Abstract
Objectives
The Lykoi is a recently recognised feline breed with a unique coat phenotype caused by six variants of the Hairless (HR) gene. A specific severe cutaneous comedonal syndrome has been reported in this breed. The aim of the study was to carry out a clinical, histopathological and genetic characterisation of this syndrome.
Methods
Prospective data collection included clinical evaluation, skin biopsies for histopathology, trichoscopic examination of hair shafts, blood sampling for haematology and biochemistry, cutaneous swabs for bacteriological culture, and buccal swabs for DNA extraction and genotyping.
Results
In total, 11 privately owned Lykoi cats were enrolled, including those with varying degrees of skin involvement and one healthy cat. Cats were aged 1.5–7 years at the time of diagnosis. Clinical presentation was characterised by papular lesions and comedones, ranging from severe generalised forms (n = 6/10) to moderate regional forms (n = 2/10) and to mild scattered forms (n = 2/10). Histopathological lesions from skin biopsies revealed infundibular follicular cysts (n = 9/10), sebaceous gland (duct) cysts (n = 10/10) and sweat gland cysts (n = 5/10). Genetic analysis identified 5/6 described HR variants among the 10 affected Lykoi and the control Lykoi.
Conclusions and relevance
This cutaneous syndrome shows a variable clinical severity that is not fully explained by genotype alone. The consistent presence of cysts from all three adnexal structures, even in clinically unaffected skin, supports the recognition of a novel skin condition, which we propose to name cutaneous adnexal polycystic syndrome (CAPS) in Lykoi cats. Further research is needed to elucidate its pathogenesis.
Introduction
The Lykoi is a recently recognised feline breed first reported in 2011 and characterised by a distinctive coat phenotype, without undercoat and with a unique black-and-white roan pattern. Controlled outcrossing with black domestic shorthair cats was introduced by breeders to preserve genetic diversity and maintain the desired appearance. 1
At the present time, six distinct loss-of-function variants in the Hairless (HR) gene are described in this breed and are named TN (Tennessee), TX (Texas), NC (North Carolina), VA (Virginia), Ca (Canada) and Fr (France), according to their geographical origin. 2
In humans, pathogenic variants of the HR gene cause several hereditary forms of alopecia, including atrichia with papular lesions, a congenital disorder with irreversible hair loss shortly after birth followed by papular eruptions. Histopathology reveals comedo-like dermal cysts filled with laminated keratin in areas previously occupied by hair follicles. 3
The occurrence of a severe comedo syndrome in a Lykoi prompted the hypothesis of a comparable dermatosis in this breed, potentially associated with HR dysfunction.
The aim of this study was to provide a comprehensive clinical, histopathological and genetic characterisation of this cutaneous cystic syndrome in Lykoi cats.
Materials and methods
Case selection
A prospective, descriptive study was conducted to address the objectives outlined above. A total of 10 client-owned Lykoi cats were enrolled through outreach to the French Lykoi Breed Club, the Fédération pour la gestion du Livre Officiel des Origines Félines (LOOF), The International Cat Association (TICA) and a network of veterinary dermatologists via the VetDerm mailing list (
Cats of any age, sex or lineage were eligible for inclusion, regardless of the severity of papular and comedonal lesions. A comprehensive data set was collected for each subject. Clinical data included age at lesional onset, progression over time, lesional morphology and severity, and lesional distribution. Various sample types were collected to enable complementary diagnostic investigations, as detailed below.
Phenotypic characterisation
Each cat underwent a complete physical and dermatological examination. For every individual, the presence, colour, size and distribution of papules were documented.
Clinical severity was categorised as follows:
An apparently clinically healthy female Lykoi cat presented for spaying was selected as a clinically unaffected control.
Haematology and biochemistry
Whole-blood samples were collected from each cat for complete blood count, serum biochemistry profiling and total thyroxine (T4) measurement. Screening for feline leukaemia virus (FeLV) antigen and feline immunodeficiency virus (FIV) antibodies was performed using standard assays.
Parasitic and fungal investigations
Skin scrapings for ectoparasites, trichoscopy, Wood’s lamp examination and fungal culture on Sabouraud’s agar were performed.
Trichoscopic examination
Trichoscopic evaluation was carried out using trichograms prepared from approximately 30 hairs collected from dorsal and abdominal regions. Samples were mounted in lactophenol and examined under light microscopy at × 200 magnification to detect any abnormalities in the hair shaft.
Bacteriological examination
Deep swabs were obtained from severe cases and collected from the centre of large ulcerated black comedones for bacteriological culture and antimicrobial susceptibility testing, ensuring sampling was taken from the depth of the lesion to minimise surface contamination.
Histopathological investigation
Full-thickness skin biopsies were obtained under general anaesthesia. A standardised anaesthetic protocol was employed to ensure optimal conditions and effective analgesia during and after the procedure, although the specific protocol varied slightly depending on the clinician. In most cases, sedation was induced with dexmedetomidine chlorhydrate (20 µg/kg IM, Sedadex; Dechra) in combination with butorphanol (0.2 mg/kg IM, Torbugesic; Zoetis), followed by maintenance with inhaled isoflurane gas.
Biopsy sites were selected based on clinical presentation. For clinically affected Lykoi cats, samples were taken from visibly affected areas of the trunk, typically areas characterised by white and black papules, as well as from a less affected area on the ventral abdomen. In the control Lykoi, the skin specimen was collected from the edge of the laparotomy incision on the flank during spaying, corresponding to a site anatomically comparable to that sampled in clinically affected individuals. Skin biopsies were obtained using an 8 mm biopsy punch and fixed in 10% neutral buffered formalin.
Tissue samples were paraffin-embedded, sectioned at 5 µm and stained with haematoxylin and eosin. Slides were examined by light microscopy, imaged digitally and lesions were qualitatively scored; periodic acid–Schiff staining was also performed to detect potential fungal elements.
Genetic sampling, DNA extraction and genotyping
Genetic analysis was performed using buccal swab samples. The swabs were sent to the Felome laboratory (www.felome.fr) for DNA extraction and genotyping of the six described HR variants. 2
Ethical considerations
This prospective study was conducted with informed owner consent for all affected and control (apparently unaffected) Lykoi cats, and all procedures complied with accepted standards of animal care and welfare.
Results
Anamnestic findings
In total, 11 Lykoi cats were enrolled in this prospective study: 10 affected cats and one control cat, consisting of seven males and four females. All females were intact, whereas four males were neutered and three were intact.
The age range at the time of diagnosis was 1.5–7 years (median 4.75, mean 4.5). The onset of skin lesions was reported between the ages of 1 and 4 years, with most cats (n = 7/10) developing noticeable clinical signs from mid-adulthood onwards (3 years of age, median 3.25, mean 2.85).
Among affected cats, the population was geographically diverse, comprising seven cats from France and one each from Belgium, the UK and Canada. The control Lykoi was an intact 3-year-old female cat from France.
Clinical findings
Clinical presentations ranged from severely affected individuals to cats without apparent skin abnormalities, with no apparent association to coat colour. None of the Lykoi cats exhibited systemic clinical signs, and manipulation of the lesions did not induce pruritus or discomfort. Pain evaluation based on the Glasgow scale resulted in a score of zero for all cats (Table 1).
Signalment, age and clinical presentation of the included cases

F = female; M = male; MN = male neutered
Severe form
Six Lykoi cats were classified as severely affected, with ages in the range of 1.5–7 years (mean 4.5). In these cats, alopecia was more pronounced on the trunk and they exhibited widespread cutaneous lesions, predominantly consisting of numerous white or black papules and nodules of varying size, on the trunk, limbs and head. On the trunk, the papules were coalescent and formed voluminous nodules or plaques, which were open or not (Figures 1 and 2). Lesions occasionally exceeded 5 cm in diameter. Deep haemorrhagic ulcers, sometimes measuring up to 1 cm, were also noted (Figure 3). Less commonly, on the trunk, among numerous small white papules, some small black papules were surrounded by an erythematous circular ring (Figure 4). The limbs exhibited a high density of papules, typically exceeding 100 per limb. These lesions had a diameter in the range of 1–5 mm.

Severe form: note the generalised distribution of numerous papules, comedones and nodules of variable size over the trunk. Some nodules are ulcerated (blue arrows)
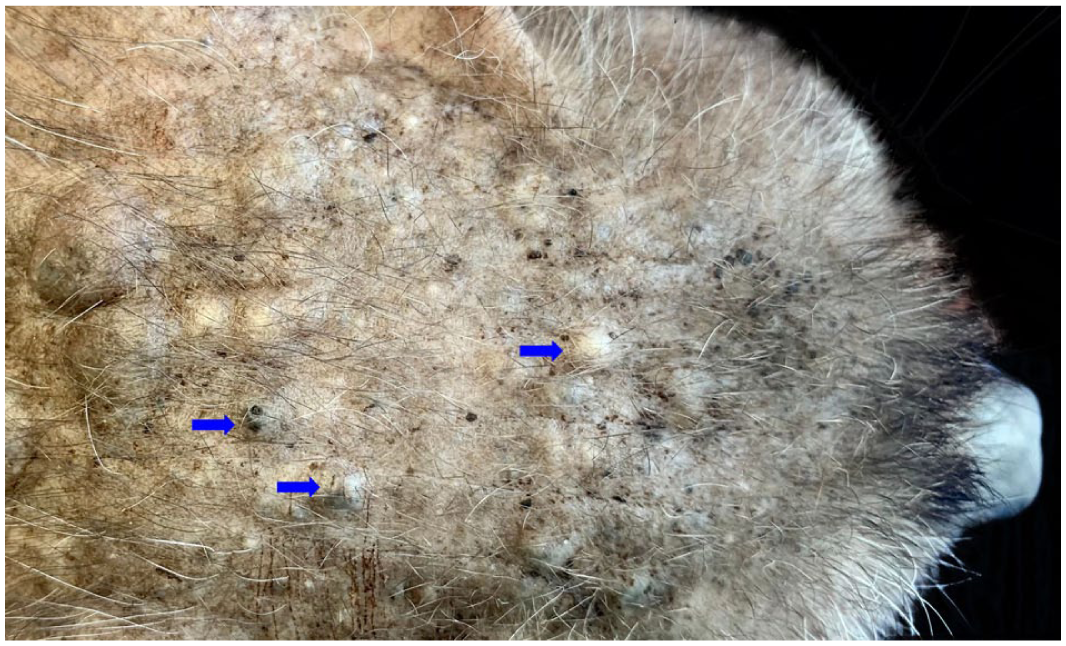
Severe form (same cat as in Figure 1): note the significant number of comedones, papules and some nodules distributed across the forehead (blue arrows)

Severe form (same cat as in Figure 1): note the significant number of large comedones and papules. Some large nodules are ulcerated and covered by crusts (blue arrows)

Severe form (same cat as in Figure 1): note the significant number of small white papules and some small black papules. A black papule is surrounded by an erythematous circular ring associated with a secondary pyogranuloma (blue circle)
Moderate form
Two Lykoi cats, aged 4 and 5.5 years, were classified as moderately affected. Lesions were more localised or regional, with involvement mainly on the limbs. The trunk was less affected than in the severe cases. Papules were numerous, white or black, small to medium in size (1–5 mm) and remained discrete (non-coalescent). Fewer than 50 lesions were typically observed per limb (Figure 5).

Moderate form: note the significant number of small white and black papules exclusively limited to a distal extremity
Mild form
Two Lykoi cats, aged 1.5 and 7 years, exhibited fewer than 20 papules, white or black, each less than 5 mm in diameter, sparsely distributed across both the trunk and limbs.
Control Lykoi
One cat, aged 3 years at the time of sampling, showed no visible clinical lesions. Skin was collected during spaying from the edge of the laparotomy incision on the flank. This cat’s coat appeared denser than that of the other Lykoi cats included in the study.
Biochemistry and haematology investigations
Serum biochemistry profiles were within reference intervals (RIs) in all Lykoi cats.
Haematological analysis revealed a mild to moderate neutrophilic leukocytosis in four Lykoi cats. This finding was observed in both moderately and severely affected cats.
Screening for FeLV antigen and FIV antibodies was negative in all Lykoi cats.
Total T4 concentrations were measured in eight Lykoi cats and found to be within the RI (Table 2).
Laboratory and ancillary diagnostic findings
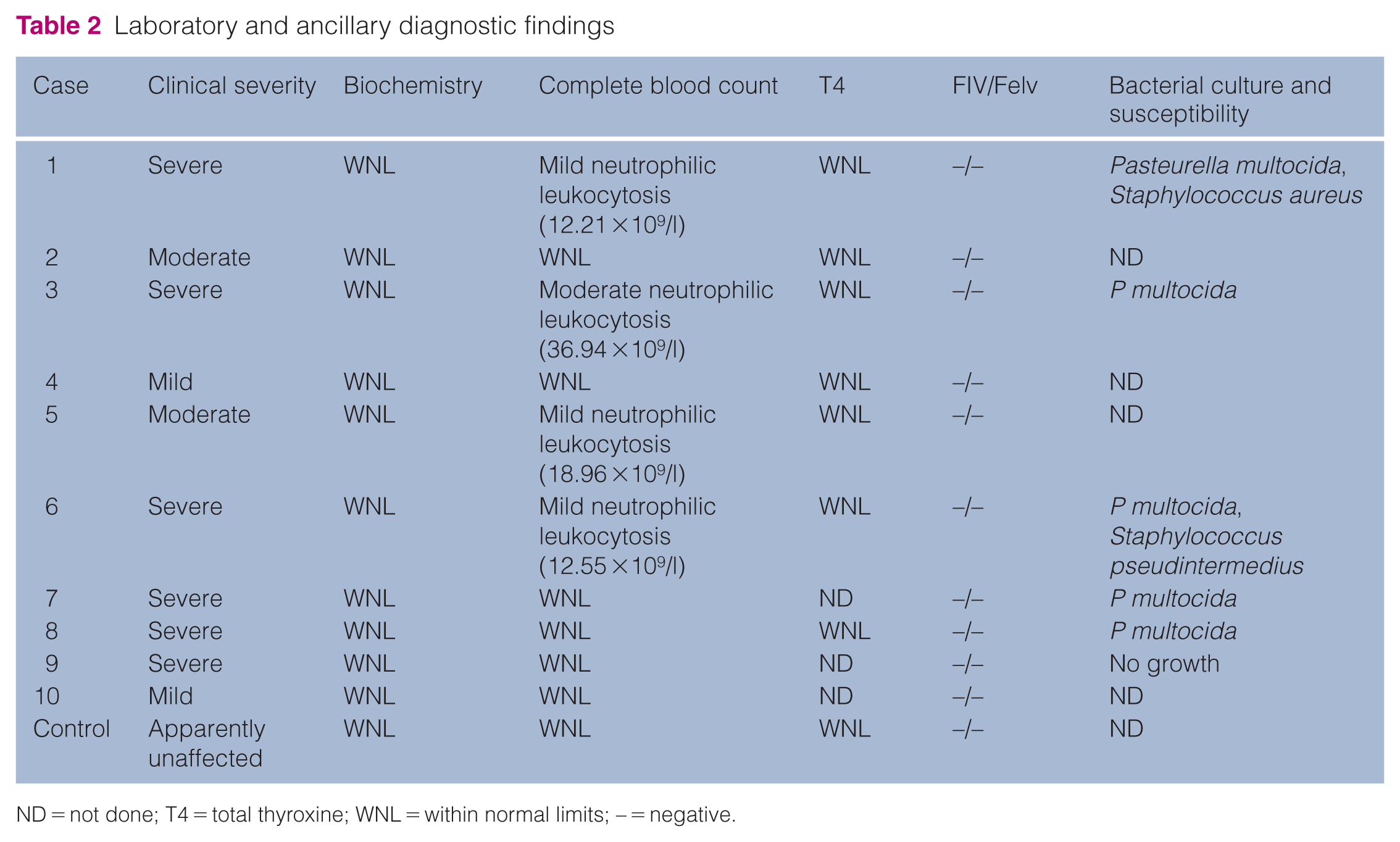
ND = not done; T4 = total thyroxine; WNL = within normal limits; – = negative.
Parasitic and fungal investigations
Skin scrapings for ectoparasites and trichoscopy, Wood’s lamp examination and fungal culture on Sabouraud’s agar were all negative (n = 11/11).
Trichoscopic examination
For each Lykoi, a total of 30 hair shafts, collected from the back and, if possible, the ventral abdomen, were examined by trichoscopy. No structural abnormalities of the hair shafts (notably, hair shaft dysplasia) were observed.
Bacteriological examination
Among the six severely affected Lykoi, five yielded positive deep swab cultures for Pasteurella multocida, all of which demonstrated broad antibiotic susceptibility. In addition, two cats were also positive for Staphylococcus pseudintermedius and Staphylococcus aureus, respectively. One Lykoi tested negative.
Histopathological findings
In lesional areas, skin biopsies revealed cystic lesions in all affected Lykoi (n = 10/10), irrespective of clinical severity.
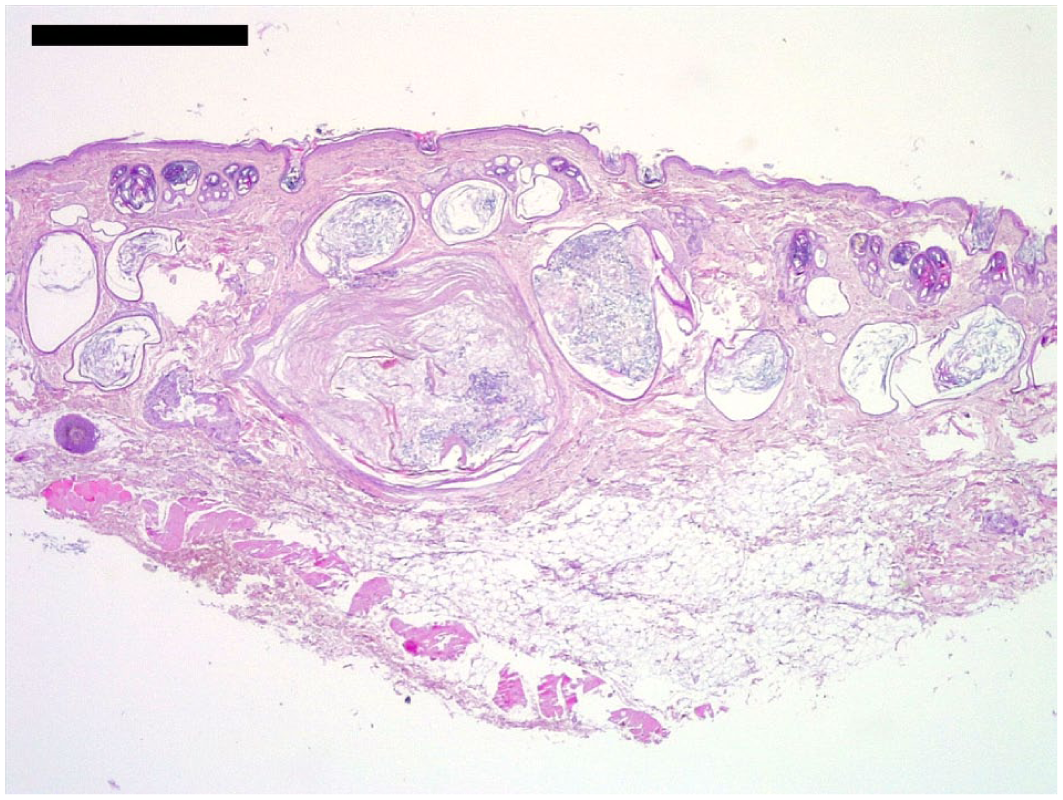
In 9/10 affected Lykoi, dermal infundibular follicular cysts were observed (Figure 6). These were lined by a keratinising stratified squamous epithelium with a well-developed granular layer and contained concentrically laminated infundibular keratin. Some of these cysts approached or breached the surface, but the majority did not have an actual epidermal opening. These cysts were interpreted as comedones (when opened) or, more frequently, as true infundibular follicular cysts (when closed). Samples obtained from regions described as ‘white comedones’ showed closed infundibular follicular cysts, whereas biopsies of ‘black comedones’ revealed larger opened follicular cysts (comedones).

Low magnification of a large follicular cyst. Haematoxylin and eosin. Bar = 1000 µm
In all 10 affected Lykoi, sebaceous duct cysts were observed (Figure 7). They were lined by a poorly keratinising squamous epithelium, with keratohyalin granules that were absent to sparse. They frequently displaced surrounding sebaceous glands peripherally and, in some cysts, one or more sebaceous glands were in direct contact with the lumen. They were optically empty or filled with a small amount of pale, amphophilic, amorphous to finely granular material, consistent with sebum.

Detail of a sebaceous duct cyst displacing sebaceous glands peripherally. Haematoxylin and eosin. Bar = 200 µm
In half (n = 5/10) of the affected Lykoi, sweat gland cysts were observed (Figure 8). They were lined by a typical cubic to flattened apocrine secretory epithelium. They were optically empty or filled with amphophilic, slightly brown, amorphous material. In three of these five Lykoi, these cysts were surrounded by or filled with few pigmented macrophages and/or neutrophils (mild perihidradenitis and hidradenitis).

Detail of a sweat gland cyst. Haematoxylin and eosin. Bar = 200 µm
In a few affected Lykoi (n = 3/10), there were dermal foci of pyogranulomatous inflammation intermingling with keratin lamellae or sebaceous-like material, consistent with cyst rupture eliciting secondary foreign body inflammation (Figure 9).

Detail from a pyogranuloma. Focal inflammation with neutrophils, foamy macrophages and a few multinucleated giant cells with haemorrhage, consistent with a rupture of a sebaceous duct cyst. Haematoxylin and eosin. Bar = 200 µm
Besides cysts and foci of secondary dermal inflammation, the epidermis, dermis and adnexa were otherwise within RIs considering the breed.
The histopathological severity, particularly the size of the cysts and the occurrence of secondary inflammatory lesions, varied among the studied population and was generally proportional to the clinical severity of the lesions.
Biopsies were collected from clinically unaffected skin of all 10 affected Lykoi. In these areas, the epidermal and dermal architecture appeared almost normal, with only a few small (<1 mm) infundibular follicular and sebaceous ductal cysts.
In the control Lykoi, histopathological changes from the skin of the flank were minimal. Hair shafts were more abundant than in other cases, and no infundibular follicular cysts were observed. The only observed cystic lesions consisted of rare, small (<1 mm) sebaceous duct cysts (Figure 10).

Skin of a clinically unaffected Lykoi cat at low magnification. A small, solitary sebaceous duct cyst is visible. Haematoxylin and eosin. Bar = 1000 µm
In the clinically severe form (n = 6/10), follicular infundibular and sebaceous duct cysts were systematically observed (n = 6/6). Sweat gland cysts were observed in fewer than half of the severely affected Lykoi (n = 2/6) (Figure 11). In the clinically moderate form (n = 2/10), follicular infundibular and sebaceous duct cysts were observed in both Lykoi (n = 2/2), with one moderately affected Lykoi having sweat gland cysts (n = 1/2).
Skin of a severely affected Lykoi cat at low magnification. The dermis is crowded with numerous follicular and sebaceous duct cysts. Haematoxylin and eosin. Bar = 1000 µm
In the mild form (n = 2/10), follicular infundibular cysts were observed in one Lykoi. Sebaceous duct cysts and sweat gland cysts were observed in both Lykoi.
Four Lykoi presented the three types of cysts: infundibular follicular cysts, sebaceous duct cysts and sweat gland cysts. Of these four Lykoi, half (n = 2/4) had a severe clinical form, one had a moderate form and the last was considered mild.
Genetic features
Genotyping of the six known HR variants identified five variants among the cats studied; the NC variant was not found in any cat. The two most frequent variants were TX and VA. Two affected cats were homozygous TX/TX and two were homozygous VA/VA, while one affected cat was homozygous TN/TN. The remaining six cats were compound heterozygous for combinations of the TX, VA, TN, Ca and Fr variants; the control cat was heterozygous TX/VA. Thus, no predominant genotype was identified among the 11 cats (Table 3).
Distribution of HR gene variants according to clinical form

Alleles were named according to Buckley et al 2
Discussion
To the best of our knowledge, this is the first report of a cystic cutaneous syndrome in the Lykoi breed. Given the coexistence of infundibular follicular, sebaceous duct and sweat gland cysts, along with its presumed genetic basis, we propose the term cutaneous adnexal polycystic syndrome (CAPS) of the Lykoi.
Cutaneous cystic syndromes of genetic origin have been reported in humans and other species, including dogs and rats. 4 In humans, atrichia with papular lesions (APL) is a rare autosomal recessive disorder caused by loss-of-function mutations in the HR gene.5,6 It is characterised by complete alopecia within the first year of life and progressive keratin retention, forming papular cystic lesions. 7 Sebaceous glands remain intact but disconnected from follicles, and sebaceous or sweat gland cysts are absent. 3 Lykoi CAPS and human APL share the presence of infundibular follicular cysts and papules in alopecic areas but differ in two major respects. First, CAPS also involves sebaceous duct and sweat gland cysts, which are absent in APL. Second, the pathophysiology diverges, as human cysts result from defective trichogenesis linked to HR mutations, whereas Lykoi cats retain partially functional follicles and show broader dysfunction of the pilosebaceous–apocrine unit. 8
An autosomal dominant comedo syndrome, unrelated to HR, has also been described in humans. 9 It results from PEN2 (PSENEN: Presenilin Enhancer, Gamma-Secretase Subunit) mutations affecting the gamma-secretase complex.10 –12 Clinically, it presents with papules and comedones from childhood, sometimes evolving into pustular acneiform lesions. 12 Histopathology reveals follicular cysts without parakeratosis, while sebaceous glands are often atrophic or absent. 9
In dogs, Schnauzer comedo syndrome is considered hereditary but remains genetically unconfirmed. 13 It is thought to result from defective infundibular cornification and presents with comedones, papules and keratoseborrhoeic lesions along the dorsal midline. 14 Histopathological examination reveals keratin and sebum plugs obstructing the follicular ostia, leading to cyst formation, without involvement of the sebaceous or sweat glands. 15
In hairless breeds of dogs, such as the Chinese Crested, Mexican Hairless and Peruvian Hairless, infundibular follicular cysts may be associated with the hairless phenotype caused by a FOXI3 (Forkhead Box I3) mutation. 16 These cysts, typically located on the trunk, may develop before 1 year of age and are characterised by dilated infundibula filled with lamellar keratin. Comedone formation is attributed to impaired keratin elimination in dysplastic follicles, and no sebaceous or apocrine gland cysts have been identified.15,16
In cats, infundibular follicular cysts have only rarely been reported in hairless or rexoid breeds, including the Cornish Rex, German Rex, Devon Rex, Sphynx and Selkirk Rex.17 –20 Several genes affecting hair morphology have been identified, notably a KRT71 (Keratin 71) variant in the Sphynx associated with abnormal follicle and hair shaft development. 21 Despite sparse hair in this breed, histopathology reveals follicular dysplasia and abnormal cornification without reduced follicle number or cyst formation. 22
In 2016, LeRoy et al 1 described Lykoi skin as hypotrichotic, with miniaturised, dilated and dysplastic follicles often containing comedones and perifollicular to mural lymphocytic infiltrates. This phenotype results from six distinct loss-of-function variants in the HR gene inherited in an autosomal recessive manner and is not associated with systemic disease. These variants, named TN, TX, NC, VA, Ca and Fr, are localised in exons 3, 8, 10, 11 and 18, with the exon 8 TX variant being the most prevalent in founder lines. 2
Recently, numerous Lykoi cats worldwide have been reported with extensive cutaneous cysts. The skin lesions observed in mid-adulthood appear to be characteristic of this syndrome in this breed; moreover, this syndrome has never been described before. None showed systemic signs, pain or pruritus, but neutrophilia and secondary P multocida infection were noted in severe cases, occasionally improving with antibiotics. By analogy with PEN2 comedo syndrome in humans, severely affected Lykoi may develop chronic lesions with secondary bacterial colonisation. 12
Trichoscopy was normal in all cats, with no evidence of hair shaft dysplasia. Histopathological examination revealed three types of cysts: infundibular follicular, sebaceous duct and sweat gland cysts, which were sometimes associated. Severe and moderate forms consistently exhibited follicular and sebaceous duct cysts, while sweat gland cysts were less frequent. In mild cases, at least one cyst type was present. Compared with prior descriptions, follicular cysts were generally larger, sebaceous duct cysts (reported here for the first time) were consistently observed and sweat gland cysts were present in nearly half the cats. 1 It is possible that the formation of these three types of cysts reflects a common pathomechanism, namely a defect in the abutment and, consequently, in the drainage pathways of the cutaneous appendages, leading to the progressive accumulation of material and ultimately to cyst formation. Notably, small cysts were also found in clinically unaffected skin and in the clinically normal control Lykoi, suggesting subclinical histopathological lesions and questioning whether truly unaffected Lykoi cats exist.
Genotyping of the six published HR variants revealed no discernible pattern of variant distribution among the three classes of affected cats (Table 3). All six variants are loss-of-function variants: two nonsense variants, a duplication, an insertion and two splice site variants, all predicted to produce a truncated protein with no or highly reduced activity. 2 The HR gene encodes a lysine demethylase and nuclear receptor co-repressor involved in hair growth that acts as a transcriptional co-repressor of multiple nuclear receptors, including thyroid hormone receptor, the retinoic acid receptor-related orphan receptors and the vitamin D receptors. HR protein also interacts with histone deacetylases. In the Lykoi breed, several foundation lines were crossed, leading to the birth of compound heterozygous cats for the six variants. 2 The identification of recurrent cases of CAPS in Lykoi cats raises the question of whether there is a syndrome associated with changes in the Lykoi’s coat due to a loss of function in HR. Is this syndrome associated with any loss of function in the HR gene in cats or is there a lineage effect, that is, a predisposition to the syndrome in cats carrying certain genotypes for the various HR variants? Although we did not identify a preferential genotype in affected cats, our study included too few cases to answer this question. It therefore appears necessary to conduct a genotype–phenotype analysis for CAPS including cats affected by the three forms and unaffected cats aged older than 3 years to consider the late onset of clinical signs in some individuals.
Conclusions
The identification of CAPS in Lykoi cats raises other questions, such as the possibility of treatment, the use of affected cats and their relatives for breeding, the implementation of breed-wide monitoring to assess the prevalence of the syndrome and the future of the breed, even though there does not appear to be any impact on the wellbeing of cats affected by CAPS (no systemic signs, pain or pruritus).
Footnotes
Acknowledgements
The authors thank Christine Boulanger for providing access to her Lykoi cats and for sharing her knowledge of the breed.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received financial support from LOOF-AGRIA; Boehringer Ingelheim Animal Health France and Santévet for this study.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
