Abstract
Purpose: The present study was conducted to determine the effect of odor stimulation with breast milk (BM) applied to premature newborns before and during enteral feeding on feeding cues, transition time to oral feeding and abdominal perfusion. Design: This study was a parallel group pretest-post test randomised controlled trial (RCT). Methods: The study was conducted in the neonatal intensive care unit (NICU) in Türkiye. Study data were collected from 32 premature newborns between October 2020 and December 2021. Newborns were randomly assigned to either intervention (n = 16) or control (n = 16) groups. Odor stimulation with BM was applied to newborns in the intervention group (IG) before and during enteral feeding for 3 days. Data were collected with the Newborn Information Form, Abdominal Perfusion Follow-up Form, and Feeding Cues Follow-up Form. Yates corrected and Fisher chi-squared test, Mann-Whitney U test, Cohen Kappa statistics, Permanova analysis, and Wilcoxon test were used in the analysis. The study was registered in ClinicalTrials.gov (NCT04843293). Results: The frequency of feeding cues in newborns who were exposed to the odor of BM, was higher than in the control group (CG) (p < .05). The transition times to oral feeding were similar in newborns in the IG and CG (p > .05). Abdominal perfusion level of newborns in the IG was higher than the CG (p < .05), and the level of abdominal perfusion showed a significant difference in terms of group*time interaction (p < .05). Conclusion: Our findings suggest that odor stimulation with BM will be beneficial in improving the digestive functions of premature newborns.
Introduction
Successful and safe oral feeding of premature newborns is affected by many factors such as the maturation of their neurological and gastrointestinal (GI) systems and the coordination of sucking-swallowing-respiratory functions (Lau, 2016). Sucking-swallowing-respiratory coordination in newborns develops between 32 and 33 weeks gestational age (GA; Bertoncelli et al., 2012; Cao Van et al., 2018). Therefore, premature newborns born before the 32nd week of GA usually need to be fed enterally or parenterally temporarily until their rhythmic breathing behavior during feeding matures (Beker et al., 2017; Cao Van et al., 2018). However, nutritional problems such as feeding intolerance, vomiting, decreased weight gain, abdominal distention, and prolonged transition to oral feeding can be seen in premature newborns (Baldassarre et al., 2019; Beker et al., 2017; Indrio et al., 2011; Lucchini et al., 2011; Thomas et al., 2018). Therefore, one of the main goals of neonatal intensive care unit (NICU) nurses is to help premature newborns develop oral feeding skills (Thoyre, 2007). In order for newborns to develop oral feeding abilities, it is recommended that the nurse evaluate the baby’s behavioral cues before each feeding (Gözen & Aykanat Girgin, 2017). Newborn licking their own lips, orienting to the gentle touch around the mouth/lips, opening the mouth (Gözen & Aykanat Girgin, 2017), being silently alert, and sucking the hand or fist are reported feeding cues (Watson & McGuire, 2016). The nutrition provided by following the feeding cues reduces the transition time to full oral feeding compared with a traditional diet (Pickler et al., 2015; Watson & McGuire, 2016), shortens the mother and child hospital stay (McCormick et al., 2010; Pickler et al., 2015), improves food intake (Kirk et al., 2007), decreases nutrition-related adverse events (Puckett et al., 2008), and decreases staff workload by improving parents’ feeding skills (Kirk et al., 2007).
The delay in the acquisition of oral feeding skills is a key factor that prolongs the hospital stay of newborns (Jadcherla et al., 2010). Newborns who can be fed effectively and safely by breast or bottle and are deemed medically stable may be discharged from the hospital (Breton & Steinwender, 2008). Problems in oral feeding skills of premature newborns may continue even after discharge (Shaker, 2013). Therefore, in recent years, the focus has been on improving the oral feeding skills of premature newborns and on interventions that can shorten the transition period to oral feeding. One of the methods applied to improve the oral feeding functions of newborns is olfactory stimulation (Schriever et al., 2018). It is known that food odors are stimulants to initiate the cephalic phase (CP) of digestion (Smeets et al., 2010). The CP includes the digestive and metabolic responses caused by the central nervous system that prepare the organism for digestion, absorption, and metabolism of nutrients (Smeets et al., 2010). With the CP response, intestinal motility is increased, digestive enzymes are secreted, nutrient absorption is facilitated, and blood glucose level is controlled (Eliasson et al., 2017; Smeets et al., 2010). In premature newborns, when fed enterally or parenterally, exposure to the odor of food which initiates the CP of digestion is eliminated (Muelbert et al., 2019). In studies evaluating the effects of olfactory stimulation on feeding of premature newborns, the effects of BM feeding in premature newborns, transition to oral feeding (Beker et al., 2017; Khodagholi et al., 2018), shorten the duration of hospital stay (Muelbert et al., 2019; Yıldız et al., 2011), and digestive function is positively influenced by reduction of the residual gastric volume have been reported (Lee, 2019).
In recent years, Near Infrared Spectroscopy (NIRS) has been used to evaluate the effect of odors on newborn child health. Regional tissue perfusion of premature newborns can be evaluated with NIRS (Garvey & Dempsey, 2018; Wolf et al., 2012). Food odors stimulate the CP response of digestion and differences occur in the activity of the digestive system organs in the abdominal region (Eliasson et al., 2017). Although the positive effects of BM odor on nutrition are known (Beker et al., 2017; Lee, 2019; Yildiz et al., 2011), no study has explored the effect of BM odor on the CP response and therefore, on the level of abdominal perfusion (rSO2). For this reason, our study was conducted to determine the effects of odor stimulation with BM applied before and during enteral feeding on feeding cues, transition time to oral feeding, and abdominal rSO2 level.
The research hypotheses are as follows: Breast milk odor applied before and during enteral feeding in premature newborns (H1) increases the frequency of feeding cues; (H2) shortens the transition time to oral feeding; (H3) increases abdominal rSO2 level; and (H4) positively affects daily weight gain, number of the premature newborns vomits, the number of times the premature newborns defecates, and the duration of hospital stay.
Methods
Design
This study was designed as a pretest-post test randomised, controlled experimental study, using a parallel group.
Participants
The study was conducted in the NICU of a university hospital in Türkiye, between October 2020, and December 2021. The adequacy of the sample size was evaluated by post hoc power analysis using abdominal rSO2 data, which is one of the main hypotheses of the study. The study was terminated with 32 infants, as sufficient power at 95% confidence (1-α), 95% test power (1-β), and effect size d = .452 that were obtained with the data from a total of 32 infants of which 16 were in the intervention group (IG) and 16 in the control group (CG).
The study included premature newborns who were 28–36+6 GA, had a birthweight >1000 g, fed by intermittent infusion (only the gravity method is used in their feeding) method. Those infants who were continuously infusion fed, slow bolus or parenteral fed, or full oral fed, or had nasal obstruction or mechanical ventilation/CPAP support during the study period were excluded from the study. In addition, participants receiving treatment affecting GI function during the study period or those who had any medical diagnosis (congenital anomaly, metabolic disease, asphyxia, sepsis, hyperbilirubinemia, GI bleeding, anatomical GI anomaly, GI disease and infection), were excluded from the study. Infants who switched to full oral feedings for 24 h during the intervention in the study would have been excluded from the study, but there were no fully orally fed infants during the intervention period of the study.
Randomisation and Blinding
The stratified randomisation method was used to assign the infants to the IG and CG. To ensure homogeneity, participants were randomised and stratified by postmenstrual age (PMA) (28–33+6 and 34–36+6) and by type of feeding (BM and mixed feeding [BM + formula milk]) (Neal-Kluever et al., 2019; Pillai et al., 2018). Blocks were created with the blocked randomisation method, after which the newborns were randomly assigned to IG or CG using the random numbers table (www.random.org). The clinical nurse implemented the randomisation table. The group to which each new case was assigned was not disclosed to the researcher until the initiative began. However, since the researcher participated in monitoring the implementation of the initiative and actively conducting the data collection process, researcher blinding could not be fully applied. In order to avoid bias in the evaluation of the data, the study groups were coded as A and B instead of IG or CG in the database, and the data were analysed by an independent statistician. Study phases and study reporting were conducted according to The Consolidated Standards of Reporting Trials (CONSORT) guidelines (Figure 1) (Boutron et al., 2017). Trial CONSORT flow diagram.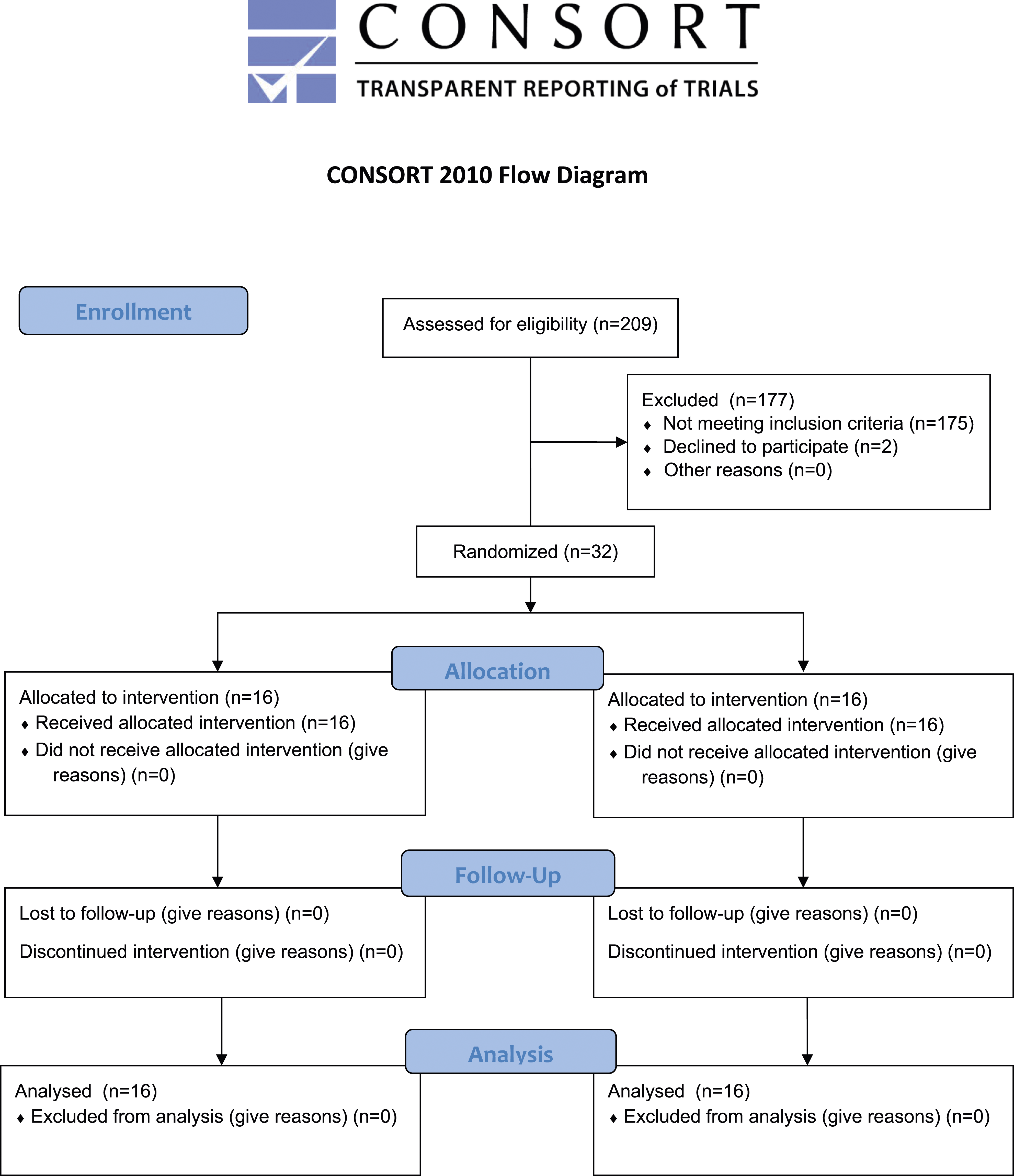
Data Collection Tools
The data collection forms used in the study were prepared in line with the literature. The forms were finalised after receiving the input from five experts in the field of child health and disease nursing.
Newborn Information Form
The Newborn Information Form consisted of questions about the newborn’s gender, type of delivery, GA at birth, birthweight, PMA, transition time to oral feeding, discharge time, daily body weight, daily numbers of vomiting and defecation (Neal-Kluever et al., 2019; Pillai et al., 2018; Schriever et al., 2018; Yildiz et al., 2011).
Abdominal Perfusion Follow-Up Form
The abdominal perfusion form contained information about the abdominal regional tissue oxygenation (rSO2) value of newborns. rSO2 measurements are recorded before feeding (T0) and 10 (T1), 30 (T2), 60 (T3) and 120 minutes (T4) after feeding. The INVOSTM 5100C NIRS device was used to evaluate the abdominal rSO2 level of newborns. NIRS is a non-invasive method that determines rSO2 by measuring the concentration of oxyhemoglobin and deoxyhemoglobin in the tissue with an infrared light source (Chakravarti et al., 2008). With the continuous use of NIRS at the bedside, it can provide early diagnosis of hypoperfusion and hypoxia in the regional tissue without irreversible tissue damage (Jeon, 2019; Martini & Corvaglia, 2018). Monitoring abdominal perfusion with NIRS provides an important opportunity to evaluate the oxygenation of the intestines, which constitutes a large part of digestion (McNeill et al., 2011).
Feeding Cues Follow-Up Form
The feeding cues form was developed by the researchers during the study planning and data collection process because there is no Turkish valid and reliable scale evaluating feeding cues. The form included the physiological and behavioral hunger symptoms of infants, which are accepted as criteria for starting oral feeding in infants and maintaining oral feeding, successfully and reliably (Holloway, 2014; Kirk et al., 2007; Ludwig & Waitzman, 2007; White & Parnell, 2013). The form consists of nine items and the presence of cues is evaluated based on observation. The cues in the form are as follows: being in the range of normal vital signs (Cue 1: C1), opening your mouth (C2), opening your eyes (C3), bringing your hands to your face and/or mouth (C4), sticking your tongue out of your mouth (C5), moving your arms and/or legs (C6), moving the head to the right/left (C7), orienting to the gentle touch around the mouth/lips (C8), and keeping the body in a flexed posture (C9) (Gözen & Aykanat Girgin, 2017; Holloway, 2014; Kirk et al., 2007; Kurt Sezer & Küçükoğlu, 2020; Ludwig & Waitzman, 2007; White & Parnell, 2013). Evaluation of the form was performed independently by the researcher and an observer (NICU nurse) in order to avoid bias. As a result of the study, the inter-observer agreement was examined, and it was found that the agreement was between .853 and 1000 on item basis (p < .01). The general agreement among the observers was .892 (p < .01).
Data Collection
At the beginning of the study, the introductory information in the “Newborn information form” was taken from the medical records by the researcher. Body weight, number of times vomited and defecated were obtained from nurse records after 24-h periods. Body weight measurement was performed using a portable digital weighing device sensitive to 20 g. The newborns were weighed after changing nappies and removing their clothes. The clean nappy weight was determined and not included in the total weight calculation. Newborns were follow-up for three consecutive days. The time (days) between the end of the 3-day follow-up of the newborn and the transition to full oral feeding was calculated as the transition period to oral feeding. Oral feeding for 1-day (24 h) was considered as full oral feeding. Similarly, the period (days) between the end of the study follow-up and the day of discharge was considered as the discharge time.
After routine care, the premature newborns were placed in the supine position. The infraumbilical region (just below the umbilicus) is recommended for sensor placement in order to best measure intestinal perfusion with the mesenteric artery (McNeill et al., 2011). Therefore, the NIRS sensor was placed in the premature newborns infraumbilical region by the NICU nurse to evaluate abdominal rSO2 (Figure 2). rSO2 values were recorded just before the newborns in the IG started feeding. Then, olfactory stimulation was started. One minute after odor stimulation was started, the feeding cues follow-up form was completed while odor stimulation continued. Premature newborn feeding was started, and olfactory stimulation continued throughout the feeding period. rSO2 values in the CG were recorded just before feeding. After rSO2 was recorded, the feeding cues follow-up form was completed, and feeding was started. rSO2 values used to evaluate abdominal perfusion were recorded with the NIRS monitor in both groups before feeding and at 10, 30, 60, and 120 min also after feeding. The flow chart of the study is presented in Figure 3. NIRS sensor location in the study. Flow chart of the study.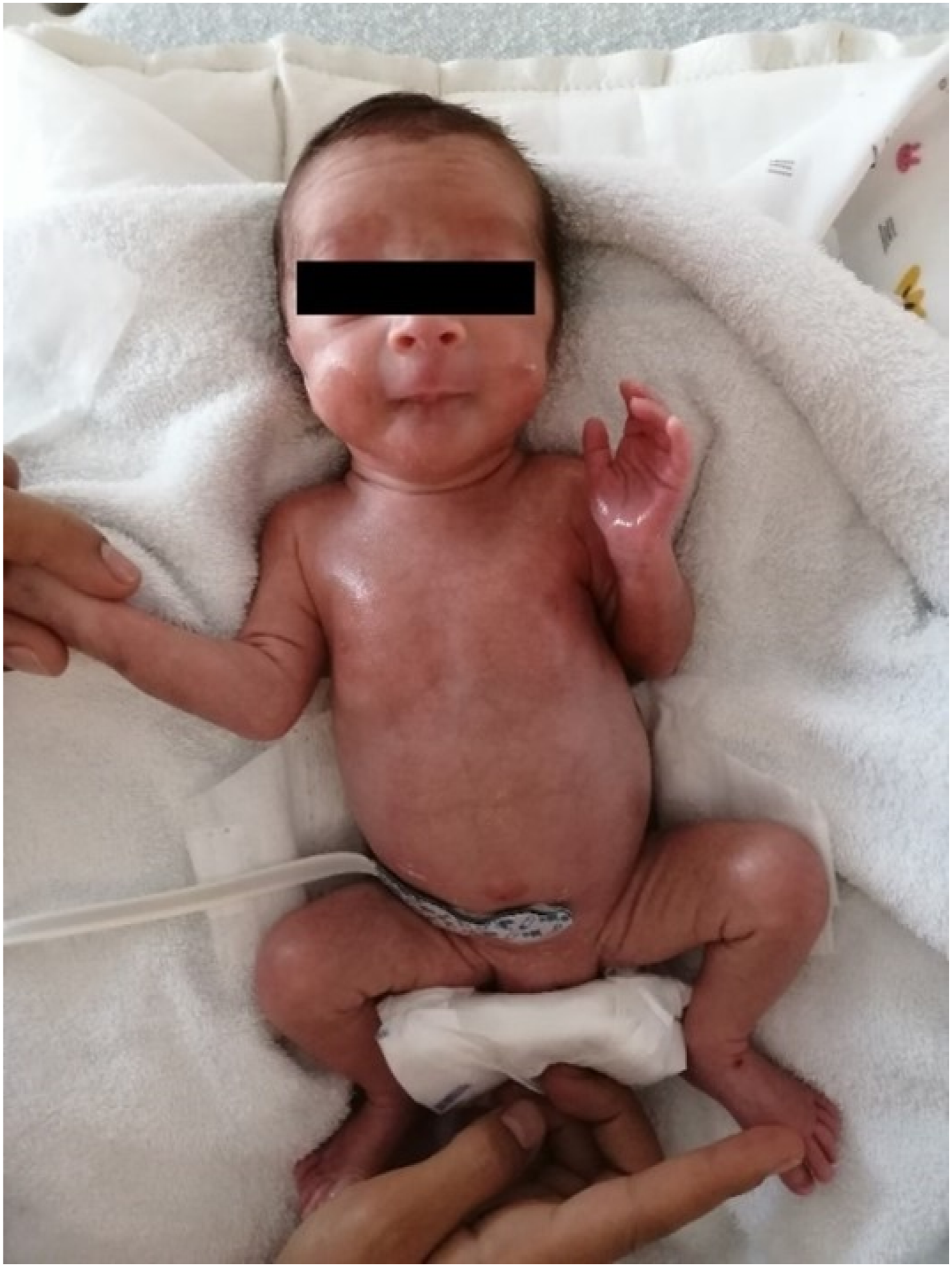
Breast Milk Odor
It has been reported that BM (10–15 drops) added to wet sterile gauze can be used for odor stimulation (Yildiz et al., 2011). For this reason, odor stimulation was performed by dripping enough (about 1 cc) BM to wet the nappy on sterile gauze with BM provided by each baby’s own mother.
Implementation of the Initiative
Premature newborns in the IG were given odor BM stimulation before three consecutive feedings during the day for 3 days. Odor stimulation was started 1 min before the start of enteral feeding and was continued until the feeding finished. Sterile gauze dripping with BM was placed in the incubator close to the newborn’s nasal septum (1.5–2 cm) (Iranmanesh et al., 2015) and not touching the newborn’s skin (Figure 4). Odor stimulation was conducted with the incubator covers closed so that the premature newborns in the incubator did not experience heat loss. The application of BM odor to the premature newborns whose feeding was completed was terminated and the gauze was removed from the incubator. A new sterile gauze was used for odor stimulation at each feed. These procedures were repeated in three feedings during the day for 3 days. Thus, odor stimulation was performed with BM for a total of nine times. The premature newborns in the CG were fed according to the routine of the clinic and no intervention was applied other than routine practice. The follow-up of premature newborns in this group was mirrored the steps applied to newborns in the IG. Odour stimulation with breast milk.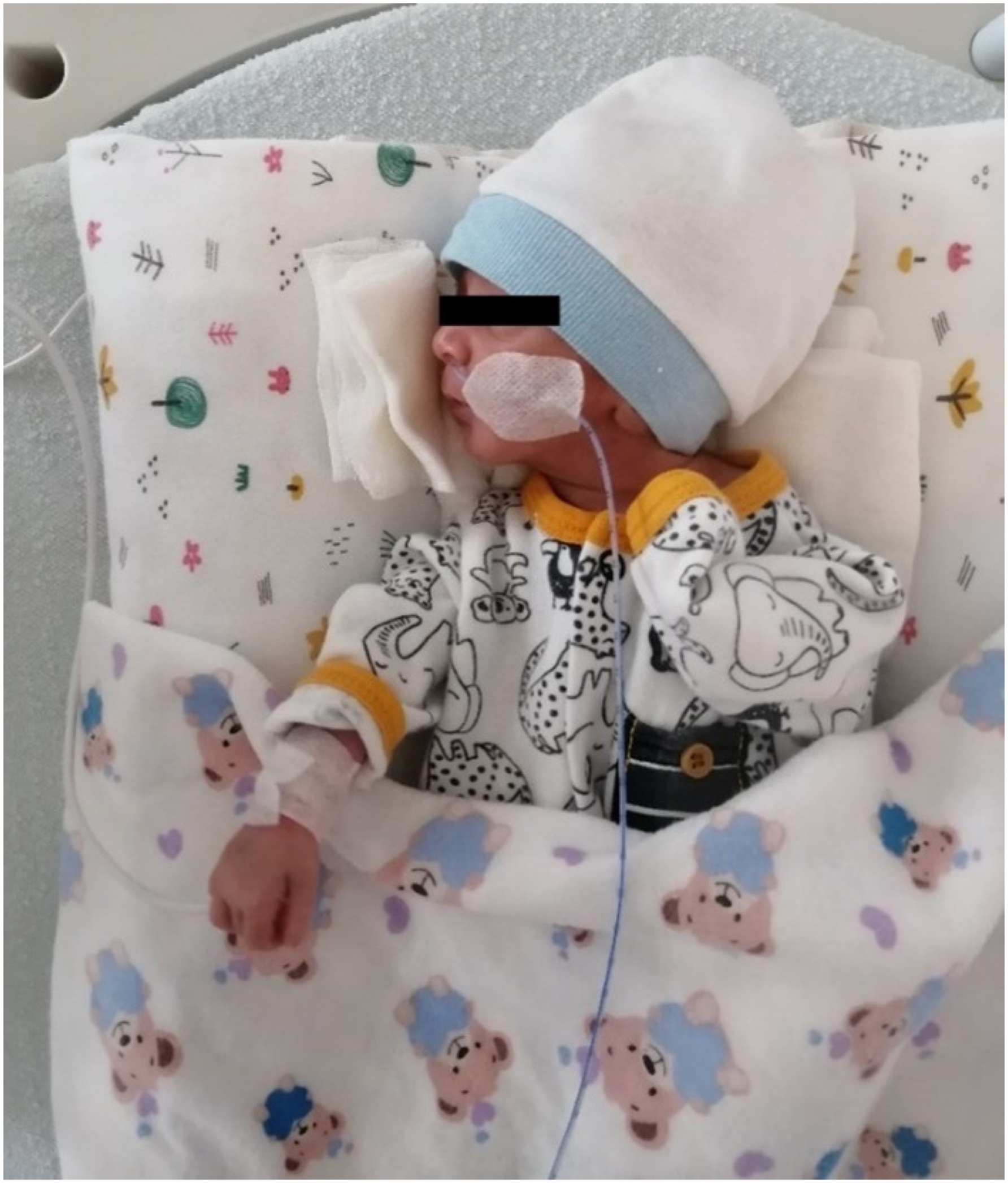
Data Analysis
Data analysis was performed with SPSS 24.0, PAST 4.04, and G*POWER 3.0.1. For the analysis of descriptive data, the number, percentage, and mean were used. Yates corrected and Fisher chi-squared tests and Mann-Whitney U test were used to compare socio-demographic characteristics of IG and CG. Fisher and Yates corrected chi-squared tests were used to compare groups according to feeding cues. The Mann-Whitney U test was used to compare the transition times of the IG and CG to oral feeding. The PERMANOVA analysis was used to compare the abdominal rSO2 value averages. The Mann-Whitney U test was used to compare the groups with each other according to time and the Friedman test was used to compare the groups according to time within themselves. When a difference was found in the Friedman test, the corrected Wilcoxon test was used to determine which measures caused the difference. Friedman test was used to compare the daily weight gains of infants, Wilcoxon analysis with Bonferroni correction was used for further analysis, and Mann-Whitney U test was used to compare groups. Friedman test was used to compare the mean daily vomiting, defecation frequency, and discharge time of the babies within the group for which the Mann-Whitney U test was used to compare the groups with each other. The significance level was accepted as p < .05.
Ethics
Prior to the study, ethics committee approval was obtained from the Non-Interventional Clinical Research Ethics Committee of the local state university with the decision number 2020/3, dated 06/07/20, and institutional permission was obtained from the hospital where the study was conducted. Before the new-borns who fit the sampling inclusion criteria were admitted to the study, the necessary information about the study was presented to the parents. The informed consent form of volunteers was read by parents and their written permission was obtained before recruiting the premature newborns for the study. The study is registered in ClinicalTrials.gov (NCT04843293).
Results
Descriptive Characteristics of Premature Newborns (n = 32).
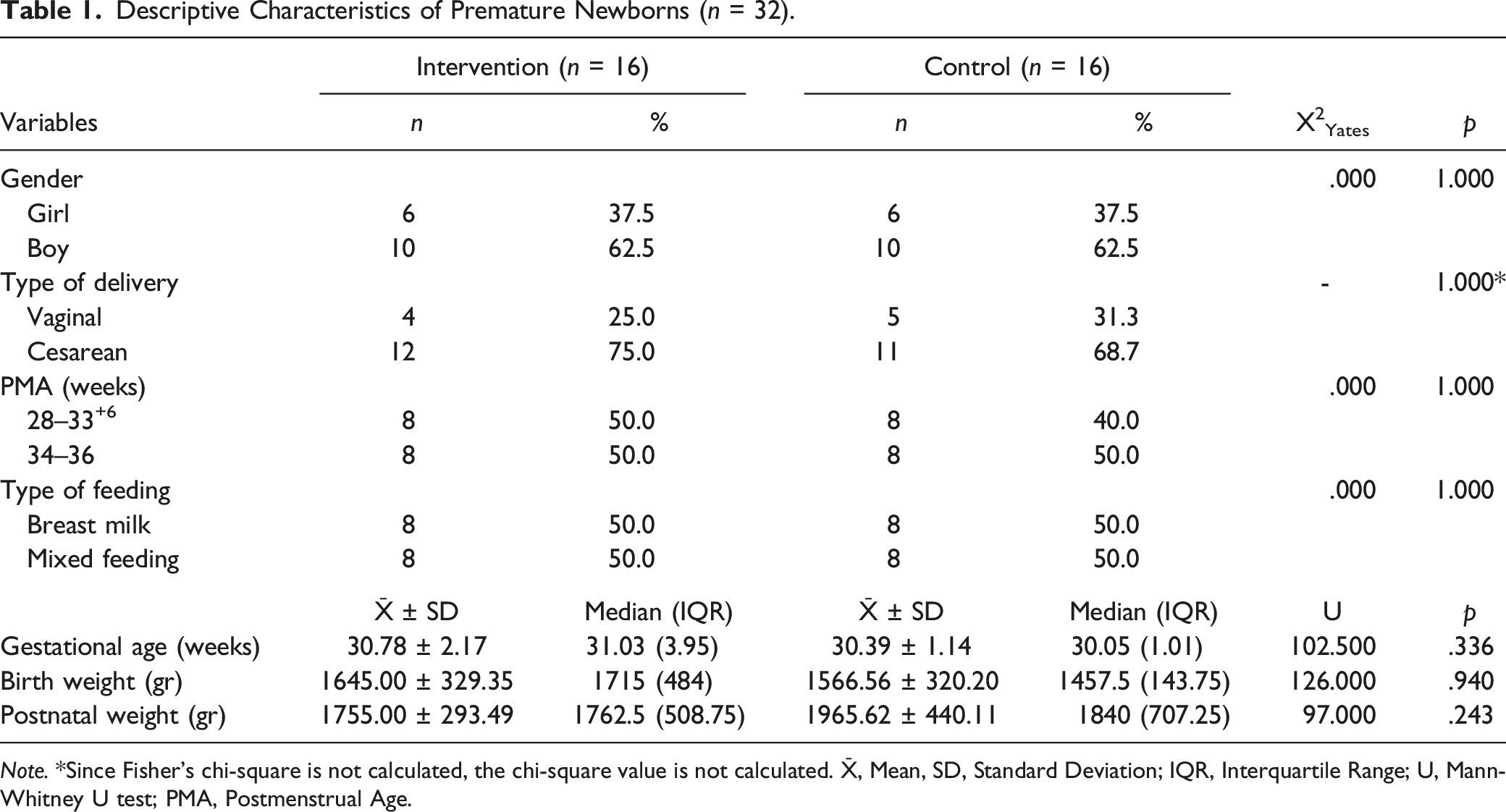
Note. *Since Fisher’s chi-square is not calculated, the chi-square value is not calculated. X̄, Mean, SD, Standard Deviation; IQR, Interquartile Range; U, Mann-Whitney U test; PMA, Postmenstrual Age.
Comparison of Feeding Cues.
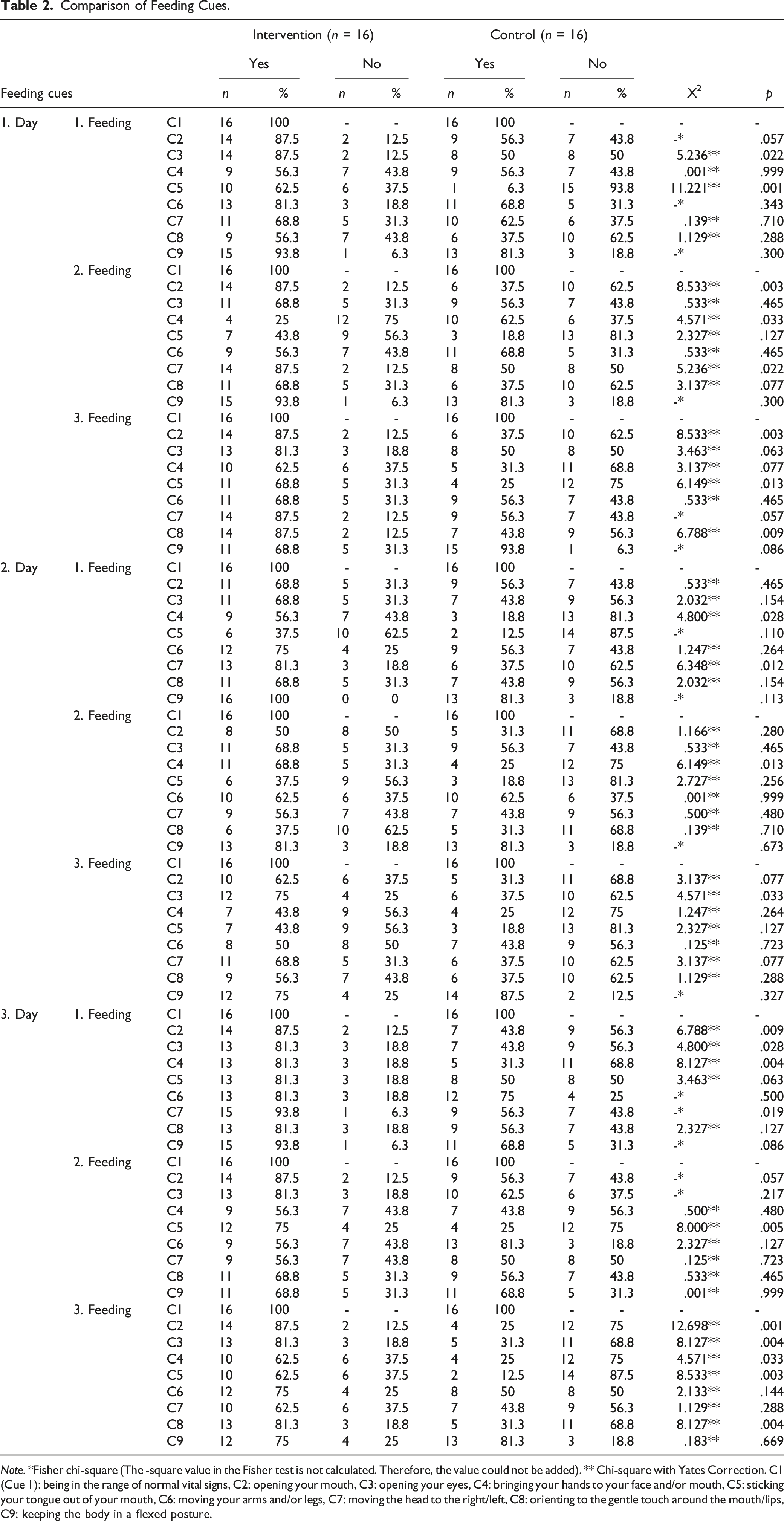
Note. *Fisher chi-square (The -square value in the Fisher test is not calculated. Therefore, the value could not be added). ** Chi-square with Yates Correction. C1 (Cue 1): being in the range of normal vital signs, C2: opening your mouth, C3: opening your eyes, C4: bringing your hands to your face and/or mouth, C5: sticking your tongue out of your mouth, C6: moving your arms and/or legs, C7: moving the head to the right/left, C8: orienting to the gentle touch around the mouth/lips, C9: keeping the body in a flexed posture.
Comparison of Abdominal Perfusion (rSO2) Values.
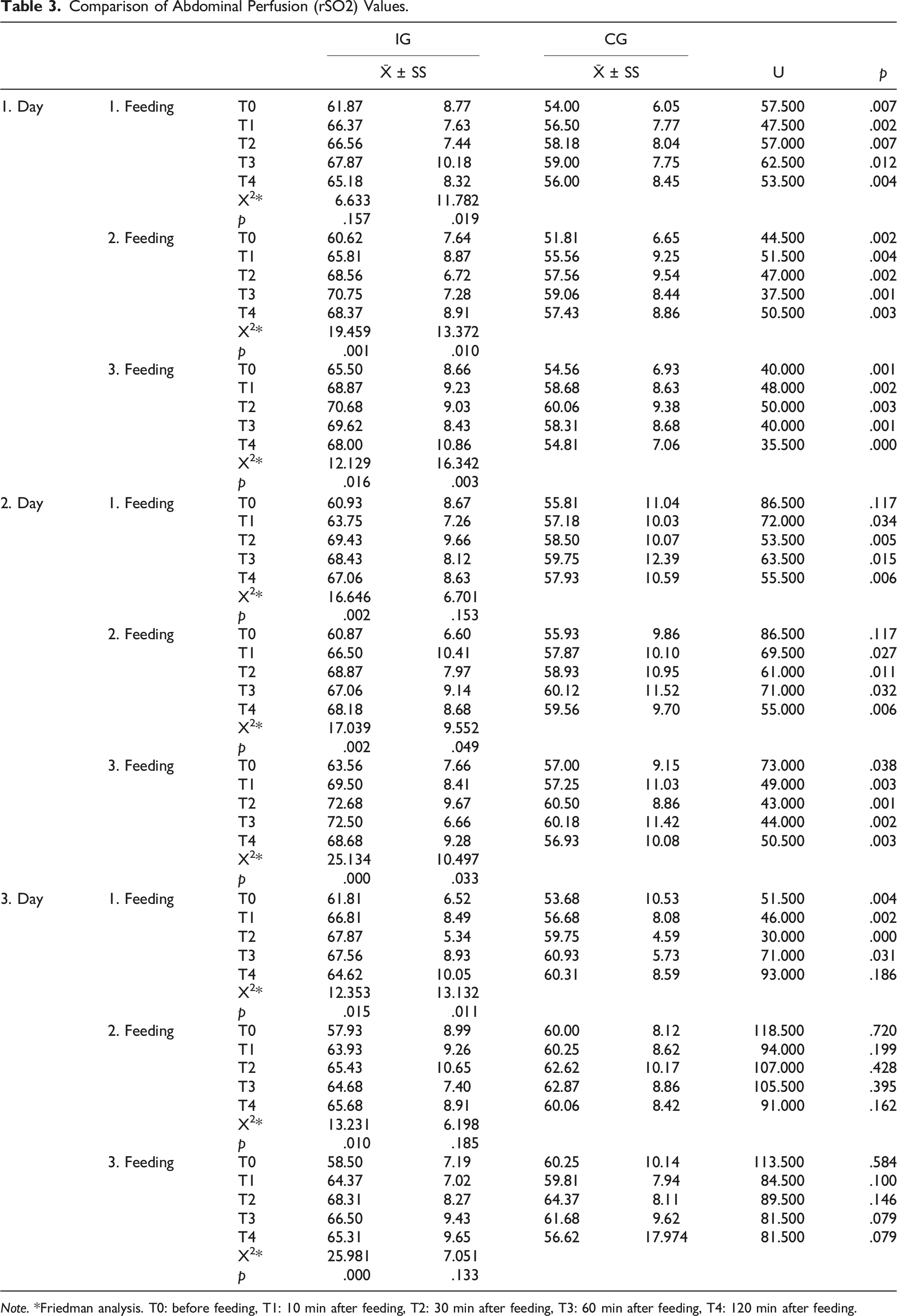
Note. *Friedman analysis. T0: before feeding, T1: 10 min after feeding, T2: 30 min after feeding, T3: 60 min after feeding, T4: 120 min after feeding.
Comparison of Abdominal Perfusion rSO2 Values by Group and Time.

Note. *Permanova Analysis.
Comparison of Daily Weight Gain, Daily Defecation, and Vomiting Frequency of Premature Newborns by Group (n = 32).
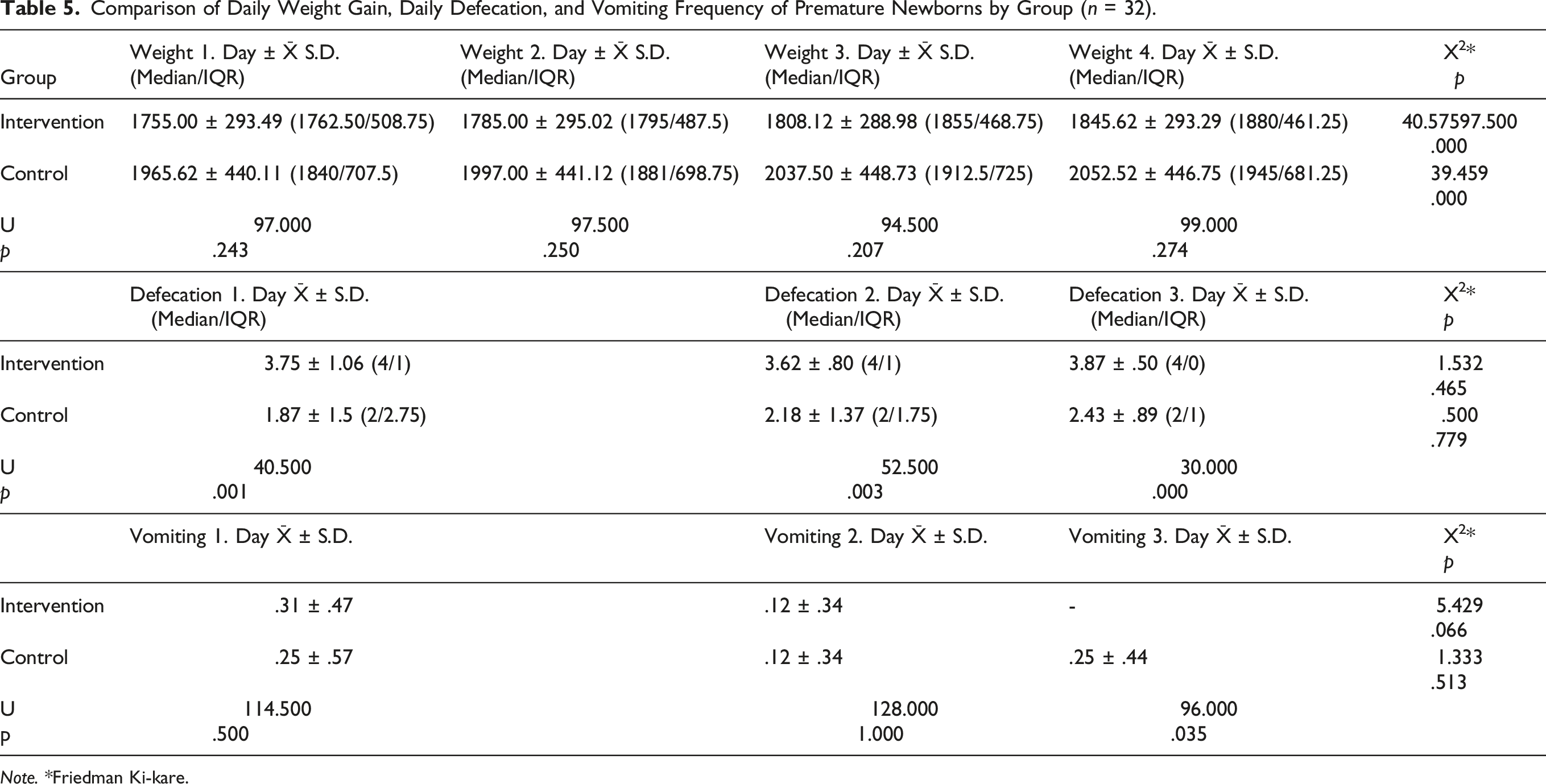
Note. *Friedman Ki-kare.
The transition time to oral feeding in the IG was 8.87 ± 4.39, while the transition time to oral feeding of the CG was 11.68 ± 4.39, which was not statistically significant (p > .05). Also, the discharge time of the infants in the IG was 12.43 ± 4.81 days while the discharge time of the infants in the CG was 13.56 ± 4.42, which was not statistically significant (p > .05).
Discussion
In this study, we aimed to determine the effect of BM odor applied before and during enteral feeding with feeding cues, transition time to oral feeding, and abdominal perfusion in premature newborns. Our findings show that the frequency of feeding cues was higher in premature newborns in the IG to whom BM odor was applied compared with newborns in the CG. It is known that newborns react behaviorally to the smell of BM (Loos et al., 2019), exhibit odor-oriented behavior, when seeking reflex (Hym et al., 2021), and BM odor causes an increase in oral activity and helps with sucking (Khodagholi et al., 2018). Marlier and Schaal (2005) studied the behavioral effects of the smell of BM and formula milk on term newborns in their studies showing that oral activity including mouth opening, tongue out of the mouth, licking, and sucking hands increased in newborns who smelled BM, and the reaction to the smell of BM was also observed more frequently. Park and Im (2020) studied the effect of the smell of BM applied to premature newborns, eight times a day for 3 days and for 1 h in parallel with the feeding plan on physiological and behavioral conditions. They found that behavioral status scores were positively affected in the group that could smell BM compared to the CG. Hym et al. (2021) in their study to observe the crawling and searching behavior of newborns for the odor of BM, showed that the frequency of arm-leg movements and searching behavior defined as dizziness, was significantly higher in newborns exposed to BM odor from their mothers. However, further studies are needed to reveal the feeding cues in newborns provided with the odor of BM.
In our study, the transition times to oral feeding for newborns in the IG and CG were similar. Beker et al. (2017), reported that the application of BM flavor simultaneously with the odor of BM before each feeding to premature newborns does not have an effect on the transition time to oral feeding. In another study, odor stimulation with BM was started 5 min before the start of enteral feeding in premature newborns and stimulation was continued for a total of 10 min until 5 min after enteral feeding. It was found that the intervention did not affect the transition period of newborns to oral feeding (Lee, 2019). According to Yildiz et al. (2011), BM odor stimulation applied three times a day during enteral feeding to premature newborns until they start oral feeding, shortened the transition time to oral feeding by approximately 3 days. Similarly, Iranmanesh et al. (2015) reported that odor stimulation with BM applied to premature newborns during enteral feeding four times a day until they start full oral feeding, shortened the transition time to oral feeding by a further 10 days. The difference between the results of our study in terms of the transition time to oral feeding may be due to the disparity between the frequency of BM odor stimulation during the day and the total application time.
We observed that abdominal rSO2 levels were found to be significantly higher in premature newborns who were given odor stimulation with BM compared to the CG. However, it was observed that the rSO2 values of preterm infants in the IG and CG showed statistically significant differences according to the group and group*time interaction. There are limited studies examining the effect of BM odor on regional tissue perfusion in premature newborns. Previous studies have shown that the cerebral region was used as a regional rSO2 assessment (Aoyama et al., 2010; Muelbert et al., 2021), and there were no studies revealing the effect of BM odor on abdominal rSO2 level in premature newborns. Aoyama et al. (2010) investigated the effect of 30-s olfactory stimulation with BM and formula milk on cerebral rSO2 level in term newborns, whereby it was determined that the odor of BM significantly increased cerebral rSO2 levels by activating the orbitofrontal cortex compared with the odor of formula milk in newborns. This result shows that newborns can distinguish BM odor from formula milk and other odors. Cues such as smell, taste, image, and food expectation cause CP and generate hormonal and neural signals, thus preparing the GI system for the digestion and absorption of nutrients (Smeets et al., 2010). Stimuli to the sensory cortex with olfaction create many autonomic responses such as vagal activation and the release of digestive enzymes (Smeets et al., 2010). As a result, it is inevitable to see an increase in perfusion in the abdominal region where the digestive organs are located. Our study provides a novel and important finding whereby we are the first to show that exposure to the smell of BM leads to an increase in abdominal rSO2 levels.
In our study, no statistically significant difference was found between the daily weight gain of premature newborns in the IG and CG, which was corroborated by previous studies (Khodagholi et al., 2018; Yıldız et al., 2011). In addition, the number of daily defecations of premature newborns in the IG was significantly higher in every 3 days compared to the CG. There are currently no studies that investigate the effect of BM odor on the number of defecations in premature newborns. It is known that parasympathetic stimulation in the digestive system ensures the secretion of digestive enzymes, increases motility in the stomach and intestines, and accelerates peristalsis (Browning & Travagli, 2014). In addition, it has been reported that vagal activity, which causes autonomic responses in the digestive system, increases with olfactory stimulation (Smeets et al., 2010). Therefore, in this study, it is thought that the odor of BM increases parasympathetic activity and vagal tone in newborns, thus increasing gastric motility and facilitating defecation.
When the effect of odor stimulation with BM was correlated to the frequency of vomiting according to days was evaluated, we observed that while there was no difference in the first 2 days, there was statistically significant less vomiting in the IG on the third day. There are no studies that have observed our findings as none have evaluated the effects of BM odor on the frequency of vomiting in premature newborns. In a study by Lee (2019), applying odor stimulation with BM, 12 times for 15 days to premature newborns significantly reduced gastric residual volume. This can be interpreted as the smell of BM improving the digestive function of newborns, facilitating the digestion of food, and reducing the frequency of vomiting. It is thought that the decrease in frequency of vomiting in newborns who underwent intervention in the study is related to the mechanism of increased frequency of defecation.
Here, we determined that odor stimulation with BM did not create a statistical difference between the IG and CG in terms of discharge times. However, in two studies with similar methodology as this study, premature newborns who were administered BM odor were discharged earlier than those who did not (Iranmanesh et al., 2015; Yildiz et al., 2011).
Limitations
We determined that the enteral feeding period of newborns took an average of 10–30 min in direct proportion to the feeding volume. Considering that the intervention was performed during enteral feeding, the duration of the intervention could not be standardised because the normal flow of the enteral feeding period could not be interfered with, and this situation was considered as a limitation. Limiting the intervention to only three feeds a day for 3 days, and the inability to blind the researcher because the researcher participated in the implementation of the intervention and data collection process, can also be presented as a limitation of the study. Furthermore, we did not measure the amount of feeding that occurred, which would benefit the study and allow us to form relationships between amount of milk consumed with vomiting and defecation among other factors. The fact that the postnatal age at which the infants were enrolled in the study was not standardized for each infant can be said to be another limitation of the study. In addition, the size of the study sample was small and may have limited our ability to detect small significant differences.
Conclusion
In our study, we determined that olfactory stimulation applied with BM before and during enteral feeding in premature newborns increased the symptoms of hunger, increased the level of abdominal perfusion, decreased the frequency of vomiting by facilitating digestion, and increased the frequency of defecation. However, the odor of BM did not affect the transition time to oral feeding, daily weight gain, nor the discharge time. Our study is the first showing that exposure to BM odor causes an increase in abdominal rSO2 levels, which has important implications for improved feeding by newborns. We recommend BM odor stimulation be applied to newborns in clinics to stimulate the hunger of premature newborns, to encourage the development of feeding behaviors, and to improve their digestive functions. Our study findings suggest that the odor of BM is a useful intervention in improving the nutritional functions of premature newborns. For this reason, it is seen as a practical, economical, and safe practice for NICU nurses to include odor stimulation with BM in their care practices. Further studies are recommended to standardise the frequency of application of olfactory stimulation during the day, total application time, and to determine the effects on newborns. In addition, it is recommended to conduct studies with large sample groups in which nutrient volumes are considered and NIRS data are compared with other parameters such as oxygen saturation and cerebral oxygenation.
Footnotes
Acknowledgments
This article is extracted from a master’s thesis (thesis number: 10437074) conducted by Adalet Yucel. We would like to express our gratitude to Selcuk University Scientific Research Projects (project number: 20202017) for their financial support in the conduct of this study. The findings of this study were presented at the third International Mediterranean Pediatric Nursing Congress held in Ankara, Türkiye.
Author Contributions
Yucel, A contributed to conception and design contributed to acquisition, analysis, and interpretation drafted manuscript critically revised manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy. Kucukoglu, S contributed to conception and design contributed to acquisition, analysis, and interpretation drafted manuscript critically revised manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy. Soylu, H contributed to conception contributed to interpretation drafted manuscript critically revised manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Selçuk University Research Foundation (project number 20202017).
Ethical Approval
Prior to the study, ethics committee approval was obtained from the Non-Interventional Clinical Research Ethics Committee of the local state university with the decision number 2020/3, dated 06/07/20, and institutional permission was obtained from the hospital where the study was conducted. Before the newborns who fit the sampling inclusion criteria were admitted to the study, the necessary information about the study was presented to the parents. The Informed Consent Form was read by parents and their written permission was obtained before recruiting the PN for the study. The study is registered in ![]() (NCT04843293).
(NCT04843293).
