Abstract
Purpose
The purpose of this study was to assess the use of topical tacrolimus ointment in preventing rejection in high-risk corneal grafts, when added to the standard immunosuppressive regimen.
Methods
We conducted an observational, retrospective study using clinical data of high-risk patients subjected to penetrating keratoplasty, who were treated with topical tacrolimus ointment 0.2 mg/g twice a day plus topical dexamethasone 0.1 mg/ml 6 id and compared it with a similar control group treated with topical dexamethasone 0.1 mg/ml 6 id alone. High-risk status was attributed to patients with previous ipsilateral corneal graft failure, two or more quadrants with corneal neovascularization or an infectious or inflammatory corneal disease.
Results
We analysed 53 patients in the trial group versus 53 patients in the control group, with similar age, baseline diagnosis and risk factors, and median follow-up times of 30 and and 24 months, respectively. Survival analysis showed a higher graft survival rate at all follow-up periods for patients treated with topical tacrolimus (p < 0.01). No adverse reactions were reported.
Discussion
This study shows that topical tacrolimus ointment increases the survival rate of the graft if added to the previous topical steroid regimen in high-risk patients.
Conclusion
Topical tacrolimus is safe and effective in prolonging graft survival in high-risk patients.
Introduction
Corneal grafting is the world's most performed and most successful allogenic transplant: the cornea's unique immunological privilege allows high survival rates in lower risk cases (90% at 1-year and 55% 15-year rates). 1 loss of said privilege in higher risk cases decreases survival rates to 30% over 10 years, with little improvement over the last decades, yielding comparable long-term success to cardiac, renal and hepatic transplants. 2
Graft immune rejection remains the leading cause for corneal graft failure. 3 Several factors have been associated with increased graft rejection risk, such as pre-existing glaucoma, lens status, Human Leucocyte Antigen mismatches, among others.2,4,5 The incidence of rejection appears to increase strongly with three risk factors: corneal neovascularization, namely the number of affected quadrants, density, and activity of neovessels; the number of previous ipsilateral corneal grafts; and an infectious or inflammatory disease that otherwise compromises corneal immune privilege. 5
In most corneal transplants, but more so in such high-risk cases, long-term topical or systemic immunosuppression is warranted. Current standard of care is based on topical corticosteroids, but its chronic use is associated with adverse effects such as increased intraocular pressure and cataract development. 6 Other alternatives or adjunctive therapies have been studied, such as cyclosporine A (CsA) and mycophenolate mofetil (MMF). However, significant effectiveness in increasing graft survival has not been demonstrated for topical use, and its adverse effects may outweigh the benefits for systemic use.7,8
Tacrolimus, or FK506, is another alternative immunosuppressant obtained from the bacteria Streptomyces tsukubaensis and first isolated in 1984. 9 It supresses production of T lymphocytes and IL-2 receptors, reducing pro-inflammatory cytokines, and has been successfully used in the prevention of rejection for hepatic, renal and cardiac allogenic grafts. Its topical formula was designed for the treatment of atopic dermatitis and is used off label in the prevention of corneal graft rejection and other ophthalmological maladies. 10
Its systemic use is associated with side effects such as hypertension, diarrhoea and hepato-, nephron- and neurotoxicity. When administered topically in the ocular surface it demonstrates high corneal concentration and adequate aqueous humour distribution without being detected in the bloodstream, thus decreasing the systemic adverse effects. 11 Some of its side effects may include headaches, fever, myalgias and ocular itching, but it is generally well tolerated. 10 The effects of long-term use of topical tacrolimus are not well documented, and rare cases of malignancy have been associated with dermatological use, but without any evidence regarding ocular use.
Recently, topical tacrolimus has emerged as a viable alternative and shown promise in earlier studies as a safe and effective treatment in prolonging high-risk graft survival when combined with topical corticosteroids.12–15 However, some of these studies have significant limitations, such as small sample sizes (Zhai L.), limited scope in indications and significant sample differences between trial and control groups (Magalhães O.) or relatively short post-transplant follow-up (Faramarzi A). Moreover, many of these studies aim to compare the effectiveness of topical tacrolimus against that of other experimental immunosuppressive strategies, such as oral immunosuppression with MMF or topical CsA, and do not directly compare topical tacrolimus combined with topical corticosteroids to the standard of care (topical corticosteroids alone in the long term).
The objective of this study is to evaluate the impact of topical tacrolimus in graft survival when added to the standard of care topical corticotherapy.
Methods
A retrospective observational study was conducted, with data being collected from clinical files and subsequently anonymized. Approval by the Ethics Committe of our institution was obtained. One hundred and six high-risk corneal penetrating keratoplasty (PKP) recipients were selected. Inclusion criteria were a history of previous ipsilateral corneal graft failure, two or more quadrants with corneal neovascularization or an infectious or inflammatory corneal disease. No exclusion criteria were defined, with death (by other causes) and loss of follow-up being considered as endpoints of the study.
Fifty-three of these patients were treated post-operatively with the standard of care in our institution: oral methylprednisolone tapered in two to three weeks after the graft surgery and long-term topical dexamethasone 0.1 mg/ml, starting at 6 id (control group). In the other half (53 patients) tacrolimus ophthalmic ointment 0.2 mg/g was added twice a day to the standard corticotherapy (trial group). Both groups completed one week of topical ofloxacin post-operatively, as well as 3 days of topical azithromycin after suture removal and complementary treatment with artificial tears (sodium hyaluronate 0.3%). Patients with glaucoma maintained their preoperative topical therapy, including topical beta-blockers, prostaglandin analogues, topical or oral carbonic anhydrase inhibitors or topical brimonidine. The ointment was produced in the hospital pharmacy specifically for ophthalmic use in 10 mg tubes containing 2 mg of tacrolimus monohydrate (supplied by Acofarma®) and 8 mg of purified vaseline, achieving optimal and durable concentration in the ocular surface.
Medical records were selected from our database of corneal transplant surgeries and separated into a trial group (treated with topical tacrolimus), and a control group (corticosteroids). The trial group consists of 53 high-risk patients, 25 male and 28 females, with a median age of 64 (range 53.5 to 77.5), subjected to penetrating keratoplasty between July 2015 and December 2021. The patients from the control group consist of 53 equally high-risk patients, 25 male and 28 females, with a median age of 64 (range 50 to 77), subjected to PK between January 1985 and March 2019, and were selected by date of transplant (from the most recent to the oldest) until the same number as the trial group was reached.
Patient gender, age, transplant technique and indication, number of neovascularized quadrants, intraocular lens status, number of previous grafts and the co-existence of uveitis, glaucoma or blood transfusion history were registered. The existence of anterior synechia and chemical burns were also evaluated. These factors were used to quantify risk through a score proposed by Hicks, 16 in which every factor is awarded a score depending on its level of importance as decided based on probability of graft survival in the first year.
Post-operative data included the duration of follow-up, signs of rejection during follow-up, its response to topical treatment, local and systemic side effects, and finally whether and when transplant failure occurred. Study duration allowed us to reach a median post-operative follow-up of 30 months (12; 43.5) for the tacrolimus group and 24 months (11; 54) for the control group, with no statistically significant difference between groups (p = 0.75).
The primary endpoint was graft failure during follow up. Death or discontinuation of therapy due to adverse effects were secondary endpoints.
Statistical analysis was performed using IBM SPSS 25. For discrete variables, values are presented as number of cases (percentage of total); for quantitative variables, normality was tested using the Shapiro-Wilk test. For normally distributed variables, results are presented as average ± standard error. For non-normally distributed variables, results are presented as median (25th centile; 75th centile). Comparison between discrete variables was performed with the chi-square test, between two normal distributions with the t-student test, whereas between non-normal distributions the Mann-Whitney test was used. Survival analysis was performed using the Kaplan-Meier procedure and the Log-Rank test. Differences between groups were considered statistically significant for p < 0.05.
Results
The trial group consisted of 53 high-risk patients, 25 male and 28 females, with a median age of 64 (range 53.5 to 77.5), subjected to penetrating keratoplasty between July 2015 and December 2021 (20 to the right eye and 33 to the left eye). The control group consisted of 53 patients, 25 male and 28 females, with a median age of 64 (range 50 to 77), subjected to PKP between January 1985 and March 2019 (25 to the right eye and 28 to the left eye). 3 of these patients were subjected to keratoplasty in both eyes. Patient age did not differ significantly between groups (p = 0.59). Sample characteristics are described in Table 1.
Sample demographic characteristics (age, gender and eye) of tacrolimus and control groups.
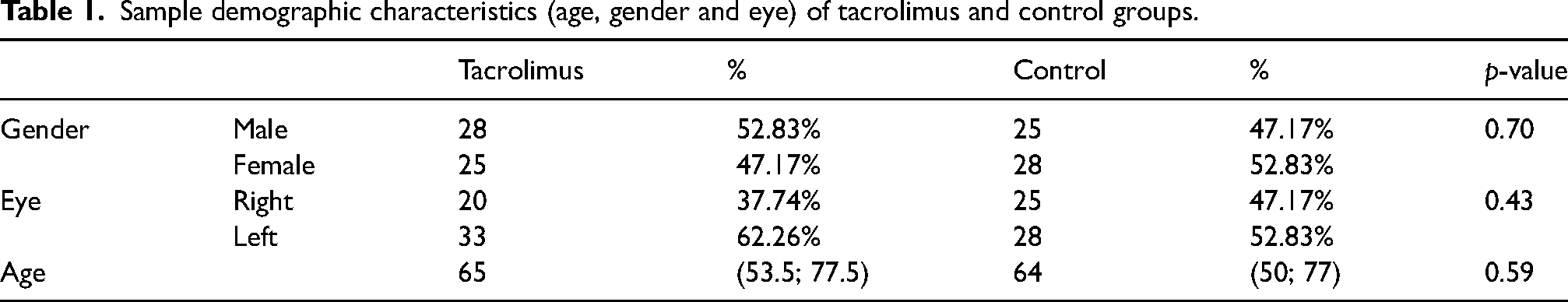
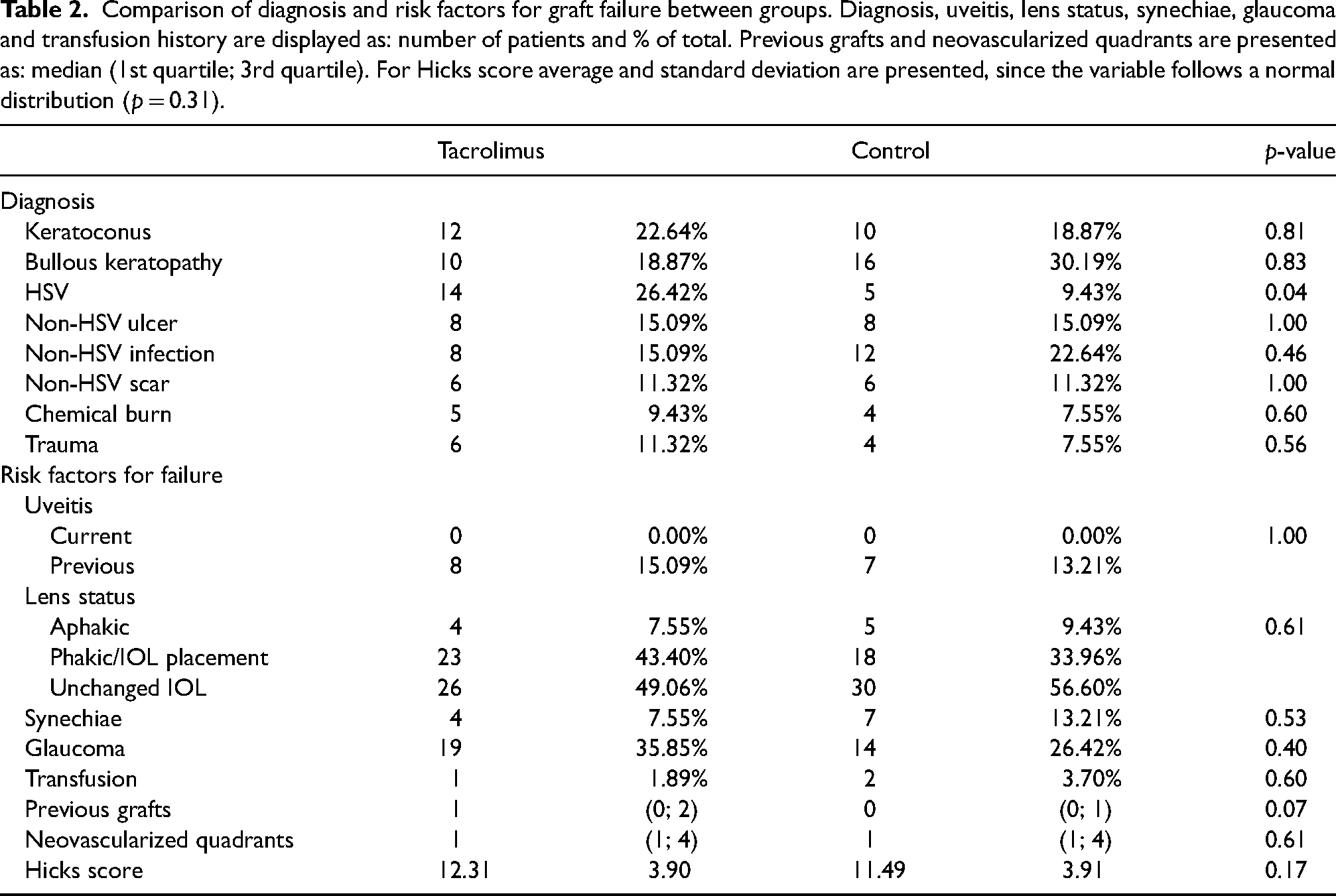
Table 2 compares the baseline diagnosis, concomitant disease and other risk factors for graft failure, as established by the Hicks score, between the trial and control groups, as well as the Hicks risk score. No statistically significant difference between patients in the tacrolimus and the control group were found with regards to these factors, except for a higher prevalence of herpetic (HSV) keratitis in the trial group (14 patients in the trial group and 5 in the control group, p = 0.04). The most common diagnosis in the trial and control groups were bullous keratopathy, keratoconus corneal infections (HSV, bacterial and fungal infections). The most prevalent concomitant risk factors for graft failure were corneal neovascularization, glaucoma, and unchanged intra-ocular lens during procedure. Average Hicks score was 12.31 ± 3.90 for the trial group and 11.49 ± 3.91 for the control group, a non-statistically significant difference (p = 0.17).
Comparison of diagnosis and risk factors for graft failure between groups. Diagnosis, uveitis, lens status, synechiae, glaucoma and transfusion history are displayed as: number of patients and % of total. Previous grafts and neovascularized quadrants are presented as: median (1st quartile; 3rd quartile). For Hicks score average and standard deviation are presented, since the variable follows a normal distribution (p = 0.31).
Median follow-up after the transplant was 30 months (12; 43.5) for the tacrolimus group and 24 months (11; 54) for the control group, with no statistically significant difference between groups (p = 0.75). Graft failure during follow-up, due to rejection, recurrence, or other complications, occurred in 19 (35.85%) of the patients treated with tacrolimus, as opposed to 43 (81.13%) in the control group (p < 0.01). Odds-ratio for failure in the control group was 7.70 (3.17–18.70 95% confidence interval). Median failure time was 10.5 (4; 21) months for the former, and 12 (6; 24) for the latter, a non-statistically significant difference (p = 0.67). These results are shown in Table 3. No statistically significant difference was found regardind failure risk factors between groups.
Comparison of follow up time and graft failure time in months between tacrolimus and control groups.

Survival analysis found a statistically significant difference between the groups, with the Log-rank test yielding a p-value of 0.024, with a higher graft survival rate at all periods in the follow-up for patients treated with topical tacrolimus. Transplant failure rate at 6 months follow-up was 5.70% for the trial group and 15.30% for the control group; at 12 months it reached 18.00% for the trial group and 25.80% for the control group; at 24 months it was 20.70% for the trial group and 45.50% for the control group. Figure 1 provides a complete graphic representation of the survival analysis.
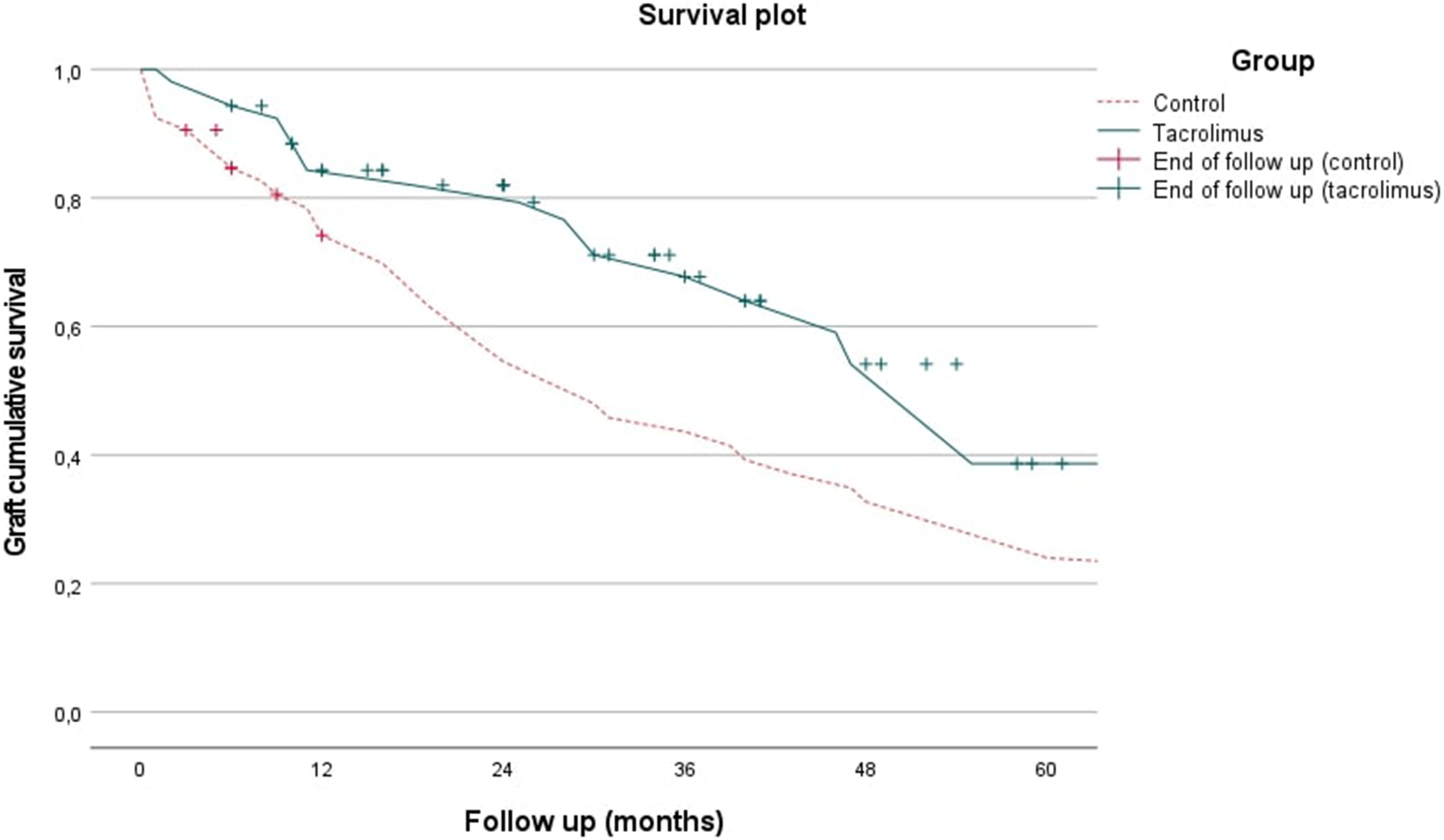
Graft survival plot for trial and control groups.
No adverse drug reactions to topical tacrolimus were reported.
Discussion
High-risk corneal transplant recipients present a challenge in terms of post-operative care, with no current consensual strategy for fighting rejection and improving long-term graft survival. To estimate transplant prognosis and rejection risk, Hicks et al proposed the Risk Score System, 15 which significantly correlates with graft failure. A recent study 17 further indicated that a score from 5 to 8 points translates low risk, a score from 9 to 13 moderate risk, and above 13 points high risk for corneal grafting, and recommended systemic immunosuppression for the latter group. As our results show, nearly all patients included in this study would be considered of moderate or high risk according to this method. A separate study further highlighted the importance of corneal neovascularization, 18 which was present in most of the patients included in this study (70.37% of the study group and 61.54% of the control group). Therefore, our inclusion criteria selected truly high-risk patients, which allow for comparisons with other similar studies.
Such studies have evaluated failure risk in high-risk grafts with several alternative or adjunctive treatment strategies to long-term corticotherapy. A randomized controlled trial studied the efficacy of topical CsA in graft failure prevention, with disappointing results. 7 Other studies evaluated the merits of systemic immunosuppression, with benefits mostly outweighted by the risks. 8
Tacrolimus, with its ability to supress T lymphocyte proliferation and reduce the levels of pro-inflammatory cytokines, has long been hypothesised as a valuable topical immunosuppressant in high-risk corneal grafts, and our study aims to cement its place as an efficacious long-term adjuctive therapy to topical corticotherapy.
Retrospective study design, although sacrificing some study power compared to a double blinded randomized controlled trial, allowed us to provide the best possible care to our patients throughout the duration of the study and better integrate it in normal hospital activity. Moreover, one of the great risks of retrospective designs, which is the heterogeneity of trial and control groups, was not verified in our study. Indeed, Table 2 shows that the groups are similar in nearly every aspect, and the most important risk factors for graft rejection are consistently present in both groups without a statistically significant difference between them. In fact, the only statistically significant difference was an increased prevalence of history of herpetic keratitis in the tacrolimus group, which would confer it a higher risk of graft failure compared to the control group, further strengthening our positive results.
This homogeneity between groups, along with larger sample sizes, broader inclusion criteria and longer median follow-up time, represents an important improvement on previous studies conducted on the efficacy of topical tacrolimus in high-risk corneal grafts.12–15
Different follow-up times in and between the groups was one of the great concerns with the study, since a few patients in the trial and control groups were transplanted as late as December 2021. Although these were not found to be significantly different between groups (Table 3), this may have impacted the late failure numbers. This problem was circumvented by using survival analysis through the Log-rank test, which is considered more adequate than the Breslow test for comparing subjects with different follow-up times. However, statistical power is lost and the survival plot progressively loses reliability as the follow up time increases.
Another limitation is that, due to logistical reasons, many patients did not effectively start therapy with topical tacrolimus for up to two weeks after the transplant, which might have led to an increase in early post-operative graft failure in the trial group. Moreover, other small treatment differences between patients, such as different administration of artificial tears and glaucoma topical medication, may have contributed to some heterogeneity in ocular surface homeostasis, which may have impacted the results in subtle but unpredictable ways.
In conclusion, this study successfully showed that the addition of tacrolimus ophthalmic ointment 0.2 mg/g twice a day to the standard immunosuppressive regimen (topical dexamethasone 0.1 mg/ml 6 id) yields significantly better short and long-term corneal graft survival rates than topical corticotherapy alone for patients with high risk of graft failure. In this context, it is one of the first studies to compare tacrolimus plus corticosteroid versus corticotherapy alone, and it is also one of the largest studies available regarding the use of topical tacrolimus in corneal grafting. A previous study by Zhai et al. 12 pointed to superiority in the efficacy and safety of topical tacrolimus alone versus topical cyclosporine alone in high-risk cases and is of great interest for corticoid intolerant patients, but lacks the general clinical reproducibility for patients in which all drug classes are an option.
This study points to topical tacrolimus with associated topical corticosteroids being an extremely promising therapy in high-risk corneal graft receptors, showcasing excellent graft survival rates with no adverse reactions. However, longer-term studies are warranted to assess impact on late graft failure and visual acuity for this regimen to become the new standard of care for high-risk corneal grafting. Larger study designs would also be useful to document rare adverse reactions.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
