Abstract
Several mature Leghorn-type hens with the same genetic background experienced skin and feather problems in a breeder flock. There was almost-total feather loss on the head and neck, as well as thickened, scaly skin, and follicular ostia were plugged with keratin debris. Other individuals exhibited prominent subcutaneous nodules multifocally on the head. Histologic examination of the skin revealed a severe hyperplasia of follicular epithelium with hyperkeratosis and cystic dilation. Numerous clefts and vesicles were detected along the epidermis and follicular epithelium, some containing acantholytic keratinocytes. A mild heterophilic inflammation was associated with these lesions, and few gram-positive cocci were present in the keratin plugs. Bacterial culture of the skin yielded a variable amount of Staphylococcus hyicus. Immunochemistry looking for chicken IgY revealed no intercellular staining in the epidermis or follicular epithelium. All these findings supported a diagnosis of Staphylococcus-associated acantholytic epidermitis and folliculitis. This case suggests that S. hyicus could be a significant pathogen in poultry production. The close genetic relationship among affected individuals could indicate a hereditary predisposition in this line of White Leghorn laying chickens.
Vesiculobullous skin diseases result from a defect, congenital or acquired, in the cohesion of keratinocytes. 15 Hereditary disorders, autoantibodies, or proteolytic microbial enzymes can target adhesion molecules at different levels of the epidermis or at the dermoepidermal junction, leading to acantholysis, a cell–cell separation that is the hallmark of these diseases. 10 Some Staphylococcus aureus strains produce exfoliative toxins that induce acantholytic dermatitides in humans, as they target and cleave the extracellular domain of desmoglein 1 (Dsg-1), a glycoprotein of the cadherin superfamily found in desmosomes and responsible for cell-to-cell adhesion in the epidermis, especially in the superficial layers. 15 In veterinary medicine, Staphylococcus hyicus, the causative organism of swine exudative epidermitis, and Staphylococcus pseudointermedius, responsible for dog impetigo and superficial spreading pyoderma, also induce acantholysis in the epidermis. 5 Exfoliative toxins with amino acid sequences similar to those of S. aureus have been identified in these bacterial species and have been proved to target Dsg-1. 3,8,9 In birds, S. hyicus is considered a commensal organism of the skin and is generally implicated as a secondary pathogen in preexisting dermatoses. 1 We describe here a S. hyicus–associated chronic folliculitis and epidermitis with obvious acantholysis involving numerous genetically related hens.
A breeder flock intended for the production of embryonated eggs for human influenza vaccines experienced a suboptimal egg production with a normal mortality rate. Different genetic lines were present within the flock, but the problem was present in only one of them. This line is descended from a Leghorn genetic strain and was composed of 3000 breeder fowl, mostly 51-week-old females.
The owner reported that approximately 25 hens from this lot (0.3%) had almost-total feather loss on the head and neck, a thickened and scaly skin, and prominent nodules on the head. The lesions began when the hens were 28 weeks old. They first appeared around the eyes and ears and gradually spread and covered the entire head and neck. The hens were kept in cages, and the skin problem did not seem to be contagious, as the owner would normally find only one affected bird per cage. They were fed a commercial layer feed that did not contain any animal source protein. Neither males from this flock nor hens of the other 11 genetic lines had any skin problems. All affected birds were euthanized by cervical dislocation. After that episode, this specific genetic line was no longer used, and no new cases have occurred in the flock.
Thirteen hens were submitted to the Laboratoire d’épidémiosurveillance animale du Québec for necropsy and laboratory testing. These birds were in fair body condition. In some hens, there was a small ovary with no mature follicles (inactive ovary). There was partial to total feather loss on the head, neck, and breast and at friction points (wings, thighs). Follicular ostia were plugged with keratin and distinctly raised over the surrounding and crusted epidermis (Fig. 1). There were large nodules (up to 3 cm) in the subcutis of the head of some individuals (Fig. 2). In most hens, the external auditory canal was filled with keratin and had a thickened and irregular mucosa. No internal lesions were seen in all individuals.
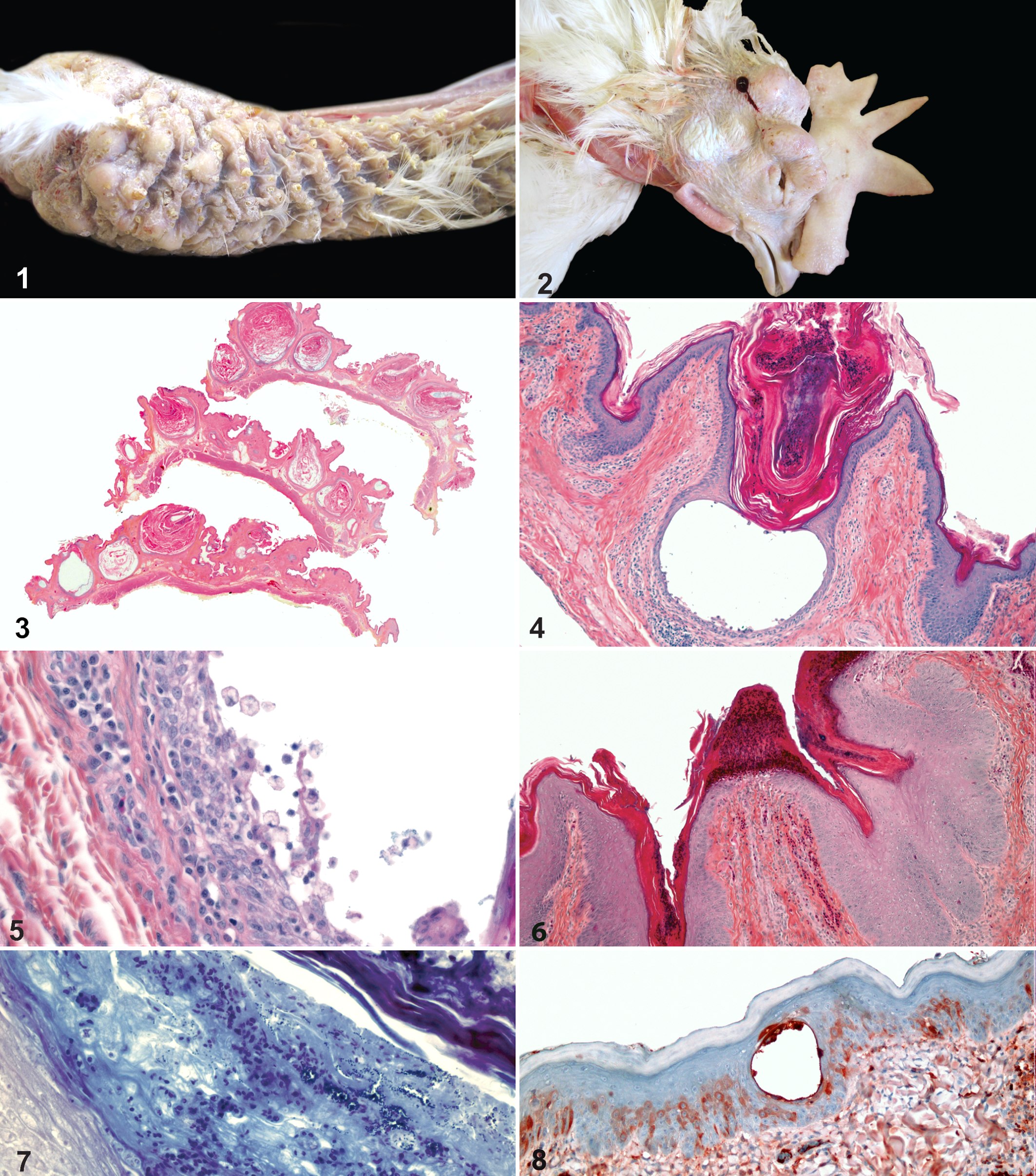
Head and neck, fowl. There is almost-complete feather loss, and follicular ostia are plugged with keratin and distinctly raised over the surrounding and crusted epidermis.
Tissues collected during postmortem examination were fixed in 10% neutral buffered formalin and routinely processed and stained with hematoxylin, phloxine, eosin, and saffron. A Goodpasture gram and a Gomori methenamine silver stain were also performed on skin sections. Microscopic examination of the skin revealed severe follicular keratosis, leading to the formation of cysts with destruction and loss of feathers (Fig. 3). Keratin plugs were often infiltrated by heterophils and huge vesicles that contained clear fluid, and some exfoliating cells (Fig. 4) were multifocally present in the follicular wall. The follicular epithelium, which was irregularly and moderately to severely hyperplastic, showed multifocal intercellular edema (spongiosis) and dissociation of keratinocytes, with vacuolar degeneration of epithelial cells and release of acantholytic cells (Fig. 5). The superficial epidermis showed a mild to moderate acanthosis with rare crusts of granulocytic inflammation similar to those of porcine exudative epidermitis (Fig. 6). Some heterophils were also infiltrating the pulp of residual feathers and were present in the follicular lumen, along with a small amount of large gram-positive cocci (Fig. 7). No fungi were detected with the Gomori methenamine silver stain. The superficial dermis showed a diffuse and mild fibrosis, as well as a very mild lymphoplasmacytic perivascular and perifollicular inflammation. At the external auditory canal level, the same changes were noted, but bacteria were more abundant and the inflammation marked, with foci of heterophilic epidermitis and adenitis with extension to the adjacent bone (osteomyelitis). The large nodules noted macroscopically in the subcutaneous tissue were heterophil granulomas centered on numerous gram-positive cocci. No remnants of follicular epithelium were present around these subcutaneous granulomas, and there was no inflammation in the overlying skin. No significant changes were noted in other organs.
Routine bacterial culture of the skin of 3 individuals yielded moderate to heavy growth of S. hyicus. S. aureus was also present in moderate quantity in the skin of 1 hen. Rare S. aureus or Staphylococcus spp. were also cultured from the liver of several birds.
Paraffin-embedded skin sections of sick and age-matched normal hens were cut in 3-μm sections and routinely prepared for a streptavidin–biotin peroxidase method. Polyclonal rabbit anti-chicken IgY (IgG) (Jackson ImmunoResearch, West Grove, PA) at a 1:1000 concentration was used as a primary antibody and biotinylated horse anti-mouse/anti-rabbit serum (Vector Laboratories, Burlington, Ontario, Canada) as the secondary one. The technique revealed a diffuse intracytoplasmic staining for IgY in individual or small groups of epidermal and follicular keratinocytes. The reaction was especially marked in exfoliating cells (Fig. 8). No intercellular staining was apparent. IgY deposition was not found in the normal hen’s skin.
S. hyicus is poorly pathogenic in fowl and considered as a commensal organism of the skin. 1,14 To our knowledge, this is the first case where this bacteria is positively associated with an acantholytic dermatitis in the domestic fowl. Hoffman (1939) described a vesicular dermatitis of the head and neck in White Leghorn chickens, from which staphylococci were isolated. 7 However, the article did not identify the bacterial species, only that it was a nonpigmented Staphylococcus. Many staphylococci isolated from animals, including S. hyicus, are nonpigmented bacteria, and even Staph aureus could appear nonpigmented, as the color of the colonies is influenced by the composition of the culture media and the host species from which it has been isolated. 12
At least 6 exfoliative toxins can be produced by strains of Staph hyicus, and of those, 4 have been shown to target porcine Dsg-1. 2,9,10 Lysis of the extracellular domain of this cadherin family glycoprotein leads to acantholysis, a characteristic lesion of porcine exudative epidermitis as well as other staphylococcal dermatitides, such as human bullous impetigo and staphylococcal-scalded skin syndrome. 10 It has been demonstrated that at least 1 exfoliative toxin of S. hyicus can experimentally induce exfoliation in the skin of fowl 10 days old or less. However, the same toxin failed to produce lesions in birds aged 15 to 20 days. 13 Thus, the toxin seems to recognize and digest fowl Dsg-1 as well as porcine Dsg-1, but affinity of the toxin for the avian protein appears to be age-related. A recent study raised the possibility that at least 2 of 7 single nucleotide polymorphisms in the porcine Dsg-1 gene could have an effect on the susceptibility of the animals to develop exudative epidermitis. 2 As the only birds with lesions in this flock were of the same genetic line, we hypothesized that a particular conformation of the Dsg-1 gene could have favored the action of S. hyicus exfoliative toxin(s) on this protein in mature hens. Unfortunately, further investigation was not possible, as this genetic line was eliminated.
Alternatively, a preexisting hereditary vesiculobullous skin disease could have been present in these birds, with S. hyicus involved only as a secondary invader. The only report of such a disease in birds involved ostriches, which developed splitting of the epidermis and bullae, lesions more similar to epidermolysis bullosa than to staphylococcal dermatitides. These lesions appeared at a very young age (few days old), which is expected with hereditary genetic skin diseases. 4
The lesions present in our case show similarities with those described in a pemphigus-like dermatosis in a fowl. 6 A nice intercellular fluorescence of antibodies was demonstrated in that bird, the hallmark lesion of pemphigus foliaceus. 5,6 To rule out pemphigus, we performed immunohistochemistry for avian IgY (bird IgG) on the skin of our birds. Only occasional intracytoplasmic staining was observed in the epidermis and follicular epithelium and was interpreted as nonspecific (T. Olivry, personal communication). Interestingly, in the bird with confirmed pemphigus, there was an overgrowth of bacteria, and although antibiotic susceptibility was determined, no bacteriology culture results were indicated. 6 It could have been interesting to see if a Staphylococcus sp. were present.
Differences exist between the histologic lesions observed in the hens of the present report and those produced by toxigenic staphylococci in mammals. In contrast to the present case, where most of the lesions involved follicles, the usual target of the toxins in mammals is the epidermis, although follicular epithelium can be affected in canine and porcine Staphylococcus-associated dermatitides. 5 The reason for this difference in location is unknown. The avian follicular epithelium might be more sensitive to bacterial toxins than the epidermis, or a specific genetic defect could be affecting the follicular epithelium of these birds.
Acantholysis was especially obvious in this case compared with mammalian staphylococcal dermatitides, in which pustules form very early in the disease, thus making acantholysis difficult to detect. 5,10 In contrast to mammals, in which lysis of intercellular bonds in the keratinizing process begins in the upper parts of the stratum granulosum, this phenomenon occurs at a low level in the stratum basale of birds. 4,11 This could possibly reduce the strength of intercellular cohesion and facilitate acantholysis. The inflammation was also minimal compared with what is usually present in mammals. In the present case, the response to the infection was rather a severe hyperkeratosis. Similarly, hyperkeratosis was a major feature in the pemphigus report in a fowl, even if heterophilic inflammation were more prominent than in our case. 6 This could indicate a specific manner of bird skin to react to an insult.
In conclusion, S. hyicus was associated with an acantholytic folliculitis and epidermatitis in middle-aged fowl under natural conditions. As these birds were all of the same lineage, a genetic predisposition is most likely. Avian acantholytic dermatitides also display a specific pattern of lesions compared with mammals.
Footnotes
Acknowledgements
We thank Claude Paquet and Dr Monique Doré for technical and professional assistance with immunohistochemistry, Drs André Dallaire and Malcom Gains for kindly giving comments on the manuscript, and Marco Langlois for technical assistance with the pictures.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
