Abstract
Purpose
To determine differences in inflammatory markers expressed in diabetic macular edema (DME) patients with and without obstructive sleep apnea (OSA).
Methods
This was a prospective, cross-sectional study. Patients with treatment naive DME were enrolled in the study. They were stratified into 2 groups based on Apnea Hypopnea Index (AHI) from overnight polysomnography: OSA + (AHI ≥ 15) and OSA – (AHI<15). Multiplex immunoassay was performed for aqueous and serum cytokines including VEGF, placental growth factor (PGF), ICAM, IL2, IL3, IL6, IL8, IL10, IL17, vascular cell adhesion molecule-1 (VCAM1), monocyte attractant protein-1 (MCP1), epidermal growth factor (EGF) and platelet derived growth factor (PDGF). Statistical significance was defined as p < 0.004 using Bonferroni correction.
Results
32 DME positive patients were enrolled in the study; of which 17 patients were OSA + and 15 OSA-. The OSA + cohort had significantly higher levels of serum EGF (p = 0.003), and trended towards higher levels of most serum cytokines including ICAM and IL6. OSA- cohort had significantly higher levels of aqueous IL17 compared to the OSA + cohort (2.97 ± 1.7 vs. 1.4 ± 0.46 pg/mL, p = 0.004). There were no significant differences in other aqueous cytokines.
Conclusions
OSA + group trended towards higher levels of most serum inflammatory markers, suggesting a greater pro-inflammatory state. However, they did not have significantly greater level of aqueous cytokines.
Introduction
Obstructive sleep apnea (OSA) is characterized by intermittent nocturnal hypoxemia, arousal, fragmented sleep and daytime sleepiness. It has been shown to increase the risk of cardiac and vascular disease through multiple mechanisms including sympathetic hyperactivity, metabolic dysregulation, and activation of oxidative stress and inflammatory pathways. 1 Studies have reported OSA as a risk factor for macro-angiopathies including hypertension, coronary artery disease and stroke, as well as micro-angiopathies such as renal and retinal disease. 1
Diabetic retinopathy is a leading cause of blindness in the working age group, affecting 93 million people worldwide. Diabetic macular edema (DME) is a sight threatening complication and the most common cause of visual loss in patients with diabetes. The pathogenesis involves disruption of blood-retinal barrier, leading to accumulation of fluid within the intraretinal layers of macula. 2 OSA is frequently associated with diabetes with prevalence ranging from 23 to 86%. 3 However, the relationship between OSA and DME is not well defined. The retina is particularly susceptible to hypoxia, being one of the most metabolically active tissues. 4 It has been shown in animal studies that exposure to greater level of intermittent hypoxia is associated with increased level of inflammatory cytokines. 5 Multiple inflammatory mediators have been found to be elevated in DME, including VEGF, VCAM-1 and IL-6.6,7 The chronic intermittent hypoxia in OSA may worsen retinopathy by further activating inflammatory and oxidative stress pathways.
Previous studies assessing OSA and diabetic retinal disease have been inconsistent,8–14 with variable findings.9,12,13 One of the challenges in comparing these studies is the variation in methods, and high degree of heterogeneity in the devices used to record sleep measurements. The current gold standard for diagnosis of OSA is overnight polysomnography, however, many of the previous studies used portable home devices/questionnaires and differed in their diagnostic criteria for OSA.
To date, there has been no study examining directly the inflammatory mediators implicated in OSA and DME. Knowledge of the cytokine differences will provide insight into the biochemical relationship (if any) of OSA and DME, and further understanding of the pathophysiology.
Methods
This was a single centre, prospective study. Ethics approval was obtained from William Osler Health Research Ethics Board and registered on clinicaltrials.gov (ID # NCT04648618). 32 patients with a new diagnosis of DME were enrolled in the study from 2018 to 2020.
Study participants
Patients with type II diabetes mellitus and treatment-naive DME undergoing first intravitreal injection were recruited for the study. If a patient presented with bilateral DME, the eye with the worse vision was included in the study. Exclusion criteria included any contraindications to intravitreal injection such stroke within the past month, ocular/periocular infection, active intraocular inflammation and hypersensitivity to components of intravitreal injection; any other retinal or macular pathology besides diabetic retinopathy, any intraocular surgery or laser within the past 3 months, any use of systemic immunosuppressive or anti-inflammatory medications within past 30 days, as well as pregnancy or breastfeeding women.
Polysomnography
All study participants were consented for overnight polysomnography and assessment by a sleep specialist to determine their OSA status. Respiratory data collected included apnea hypopnea index (AHI), Epworth sleepiness scale, mean and minimal O2 saturation. OSA positive (OSA + ) was defined as AHI ≥ 15, and those with AHI
Study protocol
All patients underwent baseline ophthalmologic examination including best-corrected visual acuity, intraocular pressure, slit lamp exam, dilated retinal exam, fluorescein angiography and optical coherence tomography (OCT; Cirrus HD-OCT; Carl Zeiss Meditec AG, Jena, Germany). Each standard domain OCT was reviewed by the study investigators to confirm diagnosis of DME and to exclude other macular pathology. Central foveal thickness was calculated by Cirrus OCT. Fluorescein angiography was performed to rule out other retinal pathology and to categorize severity of diabetic retinopathy from mild, moderate and severe non-proliferative to proliferative diabetic retinopathy (PDR). 15
Both serum and aqueous fluid samples were obtained from each patient prior to initiating any treatment with the standard of care anti-VEGF treatment. 2 mL of blood was collected in pyrogen/endotoxin-free tubes and allowed to sit at room temperature for 15–30 min to clot. This was spinned at 1000–2000 × g for 10 min in a 4°C refrigerated centrifuge to separate the cells. The serum sample was transferred to a chilled clean polypropylene tube with a sterile pipette. Aqueous fluid samples were obtained using standard sterile techniques: a 30-gauge needle was passed into anterior chamber through the temporal limbus, and approximately 0.1 mL of aqueous fluid was collected. Multiplex immunoassay (Merck KGaA, Darmstadt, Germany) was performed at Princess Margaret Genomics Centre (University Health Network, Toronto). The following cytokines were identified from literature review to be potentially relevant to the pathophysiology and were collected: VEGF, placental growth factor (PGF), ICAM, IL2, IL3, IL6, IL8, IL10, IL17, vascular cell adhesion molecule-1 (VCAM1), monocyte attractant protein-1 (MCP1), epidermal growth factor (EGF), and platelet derived growth factor (PDGF).
Statistical analysis
Demographics of the study population were presented through descriptive statistics. Clinical characteristics were compared using Two-Tailed T tests, Mann Whitney test or Chi squared test accordingly. The level of aqueous and serum cytokine between patients with and without OSA were compared using Mann Whitney test assuming nonparametric distribution. Bonferroni correction was applied for multiple comparisons. Statistical significance was defined as p < 0.004 (0.05/13).
Results
Demographics
32 patients with antiVEGF-naïve DME were enrolled in the study, of which 17 patients were classified as OSA positive, and the balance were classified as OSA negative. The mean age was 62 ± 9.3 and 60 ± 7.8 in the OSA + and OSA- cohorts, respectively (Table 1). In the OSA + cohort, 94% (16/17) of patients were male, and majority of patients had hypertension and dyslipidemia (71%, 12/17 for both). In the OSA- cohort, 47% (7/15) patients were male, while 80% (12/15) and 47% (7/15) of patients had hypertension and dyslipidemia respectively. Greater proportion of patients in the OSA- cohort were pseudophakic and had prior macular focal laser (47% vs. 24%, p = 0.03 for both comparisons). Majority of patients had remote history (>3 months) of cataract surgery and focal laser (82% for cataract surgery, 100% for focal laser). Two patients in the OSA- group had cataract surgery at 2 months prior to injection. The duration of diabetes was similar between both cohorts (16 ± 6.8 vs. 16 ± 7.8 years, p = 0.9). There were no complications performing aqueous chamber paracentesis.
Demographics of patients with and without OSA.

Clinical characteristics
Baseline clinical characteristics are illustrated in Table 2. Compared to patients with OSA + , the OSA- cohort had better vision and lower central retinal thickness, although this was not statistically significant (0.62 ± 0.53 vs. 0.39 ± 0.27 logMAR, 432 ± 139 vs. 373 ± 81 µm). The majority of patients had moderate non-PDR in both cohorts: 11/17 (65%) in the OSA + cohort; 10/15 (67%) in the OSA- cohort.
Clinical characteristics of patients with and without OSA.

*NPDR: non-proliferative diabetic retinopathy.
** PDR: proliferative diabetic retinopathy.
With respect to sleep metrics, the mean AHI in the OSA + cohort was 43 ± 29, with 8 patients categorized as moderate OSA and 9 as severe. The mean AHI in the OSA- cohort was 7.2 ± 4.5. Compared with the OSA + cohort, the OSA-cohort had significantly higher minimal O2 saturation and lower BMI (80 ± 7.6 vs 87 ± 5.3%, p = 0.02; 30 ± 4.5 vs. 25 ± 2.7, p < 0.05). The mean O2 saturation was similar for both cohorts.
Cytokines
OSA- cohort had significantly higher levels of aqueous IL17 compared to the OSA + cohort (2.97 ± 1.7 vs. 1.4 ± 0.46 pg/mL, p = 0.004). (Table 3) The OSA + cohort had significantly higher levels of serum EGF compared to the OSA- cohort (165 ± 183 vs. 18.7 ± 9.3 pg/mL, p = 0.003). There were no other significant differences in aqueous and systemic cytokines between OSA + and OSA- cohorts.
Aqueous and serum cytokines (pg/mL).
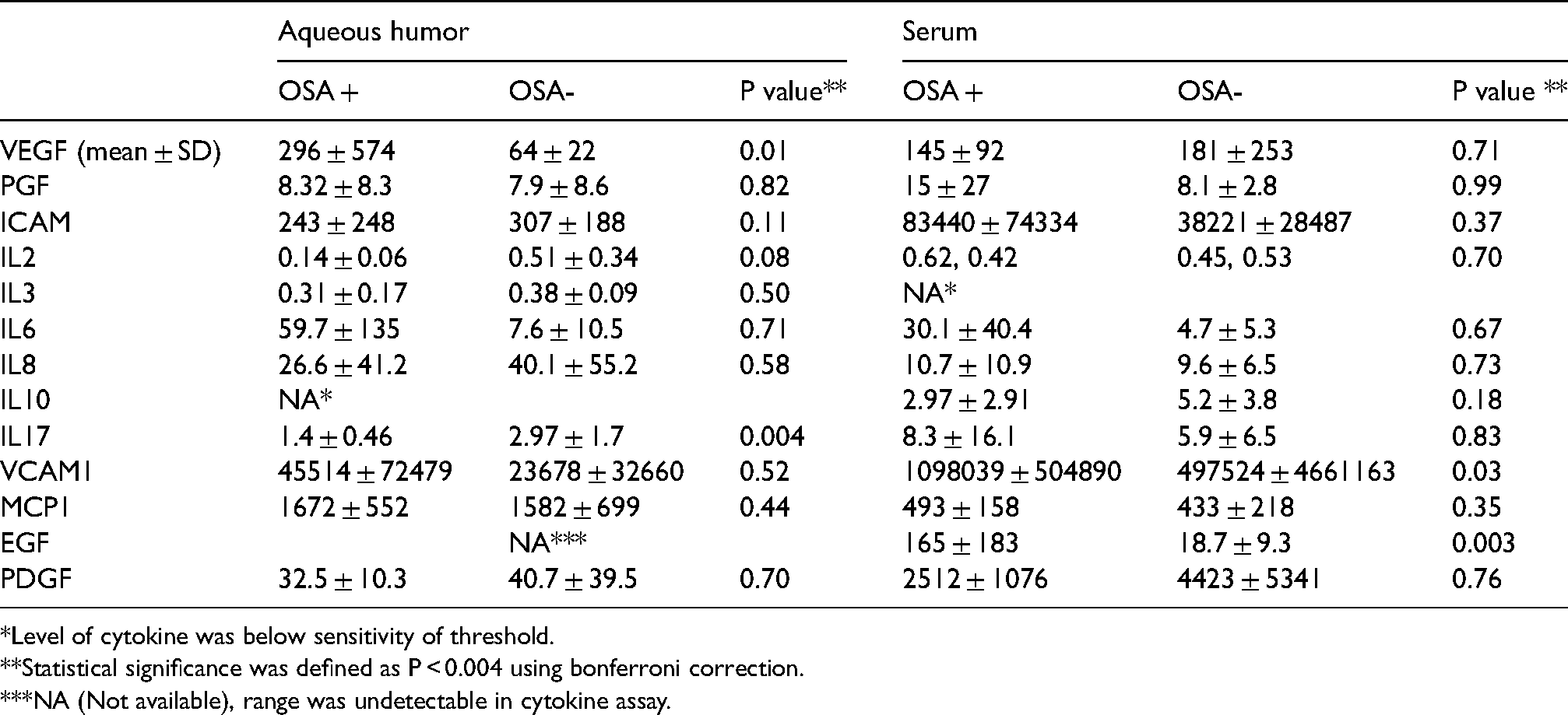
*Level of cytokine was below sensitivity of threshold.
**Statistical significance was defined as P < 0.004 using bonferroni correction.
***NA (Not available), range was undetectable in cytokine assay.
Discussion
DME has been increasingly recognized as a consequence of interaction of several inflammatory cytokines. Several studies have investigated the role of cytokines in pathophysiology of DME. Aqueous VEGF, IL6, and MCP1 have consistently been found to be elevated in patients with DME.16–18 IL6 is a pro-inflammatory cytokine that has been shown to increase vascular permeability and promote angiogenesis. 19 It correlates with severity of diabetic retinopathy as well as DME. 20 IL6 may increase vascular permeability both directly or indirectly by upregulating VEGF. 21 Meanwhile, MCP1 induces monocyte migration to sites of inflammation produced by tissue hypoxia. 22 Activated monocytes differentiate into macrophages, which secrete additional cytokines including VEGF, TNF-alpha, angiopoietin, and interleukins. 19 MCP-1 has also been shown to mediate the gene expression of VEGF-A. 23 Other cytokines have been less consistent, with variable results in studies. 19
Studies have also investigated the role of inflammatory cytokines in OSA. A previous meta-analysis of 51 studies showed elevated levels of serum IL6, IL8, ICAM and VCAM in patients with OSA. 24 Interestingly, the intermittent hypoxia in OSA has been found to induce expression of IL-6 and macrophage polarization, contributing to the inflammatory cascade. 25 An in vitro study also showed release of TNF- α, IL8, and IL6 with intermittent hypoxia. 26 Intermittent hypoxia is likely more inflammatory than stable hypoxia, as the cycles of hypoxia and reoxygenation can promote formation of reactive oxygen species and promote oxidative stress. 27 Thus, the hypoxic state in OSA may serve as an additional stimulus for the inflammatory pathway in DME.
This is the first study to investigate the biochemical relationship between OSA and DME in a prospective fashion. We report that compared to those without OSA, patients with OSA had significantly higher levels of serum EGF and trended towards higher levels of most serum cytokines. However, we did not find significantly higher levels of aqueous cytokines in the OSA + group. Interestingly, patients with OSA had significantly lower levels of aqueous IL-17, suggesting that various cytokines may have a different role in the pathogenesis of DME in patients with and without OSA.
We found higher mean levels of aqueous VEGF, IL6 and VCAM1 in patients with OSA. However, this was not statistically significant likely due to small numbers and wide range of standard deviation. Interestingly, in a previous study by Roh et al., after injection of bevacizumab in patients with DME, recurrence of DME was associated with higher level of IL6 and not VEGF, suggesting that macular edema may be influenced by IL6 independent of VEGF levels. 28 IL6 has also been studied in OSA. In a meta-analysis from 2013, the levels of inflammatory cytokines have been found to be higher in patients with OSA with standardized pooled mean difference of 2.16 pg/mL for IL6. 24 It is likely that certain cytokines such as IL6 may play a distinct role in the mediating the inflammatory pathway along with VEGF. Although the level of aqueous IL6 was not statistically different in our study, there was a wide range in the level of IL6 in OSA + patients up to 453 pg/mL. Thus, it is possible that cytokines such as IL6 may play a role in pathogenesis of DME in a subgroup of patients with OSA. Further study with greater number of patients is needed to increase power and determine the significance of these trends in aqueous cytokines.
In addition, level of aqueous IL-17 was found to be significantly lower in patients with OSA. IL-17 is produced by Th17 cells and has been reported to impair function of Muller cell. 29 It is likely that there are differences in pro-inflammatory pathways in patients with and without OSA, and that IL-17 likely plays a lesser role in mediating DME in patients with OSA. Further studies with greater power are needed to determine clinically significant differences, and whether treatment of OSA can reverse the inflammatory cascade.
The strengths of our study include involving patients who were treatment-naïve to both DME and OSA, thereby, limiting any confounding impact of treatment on the expression of their inflammatory cytokines. Further, classifying OSA patients with the use of a gold standard test allows for more meaningful comparisons. In terms of limitations, The OSA- cohort had a trend towards a greater incidence of remote (>3 months) cataract surgery and macular grid laser, which may impact the expression of inflammatory cytokines. Finally, as an exploratory study, our study was limited by small sample size and wide standard deviation. Increasing the sample size in future studies may yield more understanding into the cytokine expression patterns.
In conclusion, treatment-naïve DME patients with OSA had greater pro-inflammatory state with higher levels of most serum cytokines including serum EGF. However, they did not have greater levels of inflammatory aqueous cytokines. Patients with OSA also had lower levels of aqueous IL-17, suggesting that IL-17 likely plays a lesser role in mediating DME in patients with OSA. A greater understanding of the biochemical relationship between OSA and DME may provide further insights into novel therapeutic options.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
