Abstract
Case series summary
Fatal feline infectious peritonitis (FIP), caused by feline coronavirus (FCoV), can now be cured with GS-441524. Only a few unexpected clinical and laboratory observations have been reported with treatment, including lymphocytosis, eosinophilia and long-term persistence of abdominal lymphadenomegaly. Yet immune overstimulation associated with FIP might have negative long-term consequences. This report describes four cases of large-cell lymphoma (LCL) arising within 2 years of FIP diagnosis and successful treatment with legally sourced oral GS-441524 (15 mg/kg q24h), representing an incidence of 2.0% (n = 4/202) in a large treatment cohort. At LCL diagnosis, two cats were aged under 2 years, one was aged 8 years and one was aged 13 years. All cats showed weight loss, three had hyporexia and two had chronic vomiting; all tested negative for feline leukaemia virus and feline immunodeficiency virus. LCL was diagnosed by histology (n = 3) or cytology (n = 1) at 81, 365 (n = 2) and 595 days after FIP diagnosis/treatment start. The cats died a median of 15.5 days after LCL diagnosis. Neither a high FCoV viral load nor FCoV antigen, as determined by semi-quantitative RT-PCR and immunohistochemistry, respectively, was detected in any of the available samples. PCR for antigen receptor rearrangements revealed a monoclonal B-cell population in two cats, supporting a diagnosis of large B-cell lymphoma.
Relevance and novel information
The incidence of LCL reported here among cats in remission from FIP after legally sourced oral GS-441524 treatment is remarkably high compared with the general feline population. LCL should be considered a potential ‘long-FIP syndrome’ and/or a long-term complication after GS-441524 treatment. The pathogenesis of LCL in this context requires further clarification.
Keywords
Introduction
Fatal feline infectious peritonitis (FIP), caused by feline coronavirus (FCoV), can now be cured with antiviral drugs such as the nucleoside analogues GS-441524 and its prodrug remdesivir; treatment is usually well tolerated and successful.1 –12 To date, only a few unexpected clinical and laboratory observations have been reported during treatment, such as lymphocytosis, eosinophilia1,8 –11,13 and long-term persistence of abdominal lymphadenomegaly. 14 However, the clinical significance of these findings is still unclear.
Although initially an 84-day treatment course with oral GS-441524 was proposed, a recently published study demonstrated that a shorter treatment duration of 42 days is equally effective in achieving remission in cats with FIP. 1 Interestingly, the cats in the long treatment group (84 days) showed significantly higher lymphocyte and eosinophil counts and maintained elevated values for longer periods than the cats whose treatment was terminated on day 42, suggesting a potential side effect or immunostimulatory activity of GS-441524. 13 Lymphoma is a common neoplasm in domestic cats. 15 The incidence of feline leukaemia virus (FeLV)-associated intermediate- to large-cell mediastinal and nodal lymphomas (I/LCL), most often of T-cell origin, has declined compared with incidence in the 1980s, reflecting the decrease in prevalence of FeLV infections in many European countries due to widespread testing, segregation and vaccination programmes.16,17 Although LCL typically affects older cats, with a reported median age of approximately 10 years, 18 several studies have described cases in younger cats, with a peak incidence in those below 2 years of age.19,20 Rogato et al 20 described a cohort of 33 young cats (aged up to 18 months) diagnosed with intermediate (I) to LCL, of which only three cats tested positive for FeLV. Interestingly, seven of these cats (all FeLV-negative) survived for more than 2 years under chemotherapy. 20 Viruses and neoplastic processes are closely linked, both in human and veterinary medicine. 21 The association between infection with retroviruses, including FeLV and feline immunodeficiency virus (FIV) and LCL, is well established. 15 In addition, feline gammaherpesvirus (FeGHV) has been detected in an archived mesenteric lymph node from a 9-year-old male cat with intestinal T-cell lymphoma (not further classified), suggesting a possible pathogenic role, as confirmed for Epstein–Barr virus in humans.22,23 Nevertheless, several studies in cats could not demonstrate a direct association between FeGHV infection and the development of LCL.24,25 To date, there is no published evidence to suggest an oncogenic potential of feline coronaviruses. Interestingly, an increased risk of developing diffuse large B-cell lymphoma (LBCL) has been found in patients with COVID-19 with very severe respiratory signs of unknown vaccination status. 26 Although COVID-19, caused by a beta-coronavirus, cannot be directly compared with FIP, caused by FCoV, an alpha-coronavirus, both are coronavirus-induced diseases with some similarities regarding clinical manifestations and underlying pathological mechanisms.27,28 This case series is the first to report LCL developing in cats after successful treatment of FIP with oral GS-441524.
Case series description
Materials and methods
This study complied with the German guidelines for prospective studies and was approved by the Government of Upper Bavaria (reference number 55.2-2532.Vet_02-20-52) and by the ethical committee of the Centre for Clinical Veterinary Medicine of LMU Munich (reference number 261-19-03-2021). The study was conducted in accordance with Swiss law and was approved by the veterinary office of the canton of Zurich (TVB ZH124/2022, issued 2022-09-01). Owners gave their written informed consent for their cats to participate in the study.
Study cohort and FIP diagnosis
LCL was diagnosed in 4/202 (2.0%) cats that were initially diagnosed with FIP and enrolled in treatment studies of the Coronavirus in Cats and Kids consortium with two study centres in Munich and Zurich. Cats 1 and 2 were enrolled at the LMU Small Animal Clinic, Munich, Germany 1 and assigned to a cohort that was hospitalised for the first 7 days of antiviral treatment. Follow-up examinations for this cohort were scheduled on days 14, 28, 42, 56, 84, 168, 252 and 365 after treatment initiation. Cats 3 and 4 were enrolled at the Small Animal Internal Medicine Service, Veterinary University Hospital, Zurich, Switzerland and assigned to a second cohort, for which a 3-day hospitalisation period under close monitoring was planned, with follow-up examinations scheduled on days 7, 42, 84, 183 and 365 after treatment initiation.
In all four cases, diagnosis of FIP was established based on a positive FCoV semi-quantitative RT-PCR (RT-qPCR) result, with high viral load in an effusion as previously described 1 in combination with compatible clinical signs and laboratory parameters typically altered in cats with FIP. 29 Detailed signalment, clinical signs, laboratory findings and viral loads are provided in Table 1.
Signalment, clinical and laboratory parameters and treatment of four cats that developed large-cell lymphoma (LCL) after successful treatment of feline infectious peritonitis (FIP) with oral GS-441524, at the initiation of antiviral therapy (day 1) and at the time of LCL diagnosis

Data in bold are outside the reference intervals (RIs). RIs indicated in italics are applicable to cats 3 and 4
Days (after FIP diagnosis/antiviral treatment start) of diagnosis of LCL (cat 1: day 365, cat 2: 595, cat 3: 81, cat 4: 365)
Volume of effusion subjectively evaluated during abdominal/thoracic ultrasonography: – = no effusion, +/– = minimal amount of effusion, + = mild amount of effusion, ++ = moderate amount of effusion, +++= massive amount of effusion
Platelet count was estimated at 81–108 ×109/l on a blood smear; numerous macroplatelets were seen
Additional treatment was administered in parallel with antiviral treatment during hospitalisation
Fluid therapy with Sterofundin (B. Braun Melsungen) with potassium supplementation at 20 mmol/l for dehydration at an individual dosage calculated by rehydration and maintenance needs
Antibiotics: amoxicillin-clavulanic acid 15–20 mg/kg IV q8h
Probiotics were administered for diarrhoea during hospitalisation
A:G ratio = albumin:globulin ratio; AGP = alpha-1-acid-glycoprotein; DSH = domestic shorthair; FCoV = feline coronavirus; FeLV = feline leukaemia virus; FcaGHV1 = Felis catus gamma herpesvirus 1; FIV = feline immunodeficiency virus; fl = femtolitres; MCH = mean cell haemoglobin; MCHC = mean cell haemoglobin concentration; MCV = mean cell volume; N/A = not available; pg = picograms; SAA = serum amyloid A
All four cats received oral GS-441524 (15 mg/kg q24h; BOVA Specials). One cat was treated for 84 days (cat 1) and the remaining three cats for 42 days.
All four cats showed remarkable improvement of clinical and laboratory parameters as well as a reduction in viral RNA loads during the first week of antiviral treatment. All cats completed the treatment course without relevant unexpected clinical or laboratory observations.
Cats 1 and 4 were diagnosed with LCL during a scheduled follow-up after their antiviral treatment, whereas cats 2 and 3 presented as an emergency. The owners of all four cats reported clinical deterioration or appearance of new clinical signs, such as weight loss and hyporexia, indicating further diagnostics. When imaging revealed abnormalities in organs or lymph nodes, fine-needle aspiration (FNA) was used as a minimally invasive method to aid diagnostic assessment.
Laboratory analyses
Haematology was evaluated before starting treatment with GS-441524 (day 1) and on the day of lymphoma diagnosis (in addition to all scheduled regular rechecks) using an automated analyser (Cell-Dyn 3500; Abbott Laboratories [cats 1 and 2] and ProCyte Dx; IDEXX Laboratories and XN-1000V; Sysmex, validated for feline blood samples 30 [cats 3 and 4]). The analysis included haematocrit, reticulocyte count, red blood cell indices, platelet count, total white blood cell count (WBC) and differential count. Scattergrams from the Sysmex XN-1000V were checked visually and blood smear microscopically for plausibility of differential count; in case of discrepancies, such as the presence of left shift, the WBC differential count was conducted manually. Smears, which were stained with Wright–Giemsa stain in Hematek 3000 Slide Stainer (Siemens), were also checked microscopically for plausibility of platelet count, potential presence of platelet aggregates and/or macroplatelets, as well as evaluation of red and white blood cell morphology.
Serum biochemistry parameters were evaluated using an automated analyser (Hitachi 911; Roche [cats 1 and 2] and Cobas c 501 module (Roche Diagnostics AG) [cats 3 and 4]).
Serum amyloid A (SAA) was analysed using a latex agglutination turbidimetric immunoassay reaction (LZ Test SAA; Eiken Chemical) on a Cobas c 501 module clinical chemistry analyser (Roche Diagnostics). Alpha-1-acid-glycoprotein (AGP) was quantified using a spatial proximity analyte reagent capture luminescence (SPARCL) assay on the VetBio-1 analyser (Veterinary Biomarkers).23,31
FCoV viral RNA loads in blood, effusion and faeces were quantified using RT-qPCR, amplifying the 7b gene as previously described.1,11,32 Sequencing of part of the FCoV spike gene (144–618 bp) was performed as previously described. 32 Anti-FCoV antibody titres were determined by indirect immunofluorescent assay. 33
FIV antibody positivity and FeLV p27 antigenaemia were ruled out using the SNAP Combo Plus FeLV Antigen/FIV antibody point-of-care test (IDEXX Laboratories). FeLV provirus and FeGHV DNA loads were determined in blood using qPCR.24,34,35 FIV Western Blot (FIV-WB) analysis was performed as previously described.36 –38
Ultrasonographic examinations
Abdominal ultrasound was performed in dorsal recumbency after clipping the fur using the Logiq E9 ultrasound machine (GE Healthcare) and an 8 MHz microconvex probe (cats 1 and 2), or the Logiq E10s (GE Healthcare) and a 2–11 MHz XD clear broad-spectrum convex and 4–15 MHz broad spectrum linear probes (cats 3 and 4).
Post-mortem examinations
Complete post-mortem examinations were performed in cats 1, 2 and 3. All relevant tissues as well as the neoplastic masses were fixed in 10.0% buffered formalin routinely processed (trimming and paraffin wax embedding) for histological examination.
Histological, immunohistochemical and PCR analyses on the neoplastic processes
Consecutive sections (2–3 µm) from paraffin blocks prepared from the neoplastic processes were stained with haematoxylin and eosin for histological examination or subjected to immunohistochemical staining for T cells and B cells. Information on antibodies and detection methods is provided in S2 in the supplementary material. Immunoreactivity was visualised using 3,3-diaminobenzidine tetrahydrochloride dehydrate as chromogen. Sections incubated with buffer instead of the primary antibody served as negative controls, while sections from feline lymph nodes incubated with the listed antibodies served as positive controls.
PCR for antigen receptor rearrangements (PARR) was performed on paraffin sections from the tumour tissue blocks at a commercial provider (LABOKLIN).
Case 1
Cat 1 (an 11-month-old intact male Somali) initially presented with fever, thoracic effusion and abdominal lymphadenomegaly and was diagnosed with FIP. Mild lymphocytosis and eosinophilia developed during the 84-day GS-441524 treatment (Table 2). At the 1-year follow-up, the owner reported recurrent vomiting, weight loss and hyporexia. The cat had lost 900 g (23.0% of its body weight) since the previous recheck (day 252). A round, firm abdominal mass was palpated. Detailed laboratory parameters are shown in Table 1. Ultrasonography identified a massive focal circumferential thickening in the ileum with loss of layering (Figure 1a), intestinal lymphadenomegaly and a small volume of ascites, suggesting neoplastic disease or recurrence of FIP in first intention. FNA cytology of the intestinal wall was compatible with LCL.
Eosinophil count, lymphocyte count and presence of abdominal lymphadenomegaly in four cats diagnosed with large-cell lymphoma after successful treatment of feline infectious peritonitis with oral GS-441524 before starting the antiviral treatment (day 1) and on days 42, 84 and 168/183
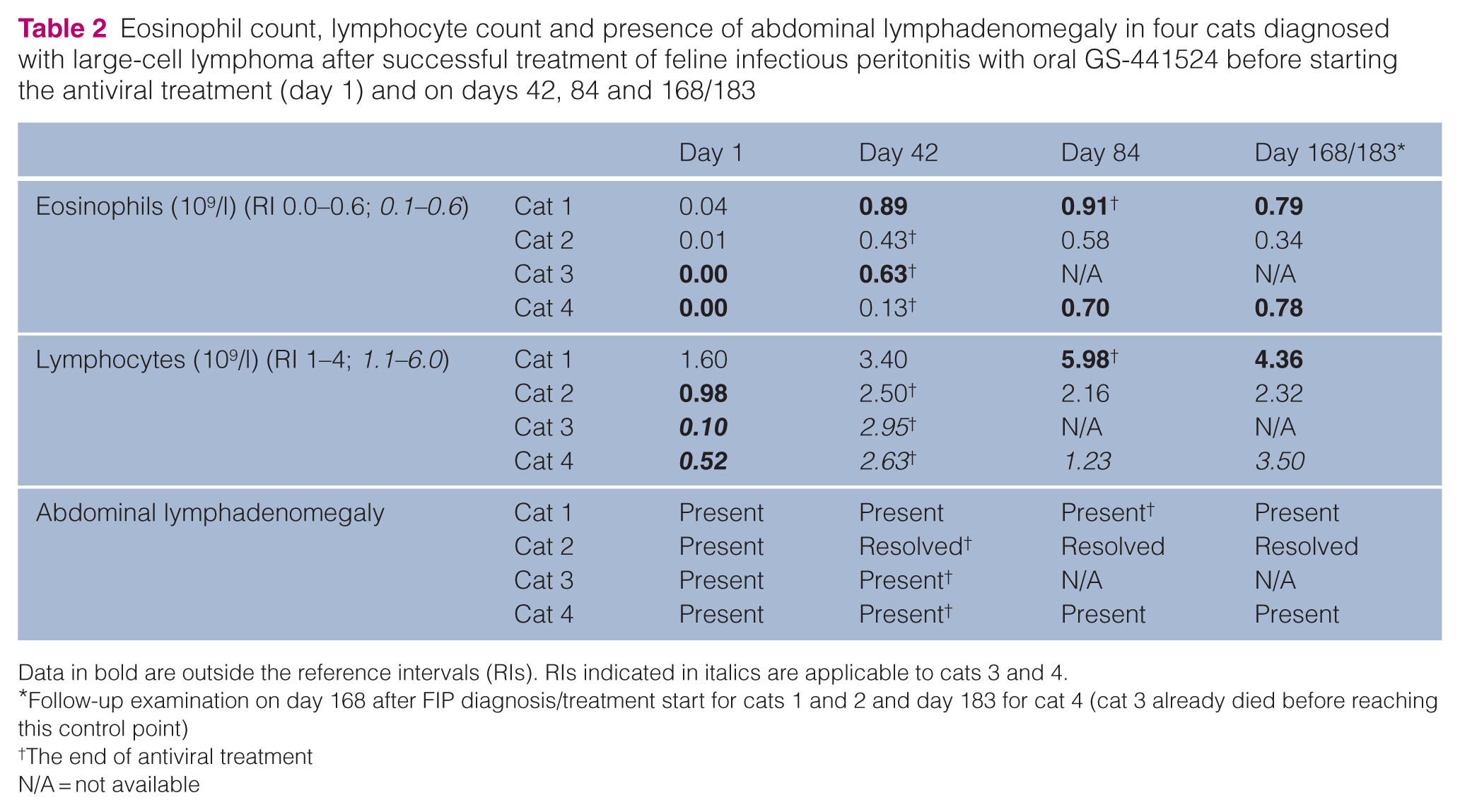
Data in bold are outside the reference intervals (RIs). RIs indicated in italics are applicable to cats 3 and 4.
Follow-up examination on day 168 after FIP diagnosis/treatment start for cats 1 and 2 and day 183 for cat 4 (cat 3 already died before reaching this control point)
The end of antiviral treatment
N/A = not available
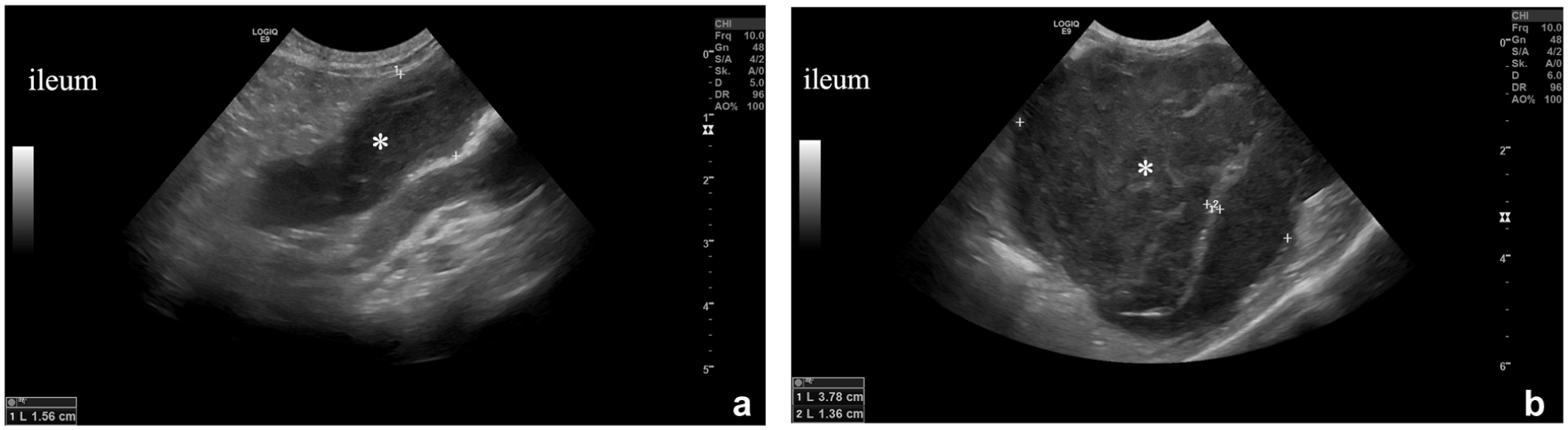
Cat 1: ultrasonographic images of the longitudinal section of the ileum. Severe thickening of the intestinal wall and loss of the typical wall layering (*). (a) On the day of lymphoma diagnosis (day 365 after feline infectious peritonitis [FIP] diagnosis/start of treatment with GS-441524) (small intestinal wall thickness up to 1.56 cm). (b) On a follow-up during chemotherapy (day 411 after FIP diagnosis/start of treatment with GS-441524) (small intestinal wall thickness 3.78 cm and 1.36 cm)
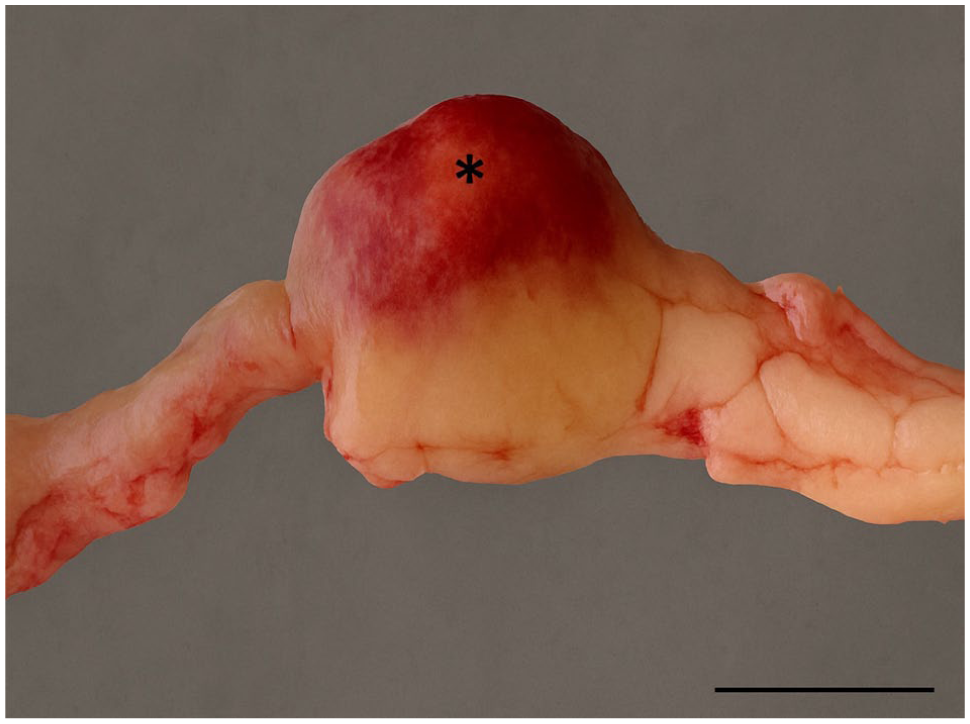
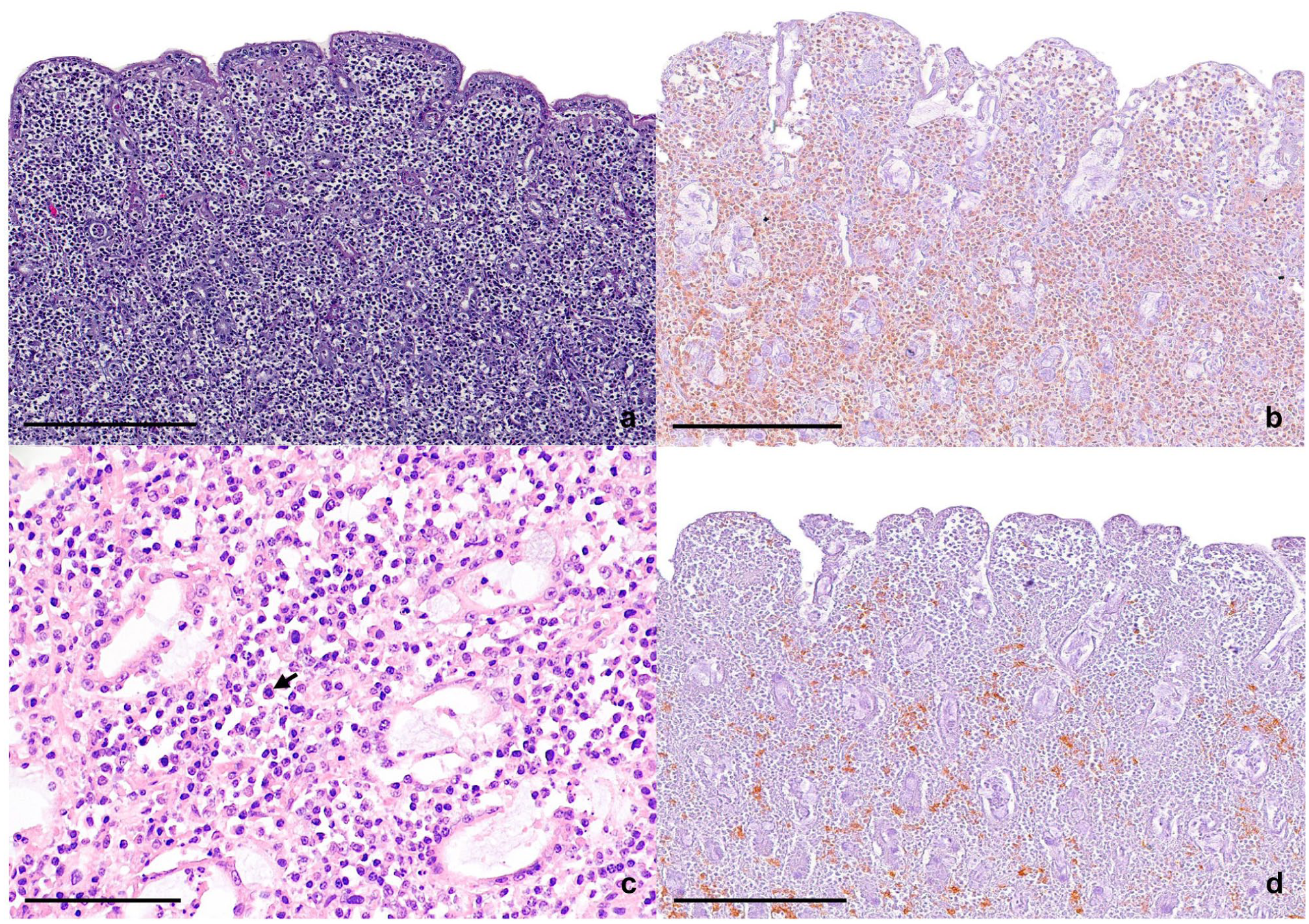
Chemotherapy was initiated with prednisolone (2 mg/kg PO q24h) followed by a single dose of cyclophosphamide 250 mg/m2 and then lomustine 40 mg/m2 every 4 weeks. Abdominal ultrasound was repeated approximately 6 weeks later and revealed a large heterogeneous mass (diameter up to 7 cm) and thickening with loss of layering in other intestinal segments (Figure 1b). Two weeks later, the cat was presented due to recurrent vomiting. As intestinal obstruction was suspected and the prognosis was considered poor, euthanasia was performed. The body was immediately submitted for a full post-mortem examination (Figure 2, Table 3). The main gross finding was a circumferential, multilobulated mass encompassing the ileum, ileocaecal valve and caecum with full thickness expansion and layer effacement of the intestinal wall as well as narrowing of the intestinal lumen (Figure 2). Other relevant findings, as well as the histological findings, are reported in Table 3. The histological and immunohistochemical examination of this mass led to the diagnosis of LBCL (Figure 3). PARR performed on tissue obtained from the mass demonstrated a clonal B-cell population with a polyclonal T-cell population; FCoV immunohistochemistry (IHC) did not detect FCoV antigen (Figure 3). In RT-qPCR, FCoV could not be detected in most tissues; only low FCoV RNA loads were detected in the mesentery, ileum and liver; however, repeated testing in triplicate was negative, indicating very low viral loads not indicative of FIP (S2 in the supplementary material).
Cytological and post-mortem findings, results of immunohistochemistry (IHC) and PCR for antigen receptor rearrangements (PARR) of four cats with large-cell lymphoma

FCoV = feline coronavirus; FNA = fine-needle aspiration; hpf = high-power field; N/A = not available (the owners of cat 4 did not agree to a post-mortem examination)

Cat 1: gross post-mortem findings. Firm, multilobulated mass (*) (7.5 × 6 × 5 cm), encompassing the ileum, ileocaecal valve and caecum, partially covered by great omentum. Bar = 5 cm

Cat 1: histological and immunohistochemistry (IHC) features of sections of an ileal segment. Immunoreactivity was visualised using 3,3-diaminobenzidine tetrahydrochloride dehydrate (DAB) as chromogen. (a) Partial loss of the overlying epithelium and a dense infiltration of lymphoid cells, expanding the lamina propria. (b) Only scattered CD3-positive T cells expanding the lamina propria. (c) Closer view of the neoplastic cells with prominent nucleoli and multiple mitotic figures (arrows). (d) CD20 displaying diffuse positivity, confirming the atypical lymphoid proliferation within the lamina propria to be of B-cell origin. Bars = 250 µm (a,b,d) and 100 µm (c). Haematoxylin and eosin stain (a,c); IHC, DAB substrate, haematoxylin counterstain (b,d)
Case 2
Cat 2 (an 8-year-old castrated male domestic shorthair [DSH]) first presented with fever, anorexia, ascites and mild abdominal lymphadenomegaly, and received a 42-day course of GS-441524 after FIP diagnosis, without severe unexpected clinical or laboratory observations (Table 2). The cat appeared healthy in physical examination at the 1-year follow-up. However, occasional vomiting was reported by the owners. On day 595, the owner reported progressive worsening of the clinical signs, including hyporexia and weight loss. Blood work revealed mild hypoproteinaemia and severe hyperbilirubinaemia (Table 1). Abdominal ultrasonography showed focal thickening of the jejunal wall with loss of layering, hepatomegaly, hyperechoic mesentery, abdominal lymphadenomegaly and a small amount of ascites (Figure 4 and S1 in the supplementary material). Gastrointestinal neoplastic disease or FIP recurrence were suspected. Cytology of the abdominal transudate was compatible with diagnosis of LCL (Figure 5).

Cat 2: ultrasonographic image of a transverse section of a jejunal segment. Severe thickening of the jejunal wall and loss of the typical wall layering (*) on day of lymphoma diagnosis (day 595 after feline infectious peritonitis diagnosis/start of treatment with GS-441524) (jejunal wall thickness up to 0.68 cm)

Cat 2: cytological features of ascites. Within massive blood contamination, multiple small lymphocytes (1–1.5× the size of an erythrocyte) with dense chromatin and scant cytoplasm. Presence of multiple intermediate (arrowheads), occasional large lymphocytes (black arrow) and few neutrophils. Medium and large lymphocytes displaying round to oval, eccentric nuclei with stippled chromatin with no or singular prominent nucleoli (not displayed in picture). Bar = 15 µm
The cat had a cardiopulmonary arrest 1 day later; a full post-mortem examination was performed immediately after death. The main gross finding was a multisegmental thickening of the jejunal wall (Figure 6). Other relevant findings, as well as the histological findings, are reported in Table 3. The histological and immunohistochemical examination of the jejunum confirmed the diagnosis of LCL and suggested a T-cell origin (Figure 7, Table 3). PARR did not find evidence of clonal proliferation within the B- and/or T-cell populations, both of which appeared polyclonal. FCoV IHC did not detect FCoV antigen on jejunal sections (Figure 3). In RT-qPCR, FCoV could not be detected in most tissues. Only low FCoV viral loads were detected in the colon and Nervus ischiadicus; however, repeated testing in triplicate was negative, indicating very low viral loads not indicative of FIP (S2 in the supplementary material).
Cat 2: gross post-mortem findings, section of a jejunal segment. Firm, nordular mass (*) within the jejunum (2.5 × 2 × 1.7 cm). Serosal surface displaying intense, bright to dark red colouring. Bar = 2 cm
Cat 2: histological and immunohistochemistry (IHC) features of sections of the jejunum. Immunoreactivity was visualised using 3,3-diaminobenzidine tetrahydrochloride dehydrate (DAB) as chromogen. (a) Intact overlying epithelium and a dense infiltration of mononuclear cells, expanding the lamina propria. (b) CD3 displaying positivity of up to 85% of atypical cells. (c) Closer view of the neoplastic cells with individual mitotic figures (arrow). (d) Patchy positivity for CD20-positive B cells. Bars = 250 µm (a,b,d) and 100 µm (c). Haematoxylin and eosin stain (a,c); IHC, DAB substrate, haematoxylin counterstain (b,d)
Case 3
Cat 3 (a 13-year-old castrated male DSH) initially presented with hyporexia and fever and was diagnosed with FIP. Eosinophilia and incomplete remission of the initial abdominal lymphadenomegaly were observed at the end of the 42-day GS-441524 treatment (Table 2). The cat was re-presented on day 81 because of a progressively worsening condition, anorexia and severe weakness (Table 1). Abdominal ultrasonography revealed abdominal lymphadenomegaly that had progressed compared with the previous examination 6 weeks earlier (end of GS-441524 treatment), as well as marked jejunal and caecal wall thickening, predominantly affecting the tunica muscularis (Figure 8 and S1 in the supplementary material). Collectively, these findings were suggestive of gastrointestinal neoplastic disease. Cytology of a mesenteric lymph node FNA was highly suggestive of LCL (Figure 9). The cat was euthanased 1 day later at the owners’ request because of its severely deteriorated general condition. A full post-mortem examination was performed. The main gross finding was a moderate to severe enlargement of the mesenteric and ileocaecal lymph nodes (Figure 10). Other relevant findings as well as the histological findings are reported in Table 3. The histological and immunohistochemical examination of the enlarged mesenteric lymph node led to the diagnosis of LBCL (Figure 11). PARR revealed a clonal B-cell population and polyclonal T-cell population, further supporting the diagnosis (Table 3). The post-mortem examination revealed mild ascites; this harboured a low FCoV RNA load. In line with this, the spleen exhibited focal thickening of the serosa due to aggregates of lymphocytes and plasma cells accompanied by dense fibrin precipitates (S1 in the supplementary material), consistent with remnants of the serofibrinous peritonitis the cat had shown upon clinical presentation with FIP 81 days ago. FCoV was not detected by RT-qPCR in any of the other tissues collected at necropsy (S2 in the supplementary material).
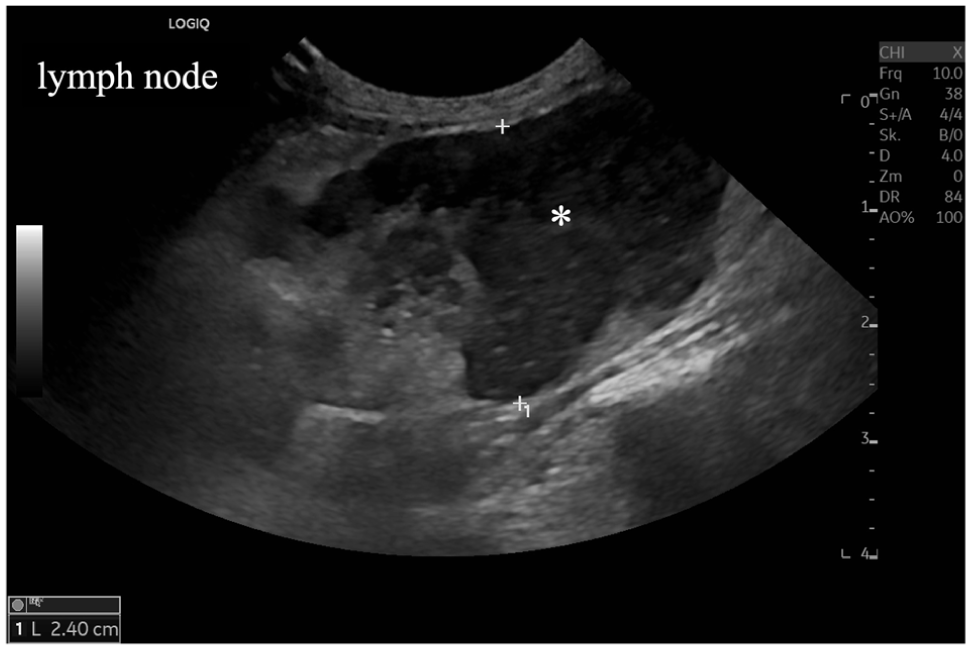
Cat 3: ultrasonographic image of a longitudinal section of a mesenteric lymph node. Severe enlargement and irregular shape of the mesenteric lymph node with heterogeneously reduced echogenicity and complete loss of normal architecture (*) on day of lymphoma diagnosis (day 81 after feline infectious peritonitis diagnosis/start of treatment with GS-441524) (lymph node diameter up to 2.4 cm)
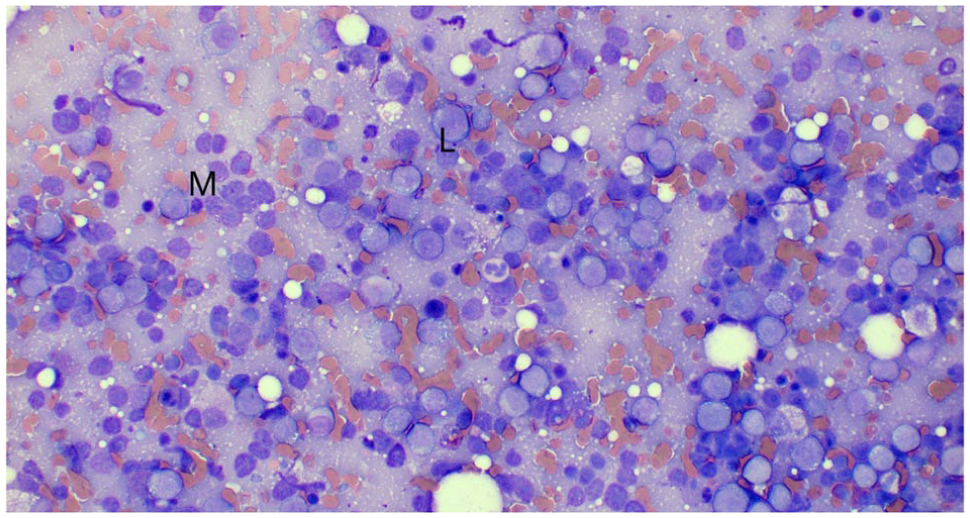
Cat 3: cytological features of a mesenteric lymph node. Increased prevalence of medium (M) to large (L) lymphocytes along with some macrophages and few non-degenerate neutrophils on a haemodiluted background with lymphoglandular bodies on day of lymphoma diagnosis (day 81 after feline infectious peritonitis diagnosis/start of treatment with GS-441524). Medium to large lymphocytes displaying oval to round and eccentric nucleus with stippled chromatin pattern and zero to one prominent nucleolus and small to moderate amount of basophilic and occasionally finely vacuolated cytoplasm. Perinuclear halo occasionally noted, as well as some mitotic figures and occasional binucleations

Cat 3: gross post-mortem findings. (a) Distal part of the jejunum with ileum, caecum and oral part of the colon and mesenteric lymph nodes. Variable enlargement of the mesenteric and ileocaecal lymph nodes (arrows). Thickening of the intestinal wall (arrowheads). (b) Cut surface of the most severely enlarged lymph node, showing focal acute haemorrhage (site of fine-needle aspirate sampling). (c) Cut surface of another enlarged lymph node, exposing homogenously beige, fleshy tissue (arrows)

Cat 3: histological and immunohistochemistry (IHC) features of a mesenteric lymph node. Immunoreactivity was visualised using 3,3-diaminobenzidine tetrahydrochloride dehydrate (DAB) as chromogen. (a) Homogenous monomorphic lymphoblastic neoplastic proliferation replacing the normal architecture. Small embedded aggregates of mature lymphocytes pointed by the arrows. (b) T cells (CD3+) forming aggregates likely representing residues of T-cell zones (arrows) and present as individual cells between the CD3-negative neoplastic cells. (c) Closer view of the neoplastic cells, with individual apoptotic cells (arrowhead) and macrophages (arrow). (d) Neoplastic cells invariably expressing the B-cell marker CD45R (clone 220). Bars = 100 µm (a,b) and 25 µm (c,d). Haematoxylin and eosin stain (a,c); IHC, DAB substrate, haematoxylin counterstain (b,d).
Case 4
Cat 4 (a 6-month-old intact female DSH) was initially diagnosed with FIP upon presentation for reduced general condition and fever. Lymphocyte and eosinophil counts remained within reference intervals during the 42-day GS-441524 treatment (Table 2). At the regularly scheduled 365-day follow-up, the cat had lost 500 g (17.0% of its body weight). A mid-abdominal mass was suspected on abdominal palpation (Table 1). Ultrasonographic examination revealed severe focal enteropathy with thickening of the jejunal wall, bilateral masses in the kidneys, moderate abdominal lymphadenomegaly and mild splenomegaly, suggesting round cell neoplasia or metastatic gastrointestinal neoplastic disease (Figure 12 and S1 in the supplementary material). FNA cytology of the jejunal mass and both kidneys was compatible with LCL (Figure 13 and S1 in the supplementary material). No FCoV RNA could be detected by RT-qPCR. L-asparaginase (400 U/kg subcutaneously) was given once, followed by palliative treatment with prednisolone (2 mg/kg PO q24h) until euthanasia was performed because of acute worsening of her general condition 1 month later. Unfortunately, the owners did not agree to necropsy.

Cat 4: ultrasonographic image of a longitudinal section of a jejunal segment. Severe focal, eccentric thickening of the jejunal wall with reduced echogenicity and complete loss of normal architecture (*) on day of lymphoma diagnosis (day 365 after diagnosis/start of treatment with GS-441524) (small intestinal wall thickness up to 1.12 cm)
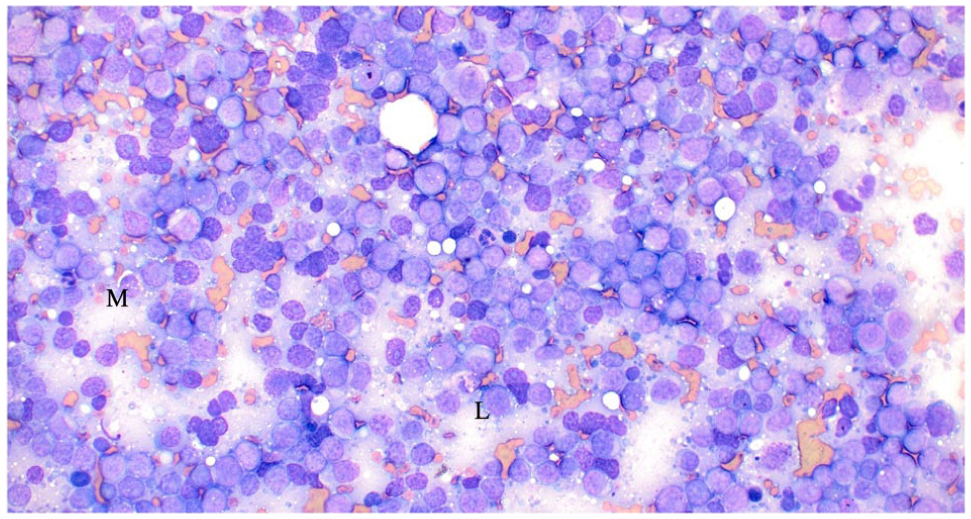
Cat 4: cytological features of a jejunal mass. Nucleated cells mostly consisting of medium (M) to large (L) lymphocytes showing eccentric, oval to round and slightly indented nucleus, stippled chromatin pattern, one frequently prominent nucleolus and small to moderate amount of deeply basophilic and finely vacuolated cytoplasm. Occasional atypical mitotic figures also observed
Discussion
This is the first report of LCL developing in cats with FIP after successful treatment with legally sourced oral GS-441524. At the time point of euthanasia, none of the four affected cats showed clinical evidence of FIP recurrence. In addition, no gross pathological or histological changes consistent with ongoing FIP were identified at post-mortem examination, and FCoV RNA was not detected by RT-qPCR in most examined tissues. In the few tissues in which FCoV RNA was detectable, viral loads were very low and therefore not indicative of FIP recurrence. Instead, all four cats had a clear cytological and/or histological diagnosis of LCL, with LBCL confirmed in two of them by PARR and IHC. In cat 2, PARR did not reveal a clonal proliferation within the B- or T-cell population. However, this does not rule out lymphoma, as the cytological and histological findings were consistent with the diagnosis of LCL and the IHC results suggest a T-cell origin. In addition, the literature states that, at least in other forms of feline lymphomas, PARR fails to detect clonal proliferation in 35.0–40.0% of cases. 39
Distinguishing LCL and FIP can prove challenging in the clinical setting. Namely, both diseases share some clinicopathological features, such as weight loss, cavitary effusions, anaemia or hyperbilirubinaemia.15,29 On ultrasonography, a focal intestinal mass, abdominal lymphadenomegaly and -pathy or reno-, spleno- or hepatomegaly can be observed in cats with LCL; this is also the case in cats with FIP.8,15,29,40,41 Reaching a definitive diagnosis quickly, however, is crucial for a timely initiation of specific antitumoural or antiviral treatment, respectively. In the four cases presented here, there was no clinicopathological evidence of a neoplastic process at the time of FIP diagnosis and upon termination of the antiviral treatment. LCL therefore most likely developed thereafter. Lymphadenomegaly persisted in 3/4 cats after completion of antiviral treatment. It cannot be fully excluded that the development of LCL was already ongoing and that an earlier diagnostic work-up might have resulted in an earlier diagnosis in these cats. Physical examination and laboratory findings were unremarkable, however, and persistent abdominal lymphadenomegaly has been described commonly in cats in long-term remission after antiviral treatment. 14 Therefore, FNAs were not considered immediately clinically indicated.
Lymphoma is a common neoplasm in domestic cats. 15 The true annual incidence rate of lymphoma in cats is currently unknown. Despite a sharp drop in FeLV-associated mediastinal and nodal LCL since the widespread integration of FeLV testing, segregation and vaccination programmes in the late 1980s, the overall prevalence of (not FeLV-related) lymphoma is increasing. 16 This is due to an increase in the number and relative frequency of the alimentary (and in particular the intestinal) form. 42 Reported incidences of feline gastrointestinal lymphoma of either small- or large-cell type are in the range of 0.04–0.2%. 15 The incidence of LCL reported here (2.0%) is therefore markedly higher than what would be expected in the general feline population.
Two cats were diagnosed with an LBCL. In the general cat population, one study found a B-cell lymphoma in 70.0% of the affected cats, whereas only 26.0% had a T-cell lymphoma. Among these cats, 72/109 (53/72 B-cell origin) neoplasms were classified as intermediate-grade LCL and 26/109 (18/26 B-cell origin) neoplasms as high-grade LCL. T-cell lymphoma tended to occur more frequently in younger cats and was more often associated with mediastinal lymphoma and leukemia. 43 Moreover, feline LCL typically occurs in older cats, with a median age of 120 months. Nevertheless, occurrence in cats younger than 24 months has also been documented. 18 This raises the question of why LCL occurred so frequently in the present cohort of cats recovering from FIP and whether this could be a long-term consequence of FIP (‘long-FIP’) or a delayed unexpected consequence of the antiviral treatment.
One mechanistic explanation might be the massive systemic inflammation associated with FIP. The link between inflammation and cellular malignant transformation was identified as early as 1863, by Rudolf Virchow. 44 The long-term persistence of abdominal lymphadenomegaly and the lymphocytosis during and after FIP treatment might reflect a prolonged immunological stimulation in cats surviving FIP. For example, abdominal lymphadenomegaly was described by Zwicklbauer et al 14 in 12/18 (67.0%) cats during a follow-up period until 48 weeks after the start of GS-441524 treatment. This prolonged immunological stimulation might contribute to the development of LCL in an otherwise predisposed cat. In humans, growing evidence indicates that severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), a beta-coronavirus, can induce chronic low-grade inflammation and thereby cause tissue damage. This leads to the hypothesis that long-term exposure to SARS-CoV-2 could predispose individuals to cancer and accelerate cancer progression. 45
Another explanation could be that FCoV itself might have contributed to LCL development. However, there is no evidence in the literature to support the hypothesis of a mutagenic role of FCoV. Interestingly, in some tissues or the ascites of the described cats, very low FCoV loads were detected post-mortem. This could indicate the persistence of FCoV as already reported in healthy cats, primarily in the colon but also in the liver, as a consequence of virus spread via monocyte-associated viremia.46,47 Recent reinfection is not fully excluded in the cats involved in the present study. Previous studies have suggested that SARS-CoV-2 can directly impair critical tumour suppressors, such as pRb and p53, and promote excessive activation of the innate immune system.48,49 Notably, COVID-19 patients with severe respiratory signs have been shown to have an increased risk of developing diffuse LBCL. 26 The reported associations are more plausibly interpreted as consequences of SARS-CoV-2 infection and post-infectious survival itself, potentially allowing chronic inflammation-driven oncogenic mechanisms to unfold, rather than effects attributable to vaccination or antiviral treatment.
On the other hand, antiviral treatment might potentially contribute to LCL development as a long-term unexpected observation. GS-441524 inhibits viral RNA transcription by competing with natural nucleosides and therefore should not have a direct mutagenic effect. Nevertheless, GS-441524 might trigger an exaggerated immune response in treated cats. This response could be reflected by the observed laboratory changes, such as lymphocytosis and eosinophilia. Indeed, these alterations were seen significantly more often in cats treated for 84 days compared with those treated for only 42 days. 1 Clinically, this immune activation could manifest as persistent lymphadenomegaly, which has been repeatedly described during and after antiviral treatment.1,8 –11,14 Zwicklbauer et al 50 demonstrated that this lymphocytosis is primarily attributed to an increase in and B cells.
Lastly, survivorship effect must be considered. Indeed, cats with untreated FIP do not generally survive long enough to develop neoplasia – especially LCL – which would manifest clinically. GS-441524 fundamentally changes the natural history of FIP by allowing long-term survival. Thus, the observation of lymphoma in survivors does not necessarily imply a FIP- or treatment-related effect but might also reflect a chronic FIP-associated immune dysregulation (or an early oncogenic ‘priming’ event during FIP) that has sufficient time to progress.
The cats in the current study had a very short median survival time of 15.5 days after LCL diagnosis, and only two cats received chemotherapy. Reported survival times for feline LCL receiving chemotherapy are highly variable. 15 In a retrospective study of 57 cats with high-grade and large granular lymphocyte alimentary lymphoma, a median survival time of 131 days was reported. 51 The reason for the poorer outcome in the cats in the current study remains unclear, and it might have been influenced by owner decisions regarding euthanasia after the emotionally disturbing and cost-intensive prior FIP treatment. However, it is conceivable that earlier detection of the neoplasm and timely initiation of chemotherapy might have resulted in a better outcome with longer survival time. Therefore, it seems essential to continue monitoring cats in remission of FIP after treatment, with particular attention to gastrointestinal signs, such as weight loss, vomiting and hyporexia. Chronic immunological stimulation might contribute to the development of LCL in these cats, and early implementation of immunosuppressive therapy might be beneficial to interrupt ongoing inflammation and prevent disease progression.
A limitation of the study is the absence of a post-mortem examination for cat 4, not allowing for full evaluation of the neoplastic process.
Conclusions
This case series describes four cats diagnosed with LCL at variable intervals after successful treatment of FIP with legally produced oral GS-441524. The high incidence of LCL in this cohort of cats (4/202, 2.0%) might reflect a long-term consequence of FIP (‘long-FIP’) and immunological stimulation or a potential long-term side effect of GS-441524 treatment. However, to demonstrate a causal association, more scientific evidence needs to be obtained and long-term prospective observational studies including large numbers of cats should be performed. Nevertheless, this series highlights the importance of extended monitoring after successful antiviral treatment, not only for the early detection of FIP relapses but also of newly appearing diseases, such as lymphoma.
Supplemental Material
S1
Additional ultrasonography, cytological and histological findings.
Supplemental Material
S2
FCoV PCR results and immunohistochemistry antibody details.
Footnotes
Acknowledgements
The GS-441524 was provided by BOVA Specials, London, UK. The authors also thank the whole Coronaviruses in Cats and Kids team, and the clinicians and nurses who participated in the daily care of the cats reported here. The laboratory work was partly performed using the logistics of the Centre for Clinical Studies at the Vetsuisse Faculty of the University of Zurich.
Supplementary material
The following files are available as supplementary material:
S1: Additional ultrasonography, cytological and histological findings.
S2: FCoV PCR results and immunohistochemistry antibody details.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was financially supported by the UZH Global Strategy and Partnerships Funding Scheme ‘A bi-center interdisciplinary study investigating the first successful oral treatment of fatal coronavirus infection in cats’ (K-53420-02 01) and UZH Scientific Research Grant, ‘Long-term study of the effect and potential side effects of oral therapy with GS-441524 in cats with feline infectious peritonitis (FIP)’ (F-53130-44-01), the Small Animal Foundation, Vetsuisse Faculty, University of Zurich (F-53130-45-01) and the Swiss Association for Small Animal Medicine (SVK-ASMPA, F-53420-10-01).
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
