Abstract
Introduction
Early initiation of anti-inflammatory therapies is recommended for dry eye disease (DED) to break the vicious cycle of pathophysiology. However, there is limited guidance on how to implement topical ciclosporin (CsA) and corticosteroid treatment into clinical practice. This expert-led consensus provides practical guidance on the management of DED, including when and how to use topical CsA.
Methods
A steering committee (SC) of seven European DED experts developed a questionnaire to gain information on the unmet needs and management of DED in clinical practice. Consensus statements on four key areas (disease severity and progression; patient management; efficacy, safety and tolerability of CsA; and patient education) were generated based on the responses. The SC and an expanded expert panel of 22 members used a nine-point scale (1 = strongly disagree; 9 = strongly agree) to rate statements; a consensus was reached if ≥75% of experts scored a statement ≥7.
Results
A stepwise approach to DED management is required in patients presenting with moderate corneal staining. Early topical CsA initiation, alone or with corticosteroids, should be considered in patients with clinical risk factors for severe DED. Patient education is required before and during treatment to manage expectations regarding efficacy and tolerability in order to optimise adherence. Follow-up visits are required, ideally at Month 1 and every 3 months thereafter. Topical CsA may be continued indefinitely, especially when surgery is required.
Conclusion
This consensus fills some of the knowledge gaps in previous recommendations regarding the use of topical corticosteroids and CsA in patients with DED.
Keywords
Introduction
Epidemiology, burden and pathophysiology of DED
Dry eye disease (DED) is a relatively common heterogeneous disease of the ocular surface affecting approximately 11–30% of individuals in Europe.1–5 The disease has been shown to have a substantial impact on vision, quality of life (QoL) and work productivity, and carries a physical and psychological burden.6–9
DED is a multifactorial disease with several pathological processes leading to ocular surface damage. Numerous causes and risk factors of DED exist and lead to loss of tear film homeostasis and subsequent activation of biological cascades detrimental to the ocular surface. 10 Tear hyperosmolarity, considered to be the primary event in DED, is either caused by increased evaporation of the tear film (evaporative dry eye) or reduced tear production (hyposecretory dry eye).11–15 This hyperosmolarity can directly damage the ocular surface or cause indirect damage by triggering an inflammatory reaction.13,16–20 Ocular surface inflammation may also be of immune origin, with numerous studies showing the activation of T-cells in human specimens or preclinical models, or neurogenic, involving a sustained stimulation of the corneal nerves.16,21 These processes lead to a self-reinforcing inflammatory cycle (the ‘vicious circle’), explaining how ocular surface damage is perpetuated in DED (Figure 1).10,13,16,22,23
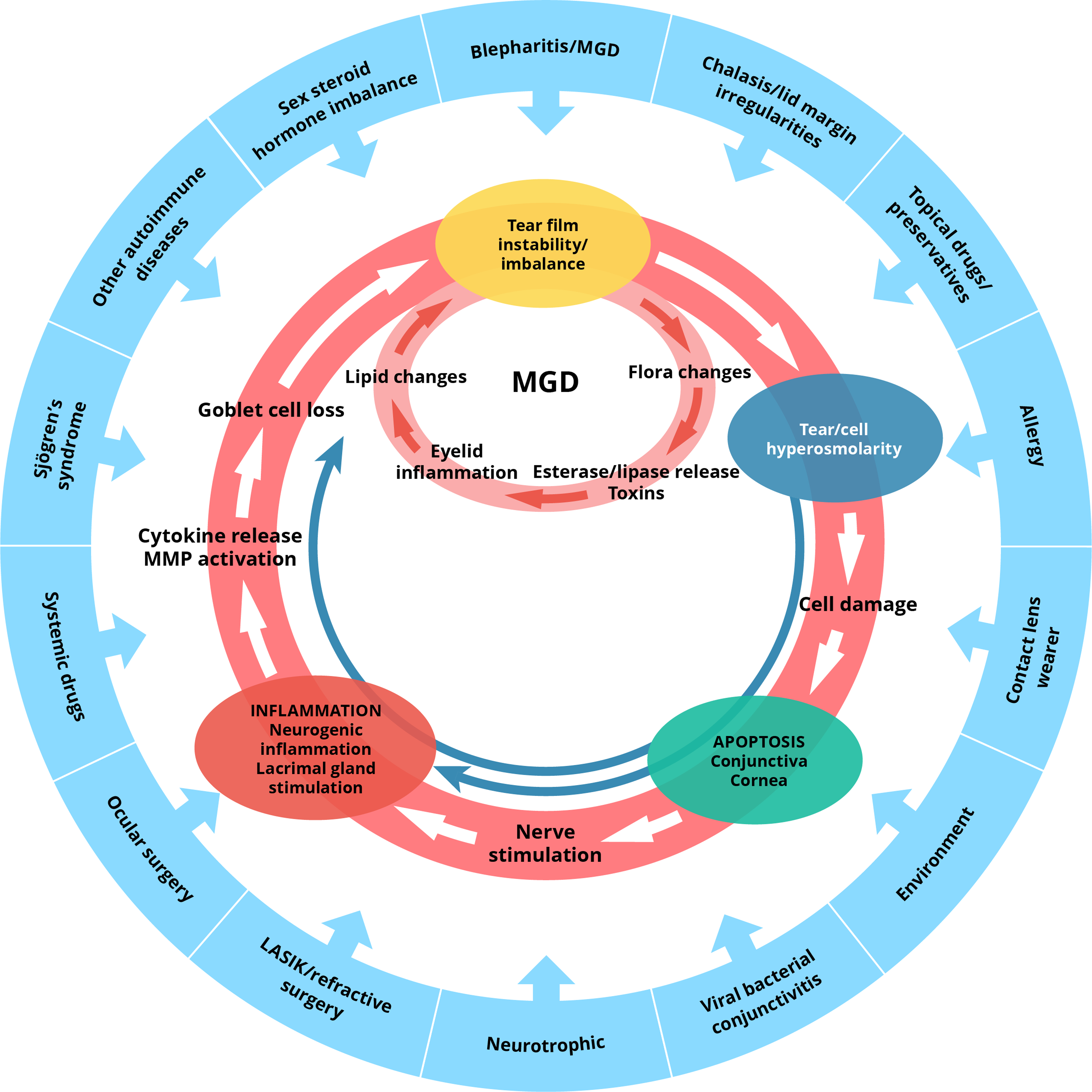
Vicious circle of DED pathophysiology. LASIK: laser-assisted in situ keratomileusis; MGD: meibomian gland dysfunction; MMP: matrix metalloproteinase. Image adapted from Baudouin C, et al. 2013. 10
Anti-inflammatory management of DED
The goal of DED management is to restore natural homeostasis at the ocular surface, which can be achieved by breaking the vicious circle of pathophysiology.13,24 Early initiation of anti-inflammatory therapy with topical corticosteroids and/or ciclosporin A (CsA) is recommended by the 2017 Tear Film & Ocular Surface Society Dry Eye Workshop (TFOS DEWS) II report. This is particularly important for patients whose tear substitutes are no longer sufficient and for whom changes in the environment do not result in an improvement. 24 Furthermore, tear substitutes are only a replacement or supplement to the natural tear film and often do not address the underlying inflammatory pathophysiology. 24 There is some evidence that tear substitutes can have an effect on ocular surface inflammation by decreasing friction or alleviating tear hyperosmolarity through non-specific (hypotonic eye drops) or specific (osmoprotectant) properties.10,25 Additional evidence suggested that emollient-based artificial tears, or those containing carmelose sodium 0.5% or a combination of hyaluronic acid and trehalose can reduce inflammation.26–28 Topical corticosteroids have consistently been shown to break the vicious cycle of inflammation in DED, providing clinical efficacy with short-term use.24,25 Nevertheless, differences regarding the incidence of side effects and evidence of efficacy in DED have been reported for the various available topical corticosteroids due to their potency and propensity to penetrate into the anterior chamber.29,30 In general, topical corticosteroids can be used only for a limited period due to potential adverse events (AEs), including ocular hypertension, cataracts and opportunistic infections.24,30 However, fluorometholone, loteprednol and hydrocortisone could be considered as they have a lower likelihood of increasing intraocular pressure (IOP) and inducing cataract formation.24,30 Numerous studies and meta-analyses have shown that CsA is an effective therapy for the management of DED, with a tolerable safety profile. In contrast to corticosteroids, CsA can be safely administered for extended periods of time.24,25 Currently, there is no clear guidance on several aspects of CsA use in the management of DED, such as when to start CsA therapy, measure treatment effects or taper treatment. There is also a lack of guidance on concomitant corticosteroid use with CsA and optimising adherence to CsA.
This consensus document was developed to address the unmet needs outlined above and provide clinical recommendations that could improve patient outcomes in DED. It covers practical guidance on the management of inflammation, how and when to measure treatment effects, and the use of CsA eye drops with or without corticosteroids to control disease. It also highlights the need for adequate patient education to optimise adherence to CsA treatment.
Methods
A steering committee (SC) of seven DED experts oversaw the consensus process. A panel of 22 additional ophthalmologists (from Belarus, Denmark, France, Germany, Hungary, Italy, Kazakhstan, Norway, Romania, Russia, Slovakia, Spain and the UK; Appendix 1) was selected to support the consensus process (Supplementary Table 1). The goal of the SC and panel was to reach a consensus agreement on the role of inflammation in DED and its practical management, particularly on the use of CsA eye drops to control signs, symptoms and disease progression.
The SC was involved in the development of a questionnaire which gathered information on the clinical practices of ophthalmology experts in the management of DED (Supplementary Table 2). The questionnaire was developed remotely via email and was based on an initial literature review to identify challenges and evidence gaps in the management of inflammation in DED. This questionnaire was subsequently distributed online to all panel and SC members using a web-based platform.
Following an analysis of the questionnaire results, the SC developed 18 relevant recommendation statements for consensus voting, covering four major areas: disease severity and progression; patient management, including treatment initiation, tapering and discontinuation; efficacy, safety and tolerability of CsA; and patient education.
Statements were voted on remotely by all 29 experts via a secure online platform and scored on a nine-point scale (1 = strongly disagree and 9 = strongly agree). A consensus was considered to be reached when ≥75% of respondents scored a particular statement between 7 and 9 (inclusive). Only one round of voting was needed to obtain a consensus on all statements.
The entire questionnaire and consensus voting process took place over 7 months (from 1 October 2020 to 10 May 2021).
Results
Disease severity and progression
A consensus was achieved for all the statements relating to disease severity and progression, with 100% agreement on the recommended criteria for use when measuring the severity of DED and for differentiating between severe DED keratitis and other forms of keratitis (Statements 1 and 2; Table 1).
Statements on disease severity and progression.

DED: dry eye disease; TBUT: tear film break-up time.
*A consensus was reached when ≥75% of respondents scored a particular statement between 7 and 9 (inclusive).
The experts came to a unanimous decision that DED severity should be measured by symptom severity, corneal and conjunctival staining, low tear production, short tear film break-up time (TBUT) and meibomian gland pathology. Furthermore, it was agreed that the degree of corneal and conjunctival staining and low tear production can help to differentiate between severe DED keratitis and other forms of keratitis (Statements 1–3; Table 1).
Evidence and interpretation
According to the literature, the factors most frequently used to evaluate the severity of DED include symptom severity (e.g., Ocular Surface Disease Index [OSDI]), corneal and conjunctival staining, tear production (Schirmer's test), TBUT, meibomian gland pathology, visual disturbances and tear hyperosmolarity.11,31–40 Of these, tear film osmolarity and corneal and conjunctival staining tend to have the greatest correlation with severe DED.33–37,39
Some of the signs of DED can mimic those of other forms of keratitis (e.g., interstitial, neurotrophic) and keratopathies (e.g., bullous); the presence of vascularisation, anterior chamber cells and flare, and stromal oedema generally set them apart from primary DED. 39 In clinical trials of CsA 0.1% cationic emulsion for DED, the key criteria used to identify eligible patients with severe DED keratitis were based on corneal fluorescein staining (CFS), the presence of ocular symptoms and the Schirmer's test.41–43
Risk factors for the development of DED
A consensus was reached on the important risk factors for disease: small fibre neuropathy, primary Sjögren's syndrome, rheumatoid polyarthritis, systemic lupus erythematosus and other factors (Statement 4; Table 1).
Evidence and interpretation
Multiple systemic diseases across many organ systems have been shown to be underlying risk factors for the initial development of DED; the strongest association is with Sjögren's syndrome.33,44 Other disorders with a high risk include rheumatological and other immune disorders, chronic pain syndromes and chronic graft-versus-host disease.33,44
Certain systemic inflammatory biomarkers (neutrophil-to-lymphocyte ratio, platelet-to-lymphocyte ratio and systemic immune-inflammation index) are elevated in patients with DED, indicating that some forms of DED might be associated with systemic inflammation.45,46 This may in part explain the prevalence of disease in these groups of patients with underlying inflammatory disorders.
Patient management, including treatment initiation, tapering and discontinuation
Stepwise approach to the management of DED
The experts agreed that, irrespective of systemic involvement, a stepwise approach to the management of patients with moderate corneal staining is recommended, using environmental control, artificial tears (ATs), lid hygiene, topical corticosteroids and/or topical CsA (Statement 5; Table 2).
Statements on patient management, including treatment initiation, tapering and discontinuation.

AT: artificial tear; CsA: ciclosporin A; DED: dry eye disease.
*A consensus was reached when ≥75% of respondents scored a particular statement between 7 and 9 (inclusive).
Evidence and interpretation
A staged management algorithm for DED has been developed as part of the TFOS DEWS II report, which recommends a stepwise approach to implementing management strategies and therapeutic options. 24 The algorithm considers disease aetiology and severity, progressing from generalised treatments for mixed aetiology to advanced and specific treatments aimed at treating the underlying pathophysiology. For patients who do not respond to a given level of management or for those with severe DED, the next level of management is recommended. In some cases, the initial therapy may be continued in combination with any additional therapies. Recommended management approaches for early-stage disease are generally conventional, low-risk and commonly available therapies, such as over-the-counter lubricants. Management progresses to the use of advanced therapies for severe forms of DED. The algorithm covers treatments for tear insufficiency and meibomian gland dysfunction, anti-inflammatory medications (including corticosteroids and topical CsA), surgical approaches, dietary modifications, environmental considerations and complementary therapies.
Treatment initiation with topical CsA
A consensus was achieved for all four statements regarding CsA treatment initiation (Statements 6–9; Table 2). The experts agreed that a decision to use CsA eye drops for the management of inflammation in DED should be based on the severity of corneal and conjunctival staining, presence/absence of immune-related systemic disease, rapid response to corticosteroid therapy and inadequate control while receiving standard-of-care treatment. However, the panel also thought that early initiation of topical CsA treatment should be considered to manage inflammation in patients presenting with risk factors for developing severe DED. In DED, the panel considered corneal staining, conjunctival staining, conjunctival hyperaemia and reduced aqueous tear production as important markers of ocular inflammation (Statement 8; Table 2). However, the absence of severe corneal staining should not prevent the initiation of topical CsA eye drops if other risk factors are present (Statement 9; Table 2).
Evidence and interpretation
Just like topical corticosteroids, topical CsA is indicated for the treatment of DED as second-line therapy when tear substitutes, lid hygiene and environmental control have not been effective.24,30,47,48 The importance of addressing inflammation early in the disease course has been shown in other inflammatory diseases. It is thought that early treatment with CsA in ulcerative colitis may improve outcomes and reduce morbidity. 49 CsA has been shown to have a reasonably rapid onset of action in diseases with an inflammatory origin, such as psoriasis, atopic dermatitis and Behçet's syndrome, with a significant improvement observed within a few weeks.50–52 It is known that inflammatory events occurring anywhere in the body can lead to tissue damage due to tissue and vascular changes, disturbance of function, release and accumulation of reactive oxygen species and translocation of plasma to adjacent tissues.53,54
Timing of CsA treatment assessment
The experts agreed that treatment response in individual patients should be evaluated after 1 month and then every 3 months to confirm efficacy (Statement 10; Table 2). The panel recommended that ATs should be used in combination with CsA (Statement 11; Table 2).
Evidence and interpretation
Initial responses to CsA treatment may be evident within the first month of therapy, demonstrated by improvements in some signs and symptoms of DED, but are typically seen after 6–8 weeks.41–43,55–62 These responses are usually maintained or further improved after 3–6 months of treatment.41–43,55,56,59–64
Administration of ATs 15–20 min before and/or after administration of CsA leads to significant improvements in the symptoms of burning, stinging and irritation previously experienced with CsA instillation. 65
Co-treatment with topical CsA and corticosteroids
There was more than 95% agreement on the statement relating to the frequency of examination of patients receiving combined topical CsA and corticosteroid therapy, and almost 90% agreement on the statement relating to bridging CsA treatment with corticosteroids (Statements 12 and 13; Table 2).
Evidence and interpretation
Initiating short-term treatment with corticosteroids at the same time or shortly before starting topical CsA therapy (bridging) leads to good outcomes.30,66–68 Patients have been shown to experience a rapid improvement in the signs and symptoms of DED as well as improved local tolerance to CsA. Corticosteroids were administered for 4–8 weeks and then gradually tapered. These clinical trial data support the experts’ opinion on the benefits of bridging CsA treatment with corticosteroids, whereby the relatively slow onset of the anti-inflammatory effect of CsA is offset by the relatively fast onset of the anti-inflammatory effect of a topical corticosteroid. The experts advocate that topical CsA should be initiated at the same time as corticosteroid treatment, or up to a week afterwards, and that the corticosteroid dose should be tapered off based on changes in the signs and symptoms of inflammation (Figure 2).
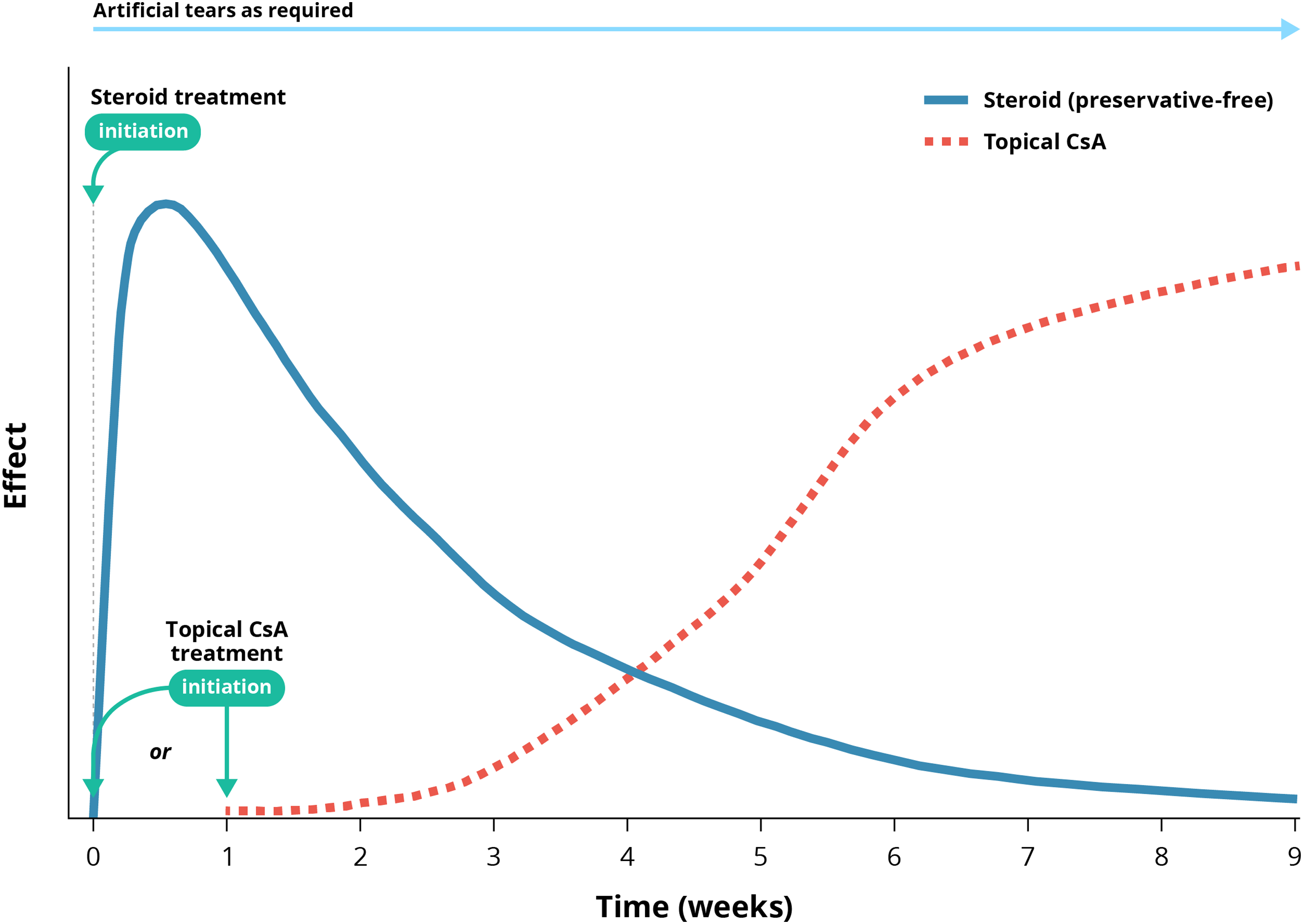
The main reason for limiting the use of topical corticosteroids is that they are known to be associated with AEs with long-term use, such as increased intraocular pressure (IOP) and cataract formation; this can even be encountered with short-term use.24,25,30,68–71 It is therefore important to monitor patients treated with corticosteroids. In clinical trials of combination treatment with corticosteroids and topical CsA, patients underwent ophthalmic examinations and completed the OSDI evaluation every 2–4 weeks.66–68 Other studies have recommended closely monitoring IOP during topical corticosteroid therapy after 1–2 weeks of treatment.30,71 The experts acknowledged that there is no fixed interval for examining patients receiving combination therapy, and instead, examination frequency should be based on other factors, such as the corticosteroid type/concentration, frequency of instillation and comorbidities.
Less potent/less penetrative corticosteroids may correspond with a lower risk of increased IOP and cataracts than high potency formulations 24,30,68 and could be considered as alternative treatment options to reduce the risk of AEs.
CsA treatment tapering and discontinuation
Almost 90% of experts agreed with the statement regarding the duration of CsA therapy in the event of sustained improvement, and discontinuation in the event of poor tolerance, a lack of efficacy or patient choice (Statement 14; Table 2). The panel agreed that CsA treatment does not need to be stopped if a patient requires surgery (Statement 15; Table 2).
Evidence and interpretation
As a chronic disease, the management of DED is based on long-term treatments, ultimately aiming to restore homeostasis of the ocular surface.16,24,25,70 Long-term studies have shown that chronic use of topical CsA can provide ongoing benefits to patients with DED.24,30,72–75 Patients may even experience complete resolution of signs and symptoms during long-term treatment and may opt to reduce the dosage or discontinue treatment altogether. Although there is a risk of relapse in certain patients, the majority (61%) did not relapse after experiencing DED improvement with CsA during a 24-month follow-up analysis of the SANSIKA study. 72 Therefore, the experts advise that CsA treatment can be continued indefinitely if it remains well tolerated and effective; tapering and stopping treatment is also an option if a sufficient and sustained improvement is achieved.
Long-term studies of up to 24 months have demonstrated the long-term tolerability of CsA, with no specific long-term safety concerns identified.63,72,73 Furthermore, the incidence of systemic treatment-related AEs was very low in clinical trials, and there were no reports of serious systemic effects.63,72,73,76–78 Nevertheless, the panel recommends that CsA should be discontinued in the event of poor tolerance or lack of efficacy.
DED is a common comorbidity in patients with glaucoma and cataracts, and ophthalmic surgery for these conditions can potentially worsen DED symptoms. At the same time, ocular surface disease can have a negative impact on surgical outcomes; therefore, optimisation of the ocular surface prior to surgery is important to ensure better outcomes.70,79–81 These data support the panel consensus on continuing CsA treatment if a patient with DED is undergoing surgery.
Efficacy, safety and tolerability of topical CsA
Efficacy of topical CsA
In total, 86% of experts agreed with the statement that CsA efficacy can be assessed from as early as 1–3 months after the initiation of treatment (Statement 16; Table 3).
Statements on efficacy, safety and tolerability of CsA.

AT: artificial tear; BUT: break-up time; CsA: ciclosporin A.
*A consensus was reached when ≥75% of respondents scored a particular statement between 7 and 9 (inclusive).
Evidence and interpretation
The immune modulator CsA has gained prominence as a steroid-sparing topical therapy that leads to positive outcomes for patients with DED. Data have showed that an initial response to CsA may be evident within the first month of treatment, demonstrated by measurable improvements in DED signs and symptoms.43,55–62,82 Furthermore, in the real-world setting, a statistically significant improvement (P < 0.0001) in the severity of CFS was reported at Week 4 in patients treated with topical CsA, which was maintained until Month 12. 83
It is recommended that the outcome measures used in clinical trials for DED should be selected carefully depending on the treatment being assessed, because the sensitivity of markers or signs differ considerably depending on the tested therapy and its mechanism of action. 35 Assessing parameters and biomarkers indicative of inflammation, such as conjunctival and corneal stainability, tear film osmolarity, conjunctival hyperaemia and matrix metalloproteinase-9 levels in the tear film, provide evidence of the anti-inflammatory effect of CsA. 30 The opinion of the experts was that the main indicators of CsA efficacy are improvements in signs and symptoms, including a reduction in blurred vision, reduced ocular surface staining and improved TBUT. Table 4 shows the symptom and sign parameters that have been used to assess the efficacy of CsA in DED in clinical trials.
Sign and symptom parameters used to measure the efficacy of CsA in DED in clinical trials.
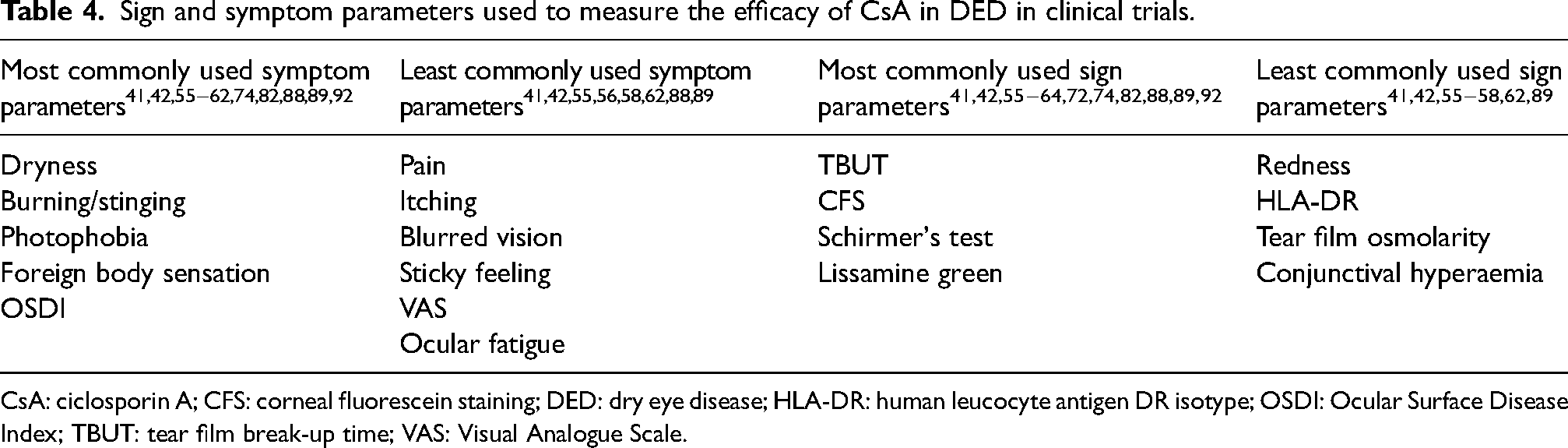
CsA: ciclosporin A; CFS: corneal fluorescein staining; DED: dry eye disease; HLA-DR: human leucocyte antigen DR isotype; OSDI: Ocular Surface Disease Index; TBUT: tear film break-up time; VAS: Visual Analogue Scale.
Safety and tolerability of topical CsA
There was almost complete agreement (96.6%) on the statement relating to the tolerability of topical CsA that only a minority of patients may need to discontinue treatment due to an AE (Statement 17; Table 3).
Evidence and interpretation
In clinical trials and in the real-world setting, most treatment-emergent adverse events (TEAEs) reported with topical CsA were ocular in nature, with the most common being instillation site pain and irritation (Supplementary Table 3). Furthermore, in several clinical trials, most TEAEs were mild to moderate in severity42,56,57,59–62,64,67,72,84–87 and few serious AEs were reported.41–43,55,59,60,62,63,72,76–78,88,89 This favourable tolerability profile meant that only a small proportion of patients needed to discontinue treatment with topical CsA due to AEs.41,43,56,59,61,62,72,85,88–90
For those patients experiencing instillation site pain and irritation upon topical CsA administration, the experts recommend that additional steps can be taken to alleviate the discomfort, such as the concomitant use of ATs and topical corticosteroids. A recent German consensus paper stated that it may be helpful to give preservative-free ATs approximately 20 min before application and a preservative-free gel 20 min after application of topical CsA, although there are no data on whether the effect of CsA is impaired by this. 30 The use of non-penetrating, preservative-free steroids before the start of topical CsA treatment or for 4 weeks in combination with CsA can improve local tolerability. 91
The panel noted the importance of informing the patient about the potential for experiencing discomfort with topical CsA treatment, as this can lead to premature treatment discontinuation. Clear communication between clinicians and patients is important to help manage treatment expectations. Physicians can recommend that patients keep the drug refrigerated so that the drops are cool upon instillation, as some patients have found this to be helpful. 30 However, the product information for topical CsA 0.1% cationic emulsion advises that eye drops must be protected from freezing and stored in the dark (in the original container); this should be made clear to patients. 48
Patient education
There was almost complete agreement (>95%) that patient education before and during topical CsA use is critical for optimising adherence to treatment (Statement 18; Table 5).
Statement on patient education.

AT: artificial tear; CsA: ciclosporin A.
*A consensus was reached when ≥75% of respondents scored a particular statement between 7 and 9 (inclusive).
Evidence and interpretation
There is limited information in the literature regarding advice that physicians should give patients who are prescribed topical CsA drops for DED. It is important to educate patients on the disease signs and symptoms, prognosis and management, time to onset of therapy effects, proper use of the medication and the treatment regimen, as well as common ocular side effects, their transient nature and how to manage them.24,30,65,93–98 Recommendations have recently been reported by a German consensus group, which are consistent with the patient education statement described herein. 30
Although studies have shown that the majority of patients and physicians (60–80%) are satisfied overall with topical CsA in DED,95,96,99–102 there is some dissatisfaction with the time to onset of effect. This may have important implications for patient adherence and treatment persistence, as the efficacy of CsA is dependent on compliance with the prescribed treatment regimen.65,101,102 Non-compliance can affect QoL, quality of vision and productivity, and lead to further dissatisfaction due to continued symptoms. 65 It has been shown that patient education may overcome adherence issues with topical CsA, particularly in relation to the need for long-term treatment of DED. 94
Compliance rates and adherence to eye drop instillation instructions were relatively high in clinical trials (rates of 76–98%).41,56,60,88 Common reasons cited by the proportion of patients who do not adhere to or persist with treatment, or who discontinue CsA, include treatment intolerance and the delayed onset of effect (Table 6).65,92,94,95,100–105 This shows the importance of informing patients that the effects of CsA are not immediately apparent and that a response may not be achieved for several weeks. Although DED is chronic in nature, with exacerbation of symptoms associated with ocular surface inflammation (DED flares), patients may suspend treatment when initial symptom relief is achieved as they may believe their condition to be well controlled.106–108
Reasons for non-compliance with CsA.

CsA: ciclosporin A; DED: dry eye disease.
In addition, patients should be advised that corticosteroids may be needed at the beginning of treatment as a bridging therapy and during possible flare-ups, and that they should continue using ATs to reduce ocular discomfort.
Before treatment with topical CsA is initiated, patients should be informed about potential AEs, such as pain and burning/stinging upon instillation. Physicians should encourage patients to persist with their treatment because these symptoms are usually transient and improve as the ocular surface improves.30,41,59,72,88,96,98 It is also important to reassure patients that topical CsA has almost no systemic effect and therefore the risk of systemic AEs is very minimal.
Discussion
This manuscript provides a European consensus (spanning 15 countries) on the management of inflammation in DED, with a particular focus on the use of topical CsA and corticosteroids. Although steroids have been the mainstay of treatment for most inflammatory conditions, including DED, non-glucocorticoid immune modulators, in particular CsA, have gained prominence as steroid-sparing topical therapies that lead to positive outcomes for patients with DED. Optimal use of topical CsA in the management of DED remains an issue of clinical relevance. There is little information to guide physicians on some aspects of treatment initiation, monitoring and stopping, as well as patient education.
The current questionnaire was conducted with the aim of filling some of the existing gaps in treatment recommendations based on real-world experience. The experts broadly agreed on all 18 statements regarding DED and topical CsA treatment, based on the criteria for reaching a consensus outlined above. Notably, there were two statements for which 100% agreement was reached, relating to the criteria for measuring the severity of DED, and the signs and symptoms used to differentiate severe DED keratitis from other types of keratitis. Potential reasons for the unanimous response are the wide range of literature and standards available to support these approaches and the similar level of knowledge and experience among the panel members. In addition, the criteria were presented clearly and in a way that was easy to apply in the clinic. It was felt that measuring the severity of DED is now well defined, although distinguishing between mild and moderate disease is less well established.
The statement with the lowest level of agreement among experts (79.3%) related to the early initiation of CsA in patients at risk of developing severe DED. Differing attitudes could reflect variable drug-funding policies and standards of care in different countries and the historical ease of access to CsA. In addition, there is still a lack of information about disease progression in patients treated with CsA. Furthermore, topical CsA is currently approved for severe keratitis only, and therefore its use in early stages of DED under an off-label indication may be a deterrent for some.
Key recommendations/practical aspects for managing DED
A stepwise approach should be used for managing DED, with the rate of progression of treatments based on disease severity. The important indicative markers of inflammation include corneal staining, conjunctival hyperaemia and reduced tear production. CsA should be initiated when there is substantial corneal and/or conjunctival staining; however, the absence of severe corneal staining should not prevent the initiation of CsA eye drops for managing inflammation in DED if other risk factors are present. For patients presenting with clinical risk factors for the development of severe DED, the early initiation of CsA eye drops should be considered. Treatment efficacy can be assessed from as early as 1–3 months after the initiation of treatment. However, even in patients demonstrating an early response, treatment should be continued and early discontinuation should be discouraged. Patients should be advised about the potential irritation associated with CsA to encourage compliance with treatment. The short-term discomfort is outweighed by the benefits, and many patients are highly satisfied with CsA treatment. Patient education is critical for optimising patient adherence to CsA. ATs should be used in conjunction with topical CsA, applied regularly throughout the day and 15–20 min before and/or after application of CsA.
Limitations of the study
As with all qualitative research, this study has limitations. The SC and panel are all based at European centres and therefore their opinions may not reflect those of individuals based in other regions, where different healthcare settings could influence perspectives. Given the anonymous nature of the questionnaire, there was not scope to obtain demographic details to provide further granularity on the results obtained. Finally, although the questionnaire format used has similarities to the Delphi approach, the achievement of a consensus on the majority of statements in the first round of voting rendered subsequent amends to the statements and revoting on revisions, required per the Delphi approach, unnecessary.
Data gaps and future research
The development of the consensus statements also highlighted areas that may benefit from further study and clarification, including:
The need to educate healthcare professionals on the importance of long-term management with anti-inflammatory agents, highlighting the impact of a lack of appropriate treatments on overall patient QoL The need to educate patients on the chronic nature of DED and the importance of long-term management The need to clearly define the patients who would most benefit from treatment with topical CsA The relevance of sharing experiences in clinical practice with regard to co-treatment/bridging topical CsA and corticosteroids
Although further research is needed in some areas, this consensus procedure has provided some key recommendations for clinicians that may be useful when prescribing topical CsA eye drops for the management of patients diagnosed with DED.
Supplemental Material
sj-docx-1-ejo-10.1177_11206721221141481 - Supplemental material for Management of inflammation in dry eye disease: Recommendations from a European panel of experts
Supplemental material, sj-docx-1-ejo-10.1177_11206721221141481 for Management of inflammation in dry eye disease: Recommendations from a European panel of experts by Elisabeth M Messmer, Sajjad Ahmad, José M Benitez del Castillo, Ewa Mrukwa-Kominek, Maurizio Rolando, Oksana Vitovska and Christophe Baudouin on behalf of a panel of European dry eye disease experts in European Journal of Ophthalmology
Footnotes
Acknowledgements
The authors acknowledge the contributions of Pasquale Aragona, Stefano Barabino, Gulnara Begimbaeva, Camelia Bogdănici, Stefano Bonini, Margarita Calonge, Elena Drozdova, David Galarreta, Gerd Geerling, Ulrike Hampel, Steffen Heegaard, Christina Jacobi, Sai Kolli, Kamila Kopálová, Marc Labetoulle, Nataliya Maychuk, Pierre-Jean Pisella, Sten Ræder, Maite Sainz De la Maza, Halina Sitnik, Edit Tóth-Molnár and Edoardo Villani for the responses given to the clinical practice questionnaire and consensus statements voting.
Author contributions
Conceptualization: EM and CB; writing—review and editing: all authors; all authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
Funding
The author(s) received financial support from Santen, in the form of medical writing support from OPEN Health.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
