Abstract
Keywords
Multiple sclerosis (MS) is a chronic neurological disease thought to be triggered by an; immune-mediated response targeting elements of the central nervous system (CNS), including both the brain and spinal cord. It is estimated that approximately one million people in the U.S. have MS, with approximately 200 new cases diagnosed each week (Goodin et al., 2016; Wallin et al., 2019). Early disease course is variable and over 80% of people with MS suffer significant disability over time, with an estimated 50% of disability occurring in young adulthood (Zurawski et al., 2019).
Fatigue is the most common presenting symptom reported by people diagnosed with MS and is a longitudinal problem for many living with this condition. Fatigue occurs in about 80% of MS patients (National Multiple Sclerosis Society, 2021), and up to 38% of people with a recent diagnosis of MS report experiencing severe, debilitating levels of fatigue (Beckerman et al., 2020; Berger et al., 2013). Fatigue consequently impacts the experience of other symptoms and quality of life (QOL). The exact cause or causes of MS fatigue are yet unknown even after years of work. One theory is that brain networks are slowed during fatiguing tasks (Capone et al., 2019), while others suggest reduced brain activity in correlation with self-report fatigue (Miller et al., 2014). Potential pathophysiological mechanisms of fatigue in MS include: structural damage of white matter (WM) and grey matter (GM); inflammatory processes within or outside the CNS; maladaptive network response due to lesions or inflammation; and metacognitive interoception of dyshomeostatic states (Manjaly et al., 2019). Fatigue in MS can also be related to factors such as insomnia, sleep disturbance due to urge incontinence or spasticity, infections, thyroid disorders, physical inactivity and deconditioning, and depression (Beckerman et al., 2020).
In people with MS, fatigue is considered a multidimensional symptom, having motor, psychosocial, and cognitive limitations (Newland et al., 2016), and as such, assessment is complex (Beckerman et al., 2020). Assessment of fatigue is further complicated by the lack of common terminology across studies and/or unclear defined parameters (Kluger et al., 2013). Fatigue can be either subjective (perceived) or related to objective changes in performance (fatigability; Kluger et al., 2013).
Subjective (perceived) fatigue can be assessed at a specific moment in time, measured through visual-analogue scales (VAS) or numerical-rating scales (Linnhoff et al., 2019). Or it can be a “trait component,” meaning a person’s predisposition for fatigue over time, and evaluated through self-report questionnaires (Linnhoff et al., 2019). The self-report questionnaires most often used to assess the perceived impact of fatigue on those with MS include the Fatigue Severity Scale (FSS), the Fatigue Impact Scale (FIS), the Modified Fatigue Impact Scale (MFIS), the Fatigue Scale for Motor and Cognitive Functions (FSMC), the Patient Reported Outcomes Measurement Information System (PROMIS)-Fatigue (MS; Cella et al., 2016), and the Wuerzburg Fatigue Inventory for Multiple Sclerosis (WEIMuS; Cook et al., 2012; Iancheva et al., 2019; Linnhoff et al., 2019). Several of these scales evaluate distinct fatigue dimensions including physical, cognitive, and psychosocial functioning (Linnhoff et al., 2019).
Fatigue that is objective, or fatigability, refers to a change in function during a cognitive or motor task (Manjaly et al., 2019; Kluger et al., 2013). Several approaches are available to evaluate objective cognitive fatigue (i.e., operationalized as performance reduction with time-on-task) among adults. The first involves the subjects performing a test paradigm several times in a row, with a comparison of performance changes made to baseline. The second and third approaches involve a pre-to-post performance decline in a specific task (task A) while inducing fatigue by either mental or physical exertion with an in-between task BA fourth involves a sustained mental effort, with a comparison of beginning performance of the cognitively demanding task with an ending performance (Linnhoff et al., 2019).
Research is currently underway to evaluate diagnostic testing to objectively assess fatigue. One systematic review (Goñi et al., 2018) found the following diagnostic tests capable of identifying neural correlates of fatigue in individuals with chronic disease: structural and functional Magnetic Resonance Imaging (fMRI); diffusion tensor imaging (DTI); and functional connectivity (FC) techniques. Specific to MS, several studies have also supported the biological assessment of fatigue using fMRI (Iancheva et al., 2019; Tanasescu et al., 2014).
Electrophysiological Techniques for Assessing Fatigue
Electrophysiological techniques are recognized as effective diagnostic tools during identification and prognostication of fatigue in MS. For example, several studies have shown that mental fatigue can result in changes in electroencephalography (EEG) signal and event-related potentials (ERP; Li et al., 2020; Linnhoff et al., 2019). Non-invasive brain stimulation (NIBS) such as transcranial magnetic stimulation (TMS) and transcranial direct current stimulation (tDCS), which employ electrical or magnetically-induced currents to stimulate the brain through the scalp, can temporarily excite or inhibit activity in target brain regions (Liew et al., 2014). NIBS has been utilized as adjunctive neuro-rehabilitative therapy (Liew). One systematic review provided evidence that tDCS can mitigate the effects of MS-related fatigue, especially cognitive fatigue; however only one of the 8 included studies used both subjective (i.e., rating scale) and objective fatigue measures (i.e., decrements in cognitive performance; Ashrafi et al., 2020). More research is needed to examine the relationship between subjective, perceived fatigue, and objective electrophysiological measurements of fatigue in MS populations.
The difficultly differentiating types of fatigue, the lack of clear understanding regarding the pathophysiologic mechanisms of fatigue, and the limited research on the use of electrophysiological techniques and NIBS to quantify fatigue in MS present obstacles to the development of effective fatigue therapies. Objective measures linked to specific brain changes are important to improve scientific understanding of mechanisms of MS-related fatigue, provide sensitive and specific outcome measures, and to facilitate the design of precise interventions that have the potential to improve fatigue and QOL for people with MS. The use of electrophysiological assessment of fatigue using EEG and NIBS is more cost effective than MRIs and likely more readily available in clinical settings. Furthermore, these present attractive options to additionally explore as objective measures of fatigue. The purpose of this literature review article is to provide a synthesis of recent research focused on the use of 3 techniques, EEG, TMS, and tDCS used during MS-related fatigue.
Method
The reviewing team conducted an initial literature review following the methodology of the Cochrane Handbook for Systematic Reviews of Interventions (Higgins et al., 2022) and the reporting guidelines from the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA; Page et al., 2021) as shown in Figure 1. An experienced health science librarian (KS), in consultation with the original review team, conducted comprehensive searches in the Cumulative Index to Nursing and Allied Health Literature (CINAHL, EBSCOhost), MEDLINE (OVID), APA PsycInfo (OVID), Scopus (Elsevier), and Web of Science (Clarivate). The team determined this grouping of databases to be the optimal combination to identify all relevant studies addressing the research question. The initial search team terms identified by the review team included: MS, fatigue, and the utilization of TDCS, TMS, and/or EEG. Next, a search string was developed by the librarian and approved by the review team. The search string included the following keywords: (“multiple sclerosis” OR “multiple-sclerosis”) AND (fatigu* OR fatiga* OR dysfunt*) AND (“transcranial direct current stimulation” OR tdcs OR “transcranial magnetic stimulation” OR tms OR electroencephalogra* OR eeg OR “electrical brain stimulation” OR (stimulat* AND/NEAR/ADJ (“direct current” OR magnet* OR electric*) AND/NEAR/ADJ (brain OR transcranial). Unique search strategies including these keywords as well as controlled vocabulary appropriate to each database were developed to ensure full utilization of keywords and of pertinent controlled vocabulary terms. Publications were date limited to 2015 January 1 and after. PRISMA flow diagram. From: Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 2021; 372:n71. Doi: 10.1136/bmj.n71.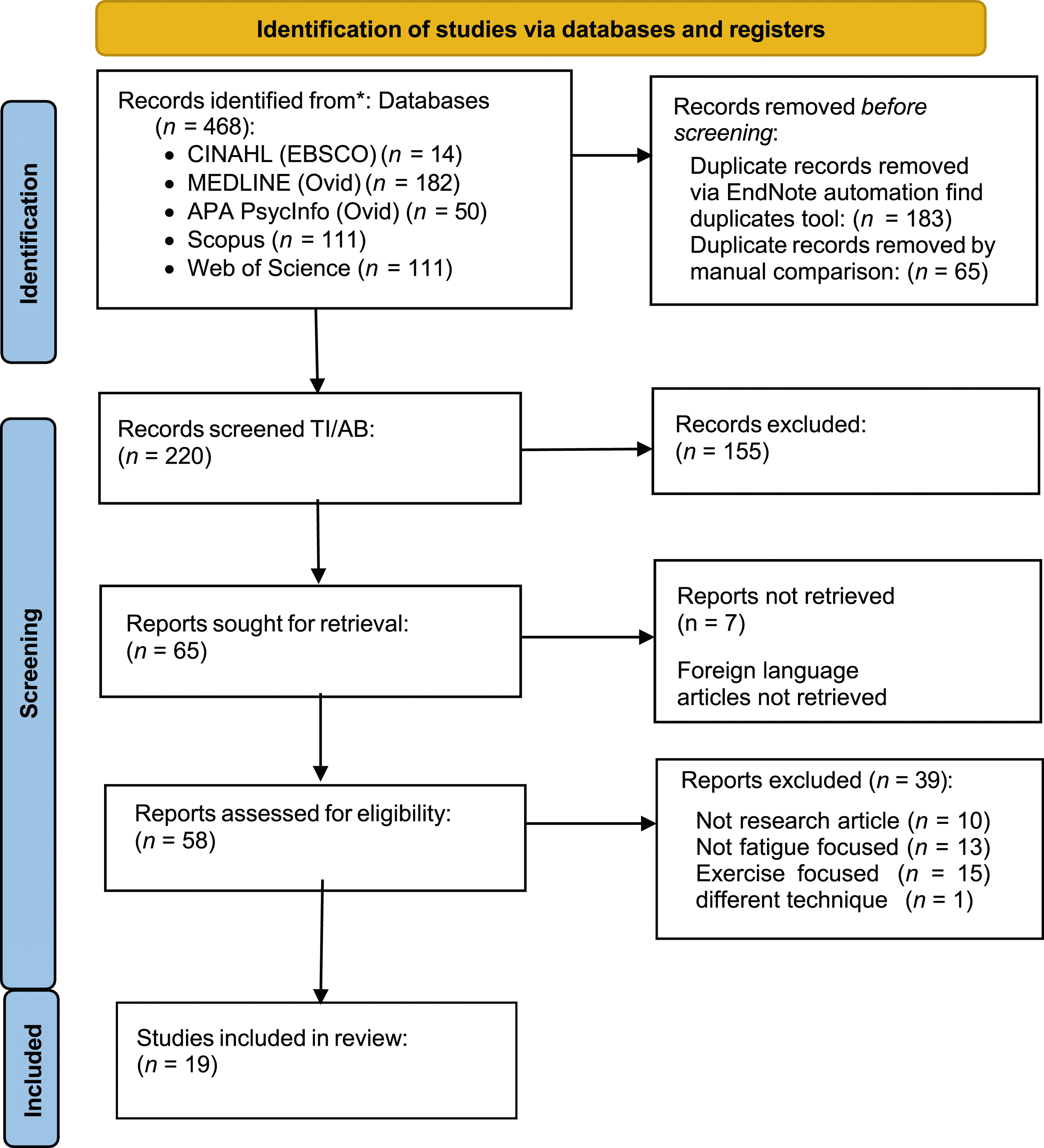
A total of 468 citations were found and exported into EndNote X9 citation managing software. The EndNote automatic tool was used to identify and to remove 183 duplicate citations. An additional manual citation screening by 2 reviewers (KH & KS) identified and removed an additional 65 duplicates. Two reviewers (KH & KS) then independently screened the remaining 220 unique citations by title/abstract for the inclusion and exclusion criteria. The TI/AB screening resulted in 65 citations retained as potentially relevant. Seven of the 65 articles were published in foreign languages; as there was no funding for translation services, they were not included. The remaining 58 articles were then retrieved and screened by 3 team members (PN, KH, & KS). Of the 58 articles, 39 were excluded at the article screening based on inclusion and exclusion criteria with 19 retained as meeting the inclusion criteria and relevance for full review.
Inclusion and Exclusion Criteria
The inclusion criteria of articles included in this literature review included research studies that used a randomized control trial (RCT); quasi-randomized, quasi-experimental, comparative control; or controlled before-and-after study design. Additionally, no restrictions were set for enrolled study participants’ MS diagnosis subtype, MS disease severity, level of MS disability, or demographic characteristics (e.g., gender, race, or ethnicity). Only research studies that identified fatigue as the primary study variable were included.
The set criteria of articles excluded in this literature review were published articles that included the following focus or format: (a) systematic reviews and meta-analyses; (b) expert opinion papers; (c) pharmaceutical and ongoing clinical trials; and (d) conference posters/abstracts. Additionally, published studies were excluded if the sample enrolled participants that were under 18 years of age.
Quality Check
Using the Johns Hopkins Guideline (Dearholt and Dang, 2012), 2 authors summarized components of each article including purpose, design, variables, subjects, measurement tools, and findings. Each author presented a matrix table via conference calls. Group consensus was obtained regarding the relevancy of each article. Once consensus was made, selected articles were graded based on the study’s design (e.g., experimental study, RCT); validity of fatigue questionnaires, and sample size. Studies were scored using a Good/Fair/Poor quality guide (Dearholt and Dang, 2012).
Results
Summaries of Studies (N = 19).
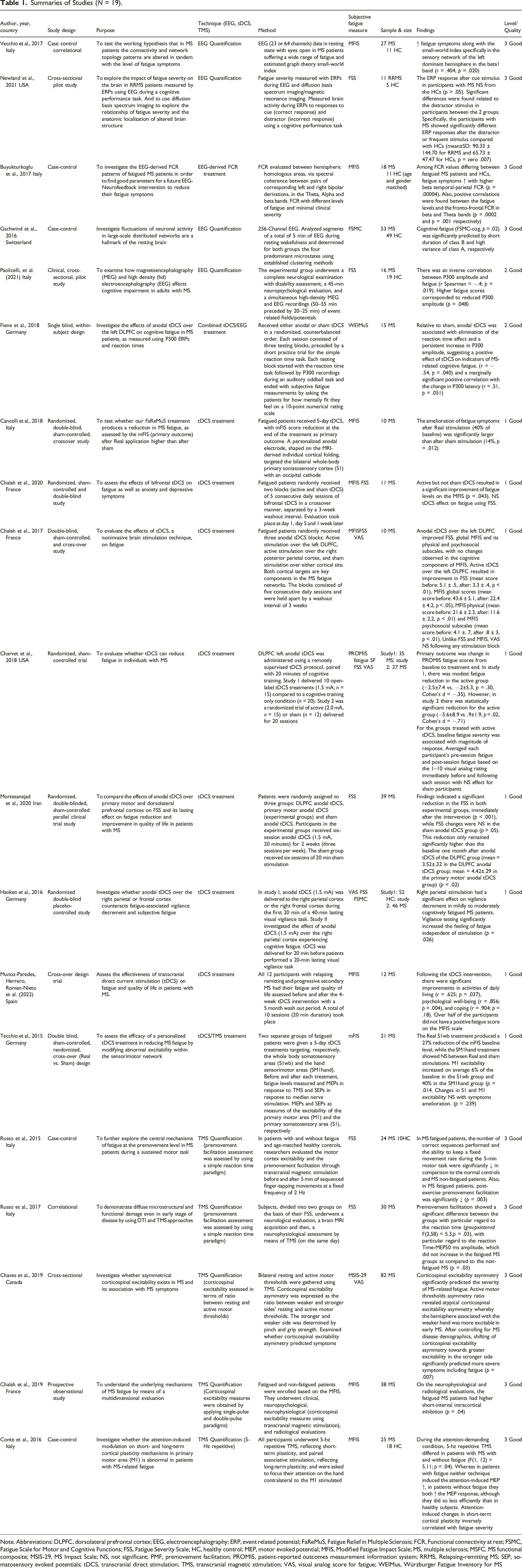
Note. Abbreviations: DLPFC, dorsolateral prefrontal cortex; EEG, electroencephalography; ERP, event related potential; FaReMuS, Fatigue Relief in Multiple Sclerosis; FCR, Functional connectivity at rest; FSMC, Fatigue Scale for Motor and Cognitive Functions; FSS, Fatigue Severity Scale; HC, healthy control; MEP, motor evoked potential; MFIS, Modified Fatigue Impact Scale; MS, multiple sclerosis; MSFC, MS functional composite; MSIS-29, MS Impact Scale; NS, not significant; PMF, premovement facilitation; PROMIS, patient-reported outcomes measurement information system; RRMS, Relapsing-remitting MS; SEP, somatosensory evoked potentials; tDCS, transcranial direct stimulation; TMS, transcranial magnetic stimulation; VAS, visual analog score for fatigue; WEIMus, Würzburger Fatigue Inventory for MS
Among the 19 studies, MFIS and FSS were the most widely used scales to assess subjective fatigue (i.e., 10 studies employed one of these 2 measures). The VAS was used in 4 studies. The FSMC was used in 2 studies. The MSIS-29, the WEIMuS, and the PROMIS Fatigue SF were each used in one study. Five studies measured fatigue using 2 or more of the scales. Additionally, among the 19 studies, investigators included other diagnostic tests. In summary these included: 7 used tDCS, 5 used EEG, 5 used TMS, one used an EEG/tDCS, and one included a combined tDCS/TMS.
EEG Studies
We identified 5 EEG studies (Buyukturkoglu et al., 2017; Gschwind et al., 2016; Newland et al., 2021; Paolicelli et al., 2021; Vecchio et al., 2017). Three of these were case–control studies (Buyukturkoglu et al., 2017; Gschwind et al.; Vecchio et al.) which collected and analyzed EEG data in fatigued MS patients, including functional connectivity at rest (FCR) between hemispheric homologous areas, and performed correlational analysis which revealed a relationship between cognitive fatigue and EEG data. Buyukturkoglu et al. found that fatigue symptoms increased with higher Beta temporo-parietal FCR, as well as a positive correlation between fatigue levels and the fronto-frontal FCR in Beta and Theta bands. Additionally, Gschwind et al. found that cognitive fatigue was significantly predicted. Vecchio et al. analysis showed increased fatigue symptoms along with the small-world index specifically in the sensory network of the left dominant hemisphere in the beta 1 band. Two cross-sectional pilot (Newland et al.) assessed event-related potentials (ERPs) during EEG using a cognitive performance task. Although the ERP response after cue stimulus in MS patients with fatigue did not differ significantly from healthy controls, there were significant differences related to the distractor stimulus and ERP amplitude among participants between the 2 groups.
tDCS Studies
Seven studies (Cancelli et al., 2018; Chalah et al., 2017, 2020; Charvet et al., 2018; Hanken et al., 2016; Mortezanejad et al., 2020; Muñoz-Paredes et al., 2022) tested the effect of tDCS on cognitive fatigue. Among these 7 studies, 6 used a cross-over design and only one was an RCT (Muñoz-Paredes et al., 2022). Investigators in all 7 studies reported fatigue was significantly improved after tDCS. Four studies (Chalah et al., 2017; Charvet et al.; Mortezanejad et al.; Muñoz-Paredes et al., 2022), using either the MFIS and FSS, or both, examined the intervention in the dorsolateral prefrontal cortex (DLPFC). One study that used a cross-over study design did not include randomization procedures (Muñoz-Paredes et al., 2022). Cancelli et al. examined the primary somatosensory cortex using a tCDS-designed stimulation treatment protocol. Chalah et al. assessed effects of bifrontal tDCS. Hanken et al. examined the right parietal or the right frontal cortex.
TMS Studies
Five of the 19 studies reviewed examined TMS and physical and cognitive fatigue correlates using various techniques of pre-motor movement (Chalah et al., 2019; Chaves et al., 2019; Conte et al., 2016; Russo et al., 2015, 2017, 2017). Investigators in the majority of these 5 studies (n = 3) provided evidence that pre-movement facilitation confirms that fatigue is associated with a disruption of brain networks involved in frontal-thalamic microstructural dysfunction of cortical and sub-cortical motor areas involved in movement preparation and execution. Among the 5 studies, fatigue symptoms were commonly evaluated using one of 2 measures (i.e., MFIS or FSS). In contrast, Chaves et al. used VAS and MSIS-29 scales with heat sensitivity and finger exercises to assess whether asymmetrical corticospinal excitability exists. Chaves et al. study results revealed weaker hand movement in fatigued MS subjects and that corticospinal excitability significantly predicted the severity of MS-related fatigue. In summary, these studies found an association with fatigue and disruption of brain networks that can be important for fatigue management.
Combined Studies
Two studies included combined tDCS and EEG techniques. In the study by Fiene et al., 2018, the investigators used a combined tDCS/EEG method to assess cognitive fatigue using the WEIMuS scale in patients diagnosed with MS patients. Fiene et al. study results provided evidence that anodal tDCS was associated with decreased reaction time effect and that there was a persistent increase in P300 amplitude. This suggests that tDCS stimulation may have lowered cognitive fatigue. In another study (Tecchio et al., 2015), the investigators used a combination of tDCS/TMS to assess the efficacy of a personalized tDCS treatment in reducing MS fatigue by altering abnormal excitability within the sensorimotor network. Using the mFIS fatigue scale, the investigators provided evidence that whole body somatosensory areas targeted by tDCS reduced fatigue levels in adults diagnosed with MS.
Discussion
Review of these studies support the notion that subjective MS-fatigue can be identified and assessed by electrophysiological techniques including EEG, tDCS, TMS, and/or a combination thereof. The number of studies we identified as pertinent to this review were small, and they used a variety of electrophysiological techniques as well as study designs and subjective fatigue measures, which makes comparison difficult. The purpose of the studies also widely varied; some studies such as Tecchio et al. (2015) and Hanken et al. (2016) assessed the efficacy of tDCS treatment on fatigue, as opposed to other investigators. Charvet et al. (2018) and Fienne et al. (2018) attempted to explore patterns and results of electrophysiological testing during cognitive performance tasks; this was more often done in the EEG studies (Newland et al., 2021). Our conclusions from this review would have been strengthened if the investigators: (a) used the same cognitive fatigue scale, such as the PROMIS Fatigue (MS) which has established content validity and robust psychometric properties that have been shown in recent research trials (Kamudoni et al., 2021); (b) used an RCT study design; and (c) had a similar purpose. It would also be interesting for researchers to employ all 3 types of electrophysiological testing within the same study design, and/or to use one specific testing alongside MRI techniques. Additionally, correlating self-report and physiological scale measures may also have value. In terms of this paper’s purpose, however, the studies reviewed collectively support the use of EEG, TMS, and tDCS to quantify and measure cognitive fatigue in persons with MS.
Our results may not be generalizable due to heterogeneity of the MS groups (subtype, age, gender, socioeconomics of the participants, and medications not being controlled). Various fatigue scales were used, although the strengths of the MFIS, FSMC, WEIMuS compared to the MSIS-21 and FSS are that the former have subscales specific to cognitive fatigue. In contrast, the FSS contains questions more focused on motor functioning (Beckerman et al., 2020; Krupp et al., 1989). The use of different self-report fatigue scales across the included studies makes it difficult to draw comparisons between cognitive and physical fatigue. Additionally, the varied study purposes (e.g., exploration vs. efficacy) used by investigators in these studies impedes making proposed generalization of the study results to other populations. Most of the studies included small sample sizes (i.e., less than 50 participants), which possibly underpowered the results. There is a need for investigators to plan and implement well designed studies (e.g., RCT, sample sizes based on a power analysis, near equal demographic representation) to support similar results if replicated by other investigators (Conde et al., 2019).
Future Directions and Conclusion
Techniques to measure fatigue in persons with MS require substantial further investigation and development. In this paper we do not seek to examine the best treatment practice for fatigue but to investigate the use of each technique as a possible biomarker using fatigue as the symptom. The common disrupted brain networks in this review using various techniques suggests a method to use for fatigue measurement. Several studies identified through this literature review were limited in number and widely varying in their measurement techniques. Even though a majority of studies were RCTs, some did not use control groups or collect data based on fatigue levels. Thus, our review does not allow for pathophysiological causality of fatigue to be established. To date, there have not been any studies that identify which of the 3 methods is better. More diverse studies are needed to understand the ways in which these techniques can be used for fatigue management. More RCTs and longitudinal studies with consistent procedures (e.g., objective measures and timing of collection) are needed in order to comprehensively assess fatigue in MS patients. Upcoming studies might benefit from adding additional time points after each condition (active vs. sham) to determine the optimal washout intervals. Furthermore, the clinical assessment in this work mostly relied on self-administered scales which are subjective in nature. Objective biomarkers (e.g., neurophysiological, or functional neuroimaging variables such as resting-state EEG) are warranted for future studies (Chalah and Ayache, 2019).
Self-reported fatigue alone has not proved useful as a reliable measure for clinical and mechanistic studies in persons with MS (Allali et al., 2018). This is especially true due to the lack of a homogeneous fatigue tool. There is a wide degree of variation in how the different self-report measures assess fatigue, and the quality, validity, psychometric properties, and reliability of these instruments also varies. Nurses should be aware of the strengths and weaknesses of the instruments they choose to use.
Fatigue is a common symptom of MS and identification of an evidence-based protocol that is useful in clinical practice to identify persons at risk is key. It is important for nurses caring for persons with MS to be aware of the noninvasive techniques available to corelate with fatigue and “picture” the brain (Cordani et al., 2020). Our review provides guidance to nurses about promising physiological approaches which with further study and development may be able to better quantify and accurately measures fatigue in persons with MS. For nurses in the practice setting, knowledge of these developing electrophysiological techniques could contribute to identification, assessment, and prognosis of fatigue in persons with MS. This can expand their repertoire and ability to educate and advise in this area about this and also about the benefits and limitations of currently available subjective assessment approaches. It can also prompt them to be watchful for future developments in this area as objective assessments eventually move into the practice environment over time
Looking to the future, well-developed, studied, and validated non-invasive physiological assessments of fatigue may become part of standard clinical practice, just as MRI approaches are now for the diagnosis and management of MS. In this future, the clinical nursing assessment of fatigue could foreseeably include the direct use of physiological assessments which could compliment or replace currently available validated self-report measures in the clinical care of fatigue in people with MS. Nurses are in a critically important position to educate and guide people with MS concerning the use of these approaches, the interpretation and clinical significance of the results to treatment. Given the lack of an established dominant approach that is available for clinical use, nurses can guide patients and families by educating about the characteristics and potential benefits of each approach and advising that these are still being developed and studied in research settings. Finally, nurses are instrumental in participating in the research and development of these approaches and in their translation and integration into clinical practice, as educators, as research recruiters and facilitators, as implementors and translators, and as scientists and investigators
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author Contributions
Oliver, Brant J contributed to design contributed to interpretation critically revised manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy Winn, Karen contributed to conception and design contributed to acquisition, analysis, and interpretation drafted manuscript critically revised manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy Newland, Pamela contributed to conception and design contributed to acquisition, analysis, and interpretation drafted manuscript critically revised manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy Hendricks-Ferguson, Verna contributed to design contributed to analysis and interpretation drafted manuscript critically revised manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy. Hamilton, Karlie contributed to conception and design contributed to acquisition, analysis, and interpretation drafted manuscript critically revised manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy. Smith, Katy contributed to design contributed to acquisition and analysis drafted manuscript critically revised manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy.
