Abstract
Objectives
This study evaluated the efficacy of a psyllium-enriched diet for the management of chronic constipation in cats.
Methods
A multicentre, controlled, blinded, 6-month trial randomly assigned client-owned cats to a gastrointestinal test diet containing 6% psyllium or similar control diet containing 0.5% psyllium (as-fed values). Inclusion criteria included two or more constipation episodes in the previous 6 months and two or more constipation signs for 14 days or longer. Constipation severity (stool consistency and frequency, and defecation pain/difficulty) was scored on days 7, 30, 60, 90 and 180. Cats could switch diets if constipation signs did not improve or the diet was not tolerated. Diet failure was defined as study withdrawal due to lack of improvement in constipation, new gastrointestinal signs or diet switch. Cats switching diets were considered as new cases in the group into which they moved, and statistical analyses (linear and linear mixed models) included them in both diet groups.
Results
Of 49 cats enrolled, two were removed for non-compliance, leaving 26 in the test group (18 assigned, eight switched from control) and 30 in the control group (29 assigned, one switched from test). Of the remaining cats, 19/26 and 11/30 completed 6 months of the test and control diets, respectively. The rate of diet failure for gastrointestinal reasons was significantly higher in the control group than in the test group (22/30 [73.3%] vs 7/26 [26.9%]; P = 0.0005). Constipation signs improved significantly from baseline in both groups at each visit after day 7. For stool consistency, improvement was significantly greater in the test group vs control group except at day 30.
Conclusions and relevance
In cats with chronic constipation, a 6% psyllium-supplemented gastrointestinal diet resulted in sustained improvement of clinical signs, a significantly lower probability of diet failure and greater improvements in stool consistency than a control gastrointestinal diet.
Keywords
Introduction
Constipation, defined as infrequent or difficult emission of hard and dry faeces, is among the top 30 most common disorders seen in cats by veterinarians in general practice. 1 In cat rehoming centres in the UK, the overall prevalence of constipation was 5.6% (range 0–19.7 in individual centres) – more than twice that of diarrhoea. 2 The aetiology of constipation is diverse and includes neuromuscular dysfunction, physical colonic obstruction, electrolyte imbalances, endocrine disease, inflammation that causes defecation to be painful, some medications, dehydration and environmental disturbances. The most common cause of severe, intractable constipation is idiopathic megacolon. 3
Constipation is often recurrent; one of the greatest risk factors is previous episodes of constipation. 4 Acute episodes of constipation are usually managed medically with enemas, intravenous or subcutaneous fluids and laxatives. 4 In the absence of a treatable cause, long-term management options include medication, dietary modification or surgery to minimise recurrence. The ideal diet will have the right amount and proportions of soluble and insoluble fibres to promote soft, easily passed stools while maintaining a high digestibility to prevent large amounts of stool being formed. 5 The husk of Plantago ovata (psyllium) is a beneficial fibre source in the treatment of constipation because of its high proportion of soluble fibre that absorbs and retains water in the colon, increases soft faecal bulk, produces a mucilaginous, lubricating gel 6 and has a low propensity for fermentation by gastrointestinal microbiota.7,8
In two similar uncontrolled pilot trials, a highly digestible, 6% (as-fed) psyllium, dry, extruded diet fed for 2 months was effective in the management of recurrent feline constipation. 9 In the first trial, which disallowed medications concurrent with the study diet, the median faecal score improved significantly, and 14/15 (93%) cats were complete responders on the basis of clinical signs. In the second trial, which permitted the continuation of medications used at baseline, the faecal score also significantly improved, and 42/51 (82%) cats had improved clinical signs. There was a significant reduction in the use of cisapride (a serotonin 5-HT3 receptor antagonist and 5-HT4 receptor agonist that stimulates gastrointestinal peristalsis) and lactulose, but not other laxatives. A meta-analysis of 16 randomised, controlled clinical trials in humans concluded that psyllium is the most effective of fibre supplements investigated for the management of chronic constipation signs. 10
This European, multicentre, controlled, blinded trial aimed to evaluate the efficacy of a gastrointestinal diet containing 6% psyllium for the control of signs of chronic constipation and prevention of recurrence in cats compared with a similar control diet containing 0.5% psyllium.
Methods
Animals and eligibility criteria
Client-owned adult cats aged 12 months or older (>24 months for Maine Coons) were recruited from 13 primary veterinary practices in France, five in Spain and one in Poland. Eligibility criteria (see Supplementary methods in the supplementary material) included recurrent constipation (⩾2 episodes of constipation in the previous 6 months, each lasting ⩾24 h) and at least two of the following signs present for 14 days or longer: dry or firm faecal consistency, infrequent emission of faeces (<1/day), straining, decreased faecal volume, vomiting or tenesmus and dyschezia. Constipation had to have a severity score ⩾5 based on an eight-criteria constipation scale (total maximum score 22) (Table 1). Cats hospitalised to correct metabolic abnormalities or evacuate faeces could be included after these procedures had been completed. Exclusion criteria (see Supplementary methods in the supplementary material) included concurrent disease, use of specified medications prescribed for conditions other than constipation and abnormal findings from blood tests or clinical examination (other than signs of constipation). Cats were withdrawn from the study if clinical signs became significantly more severe or did not improve within 1 week of receiving the study diet and if the owner and veterinarian did not agree to switching to the other study diet.
Constipation scoring system*
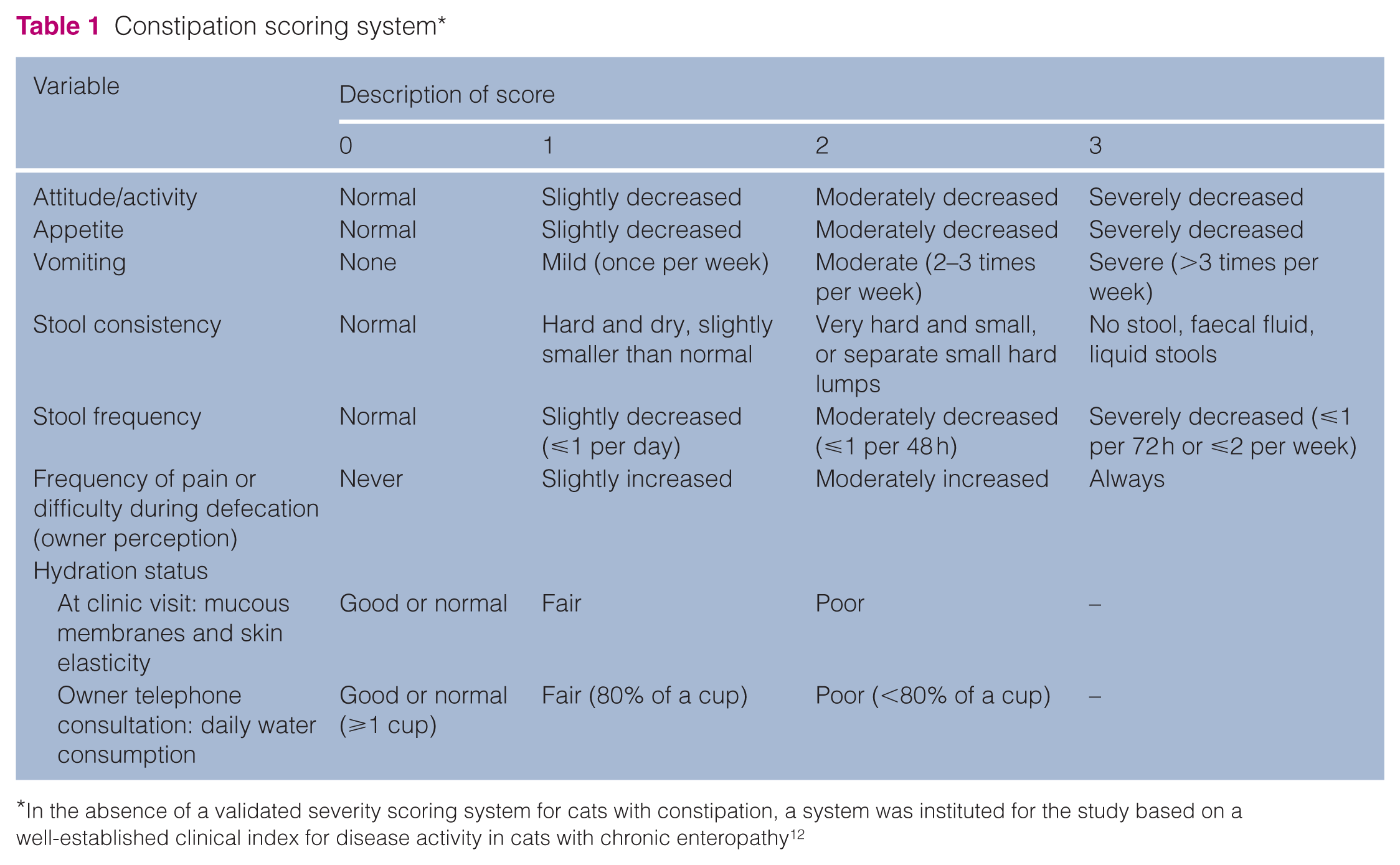
In the absence of a validated severity scoring system for cats with constipation, a system was instituted for the study based on a well-established clinical index for disease activity in cats with chronic enteropathy 12
New laxatives, enemas, prokinetic agents and functional supplements could not be initiated during the study but were permitted if they commenced at day 0 and were discontinued within 4 days. Medications ongoing at day 0 could be continued throughout the trial if necessary. Cases that initiated medication during the study were reviewed by a qualified veterinarian (the Royal Canin Pet and Product Scientific Specialist) to assess the potential effects of the medication on clinical gastrointestinal signs. If an interaction was suspected, the cat was classified as medication non-compliant and removed from the analysis.
Study diets
Enrolled cats were assigned to a dry gastrointestinal diet including 6% psyllium (test diet; Royal Canin Gastrointestinal Fibre Response) or a dry gastrointestinal diet with 0.5% psyllium (control diet; Royal Canin Gastrointestinal) to be fed for 180 days (Table 2). The algorithm for the protocol-planned 1:1 randomisation actually randomised more cats to the control group than the test group. Because of slow recruitment, there was insufficient time to rectify this. Diets were provided in coded white bags for double-blinding purposes.
Composition of study diets

Metabolisable energy was calculated using the National Research 2006 (nutrient requirements of dogs and cats) predictive equation based on crude fibre
DHA = docosahexaenoic acid; EPA = eicosapentaenoic acid; ME = metabolisable energy; NA = not applicable
There was no dietary transition period except for cats known to be sensitive to dietary changes or if vomiting or diarrhoea occurred for more than 24 h after a direct switch. Feeding guidelines used a calorie allocation of 93 kcal/kg body weight (BW)0.711, 77.6 kcal/kg BW0.711 and 62 kcal/kg BW0.711 for cats with underweight, ideal and overweight body condition scores (BCS), respectively.
Study design
Eligibility determination, enrolment and diet assignment occurred on day 0. At scheduled visits on days 0, 30, 60, 90 and 180, the veterinarian performed a clinical examination, determined the constipation score (Table 1) and measured BW (Figure 1). Owners completed questionnaires to allow assessment of diet failure with respect to stool quality. blood samples from day 0 were analysed for haematocrit, red blood cells, white blood cells, haemoglobin, urea, creatinine, glucose, total proteins, albumin, alkaline phosphate, alanine aminotransferase, thyroxine (T4), calcium and phosphorus. Owners were contacted by telephone on day 7 ± 3 to determine a constipation score. If there had been no improvement compared with day 0, a supplementary visit was performed at which the cat could be switched to the other study diet or withdrawn for medical/eligibility reasons or at the owner’s request. Veterinarians could withdraw cats or switch study diets at any other visit for the same reasons.

Study design. *Supplementary visits could be made during the study at any time if necessary. †Blood analyses could be performed at visit 1, 2, 3 or 4 or any supplementary visit at the discretion of the veterinarian
The study protocol was approved by the Royal Canin Ethics Committee (validation 111021-39) before the study commenced and owners provided informed consent.
Endpoints
Clinical disease activity was assessed by a constipation scoring system for stool consistency, stool frequency, difficulty or pain during defecation, attitude, appetite, vomiting and hydration status (Table 1). A maximum overall score of 22 represented the worst constipation severity. The study’s primary endpoints were diet failure, stool consistency, stool frequency, difficulty or pain during defecation, and total constipation score. Diet failure was defined as study withdrawal or diet switch due to gastrointestinal signs (including withdrawals due to gastrointestinal signs after a diet switch) or diet switch for diet-related reasons (inappetence or digestive intolerance), or a lack of owner-reported improvement in the quality of stools relative to before the study (stools reported to be identical, slightly less good or far less good). Secondary endpoints were medications to manage constipation signs before and during the study, BW and BCS.
Statistical analysis
A statistical power calculation (made using a t-test for considering an effect size d = 1, a significance level at α = 0.05 and a test power of 1-β = 90%) indicated that a minimum of 13 cats per diet was required. The recruitment target was increased by 20% to allow for withdrawals.
Data were analysed only for cats that met the eligibility criteria, received a study diet and were medication compliant. Each cat represented an experimental unit; any cat that switched diets restarted the study as a unique subject in the second diet group. Medication non-compliance was assessed independently for each group; if a cat became non-compliant only after switching diet, only data from the switched diet were excluded. Study endpoints were analysed on an as-treated basis. A diet group, unless explicitly specified as an assigned group, is an as-treated group comprising eligible, medication-compliant cats that received that diet either as initially assigned or after switching; hence, a switched cat could be included in both diet groups. Additional within-diet comparisons were made between cats that switched diet and those that remained on their assigned diet to the study end or withdrawal.
Baseline variables were compared between each diet group using one-way ANOVA with a fixed effect for diet group; P values were calculated using an independent t-test. Within-diet group comparisons of post-baseline constipation scores with mean day 0 scores were made using a linear model with visit as a fixed effect and the observed score as the dependant effect. Constipation scores were compared between diet groups at each visit using a linear mixed model with change from day 0 score as the dependent variable, diet, visit and diet-by-visit as fixed effects, individual cat as a random effect and the baseline score as a covariate. The same statistical methods were employed for within-diet comparisons of switching vs non-switching cats. Diet-related study discontinuations or switches were compared between diet groups by χ2 test and Kaplan–Meier analysis. All hypotheses were tested at a two-sided 0.05 level of significance. Statistical analyses used SAS version 9.4 and STATA version 18 software (StataCorp).
Results
Study population and baseline characteristics
Of the 49 cats enrolled, 19 were assigned to the test diet; of them, one became ineligible because the day 0 blood test diagnosed anaemia and one later switched to the control diet (Figure 2). There were 30 cats assigned to the control diet, of which one was non-compliant (initiated lactulose at enrolment for >4 days) and nine later switched to the test diet (Figure 2). One cat that switched from the control diet to the test diet was excluded from the test group analyses because of medication non-compliance after the switch. Accordingly, the test group comprised 26 cats (18 assigned and eight switched in) and the control group comprised 30 cats (29 assigned and one switched in).
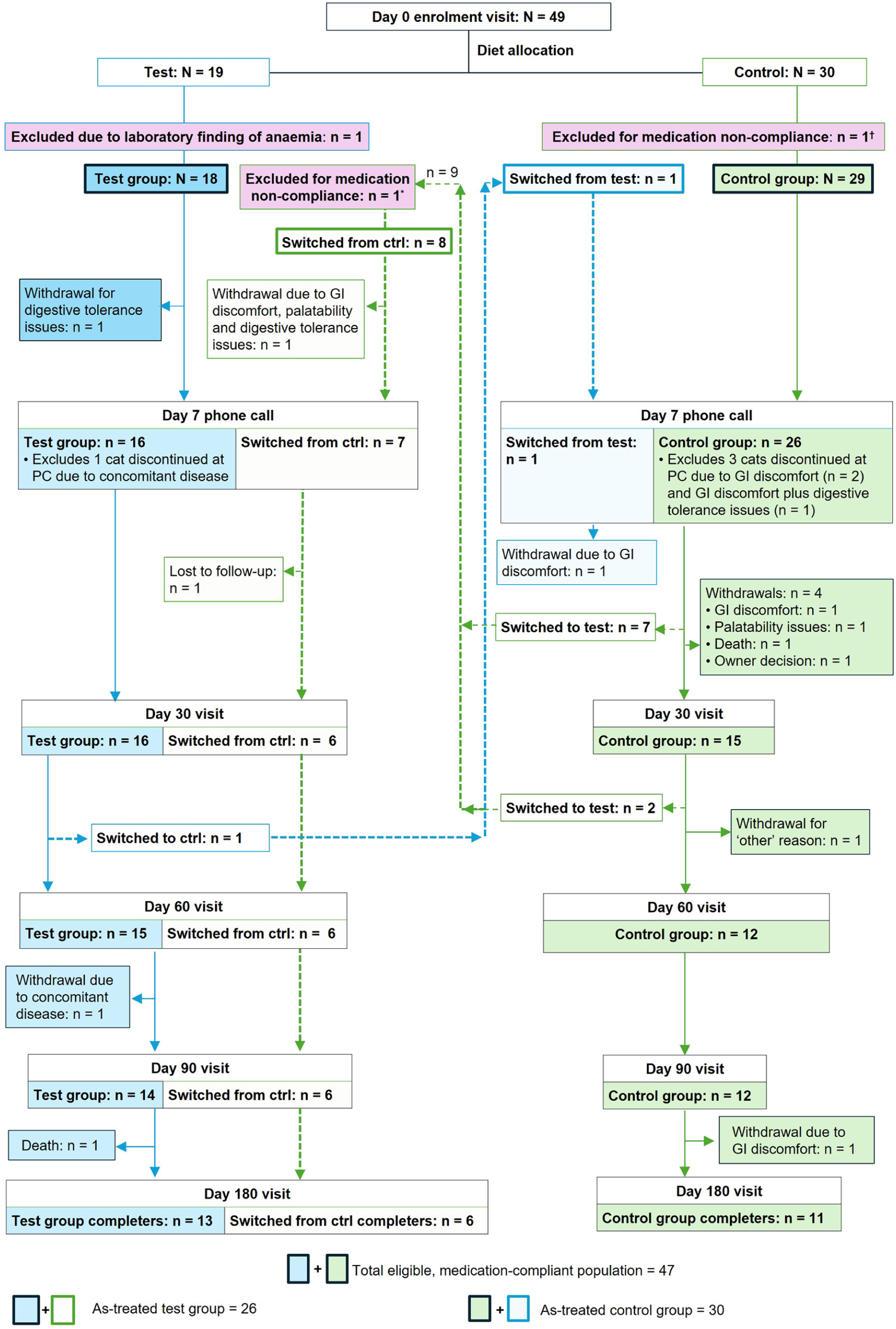
Cat disposition throughout the study and composition of the full analysis sets. *One control cat that switched was excluded from the test group full analysis set but not the control group full analysis set as it was medication non-compliant only while receiving the test diet. †One control cat was excluded from the control group full analysis set because it commenced new medication at enrolment that continued for >4 days. Ctrl = control; GI = gastrointestinal; PC = phone call
At enrolment, the proportion of cats with each sign of constipation was similar between the assigned test and control groups with the exception of tenesmus, which was reported in 12/18 (66.7%) and 11/29 (37.9%) cats, respectively (Table S1 in the supplementary material). At enrolment, gastrointestinal signs had been present for 6 months in 9/47 (19.1%) cats and more than 6 months in 38/47 (80.9%) cats. There was a history of megacolon in seven cats (two of which underwent surgery) and pelvic fracture in six cats (equally distributed between groups). Clinical characteristics at baseline were similar between diet groups (as treated) (Table 3). There were no clinically significant blood haematology or biochemistry results (Table S2 in the supplementary material). For information on previous diets and medications see Tables S3 and S4 in the supplementary material.
Baseline characteristics of the full analysis sets

Data are n (%) unless otherwise indicated. P values are for the difference between diet groups (one-way ANOVA with treatment effect unless otherwise stated)
This population comprised 18 cats assigned to the test diet plus eight cats that switched from the test diet
This population comprised 29 cats assigned to the control diet plus one cat that switched from the test diet
χ2 test
BCS = body condition score
Diet failure
The diet failure rate was significantly higher for the control group compared with the test group (22/30 [73.3%] vs 7/26 [26.9%]; P = 0.0005). The probability of a cat not having diet failure by the end of the study was 75% in the test group vs 25% in the control group (P = 0.0015 for the difference between groups) (Figure 3).

Kaplan–Meier plot of time to diet failure in the test and control groups (as treated)
Two cats were euthanased: one in the test group owing to recurring faecal impaction (the owners declined enema treatment) and one in the control group that developed a non-gastrointestinal concomitant disease and deteriorated rapidly (suspected neoplasia based on abdominal fluid analysis). Acute, concomitant diseases that caused withdrawals in the test group were severe pancreatitis (n = 1) and fever of undetermined origin (n = 1).
Constipation score
The stool consistency score, stool frequency score, pain during defecation score and total constipation score were significantly lower in each diet group at each visit compared with baseline (ie, stools became softer and were more frequent and there was less apparent pain) (Table 4 and Figure S1 in the supplementary material). The decrease from baseline in stool consistency score was significantly greater for the test group compared with the control group at each visit except day 30 (P <0.0001 at the phone call, P = 0.226 at day 30, P = 0.001 at day 60, P = 0.0245 at day 90 and P = 0.0011 at day 180). The reduction in frequency of pain during defecation was significantly greater for the test group compared with the control group at day 30 only (P = 0.043). There were no statistically significant differences between groups in the change from baseline in stool frequency.
Constipation scores in diet groups (as treated)

A lower score for stool consistency indicates softer stools, for stool frequency indicates greater frequency, for frequency of pain during defecation indicates lower frequency, and for constipation overall represents less severe signs of constipation
CFB = change from baseline (day 0); CS = constipation score; Ctrl = control; NA = not applicable
Medications for the management of signs of constipation
There were 4/26 (15.4%) cats in the test group and 7/30 (23.3%) cats in the control group that were receiving ongoing medication for constipation at day 0, of which 3/4 (75%) and 4/7 (57.1%), respectively, continued to receive at least one of these medications for at least part of the study duration (Table S4 in the supplementary material). One cat in the test group receiving ongoing metoclopramide, Consti Regul (Miloa), lactulose and psyllium at day 0 was able to decrease the dose of the last two medications from twice a day to once a day at day 90.
Body weight
BW was stable in both diet groups throughout the study. Mean (±SD) BW at day 0 and day 180 was 4.87 ± 1.45 kg (n = 26) and 4.88 ± 1.55 kg (n = 19) in the test group, and 5.16 ± 1.62 kg (n = 30) and 5.15 ± 1.36 kg (n = 11) in the control group, respectively. There were no statistically significant changes in mean BW at any visit compared with baseline. Similar and consistent BCS was observed for both groups across all visits (data not shown).
Diet switches
Only one cat switched from the test diet to the control diet; a comparison of diet-switching vs non-switching cats was therefore made only for the eight cats that switched from the control group compared with the 20 cats in the control group that did not switch. There were no significant differences at day 0 between switching and non-switching cats with respect to BCS, stool consistency, stool frequency, pain during defecation and total constipation score (Table S5 in the supplementary material). After switching to the test diet, cats had significant decreases from baseline at all study visits in stool consistency score, pain during defecation score and total constipation score (P <0.0001 for each comparison) and in stool frequency at day 180 (P = 0.017).
Discussion
This study demonstrated that a 6% psyllium-supplemented gastrointestinal dry diet was effective in managing chronic constipation in cats and confirms the results of pilot trials without control groups. 9 Over 6 months, cats fed this diet were approximately three times less likely to have experienced diet failure for reasons of efficacy or tolerability than cats fed a similar gastrointestinal diet with 0.5% psyllium.
Key signs of constipation – stool consistency, stool frequency and pain during defecation – improved significantly in both diet groups (mean values) after a week of study diet and remained significantly better than baseline throughout 6 months. However, in the control group, eight cats had to be switched to the test diet before study completion because of failure to respond. The improvement in stool consistency was significantly greater in the test group than the control group at all visits except day 30. This suggests that by sustaining a softer stool consistency over 6 months the test diet may reduce the likelihood of repeat episodes of constipation in cats with a history of more than two episodes over the previous 6 months. The improvement in signs of constipation after cats failing the control diet switched diets also suggests greater efficacy of the test diet. It might be postulated that failure of one diet could affect the likelihood of success with another diet; however, for each cat in both assigned groups there was the same option to switch diets for reasons of tolerability or lack of improvement.
Improved signs of constipation were expected in both groups because both diets were formulated for high digestibility and low residue content. The diets had a similar total fibre content and the greater efficacy of the test diet in softening stools was attributable to its higher ratio of soluble to insoluble fibre, achieved specifically by much higher levels of psyllium as a source of soluble fibre. Softening of stools due to the water-retaining and gel-forming properties of psyllium 6 has also been demonstrated in healthy cats. 11 The study diets had benefits of similar magnitude for stool frequency, pain during defecation and total constipation score, suggesting the importance of the diets’ high digestibility.
A higher proportion of cats in the test group than in the control group continued with medications ongoing at study inclusion, but the numbers were too small to ascertain a drug-sparing effect. To our knowledge this is the first controlled study of a psyllium-supplemented diet in cats with constipation, and assessment of constipation signs was more comprehensive than a previous uncontrolled study. 9 Switching between diets was permitted in the absence of an adequate response because this reflects real-life clinical practice. Combined with an as-treated analysis it allowed us to use all data from each eligible, medication-compliant cat. Limitations included the absence of a validated severity scoring system for cats with constipation. The system used in the study was adapted from a well-established clinical index for disease activity in cats with chronic enteropathy; however, more research is needed for a validated clinical tool for assessment of constipation in cats. 12 Although the randomisation protocol failed to achieve equal group numbers, clinical characteristics were balanced at baseline and blinding was maintained. It was necessary to allow the continued use of medications ongoing at enrolment for ethical reasons and owner reassurance, given that severe constipation or obstipation can be life-threatening. The objective of this study was not to identify any medication-sparing effect.
Conclusions
A gastrointestinal diet enriched with 6% (as-fed) psyllium resulted in rapid and sustained improvement of clinical signs of constipation (stool consistency, stool frequency and pain during defecation) in cats with chronic constipation. Cats did not require additional medications during the trial and some reduced or stopped current medications. Both test and control gastrointestinal diets improved stool consistency in the first month; thereafter, improvements were consistently greater in cats fed the psyllium-enriched diet, consistent with physical effects of that diet’s high soluble fibre formulation. Cats were significantly less likely to discontinue the psyllium-enriched diet because of constipation relapse or other gastrointestinal signs. Overall, the observed clinical benefits of the gastrointestinal psyllium-enriched diet are fundamental to the management of constipation and prevention of its recurrence in cats with chronic constipation.
Supplemental Material
Supplemental Material
Supplementary methods
Supplemental Material
Table S1
Signs of constipation at day 0 according to assigned diet group.
Supplemental Material
Table S2
Baseline blood variables of cats.
Supplemental Material
Table S3
Diets routinely fed to the cats before the beginning of the study.
Supplemental Material
Table S4
Medications ongoing at day 0 to manage constipation signs in each diet group (as treated).
Supplemental Material
Table S5
Baseline clinical characteristics of cats assigned to the control diet that switched to the test diet compared with those that did not switch.
Supplemental Material
Figure S1
Constipation scores in each diet group (as treated).
Footnotes
Acknowledgements
The authors would like to thank Susanna Ryan of Ryter Ltd for editorial assistance, Roland Klopper of Clindata for statistical support and the Teranga group (Cebiphar) for clinical trial management, all funded by Royal Canin.
Supplementary material
The following files are available as supplementary material:
Supplementary methods
Table S1: Signs of constipation at day 0 according to assigned diet group.
Table S2: Baseline blood variables of cats.
Table S3: Diets routinely fed to the cats before the beginning of the study.
Table S4: Medications ongoing at day 0 to manage constipation signs in each diet group (as treated).
Table S5: Baseline clinical characteristics of cats assigned to the control diet that switched to the test diet compared with those that did not switch.
Figure S1: Constipation scores in each diet group (as treated).
Conflict of interest
Julie Rochon, Hélène Charles, Vincent Biourge, Jeremy Laxalde, Mikel Goñi García-Falces and Danielle Dito are employees of Royal Canin SAS.
Funding
This study was funded by Royal Canin SAS.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
