectomized eye with ME resistant to anti-VEGF. 5
Ocular complications of dexamethasone implant can be related to the drug itself or to the injection. The first type consists especially of elevation of intraocular pressure (IOP) and cataract formation. The second type includes some less severe like conjunctival hemorrhage or vitreous detachment and others more severe, but infrequent, as retinal tear/detachment, vitreous hemorrhage (VH), endophthalmitis and cataract secondary to lens injury. Rezkallah et al. compared the safety of Dex implant in vitrectomized and not vitrectomized eyes showing no difference in IOP raise between the two groups. However they did not describe the rate of other complications in this two groups. 6 Despite the aforementioned complications, Ozurdex® injection is a safe procedure.
Case report
A 72-years old man, already followed at our ophthalmology unit since 2016 for Central Retinal Vein Occlusion (CRVO) in the Right Eye (RE), presented in March 2022 for a worsening in visual acuity in RE. Regarding systemic medication the patient was treated for Hypertension and Dyslipidemia, he was also Cardiopathic, in therapy with New Oral Anticoagulants (Apixaban). Concerning ophthalmic history, the patient performed phacoemulsification and IOL implantation in both eyes in 2013. In March 2016 he suffered from a RE CRVO with ME treated by Ozurdex® implant, with a relapse in April 2017 treated again with a single Ozurdex®. In February 2018 he performed a pars plana vitrectomy with peeling of an epiretinal membrane for lamellar macular hole in RE (Figure 1).
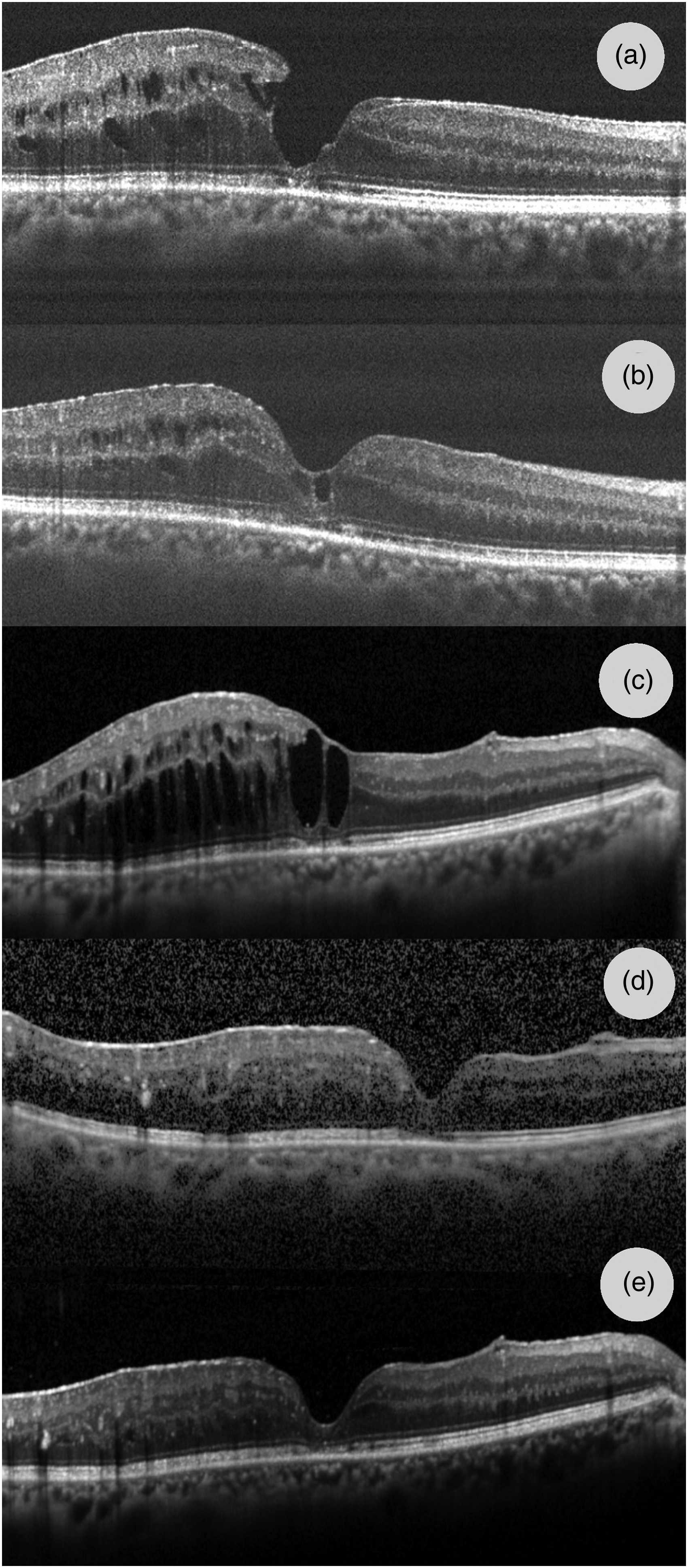
SD-OCT of the right eye. (a) Before Vitrectomy, February 2018 (b) Two months after vitrectomy (c) Before the Ozurdex® implant, July 2022. (d) 5 days after the implant. (e) After the resolution of the complication.
In July 2022 the RE VA was 70 letters (20/40), another relapse of ME was diagnosed (Figure 1). Fluorescein angiography demonstrated the absence of peripheral ischemia and gonioscopy showed the absence of neo vessels at the angle. Another Ozurdex® implant was indicated for the treatment of ME. The injection was performed in the setting of an Arc Sterile, the same described by Grassi et al. and which safety was demonstrated by Furino et al.7,8 We used a single use sterile injection set and the instruments were prepared on a sterile tray. 5% Povidone iodine (PVI) (Oftasteril 5%; Alfa Intes, Casoria, Italy) was applied on periocular skin and eyelids and instilled in the conjunctival sac, 3 min before injection. Sterile anesthetic drops were instilled multiple times. The patient was told to look upward, and the injection was performed inferior temporal, in Pars Plana, 3.5 mm from the limbus since the patient was pseudophakic, with a self-sealing sclerotomy. The Ozurdex was injected without problem, the eye was covered, and the patient dismissed.
Two days after the injection the patient referred to the emergency room whit a persistent decrease in VA in RE to hand motion, since the day after the injection when the eyepatch was removed. There was subconjunctival hemorrhage in the site of sclerotomy, the aqueous humor was murky with blood cloths and hyphema (Figure 2). The structures behind the iris were not visible. The patient reported no ocular trauma after the injection. A B-Scan ultrasound was performed that pointed out no vitreous chamber hemorrhage or any retinal tear (Figure 3). Therefore, a topical 0,15% Dexamethasone was suggested q.i.d. Five days after the injection the situation was improving, the VA already returned to 70 letters (20/40), the IOP was 14 mmHg and at OCT the solution of the ME was evident, even if the quality of the image was not good (Figure 1). Two weeks from the injection VA was 80 letters (20/25) the anterior chamber cloths were reabsorbed, the aqueous humor was clear (Figure 2), the retina was visible - confirming the absence of retinal tears - and the OCT scan confirmed the resolution of the ME (Figure 1). Topical steroid was suspended. In all this period no IOP raise was recorded.
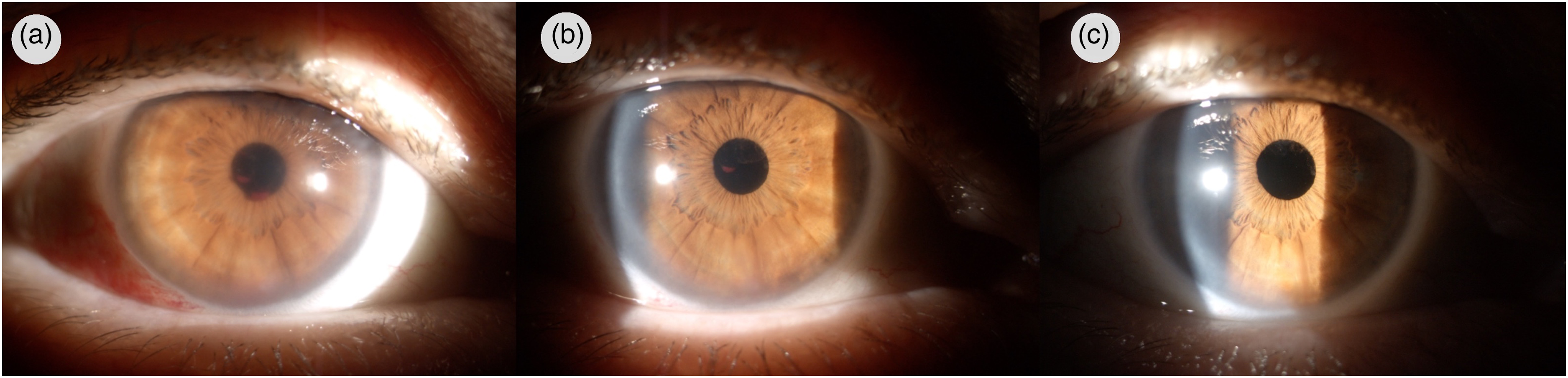
Anterior segment of the right eye. (a) 2 days from the injection. (b) 5 days after the injection. (c) 14 days after the injection.
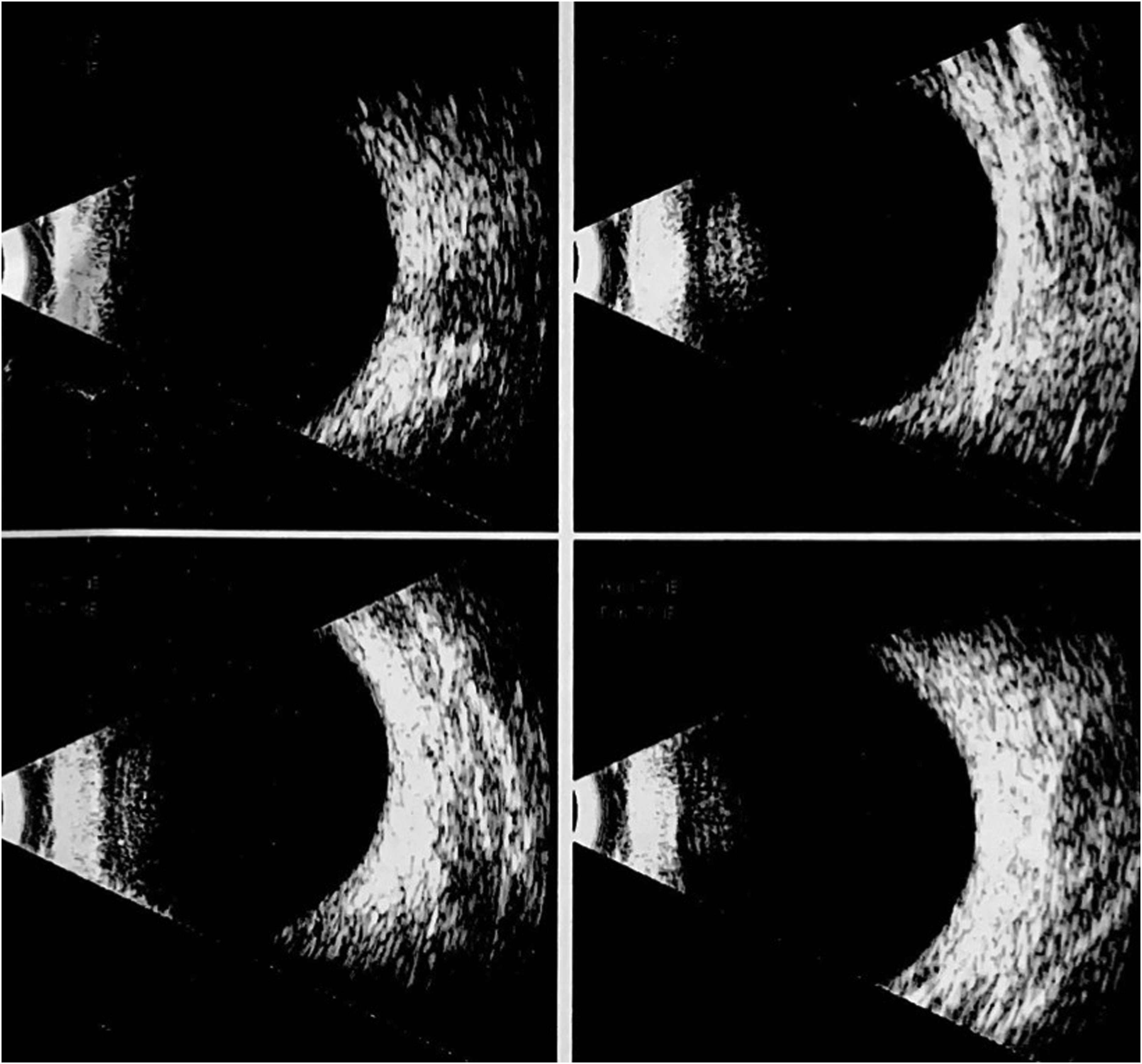
B-Scan ultrasound of the right eye, 2 days after the injection.
The Institutional Review Board of the Eye Clinic of University of Bari approved the study protocol. All clinical procedures were conducted according to the principles of the Declaration of Helsinki. The patient provided informed consent for all procedures, and their possible complications were explained.
Discussion
The peculiarity of this case is the anterior chamber bleeding, without VH. It must be said that even if we could not find any sign of VH it is impossible to establish with absolute certainty that there was not one. However, there was such a disparity between the blood in the anterior chamber and the vitreous chamber two days after the injection that we hypothesized the primary bleeding site was the anterior chamber. In addition, even if the patient was pseudophakic, the intraocular lens was in the bag and the posterior capsule was intact. Therefore ikthe integrity of the whole lens diaphragm points towards the anterior chamber as a primary site of bleeding, instead of the vitreous chamber.
To our knowledge in literature there are no cases of prevalent anterior chamber bleeding, following an Ozurdex® implant. Nevertheless, there are two cases reported of presumed sclerotomy site bleeding into the anterior chamber after the removal of a 23-gauge microcannula in 23-gauge sutureless vitrectomy. 9 Even if those are very different procedures, the 22-gauge Ozurdex needle is similar to a PPV trocar and the shape of the self-sealing sclerotomy is the same, so it is possible that also the origin of bleeding is the same i.e., the sclerotomy. A possible pathophysiologic mechanism could be a blood inflow from the sclerotomy trough the suprachoroidal space into the AC. In addition, both our case and the ones reported in literature resolved within 7 to 14 days with conservative management.
A key pathogenetic role in our case was played by the anticoagulant therapy of the patient, which were. 10 It is very likely that the intravitreal injection of an anti-VEGF drug would have not cause a bleeding, due to the smaller caliber of the needle. Nevertheless in our opinion Ozurdex® remain the best choice in this case, since anti-VEGFs half-life is reduced in a vitrectomized eye. 4 This is supported also by the complete resolution of ME after the last Dexamethasone implant (Figure 1) and by the improvement in VA.
In conclusion anterior chamber bleeding is a possible complication of Ozurdex® implant, even though it is very rare. It is not a severe condition and can be treated in a conservative way, resolving in one or two weeks without consequences.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
