Abstract
Objectives
The present study evaluated the safety and efficacy of intravenous, allogeneic uterine-derived mesenchymal stem cells (UMSCs) for client-owned cats with refractory feline chronic gingivostomatitis (FCGS).
Methods
This was a 90-day prospective, multi-site, baseline-controlled, clinical trial involving 46 cats with refractory FCGS after partial- or full-mouth extractions. Cats received two doses of 20 million UMSCs, 14 days apart. Effectiveness was evaluated at days 14, 28, 60 and 90 using owner-reported assessment of overall response (ORA), client-specific outcome measures (CSOM) of quality-of-life improvement and veterinarian-assessed global oral lesion score (GOLS) tissue healing. Safety assessments included clinical observations and clinical pathology.
Results
Treatment success rates at days 28, 60 and 90 were 61.4% (27/44), 76.3% (29/38) and 78.8% (26/33) for CSOM; 65.9% (29/44), 73.7% (28/38) and 75.8% (25/33) for ORA; and 31.8% (13/44), 47.5% (18/38) and 45.5% (15/33) for GOLS. CSOM and GOLS on day 14 after a single dose were 52.3% (23/44) and 25.0% (11/44), respectively. Cats treated within 6 months of tooth extraction had greater improvement in GOLS. There were no statistically significant differences in outcomes between cats with partial- or full-mouth extractions. Adverse events were generally mild and transient, with most related to underlying disease. All six serious adverse events were either unlikely to be due to, or in one case inconclusively attributed to, UMSC therapy.
Conclusions and relevance
Repeated intravenous UMSC therapy was well tolerated and demonstrated clinically meaningful improvement in quality of life in cats with refractory FCGS within 2 weeks of treatment. In addition, oral lesions continued to improve over time. These results support the safe and effective use of UMSCs for this life-threatening condition.
Plain language summary
Feline chronic gingivostomatitis (FCGS) is a painful, long-lasting inflammation of a cat’s mouth and gums. Cats with this disease may have trouble eating, grooming or enjoying normal activities. In severe cases, veterinarians remove many or all of the cat’s teeth, but this surgery does not always solve the problem. When the disease continues after surgery, it can be very difficult to treat, and some cats are euthanized because of poor quality of life. This study tested whether stem cells from donated cat uterine tissue could help cats with non-responsive FCGS feel better and heal. A total of 46 pet cats from multiple veterinary clinics across the USA took part. All cats had already had partial- or full-mouth extractions and medication, but their disease remained active. Each cat received two doses of stem cells by intravenous injection, given 2 weeks apart. Veterinarians examined the cats’ mouths at several visits over 3 months, and owners reported changes in their cats’ comfort and daily activities. Within 2 weeks of starting treatment, more than half the cats showed meaningful improvement in both oral healing and quality of life. By 3 months, about three-quarters of the cats had better owner-reported quality-of-life scores and nearly half had visibly improved mouth tissues on veterinary examination. Cats treated sooner after tooth extraction showed better oral healing. Side effects were generally mild and temporary. A few serious health issues occurred, but none were clearly linked to the stem cell treatment. These findings suggest that intravenous stem cell therapy can be a safe and effective option for cats with FCGS that does not improve after surgery and medical management, offering hope for a better quality of life in cats with this challenging disease.
Keywords
Introduction
Feline chronic gingivostomatitis (FCGS) is a debilitating immune-mediated disease in cats characterized by ulcerative and proliferative oral mucosal lesions, most often involving the area lateral to the palatoglossal folds. 1 Clinical signs include oral pain, difficulty eating, weight loss, lethargy and behavioral changes.2 –4 Underlying immune dysregulation can also drive extraoral manifestations, including systemic inflammation, further compromising quality of life and complicating management.5,6
Medical management and extraction of affected teeth is the current standard of treatment for FCGS. Extractions lead to clinical improvement in approximately one-third of cases; the remaining two-thirds typically require lifelong immunosuppressive or anti-inflammatory therapy.2,4,7 Although these medications may offer symptomatic relief, they are associated with toxicity syndromes and relapse is common when they are withdrawn. 8 Refractory disease often results in euthanasia of affected cats. 1
Mesenchymal stem (ie, stromal) cell (MSC) therapy is a novel treatment for cats with refractory FCGS.1,6,9 –11 MSCs possess potent immunomodulatory properties that address the complex immune aberrancies driving this and other inflammatory diseases.12,13 Previous clinical trials using adipose-derived MSCs (adMSCs) have demonstrated favorable response rates (57–72%) among cats with FCGS, but collection of adMSCs, often from the diseased patient, requires invasive techniques, is of low yield and is not conducted under strict manufacturing practices. 14 This adds morbidity and clinical complexity.6,9,10 The uterine-derived mesenchymal stem cells (UMSCs) used in this study offer a source of highly abundant cells collected through a routine ovariohysterectomy from a healthy US Food and Drug Administration (FDA)-qualified feline donor, ensuring a high level of potency, sterility, stability and purity. 15
In recent pilot studies conducted by the sponsor of the current study, a total of 29 cats with refractory FCGS were treated with UMSCs. 15 By day 90, 78% showed clinically relevant improvement in quality of life as assessed by the owners, and 50% showed at least a 25% reduction in oral lesion scores as assessed by a veterinarian. In these and other similar studies with adMSCs, substantial improvement in quality of life was observed before oral lesion improvement, likely due to rapid effects on systemic inflammation and immune dysregulation.16 –18 Mucosal healing may take more than 6 months to become appreciable and up to 18 months for full resolution. 10
The objective of this study was to evaluate the safety and efficacy of repeated, intravenously administered allogeneic UMSCs for the management of refractory FCGS.
Materials and methods
Investigators and animals
This study was conducted with board-certified veterinary dentists experienced in FCGS management. Client-owned cats with refractory FCGS that had undergone either full-mouth extraction (FME) or partial-mouth extraction (PME) were enrolled after informed consent was obtained. Based on oral radiographs, PME cats had all diseased teeth removed, while healthy teeth were left intact. Eligible cats were refractory as defined by disease persisting at least 2 months after extraction despite ongoing medical management consistent with and justified by other studies.3,6,9,10 All cats discontinued immunosuppressants at least 14 days before treatment to accurately evaluate the time to effect and clinical impact of treatment.
Investigational drug
Cells were derived from uterine tissue after collection via a routine ovariohysterectomy from a healthy, specific pathogen-free, FDA-qualified, feline donor. 15 After harvest, cells were cultured and expanded under Good Manufacturing Practices (GMP) conditions. Each 2.0 ml dose contained 20 million viable UMSCs in a suspension containing cryoprotectant with 5% dimethyl sulfoxide. The cells were not preconditioned and were packaged in a polymer vial compatible with cryogenic temperatures and demonstrated not to interact with the drug product. The cells were stored in liquid nitrogen following the manufacturer’s instructions, shipped on dry ice, and either held in dry ice or transferred to a –80°C freezer for clinic storage. The cells were thawed at room temperature immediately before administration. Thawed UMSCs (2.0 ml) were diluted in sterile saline (20.0 ml total volume) and administered by slow injection via an intravenous catheter in four fractions (0.5 ml cell suspension diluted to 5.0 ml with sterile saline), injected at approximately 0, 10, 20 and 30 mins. No extension sets or infusion pumps were used. Two doses were given, a mean of 14 ± 3 days apart (Figure 1). The final drug product met FDA-accepted prespecified criteria for characterization (major histocompatibility complex class II-deficient, genotypically and phenotypically stable, and potent for FCGS), as well as viability, purity, sterility, morphology and identity. 19

Timeline for diagnosis, extractions and uterine-derived mesenchymal stem cell (MSC) treatment. FCGS = feline chronic gingivostomatitis
Study design
This was a 90-day prospective, multi-site, single-arm, baseline-controlled, clinical field trial conducted at nine veterinary dental specialty hospitals across the USA. Each cat served as its own control since the cats were ‘refractory’ and not expected to improve further with conventional therapies. The trial was conducted in compliance with Good Clinical Practice (Veterinary International Cooperation on Harmonisation Guideline 9) and approved by an independent ethical review committee and the Institutional Animal Care and Use Committee (assurance number D16-00272 [A3433-01]).
Enrolled cats underwent a washout period for immunosuppressants before treatment. Certain medications were allowed if the cat had been receiving a consistent dose and schedule before the first dose of the investigational drug, to ensure no impact on efficacy. Cats were allowed to receive sedation for intravenous catheter placement. Cats were required to be fully awake before dosing to ensure safety could be adequately monitored. By definition of ‘refractory’, all cats were receiving medical management for FCGS – including anti-inflammatories, analgesics, appetite stimulants and gabapentin – at the time of screening. Analgesics such as gabapentin, buprenorphine and frunevetmab were allowed to continue, provided there was a lead-in period and no change in dosing, to ensure adequate analgesia. Immunosuppressive drugs, such as corticosteroids or cyclosporine, were prohibited during the study. Vaccinations were also prohibited because of their effects on the immune system, which may have confounded assessments of efficacy and safety.
Cats were monitored continuously for at least 120 mins after each injection. Follow-up assessments were conducted at days 28, 60 and 90.
Efficacy outcomes
Effectiveness was assessed primarily at days 60 (±7) and 90 (±7), but also at days 14 (±3) and 28 (±3), using three outcome measures pertaining to improvement in quality of life and tissue healing.
(1) Owner-reported overall assessment (ORA): each owner rated the cat’s overall response to treatment as greatly improved, mildly improved, unchanged or worsened. Cats rated as greatly or mildly improved were considered treatment successes.
(2) Client-specific outcome measures (CSOM) 20 : each owner identified three activities impaired by FCGS and rated each on a 5-point scale (0 = no problem, 1 = mildly problematic, 2 = moderately problematic, 3 = severely problematic, 4 = impossible). Each activity was associated with a time and place. Treatment success was defined by at least a 2-point reduction in total CSOM score, with no worsening in any individual activity; this 2-point threshold is the validated criterion.
(3) Investigator-assessed global oral lesion score (GOLS): the veterinarian scored the oral lesions using a modified Stomatitis Disease Activity Index (SDAI), which evaluated predefined criteria at eight sites in the oral cavity; each was scored as 0, 1, 2 or 3, for a total score in the range of 0–24. This index was modified from the published SDAI by assessing only oral lesion scores, without the owner assessment component. 21 Treatment effectiveness was defined by at least a 25% reduction in total score relative to baseline.
Safety outcomes
Safety was assessed through physical examinations, hematology, serum biochemistry, urinalysis, owner-reported observations and documentation of adverse events (AEs). Clinical signs were observed by the investigator during physical examination, injections, the post-treatment monitoring period and at other scheduled and unscheduled visits. AEs were recorded at the investigator’s discretion, regardless of causality. Owners were asked to observe their cats daily, were interviewed at scheduled times and study visits, and kept a diary for the first 7 days after each treatment. Cats were monitored continuously for 2 h from the start of the treatment period, with vital signs taken every 5 mins during the 40-min injection period and then every 15 mins thereafter, or more frequently if clinical abnormalities were observed. In addition, owners recorded injection-site observations in their diaries for the first 7 days after each treatment.
Statistical analysis methods
Analyses were exploratory in nature and involved the modeling of outcomes as binary events. Response rates and confidence intervals were estimated for each binary response variable. A generalized linear mixed-effects model (GLIMMIX procedure; SAS) with a logit-link was used to refine estimates, accounting for effects of site and time since extraction, as well as type of extraction. Repeated measures analysis assessed changes over time, with visit day as a fixed effect and site as a random effect. All statistical tests were conducted at the 0.05 level of significance. All analyses were performed using SAS v9.4. The site effect was not analyzed as a fixed effect; however, site was included as a random factor in the repeated measures model.
Results
Animals
A total of 57 cats were screened across nine clinical sites, with 46 enrolled. Demographics are shown in Table 1. In total, 12 cats were ineligible because of screening failures; however, three of these cats later became eligible and enrolled. An additional two cats met the eligibility criteria but were not enrolled because of the timing of study termination. These cats were enrolled in a separate study. Table 2 shows the total population completing the study, along with the evaluable population for effectiveness and the reasons for non-evaluability. All 46 enrolled cases were included in the safety analysis. For efficacy, evaluability was assessed at each time point for each variable. Evaluable status was first determined based on per-protocol inclusion and exclusion criteria: receipt of both doses of UMSCs, completion of the study to at least day 60 (time of earliest expected efficacy) and absence of major protocol deviations. Each variable was then assessed for data availability and whether prohibited medications had been administered that could have impacted assessment. Outcomes (success or failure) at the time new medication was administered were also considered.
Demographics of the study population
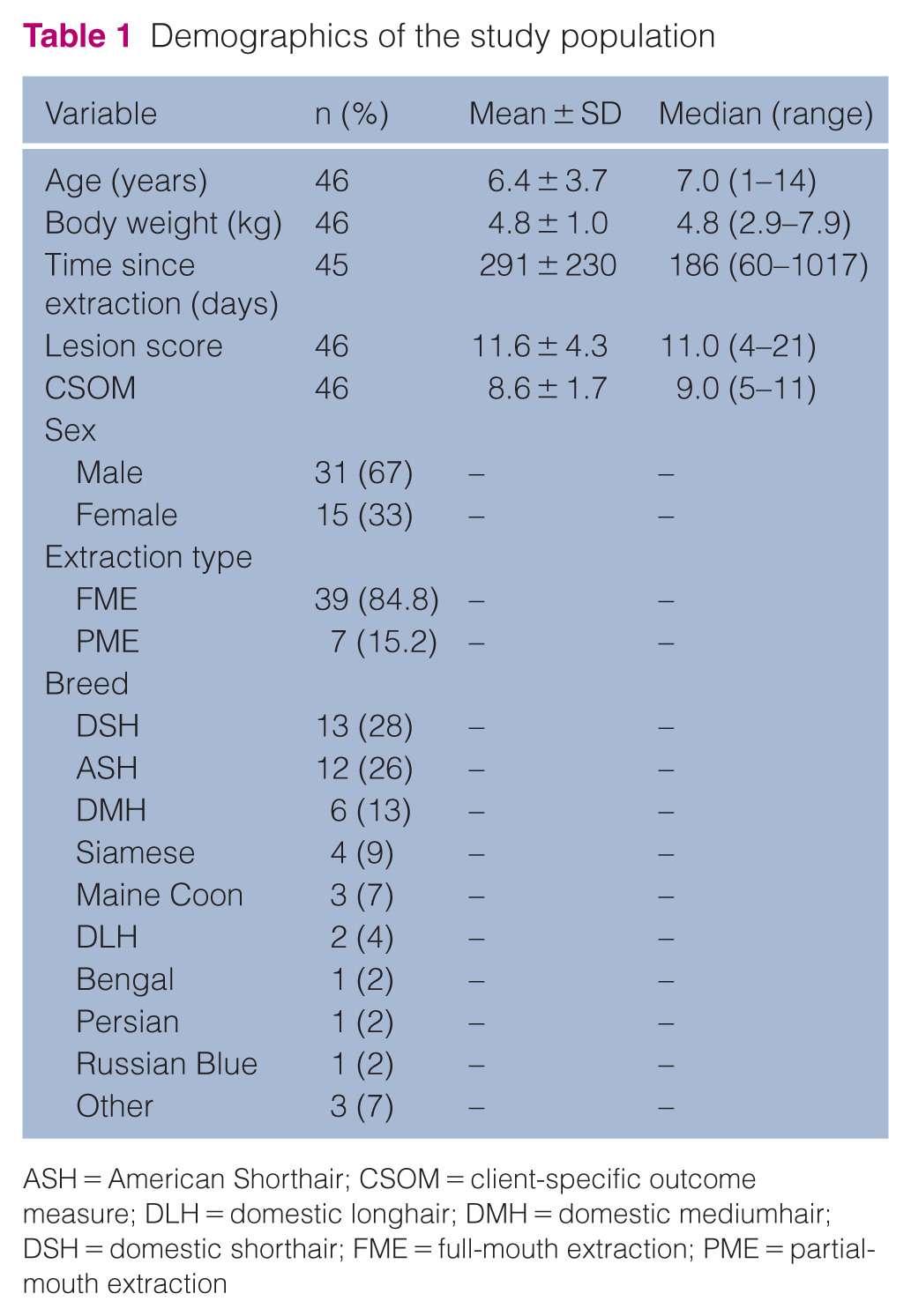
ASH = American Shorthair; CSOM = client-specific outcome measure; DLH = domestic longhair; DMH = domestic mediumhair; DSH = domestic shorthair; FME = full-mouth extraction; PME = partial-mouth extraction
CONSORT of cases in the safety and effectiveness population to day 90

Overall outcome was considered a failure if the outcome was a failure at the time of prohibited medication administration. Cats with treatment success at the time of prohibited medication administration were henceforth unevaluable
AE = adverse event
Upon enrollment, 39/46 (85%) cats had undergone FMEs and seven (15%) had undergone PMEs. A median of 186 days (mean ± SD 292 ± 230) had passed since extraction, in the range of approximately 2–34 months.
Breeds included 13 (28%) domestic shorthairs, 12 (26%) American Shorthairs, six (13%) domestic mediumhairs, four (9%) Siamese, three (7%) Maine Coons, two (4%) domestic longhairs, one each (2%) of Bengal, Persian and Russian Blue, and three (7%) other. In total, 31 (67%) cats were neutered males and 15 (33%) were spayed females. Ages were in the range of 1–14 years (mean 6.4). Cats weighed 2.9–7.9 kg (mean 4.8).
Pre-existing concurrent conditions included feline calicivirus, feline infectious peritonitis, feline immunodeficiency virus (FIV) and diabetes mellitus. In addition, 40 cats exhibited mandibular lymphadenopathy, six had heart murmurs and eight had respiratory abnormalities such as nasal discharge or tachypnea. Various other dermatologic, musculoskeletal and ophthalmic abnormalities were noted at screening but were not considered exclusionary or likely to affect study results. Oral biopsies were not required.
Administration of UMS
All enrolled cats received at least one dose of UMSCs and 44 (96%) completed the full two-dose regimen. All doses were administered within the allowed 14 (±3) day range.
Medical management
Medications used to manage refractory FCGS in enrolled cats at the time of screening are summarized in Table 3.
Historical concomitant medications for management of refractory feline chronic gingivostomatitis

Table 4 summarizes the allowed medications used in enrolled cats during the study.
Concomitant medications used during the study

Cats that received more than one type of medication in a category were counted by number of medications
Used to aid in catheter placement before administration of uterine-derived mesenchymal stem cells (UMSCs)
Used in one cat for anesthesia to biopsy oral lesions in response to a serious adverse event (University of California Davis-001)
Administered at the time of study exit at the end of study or as a rescue medication
To reverse sedation used for catheter placement before administration of UMSCs
Efficacy analysis
At days 14, 28, 60 and 90, efficacy analyses included 44, 44, 38 and 33 evaluable cats, respectively. The outcomes by study day and extraction type are provided in Table 5.
Efficacy outcomes over time and by extraction type

Data are n (%)
ORA data were not collected at day 14
CSOM = client-specific outcome measure; FME = full-mouth extraction; GOLS = global oral lesion score; ORA = owner-reported overall assessment; PME = partial-mouth extraction
The most common CSOM activities were related to grooming, appetite and social interactions, consistent with typical feline behavioral changes under disease conditions. Improvements in CSOM score were observed early in the study and continued throughout the follow-up period (Table 5, Figure 2).
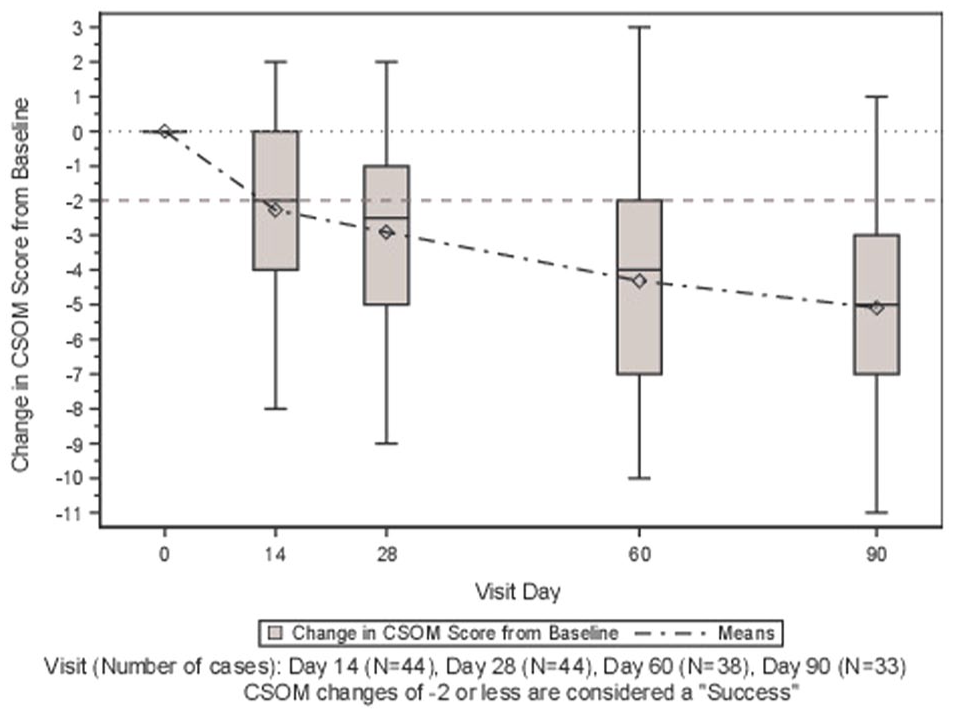
Mean change in client-specific outcome measure score from day 0 with 95% confidence interval. CSOM = client-specific outcome measure
By day 28, 61.4% (27/44) of cats achieved CSOM treatment success. This success rate increased to 76.3% (29/38) by day 60 and 78.8% (26/33) by day 90, indicating a sustained improvement in quality of life. Of note, more than half of the cats (23/44, 52.3%) showed measurable improvement as early as day 14, before receiving the second dose of UMSCs, suggesting rapid improvement in quality of life. A mean CSOM change of –2 (treatment success) for the 44 cats was achieved by day 14.
The mean CSOM score was initially 8.6 and decreased to 5.6, 4.2 and 3.4 at days 28, 60 and 90, respectively. When evaluated using a generalized linear mixed-effects model with repeated measures, CSOM success rates were estimated to be slightly higher than the raw frequencies, with estimated values of 69.9%, 83.9% and 84.9% for days 28, 60 and 90, respectively. There was some indication of an effect due to extraction type at day 90, with estimated success rate for FME and PME cats of 63.4% and 90.6%, respectively (P = 0.145).
ORA improvements mirrored the CSOM results. By day 28, 65.9% (29/44) of owners reported their cats as mildly or greatly improved. This percentage increased to 73.7% (28/38) by day 60 and 75.8% (25/33) by day 90, reflecting a continued positive trajectory. Model-based estimates were 63.7%, 68.4% and 64.6% for days 28, 60 and 90, respectively, closely tracking observed data (Figure 3).

Owner-reported overall assessment
Among cats considered treatment successes at day 90, 15/25 (60%) were categorized as greatly improved. No significant differences were observed in ORA success rates in the model-based estimate between FMEs and PMEs, and all cats with PMEs were considered treatment successes (P = 0.865). Veterinarian-assessed GOLS improved over time. Mean change in GOLS at days 14, 28, 60 and 90 was –3.3%, –10.5%, –10.4% and –18.9%, respectively. At day 90, the 95% confidence interval limits were in the range of –33.1 to –4.79.
By day 28, 32% (14/44) of cats demonstrated a reduction in GOLS of at least 25%, which increased to 47.4% (18/38) by day 60 and 45.5% (15/33) by day 90. Model-based estimates were 17.6%, 33.7% and 31.7% for days 28, 60 and 90, respectively. Statistical comparisons indicated a significant improvement between days 28 and 60 (P = 0.035), with continued improvement to day 90. Cats treated within 6 months of extractions had a higher estimated GOLS success rate (45.0%) than those treated after 6 months (20.3%).
GOLS was evaluated for correlation with clinical effect based on the CSOM and ORA, and was found to be correlated with owner assessment of improvement. By day 90, 86.7% (13/15) and 73.3% (11/15) of cats with a reduction in GOLS of at least 25% were also treatment successes for CSOM and ORA, respectively.
Viral status had no significant effect on any study outcome (P value range across outcome parameters 0.298–1.000). All cats that were viral-positive remained viral-positive at the end of the study. Age had no effect on study outcomes.
Safety analysis
There were 64 non-serious AEs among 28 cats: 32/64 (50%) received no intervention and resolved uneventfully, 8/64 (12.5%) received no intervention but were ongoing and related to FCGS, and 24/64 (37.5%) received treatment for the AE. Of the 24 that received treatment, 15 (62.5%) recovered and nine (37.5%) were ongoing and were related to FCGS or concurrent diseases. Treatments for AEs included buprenorphine, gabapentin, mirtazapine, maropitant, supportive fluids and antibiotics. Among these 64 AEs, association with the UMSCs was described by the investigator as follows: 40.6% unlikely, 40.6% inconclusive, 28.1% possible, 7.8% probable and 1.6% unclassifiable.
Non-serious AEs that occurred in more than one cat are summarized in Table 6.
Non-serious adverse events occurring in more than one cat

Likely related to a flare or temporary worsening of clinical signs
AEs occurring in a single cat included anemia, cardiac insufficiency, corneal ulceration, dandruff, dehydration, dilated pupils, elevated creatinine, exophthalmia, granuloma (at the intravenous catheter site), inappropriate urination, lethargy, leukocytosis, mild hypocalcemia, monocytosis, myopathy, periodontitis, polyuria, skin abscess, stridor, tachypnea and urine crystals.
Self-limiting injection-site abnormalities such as mild erythema or localized swelling were noted in 12 cats; these were considered mild and/or transient and likely related to intravenous catheter placement but were not necessarily reported as AEs.
Routine clinical pathology assessments, including hematology, serum biochemistry and urinalysis, showed no clinically relevant trends in abnormalities during the 90 days after the start of UMSC treatment.
A total of six serious AEs (SAEs) were reported in five cats. None were attributed to UMSC administration, as all were consistent with pre-existing conditions or concurrent treatments. SAEs are summarized in Table 7.
Summary of serious adverse events (SAEs) reported in the study

Euthanized in a follow-up study and confirmed squamous cell carcinoma; thought to be pre-existing due to the extensive nature of the lesions in a short time since treatment; the cat did not have a mucosal biopsy at the time of enrollment
FCGS = feline chronic gingivostomatitis; UMSCs = uterine-derived mesenchymal stem cells
Discussion
The findings of this pivotal study support reasonable expectation of effectiveness and demonstrate the safety and efficacy of intravenous UMSC therapy for refractory FCGS. More than 75% of cats exhibited meaningful improvements as indicated by quality-of-life owner-based assessments, while veterinarian-assessed GOLS improved in approximately half of the treated population by day 90. In addition, improvement in quality-of-life assessments (CSOM) in over half the cats by day 14 suggests that an early treatment response may be observed.
FCGS is recognized as a systemic immune-mediated condition with complications beyond the oral cavity, including behavioral disturbances.2 –4 Oral-targeted therapies alone, such as tooth extractions or medications limited to the oral cavity, may inadequately address underlying immune dysregulation. The intravenously administered UMSCs in this study normalize the systemic immune system, promoting early clinical improvement before improvement in oral lesions, as demonstrated by potency and in-use stability studies (Gallant, unpublished data).
The CSOM (79%) and ORA (76%) efficacy rates at day 90 support improved quality of life based on owner assessments. These rates are consistent with success rates for adMSCs in FCGS, although different outcome measures were used.6,9,10 The use of UMSCs sourced from an FDA-qualified donor and manufactured under GMP conditions ensures sterility, stability, purity, potency and clinical accessibility, and facilitates broader adoption with a ready-to-use product available for use on demand.
An exploratory finding of particular interest was the 90.6% estimated CSOM success rate among cats with PME. In contrast to a previous report, these findings suggest that less aggressive surgical approaches combined with MSC therapy might achieve successful outcomes with reduced surgical morbidity. 22
The clinical improvements observed likely reflect the systemic immunomodulatory properties of UMSCs. In this study, owners commonly reported increases in appetite, activity and social interactions within hours of treatment, corresponding to relatively rapid clinical improvement extending beyond the time of any expected benefit from the 20 ml dose administration (Gallant, unpublished data). This contrasts with historical reports of a delayed clinical effect after MSC administration. Differences in mode of administration, manufacturing methods, cell quality, potency, clinical evaluation metrics and outcomes may impact study results and limit comparison of results between studies and cell types. Oral mucosal lesions are known to improve over time and lag the improvement seen in the quality of life of treated cats. 10 This is a consistent finding across all studies conducted by the sponsor involving UMSCs for cats with refractory FCGS (Gallant, unpublished data). The proposed disease-modifying mechanism of action of UMCS is directly related to cell potency – rebalancing T-cell populations, inhibiting excessive T-lymphocyte expansion and enhancing CD8+ T-lymphocyte function – and may be responsible for clinical cures reported in cats 180–365 days after MSC treatment.16,17,23,24
Safety outcomes were favorable overall, with predominantly mild, transient AEs observed, and no SAEs considered possible or probable in relation to UMSC administration. These safety outcomes are consistent with those reported in a long-term study of 38 cats receiving intravenous MSC therapy for refractory FCGS. 11
One cat developed oral squamous cell carcinoma (SCC) that had an inconclusive relation to UMSC administration. This cat presented with suspicious proliferative lesions at enrollment, imaging confirmed advanced disease shortly after diagnosis, and FIV infection – a known risk factor for SCC – was present at baseline.25,26 No similar lesions appeared in other study cats. These findings collectively support that SCC in this patient was unlikely to be related to UMSC treatment and was likely pre-existing because of the extent of the lesions found soon after treatment. Biopsy is recommended in cats with proliferative FCGS before MSC therapy. 27
A limitation of the study is the low number of cats with PME. Additional studies are warranted to evaluate bolus dosing, the mechanisms by which MSCs affect systemic immunity, the significance of extraction type on treatment response and long-term tissue healing, and the role of viral dynamics in cats with refractory FCGS. Although age, days from extraction to treatment and viral status may impact the clinical presentation of FCGS, none of these factors affected outcomes in this study.
Conclusions
This prospective, baseline-controlled study provides evidence supporting the effectiveness and safety of intravenous administration of allogeneic UMSCs for managing refractory FCGS in cats receiving concurrent medical management. UMSCs may target the underlying pathogenesis of FCGS and improve the quality of life of affected cats. UMSC use may also reduce the need for drugs with unfavorable long-term safety profiles. Active studies are evaluating the long-term safety and effectiveness of UMSCs in the cats reported here.
Footnotes
Acknowledgements
This clinical study was conducted with contributions from several board-certified dental specialists serving as clinical investigators. Manuscript drafting support was provided by Will Pass DVM, of Pass Medical Writing, with compensation provided by Gallant.
Author note
The data supporting this study are on file with the study sponsor and are not publicly available. Access may be granted upon reasonable request to the corresponding author or study sponsor.
Conflict of interest
VSW, JLS, CW, NT, OS and LB are employed by Gallant. RP, CT and BA received consulting fees from Gallant. MSR was an investigator on the study.
Funding
This study was funded by Gallant, San Diego, California, USA.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals and procedures that differed from established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient. The study therefore had prior ethical approval from an established (or ad hoc) committee as stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
