Abstract
Case series summary
The medical records from 25 cats with feline plasma cell pododermatitis (PCP) and concurrent glomerular disease presenting between 2017 and 2025 were reviewed. The aim of this study was to highlight that feline patients with PCP may be predisposed to glomerular disease and to document the clinical presentations of these patients. Inclusion criteria included diagnosis of PCP by physical examination or histopathology and diagnosis of glomerular disease via renal histopathology or urine sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE). Data collected included signalment, physical examination findings, clinicopathologic parameters, imaging results, urinalysis, urine SDS-PAGE results, renal histopathology, treatment protocols and survival times. Affected cats were most commonly young (aged <5 years) and castrated males. Clinicopathologic abnormalities included marked anemia, hyperglobulinemia and proteinuria, although urine protein:creatinine ratios varied widely between patients. Renal ultrasound findings were typically non-specific. Urine SDS-PAGE consistently demonstrated evidence of glomerular dysfunction, and renal histopathology demonstrated immune complex glomerulonephritis in 14/17 (82%) cases. Immunosuppressive therapy was the primary treatment for renal disease but was often delayed owing to challenges diagnosing glomerular dysfunction as the underlying cause of renal disease. Median survival times after onset of renal disease were short, at approximately 1 month.
Relevance and novel information
Our findings suggest that PCP, particularly in young males, can be associated with concurrent glomerular disease, which is often immune mediated in nature. Urine SDS-PAGE may be useful in identifying underlying glomerular damage. Median survival times in these patients are short, emphasizing the need to closely monitor cats presenting with PCP for evidence of developing glomerular disease.
Keywords
Introduction
Feline plasma cell pododermatitis (PCP), or ‘pillow foot’, is reportedly an uncommon dermatologic disorder that causes swelling and potentially erosion of the paw pads secondary to infiltration by plasma cells.1 –5 The underlying cause of PCP is considered idiopathic or immune-mediated, as previous investigation of infectious causes failed to identify an etiologic agent. 6 Cats with PCP can have varied clinical presentations ranging from no clinical signs to severe ulceration of the paw pads causing lameness. Presumptive diagnosis is based on the patient’s history, clinical signs and gross appearance of paw pads demonstrating characteristic swelling and loss of metacarpal/tarsal pad definition, erythema, erosion and ulceration.6 –9 Diagnosis can be confirmed with cytology or histopathology demonstrating abundant plasma cells.1,7 Immune modulation via immunosuppression is the treatment of choice, although one study suggests the efficacy of doxycycline because of its immunomodulating properties. 10 Prognosis in these patients is generally considered good; however, variable responses to treatment have been noted. 1
Immune complex glomerulonephritis (ICGN) is a form of renal disease caused by antigen-antibody complex formation and deposition within the glomerulus. Deposition of complexes results in the disruption of the glomerular filtration barrier, proteinuria and severe renal disease.11,12 Feline ICGN has previously been of limited focus in the veterinary literature as it is less commonly reported than canine ICGN. However, recent studies have indicated that among cats with protein losing nephropathy, 74% had evidence of ICGN. 11 Furthermore, studies focused on feline ICGN have consistently demonstrated short median survival times in the range of 30–90 days.11,13 Interestingly, renal diseases such as ICGN and amyloidosis have been reported as rare sequela of PCP.1,2 Therefore, although PCP has a favorable prognosis, the development of concurrent renal disease represents a significant and life-threatening complication.
Currently, there are no studies focused on the prevalence or clinical features of cats with PCP and concurrent glomerular disease. Here, we report the clinical presentation, diagnostic findings and prognostic outcomes of 25 cats with concurrent PCP and glomerular disease presenting over 8 years. This case series aims to highlight PCP with concurrent glomerular disease in cats and to describe the clinical features that may help clinicians identify and treat these cases earlier.
Case series description
Case selection and data collection
The medical records of cats with both PCP and glomerular disease either presenting to the Ohio State University’s (OSU) Veterinary Medical Center or that received a clinical consult through the International Veterinary Renal Pathology Service (IVRPS) were reviewed. Identified cases were presented between 2017 and 2025. Cases were selected if the medical record included the following: (1) a working diagnosis of PCP via characteristic physical examination findings, photo documentation of paw pad lesions and/or histopathology; and (2) a diagnosis of glomerular disease via urine sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) and/or histopathology. Data collected from medical records included signalment, physical examination findings, bloodwork (complete blood count, serum biochemistry, feline leukemia virus [FeLV]/feline immunodeficiency virus [FIV] SNAP test), urine assessment (urinalysis and urine protein:creatinine [UPC] ratio), diagnostic imaging results (abdominal ultrasound), urine SDS-PAGE findings, biopsy or necropsy microscopic examination of renal tissues (hematoxylin and eosin, electron microscopy and immunofluorescence) and survival time (if applicable). Feline retroviral SNAP testing results were reported if testing was performed at the time of presentation for renal disease or within 1 year before presentation. For all other diagnostic testing, results were recorded from the first time the test was performed after identification of renal disease. Survival time was calculated from the time of renal disease diagnosis to time of death.
Results
A total of 49 cats with both putative pododermatitis and renal disease were identified. Of them, 11 cats did not have either urine SDS-PAGE or renal histopathology data, 12 cats had incomplete clinical data or did not have follow-up, and one cat had nephrolithiasis as the attributed cause of renal disease. In total, 25 cats, including 11 from OSU and 14 from the IVRPS, met all the inclusion criteria. The most common signalment was young (aged <5 years) male castrated cats (Table 1). Retroviral status was available for 18 cats, with 17 cats testing negative and one cat testing positive for both FIV and FeLV. Physical examination findings prompting a diagnosis of PCP were characteristic metacarpal/metatarsal pad lesions including swelling, loss of metatarsal/metacarpal pad definition, purple discoloration, fissures, and rarely ulceration and crusting (Figure 1). Of the 25 cases, nine had histopathology of the paws, eight of which were consistent with PCP. The remaining case had mild to moderate lymphoplasmacytic perivascular dermatitis but was not definitive for PCP, despite paw pad changes on initial physical examination. However, histopathology was performed at necropsy 120 days after immunosuppression was initiated.
Signalment information


(a–d) Normal paw pad (a) compared with characteristic gross appearance of feline plasma cell pododermatitis (b–d). Severity of paw pad lesions varies, with all cases demonstrating swelling and loss of metacarpal/tarsal definition and variations in degree of erythema, cracking, flaking and erosion. (e) Histopathology confirms severe plasma cell pododermatitis with too numerous to count plasma cells infiltrating and expanding the dermis of the paw pads, in some cases resulting in a mass-like effect (arrow heads). (f) High magnification of paw pad infiltrates demonstrates numerous plasma cells (arrowheads) admixed with scattered Mott cells (arrow) and lesser numbers of neutrophils and lymphocytes consistent with diagnosed plasma cell pododermatitis
On initial blood work, 23/25 cats had non-regenerative anemia, 25/25 were azotemic, 20/24 were hypoalbuminemic and 13/23 were hyperglobulinemic (Table 2). Urinalysis commonly demonstrated isosthenuria and proteinuria with a median UPC of 4.07. Notably, UPCs varied widely, in the range of 0.4–14 (Figure 2).
Recorded clinicopathologic parameters at first presentation

Clinical decision limit is the threshold for which an increase or decrease of the examined parameter is considered significant. In this case, the clinical decision limit was designated based on consultation with a veterinary clinical pathologist, the veterinary literature and/or consensus guidelines, and by clinical experience
HCT = hematocrit; SDMA = symmetric dimethylarginine; UPC = urine protein:creatinine ratio; USG = urine specific gravity
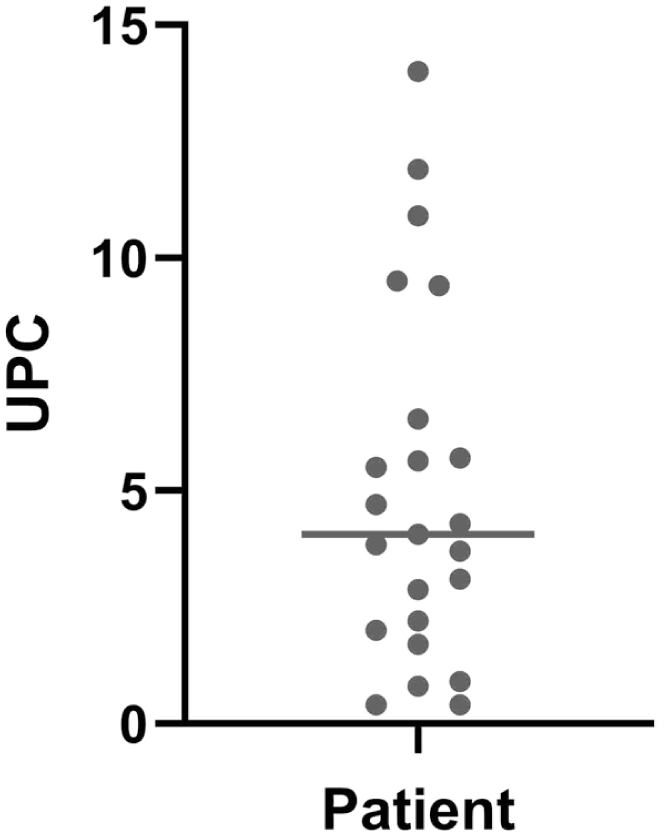
Distribution of urine protein:creatinine ratios (UPCs) from 25 cats with plasma cell pododermatitis and glomerular disease. Median UPC was 4.07 and is indicated by the horizontal line
The most common abdominal ultrasound finding was non-specific nephropathy/renal degeneration (Table 3). Less commonly diagnosed were pyelectasia and renomegaly. Urine SDS-PAGE was performed on 20 cats to determine the likely origin of proteinuria. All 20 had evidence of glomerular damage/dysfunction and 14 had a secondary component of tubular damage (Table 3). In addition, all 20 cats demonstrated molecular weight bands above 200 kDa, consistent with very large urinary proteins and significant glomerular dysfunction (Figure 3). Based on urine SDS-PAGE, the primary differential diagnosis was primary glomerular disease such as ICGN, podocytopathy or amyloidosis.
Summary of advanced diagnostic findings

ICGN = immune complex glomerulonephritis; LLC = lamda light chain; SDS-PAGE = sodium dodecyl sulfate–polyacrylamide gel electrophoresis

Representative sodium dodecyl sulfate–polyacrylamide gel electrophoresis protein banding patterns from the urine of cats with plasma cell pododermatitis and biopsy-confirmed glomerular disease, healthy cats and cats with chronic kidney disease (CKD). Glomerular disease cases show numerous high (66.3–200 kDa) and very high (>200 kDa) molecular weight (HMW/VHMW) bands, indicating glomerular damage, along with intermediate (36.5–66.3 kDa) and low (<36.5 kDa) (IMW/LMW) bands consistent with secondary tubular injury. Healthy cats show minimal proteinuria, with bands at ~55.4 kDa (albumin) and ~66.3 kDa (cauxin). Non-proteinuric CKD cats have banding patterns similar to healthy cats, whereas proteinuric CKD cats display low and intermediate MW bands suggestive of tubulointerstitial damage
In total, 17 cats had renal histopathology performed; samples were collected post mortem in 16/17 cats, with 1/17 cats receiving an ante-mortem needle biopsy as part of the clinical work-up. Of these 17 cats, 16 also had ultrastructural evaluation performed by transmission electron microscopy. Fourteen cats were diagnosed with ICGN, two with segmental glomerulosclerosis secondary to chronic podocyte injury and one with arteriosclerosis causing secondary glomerular damage (Table 3). Among the ICGN cases, membranoproliferative ICGN was the most common histologic pattern. On transmission electron microscopy, ICGN cases had electron dense deposits most frequently between the endothelium and glomerular basement membrane (subendothelial deposits) and/or within the glomerular basement membrane (intramembranous deposits) (Figure 4). Deposits were rarely noted between the glomerular basement membrane and podocytes (subepithelial). Seven cats had immunofluorescence performed; there was moderate to marked immunopositivity for lambda light chain and variably prominent staining with IgA, IgM and IgG (Table 3). No cats in this case series were diagnosed with amyloid deposition (or amyloidosis).

Electron microscopy of a glomerular capillary loop from a cat with plasma cell pododermatitis and glomerular disease. There are subendothelial electron dense deposits (arrows) located between the glomerular basement membrane and endothelium of the capillary lumen. The capillary contains a hypertrophied endothelial cell and the lumen is narrowed (asterisk). Deposits are consistent with antigen-antibody complexes and are diagnostic for immune complex glomerulonephritis
Seven cats were treated for paw pad lesions (Table 4). In many cases, a diagnosis of PCP was not made until the cat presented for renal disease. Of cats that received treatment for pododermatitis, the most common treatments were doxycycline and anti-inflammatory doses of prednisolone either alone or in combination (Table 4).
Summary of treatment types for pododermatitis and renal disease

Cats frequently received multiple agents either concurrently or following adjustment of treatment regimens, as in the case of anemia and proteinuria support
In total, 22 cats received treatment for their renal disease. Treatment regimens commonly included immunosuppression for suspected ICGN, anemia support, antiproteinuric medications and/or blood pressure regulation (Table 4). The most common immunosuppressive agents were mycophenolate and/or prednisolone, with fewer cases receiving cyclosporine. Three cats received GS-441524 for suspected feline infectious peritonitis (FIP) and three cats were started on antibiotics. The reason for antibiotic administration was unclear in the medical records and presumed to address possible lower urinary infection or pyelonephritis. Treatments for anemia included combinations of darbepoetin, erythropoietin, iron dextran and molidustat. Antiproteinuric medications included telmisartan and enalapril, and amlodipine was utilized for blood pressure control. Of the three animals that did not receive treatment for their renal disease, one was euthanized on presentation, and two decompensated and were euthanized while awaiting urine SDS-PAGE results.
Of cats that received renal disease treatment, 20/22 ultimately received immunosuppression for suspected ICGN. Initiation of immunosuppression took a median of 1 month (range 0–7) from the time of renal disease diagnosis. Finally, 21/25 cats were ultimately euthanized with a median survival time of 1 month (range 0.25–60) after a diagnosis of renal disease.
Discussion
Clinically, several features of cats presenting with PCP and glomerular disease are remarkable. Most cats in this series were young, male, castrated and retroviral negative. This signalment is consistent with previous studies evaluating ICGN and PCP individually, where young to middle-aged cats were most affected by both processes.7,10 –15 Likewise, Rayhel et al 11 previously reported a potential male sex predisposition associated with ICGN, and several studies on PCP have reported the same.7,14,15 Interestingly, only one cat in our case series was retroviral positive. This finding differs from previous studies suggesting increased prevalence of both ICGN and PCP in retroviral positive cats.12,14,16 This finding may reflect differences in geographic location and population demographics or suggest that although retroviral positivity may increase the likelihood of PCP, other underlying immune-mediated factors may stimulate the disease independently. Taken together, these findings suggest that ICGN associated with PCP should be ruled out, particularly in young, male, castrated and retroviral negative cats presenting with paw pad lesions and evidence of renal disease.
Interestingly, 23/25 cats were anemic, with some animals having hematocrits as low as 9%. It is unclear whether the anemia in these cases reflects decreased erythropoietin production from renal disease, anemia of chronic disease, an immune-mediated component or a combination of all the above. Regardless, persistent non-regenerative anemia appears to be a common presenting clinical sign in these cases. Other interesting findings on blood work included hyperglobulinemia in 13/25 cats. Previous studies have documented elevated globulins in PCP, and this finding likely reflects the underlying pathogenesis of immune complex formation and secondary ICGN in these patients.15,17 When combined with signalment information, marked anemia and hyperglobulinemia in the setting of paw pad lesions and renal disease may warrant concern for PCP and ICGN.
These cases also frequently presented a diagnostic challenge in terms of determining the underlying cause of renal disease. On ultrasound, the most common finding was non-specific nephropathy, although in six cases renomegaly was appreciated, leading clinicians to prioritize differentials such as lymphoma or FIP. Hyperglobulinemia in these cases further supported the differential of FIP. Variations in UPCs complicated these cases as some cats had UPCs as low as 0.4. In the absence of specific ultrasound findings or significant proteinuria, a final diagnosis of glomerular disease was delayed until either UPC ratios increased or the animal clinically worsened.
Urine SDS-PAGE was therefore very helpful to increase the index of suspicion for glomerular disease. In cats, evidence of high molecular weight proteins on SDS-PAGE has been shown to correspond to glomerular disease, while low molecular weights correspond to tubular damage.18 –20 All cases in this series demonstrated very high molecular weight protein bands on SDS-PAGE and several bands around the 200 kDa range, consistent with significant glomerular damage. Even in cases where UPCs were only mildly increased (0.4, 2/25), SDS-PAGE was able to demonstrate high molecular weight proteins in the urine consistent with glomerular dysfunction. One of these cases had histopathologic confirmation and the other responded positively to immunosuppression and is still alive. SDS-PAGE therefore represents a useful, non-invasive diagnostic tool that can identify a glomerular source of the proteinuria in cats.
In total, 17 cats had histopathology; of them, 14 (82%) had a final diagnosis of ICGN. In these cases, a membranoproliferative histologic pattern was most common. This finding contrasts with previous studies evaluating ICGN in cats without pododermatitis in which a membranous pattern was most frequently observed.11,12 In the human literature, primary membranous glomerulonephritis with deposits between the glomerular basement membrane and podocytes (subepithelial deposits) is reported secondary to autoantigens within the glomerulus, particularly against podocytes. 21 With membranoproliferative ICGN, deposits form between the glomerular basement membrane and capillary endothelium (subendothelial deposits), and in humans is most reported with chronic infections, autoimmune disease and monoclonal gammopathies. 22 In cats with PCP, it is possible that elevated systemic immunoglobulins due to paw pad plasma cells may circulate and become entrapped within the glomerulus, similar to the proposed mechanisms of human membranoproliferative disease. This theory is supported by the previously noted hyperglobulinemia in many of these cases. Although serum protein electrophoresis was not performed in these cases, there are previous reports of polyclonal gammopathies in cats with PCP.7,17 Additional studies focusing on the pathogenesis of immune deposit formation and characterization of hyperglobulinemia in these cats are warranted.
Two cases in this series were diagnosed with segmental glomerulosclerosis and one with arteriosclerosis. To our knowledge, these are the first reports of renal segmental sclerosis and arteriosclerosis in a cat with PCP. Evidence of podocyte foot process effacement on electron microscopy and lack of immune deposits suggests segmental sclerosis is secondary to chronic podocyte injury. Within the human literature, there are reports of podocytopathy and focal segmental glomerulosclerosis secondary to plasma cell disease, particularly multiple myeloma and rare cutaneous and systemic plasmacytosis.23,24 In these cases, a soluble circulating factor such as cytokines or abnormally charged immunoglobulins are suspected to cause direct damage to podocytes. 24 In the cat with arteriosclerosis, vascular lesions resulted in diffuse damage of the glomerulus, including podocyte foot process effacement and endothelial cell injury. Interestingly, this cat was initially febrile on presentation, diagnosed with suspected FIP and treated with GS- 41524. The cat significantly improved while on GS-41524 and only presented with signs of renal disease 4 months after resolution of initial clinical signs. It is possible that renal arteriosclerosis was secondary to resolved vasculitis from FIP. As cats are now widely being treated for FIP and achieving resolution of clinical signs, additional studies evaluating any long-term effects of disease will be needed. Lastly, no cats in this series were diagnosed with renal amyloidosis. However, on review of cases presenting to the IVRPS before 2017, there was one cat diagnosed with amyloid that was included in a previous case series on feline protein-losing nephropathy and mentioned a history of PCP. 11 In addition, other case series have noted amyloid in association with PCP (2/9 cases).2,8
Finally, given the diagnostic challenge cases of PCP and ICGN present, it is unsurprising that treatment methods varied widely. Specifically, only a few cats in this series received treatment for PCP, likely due to it not being diagnosed until examination for renal disease. These findings highlight that although PCP has a characteristic gross appearance, it may be underdiagnosed on routine physical examination in the absence of clinical signs. In cases where PCP was previously diagnosed, doxycycline and prednisolone were the most common treatments. Although some studies suggest efficacy of doxycycline against PCP lesions, it is unclear if doxycycline’s immunomodulatory properties are sufficient to prevent secondary ICGN.10,15 Further studies are needed to determine the most appropriate treatment strategy for PCP and preventing secondary glomerular disease.
Immunosuppression was the most common treatment for renal disease because of evidence of glomerular dysfunction and suspicion for ICGN. In one case, it took 7 months to initiate immunosuppression. This delay in treatment initiation likely reflects difficulty in reaching a final working diagnosis of glomerular disease, specifically ICGN. As mentioned previously, diagnostic findings leading to the prioritization of differentials, such as FIP or lymphoma, likely delayed diagnosis of glomerular disease and initiation of treatment. Delayed treatment is ultimately concerning given the poor median survival time of these patients. The longest surviving cat in this series lived 5 years after a prompt diagnosis of PCP and glomerular disease. In that case, mycophenolate was started within 1 week of initial presentation owing to suspicion of ICGN, which was ultimately confirmed on post-mortem histopathology. It is possible that this early initiation of immunosuppression helped prolong survival in this animal. However, in other cases, the degree of renal disease at presentation was so significant that immunosuppression was of minimal help despite prompt initiation. Therefore, close monitoring of cats with PCP for development of renal disease and additional studies evaluating timing and initiation of different treatment modalities are essential for improving outcomes in these cats.
Several limitations of this study should be acknowledged. First, as a retrospective case series, diagnostic work-up, treatment protocols and follow-up intervals were not standardized or consistently available across cases, making direct prognostic comparisons difficult. Prospective studies are needed to determine the most effective treatment modalities and associated outcomes in affected cats. Second, paw pad and renal histopathology were available for only a subset of cases, preventing definitive diagnosis of PCP and identification of the underlying cause of glomerular disease in some cases. Because fine-needle aspiration or biopsy of the paw pad is invasive and not routinely performed, diagnosis was often based on the characteristic gross appearance, which, while highly suggestive of PCP, is not definitive. In addition, in 1/9 cases with paw pad histopathology, findings were not definitive for PCP. In that case, 4 months of immunosuppressive therapy and reported lesion improvement may have resulted in reduced plasma cell infiltrates, complicating histopathologic interpretation. These findings underscore the need for studies evaluating histologic changes in pododermatitis lesions after immunosuppressive treatment. Similarly, renal histopathology in these cases is needed to better define the mechanisms of glomerular injury and the prevalence of specific lesions such as ICGN, segmental sclerosis and renal arteriosclerosis.
Conclusions
Overall, the aim of this case series was to highlight the association between PCP and the development of glomerular disease. We suggest that cats diagnosed with PCP should be routinely monitored via blood work, urinalysis and UPC for the development of renal disease. If renal disease does develop in PCP patients, glomerular disease, specifically ICGN, should be a primary differential. In the absence of renal biopsy, urine SDS-PAGE is a valuable diagnostic tool for determining glomerular dysfunction.
Footnotes
Acknowledgements
We gratefully acknowledge the practitioners who provided case information for the cats described in this case series.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for this study was provided by Buttons Fund for Feline Chronic Kidney Disease Research at The Ohio State University.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
