Abstract
Aim
To compare pain and stress response through cortisol and DHEA levels, implementing. Total intra venous anesthesia (TIVA) versus general anesthesia with volatile anesthetics (VOL).
Design
This is a prospective randomized correlation study with consecutive measurements.
Methods
In an ambulatory surgical center from October of 2019 to August of 2020, patients who underwent breast reconstruction with autologous fat grafting were randomized into 2 groups. Patients in the TIVA group (
Findings
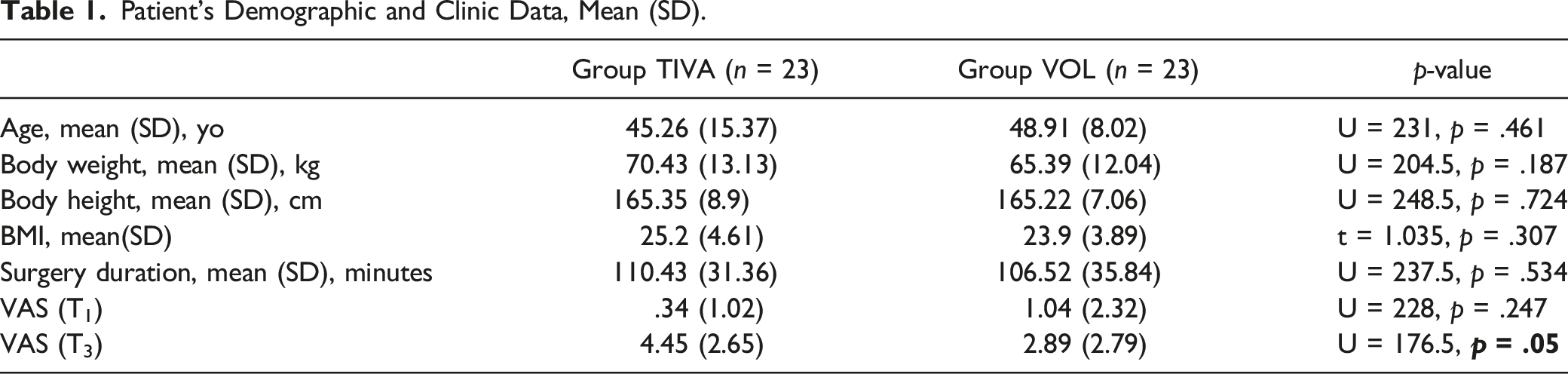
There were no statistically significant differences between the 2 groups regarding demographic features. Interestingly that there was a statistically significant difference in the vital sign monitoring where patients in the TIVA group reported with higher levels of SP(T2) and DP (T2), whilst DHEA (T1) levels was correlated positively with patient’s age and cortisol (T1) levels and negatively with DP (T3).
Conclusions
This study supports the use of TIVA as a safe and effective option for anesthesia in patients undergoing breast reconstruction with autologous fat grafting.
Keywords
Introduction
Breast reconstruction with autologous fat grafting has been associated with reduction of perioperative stress and pain levels in oncological patients (Geers et al., 2018; Turner et al., 2020). Promising results of new techniques show better control of patients’ perioperative pain and stress in clinical practice. In recent years, such techniques as the use of anesthetic agents in combination with the management of acute pain, the evolution of patient monitoring systems, the ongoing training of healthcare professionals, and the improvement of intraoperative care, have contributed to providing safer anesthesia in ambulatory surgical centers. In the Total IntraVenous Anesthesia (TIVA) technique, intravenous anesthetic agents are administrated for inducing and maintaining general anesthesia avoiding the administration of inhalation agents. While minimally invasive, surgical techniques result in the depletion of tissue damage and postoperative pain levels while reducing costs simultaneously. Ambulatory centers have been established in developed countries, ensuring shorter postoperative hospitalization, efficiency during surgery, and safe short-term anesthesia for all patients (Biricik et al., 2018).
Moreover, pain relief and the stress response in surgery are essential for patients’ safety post-recovery. TIVA was associated with a lower rate of postoperative nausea and vomiting (PONV) than the implementation of volatile anesthetics (desflurane). PONV may lead to immediate postoperative complications and delay same day discharge. TIVA is safe and effective in patients undergoing general anesthesia (Teunkens et al., 2017).
The hypothalamus-pituitary-adrenal (HPA) axis is activated during the perioperative stress response, resulting in the release of biomolecules such as ACTH and cortisol (Manou-Stathopoulou et al., 2019). However, early studies demonstrate the release of other biomolecules which are being triggered by surgical stress. The adrenal androgenic steroid dehydroepiandrosterone (DHEA) hormone has been noted to be affected by stress response (Osorio et al., 2002). Activation of the HPA axis leads to the ACTH secretion, which activates the adenylyl cyclase in the adrenal cells. As a consequence, enzymatic steps begin, leading to cortisol and DHEA biosynthesis (Kamin & Kertes, 2017). DHEA levels have been shown to be decreased over aging due to adrenopause (Samaras et al., 2013). Cortisol levels increase during surgical procedures and lead to an increase in DHEA levels (El-Sibai et al., 2017). In comparison to plasma cortisol levels, salivary cortisol is considered a reliable biomarker. Meanwhile, behavioral studies have demonstrated that salivary hormone monitoring offers well-established advantages over serum hormone testing (Malamud & Tabak, n. d.).
To the best of our knowledge, this is the first randomized trial to assess cortisol and DHEA levels in patients undergoing breast reconstruction with autologous fat grafting in an ambulatory surgical center. We aimed to compare pain and stress responses measured by cortisol and DHEA levels, implementing TIVA versus general anesthesia with volatile anesthetics (VOL) through perioperative hospitalization of patients.
Methods
Research Design and Setting
The research study was a prospective randomized correlational trial conducted between October 2019 to August 2020 in the Ambulatory Surgical Center at the Hellenic Anticancer Hospital. To assure stability of the research results, the breast reconstruction with autologous fat grafting surgery was conducted by the same plastic and reconstructive surgeons. Magellan System (Isto Biologics, Massachusetts, USA), which contains 3–10 mL of plated rich plasma was used (Medisports, Athens, Greece). The data collection and recording were done by the principal investigator.
Ethical Considerations
The research was approved by the Ethical Committee of the Faculty of Nursing of the National and Kapodistrian University of Athens, Greece (approval number: 2834/16-07–2019) and the local Scientific Committee of the Hospital, where it was carried out (approval number: 20467/10-09–2019). The Registration Number of clinicaltrials.gov is 2834/NCT04077827. In order to participate in the protocol, informed consent was given and signed by all patients. Confidentiality of information throughout the collection and processing of the data was prioritized.
Study Sample
Patients who have undergone mastectomy for breast cancer previously were eligible for breast reconstruction with autologous fat grafting in an ambulatory center. Inclusion criteria were adult patients 65 years of age or younger with ASA score I–III and Mallampati I–III classification. Normal level of consciousness and communication was required. A body mass index (BMI) below 40 kg/m2 was required. Patients with chronic pain, kidney and/or liver disease were excluded from the study.
Participants were randomized into 2 groups using the random sampling tool from SPSS 25.0 (IMB SPSS Software, Chicago, Illinois) software. In the TIVA group ( Flow-chart of participants allocation.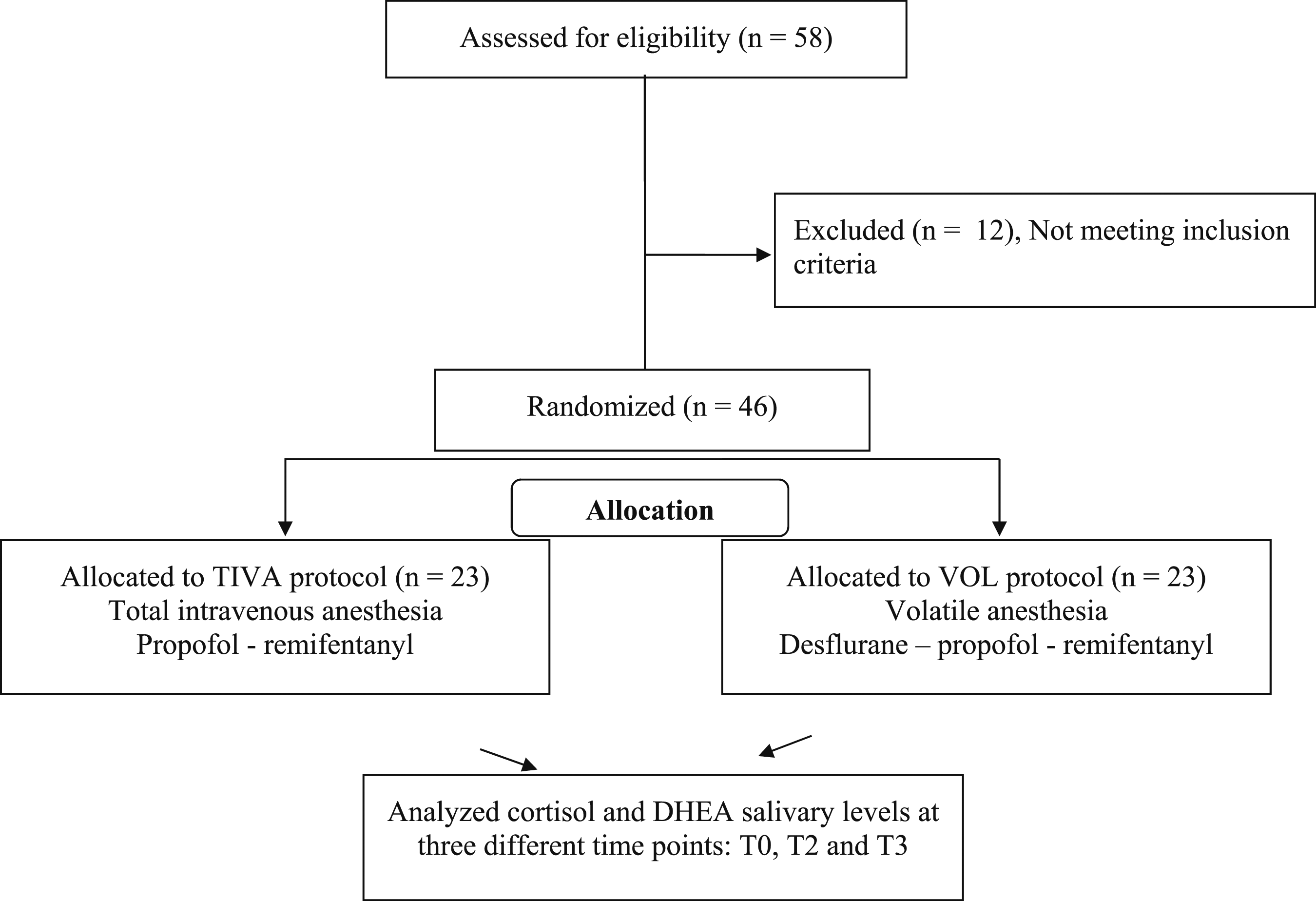
Data Collection
Demographic, anthropometric, and clinical data were recorded. Specifically, patients’ age and gender, as well as height, body weight, and body mass index (BMI) were documented. Furthermore, arterial systolic (SP) and diastolic (DP) blood pressure, heart rate (HR), and oxygen peripheral saturation (SpO2) were measured at three different time points: T1) during induction, T2) during anesthesia maintenance, and T3) in the Post Anesthesia Care Unit (PACU). Patients’ vital signs, duration of surgery and immediate post-op complications, such as postoperative shivering, hemorrhage and PONV. The fentanyl, remifentanil, and propofol doses were also reported.
Stress and Pain Evaluation Tools
Self-reported pain and stress levels were measured at 4 different time points: T0) 1 hour before induction, T1) during the induction, T2) during anesthesia maintenance and T3) in the Post Anesthesia Care Unit (PACU) recovery period. Pain evaluation was assessed using the Visual Analogue Scale (VAS 0-10) at T0 and T3 time points. VAS ranges from 0 to 10, where 0 referred to “no pain” and 10 to the “worst pain” that patient has experienced. Pain levels at time point T2 were measured with the Critical Care Pain Observation Tool (CPOT) for patients under anesthesia, which ranges from 0 (no pain) to 8 (maximum pain), assuring that intubated patients had no pain during general anesthesia (Gélinas et al., 2011). Patients’ perioperative stress response was estimated by 3 Numeric Analogue Scales (NAS: 0–10) questions at T0 and T3 time points. The scales range from 0: not at all to 10: very much, specifically “How sad do you feel right now from 0 to 10?” “How stressed do you feel right now from 0 to 10?” “How optimistic do you feel right now, about the future from 0 to 10?” (Kapritsou et al., 2017; Mpouzika et al., 2013). The same investigator collected the pain and stress response data, prior to acquiring the saliva samples for evaluating Cortisol and DHEA levels.
Evaluation of Patient’s Sedation and Depth of General Anesthesia
Patient’s sedation was assessed by the Richmond Agitation- Sedation Scale (RASS) and the Glasgow Coma Scale (GCS). The RASS was used at time point 2 (T2). It ranges from “+4, (combative)” to “-5, (unarousable)” (Sessler et al., 2002). The GCS scale was assessed at the time points T3 and T4 (T4: at the time that patients were discharged from PACU). It ranges from “3; indicating deep unconsciousness” to “15; fully awake patient” (Teasdale et al., 2014; Teasdale & Jennett, 1974). The depth of anesthesia was evaluated using the Bispectral Index (BIS), which estimates the cerebral electrical activity in anesthetized patients. BIS rates were measured at T1 and T2 and ranged between “0: flat line” and “100: awake status”. Lower scores indicate deeper anesthesia, a typical BIS score for a sedated patient is between 40–60 and is 90–100 in awakening patients (Gaowei et al., 2018).
Assessment of DHEA and Cortisol Salivary Levels
Salivary samples were collected at 3 different time points: T0) 1 hour before induction, because the fluctuations of cortisol and DHEA levels in 1 hour are not such as to affect the outcome after surgery (Azmi et al., 2021; Pelewicz et al., 2020; Upasham & Prasad, 2020); T2) during anesthesia maintenance; and T3) in the recovery period, during patients’ hospitalization in the PACU. Samples were centrifuged at 3000 r/pm for 20 minutes and were stored at −80°C. DHEA (pg/ml) and cortisol (ng/ml) salivary levels were quantified by enzyme-linked immunosorbent assay (ELISA) kits (DRG International, Inc., Germany).
DRG® Salivary Cortisol ELISA assay range was 0–80 ng/mL, while intra-assay variation was determined by repeated measurements from 4 saliva samples using the DRG ELISA kit.”; the intra-assay variation in the 4 tests was 4.52 (.12), .94 (.042), 12.79 (.23) and 17.50 (.258), respectively. Inter-assay (between-run) variation was determined by measuring commercial control samples 4 times over 3 days. Assay linearity was based on three samples (saliva) containing different amounts of analyte, serially diluted 1:64 with zero standard and tested by DRG ELISA. The linearity of the assay is .537–77 ng/mL (BioCheck, Inc).
The assay range for DRG® Saliva DHEA was 0–1440 pg/mL and the intra-assay variability was 66.41 (4.4), 318.59 (18.07), 150.44 (3.47) and 31.36 (2.51), based on repeated measurements of 4 saliva samples, respectively. Inter-assay (between-study) variation was determined by measuring 5 saliva samples in duplicate over 10 runs. Three saliva samples containing different amounts of analytes were serially diluted with zero standard and tested in DRG ELISA (BioCheck, Inc).
Statistical Analysis
Statistical analysis was carried out by SPSS 25 (IMB SPSS Software, Chicago, Illinois) and the mean standard deviation (SD) of variable values is reported. The use of the Kolmogorov-Smirnov test of Normality resulted in the use of independent samples tests, such as the
Results
Among 58 patients who were assessed against eligibility criteria, 46 patients were included in the study (2 males and 44 females). Participants underwent breast reconstruction with autologous fat grafting and were randomized into 2 groups. The TIVA group (
Patient’s Demographic and Clinic Data, Mean (SD).
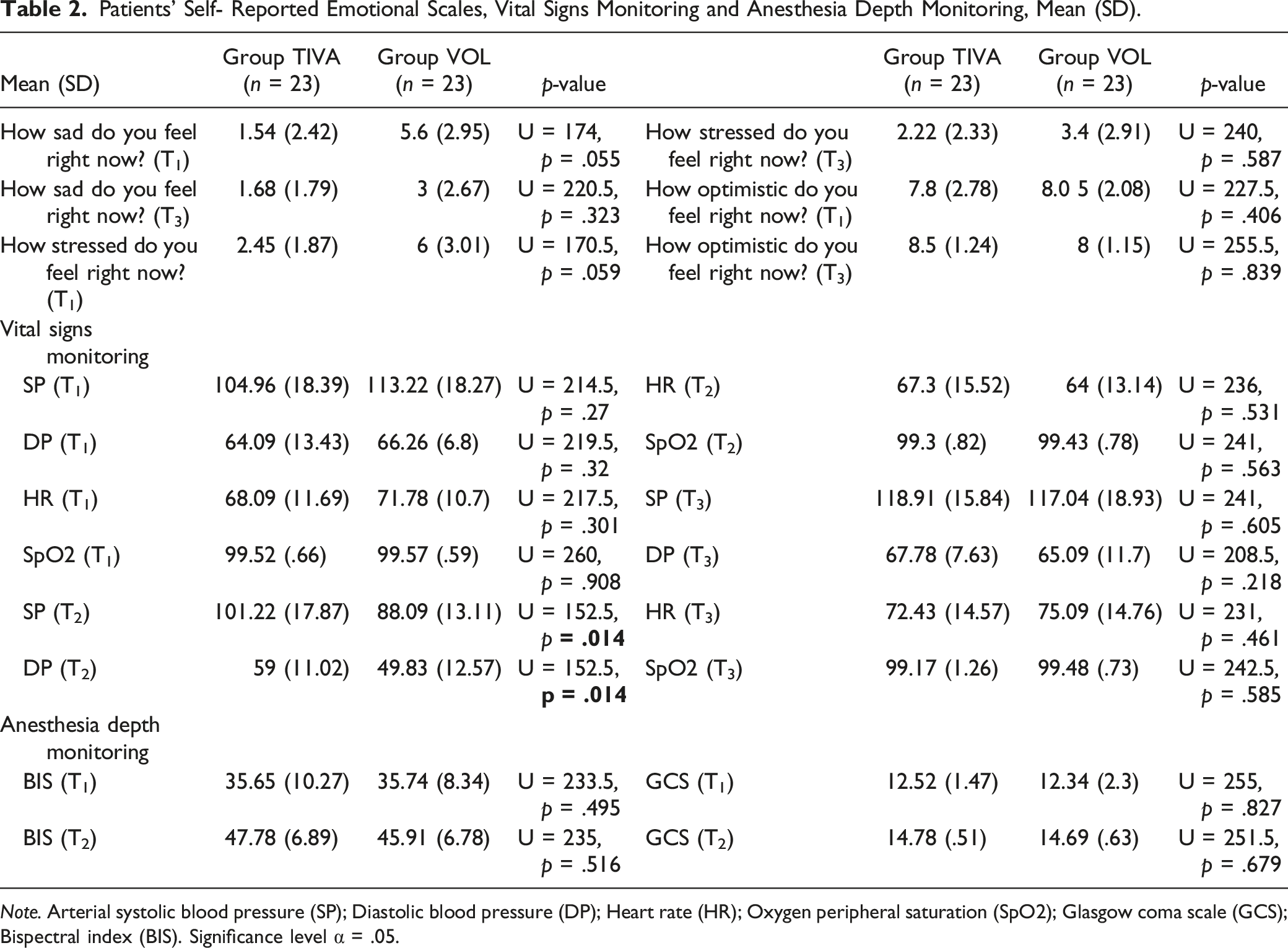
Patients’ Self- Reported Emotional Scales, Vital Signs Monitoring and Anesthesia Depth Monitoring, Mean (SD).
Assessment of Pain and Stress Levels
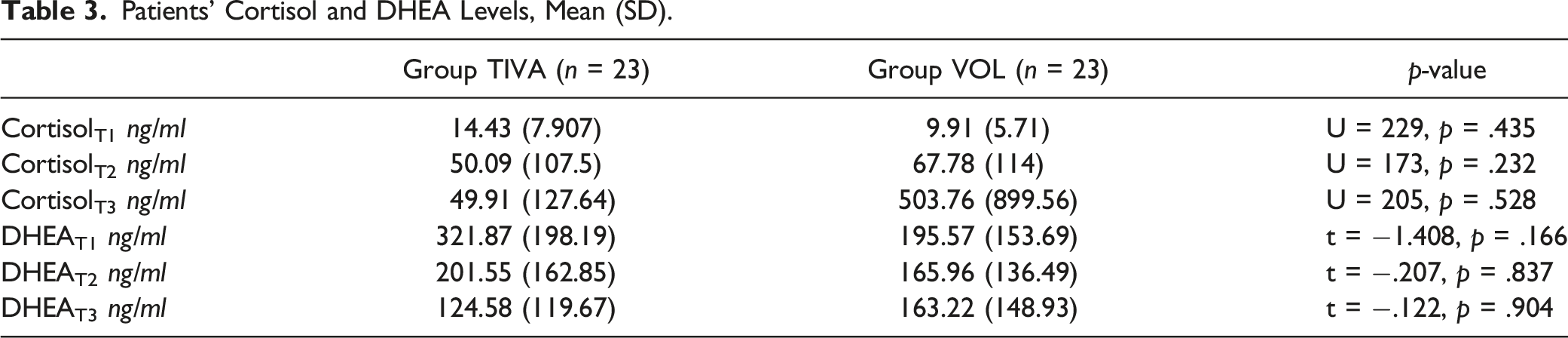
Patients’ Cortisol and DHEA Levels, Mean (SD).
Additionally, patients in the TIVA group reported higher pain levels at time point T3 on the VAS scale. Patients in both groups reported level 0 and 4 pain levels on the CPOT and RASS scales at time point T2, respectively.
In the TIVA group, there was a significant reduction in cortisol salivary levels in T2 compared to preoperative levels (z = −.217,
Bivariate Correlations Among Cortisol and Dhea Salivary Levels, Vital Signs and Anesthesia Depth Monitoring, Pharmaceutical Doses, and Stress and Pain Levels
Bivariate Correlations in TIVA and VOL Groups.

Note. *Significant level α = .01; Arterial systolic blood pressure (SP); Diastolic blood pressure (DP); Heart rate (HR); Desflurane (DES); Bispectral index (BIS).
Mixed Linear Models for Cortisol and DHEA Salivary Levels
Correlations of Cortisol and DHEA Salivary Levels.
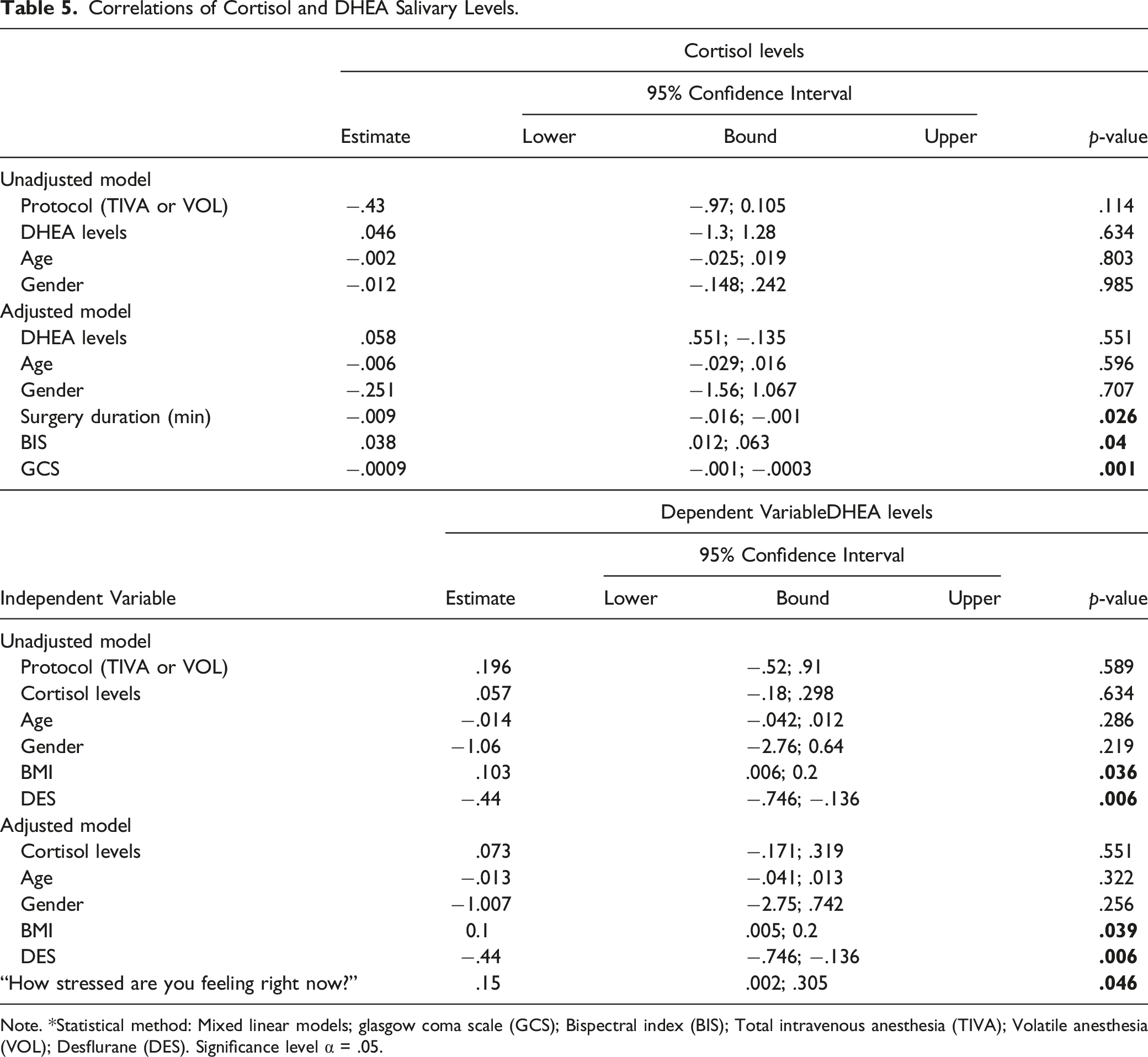
Note. *Statistical method: Mixed linear models; glasgow coma scale (GCS); Bispectral index (BIS); Total intravenous anesthesia (TIVA); Volatile anesthesia (VOL); Desflurane (DES). Significance level α = .05.
Discussion
This prospective cross-sectional study investigated HPA axis changes in patients following the TIVA protocol and general anesthesia with volatile anesthetics. Our findings revealed that older patients in the TIVA group experienced higher levels of preoperative DHEA levels. Patients who followed the TIVA protocol and had higher levels of preoperative cortisol levels appeared to have higher HR, DP, and pain levels in the PACU phase. Patients in the VOL group had higher levels of DHEA peri and postoperatively, respectively, as well as higher BIS rates, SP and DP perioperatively. In general, the study indicated that after implementation of the 2 protocols, increased stress levels led to increased DHEA levels. Patients with higher BMI were noted to have higher levels of DHEA levels, possibly affectively postoperative recovery.
In the present study, we observed that patients in the TIVA group had a decrease in DHEA levels perioperatively. This may be due to patients’ sedation after administering propofol or opioids. Lower DHEA and cortisol levels result in negative mood factors such as fatigue, depression, and anger. In contrast, our findings suggested that in the VOL, higher DHEA levels resulted in higher levels of sadness. While higher doses of propofol and remifentanil have been associated with lower stress levels (Brorsson et al., 2014), patients in the VOL group had lower propofol doses and experienced higher stress and sadness levels. This may have resulted in hemodynamic instability.
Furthermore, DHEA levels have been shown to decline with aging. By the age of 60–80 years old, the DHEA levels are 10-times lower than levels of people aged 20–30 years (Klinge et al., 2018). Previous studies have highlighted the negative correlation between DHEA levels and age in humans. In animal models, DHEA levels have been associated with shorter life span (Rutkowski et al., 2014). In our study, results are in line with current literature that DHEA levels are lower in older patients.
DHEA levels were positively correlated with BMI in the present study. However, these results are contradictory to published studies (Al-Harithy, 2003; Reyes-Muñoz et al., 2016). Investigators have highlighted that high BMI has been associated with lower DHEA levels, especially in young obese females (Al-Harithy, 2003). DHEA levels and their regulatory production mechanisms are uncertain in some cases, regarding to gender, obesity, and ethnicity (Reyes-Muñoz et al., 2016). It is of concern that patients in the present study had received chemotherapy and/or radiotherapy. In a recent study, patients who received chemotherapy reached lower DHEA levels, after treatment, especially women with breast and ovarian cancer. Probably, chemotherapy in combination with obesity affect estrogen’s metabolic path, consequently the DHEA production. Our findings that BMI was positively correlated with DHEA levels were consistent with the literature (Crean-Tate et al., 2020).
In animal models anesthetized with sevoflurane, researchers reported that treatment with DHEA supplements significantly decreased immobility time post-surgery (Moriguchi et al., 2013). However, lower anesthetic gas concentrations have been correlated with rapid recovery (Cicero et al., 2018). In the current study, patients hospitalized in day care clinics with high DHEA levels demanded lower DES doses, which led to faster recovery, allowing them to be discharged on the same day postoperatively.
Cortisol levels have been reported to be associated with BIS index values. In patients undergoing open-heart cardiac surgery, BIS index values were negatively correlated with cortisol levels, in contrast with our results, where cortisol levels were positively correlated with BIS index values (Plaschke et al., 2010). This difference may lie in the different types of surgery. Also, our results demonstrated high cortisol levels in patients that had markedly lower scores on the GCS scale. These findings contradicted recent research, which showed that critically ill trauma-patients (GCS mean 6) had significantly lower cortisol levels. One explanation could be that our patients were not in shock and sepsis (Kwok et al., 2020). TIVA protocols have been established for their efficacy and safety (Chan et al., 2016; Kim et al., 2020). Patients who participated in the TIVA protocol had higher SP, DP, HR than those in the VOL group who had more stable hemodynamic parameters. This may have been associated with the higher pain levels for patients in the TIVA group noted in the PACU phase.
We recognize the limitations of this study, including its single center nature as well as the limited number of cases. Consequently, no conclusions can be drawn, and future prospective studies are warranted.
Conclusions
In conclusion, both anesthesia types could be administered for patients with breast cancer undergoing breast reconstruction with autologous fat grafting. Patients who received TIVA anesthesia did not have higher levels of stress compared to VOL group. TIVA has shown to be safe and efficient for patients undergoing this kind of operation.
Footnotes
Author Contributions
Maria Kapritsou contributed to conception and design contributed to acquisition, analysis, and interpretation drafted manuscript critically revised manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy Evangelos Bozas contributed to conception and design contributed to acquisition, analysis, and interpretation drafted manuscript critically revised manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy Spyridon Stavrianos contributed to conception contributed to acquisition drafted manuscript critically revised manuscript gave final agrees to be accountable for all aspects of work ensuring integrity and accuracy Konstantinos Salatas contributed to conception and design contributed to acquisition and analysis drafted manuscript critically revised manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy Aris Plastiras contributed to design contributed to interpretation drafted manuscript critically revised manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy Konstantinos Pistolas contributed to conception contributed to acquisition drafted manuscript critically revised manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy Nikolaos Fyrfiris contributed to conception contributed to acquisition drafted manuscript critically revised manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy Maria Kotrotsiou contributed to conception contributed to acquisition drafted manuscript critically revised manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy Evangelos A. Konstantinou contributed to conception and design contributed to acquisition, analysis, and interpretation drafted manuscript critically revised manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Clinical Trials Registration
Clinicaltrials.gov; Registration Number: 2834/NCT04077827.
